Abstract
Arterial disease is associated with elevated serum matrix metalloproteinase (MMP)-8 concentration. We studied the role of two promoter region single nucleotide polymorphisms (SNPs) of MMP-8 gene in the arterial disease. The population comprised patients with arterial disease (n = 124) and healthy blood donors (n = 100) as a reference group for MMP-8 SNPs (−799C/T and −381A/G) genotypes and serum concentrations. Genotype frequencies for MMP-8 −799C/T SNP in arterial disease were C/C (43.5%), C/T (32.3%) and T/T (24.2%), and in the reference group they were C/C (50.0%), C/T (40.0%) and T/T (10.0%; P = 0.012). The −799C allele frequency was lower in the patients (59.7%) than in the reference group (70.0%; P = 0.023). The −799C allele showed protective effects against the arterial disease with an odds ratio [95% confidence interval (CI)] of 0.372 (0.141–0.980, P = 0.045) after adjustment for age, gender, and serum MMP-8 and TIMP-1 concentrations. Only in the reference group and whole study population (n = 224), the −799TT genotype significantly associated with an increase in serum MMP-8 concentrations (P = 0.047, 0.025). The −799C allele appeared protective against the arterial disease. The genotype may have an effect on systemic MMP-8 levels which could not, however, be seen in the arterial disease patients probably as a result of the strong inflammation involved in the disease pathogenesis.
Introduction
Matrix metalloproteinases (MMPs) form an enzyme family composed of structurally related, but genetically distinct, zinc-dependent proteolytic enzymes that can process or degrade a variety of extracellular matrix and non-matrix bioactive proteins.1,2 Collagenase-2 (MMP-8), also called neutrophil collagenase, is a member of the 23 human MMP-family and produced not only by neutrophils, but also by macrophages, endothelial cells, smooth muscle cells and fibroblasts. 3 MMP-8 is catalytically the most competent type I collagenase 3 present at inflammatory sites and plays an important role in extracellular degradation and tissue remodeling.4,5 In addition to the surrogate classical function ofMMP-8 as a mediator in connective tissue destruction, its ability to decisively process various bioactive non-matrix substrates such as chemokines, cytokines, growth factors, hormone receptors and immune mediators can exert anti-inflammatory, defensive characteristics.1,6– 12
Atherosclerosis is an inflammatory disease. 13 Circulating leukocytes adhere to the vascular endothelium and penetrate into arterial intima where they differentiate into macrophages, take up modified lipoproteins and transform to lipid-laden foam cells. The foam cells are the hallmarks of atherosclerotic lesions. 13 Atherosclerotic plaques also comprise of extracellular matrix proteins, for example type I and III collagens and proteoglycans, and vascular smooth muscle cells. 13 Various MMPs participate in different steps of atherosclerotic plaque and aneurysm formations. 14 Smooth muscle cells, endothelial cells and macrophages in atherosclerotic lesions can express MMP-8 thereby increasing their potential to rupture.3,15 It has also been demonstrated that elevated serum MMP-8 concentrations are associated with the presence of atherosclerosis, arterial disease and cardiovascular outcome. 16 – 18
MMP-8 single nucleotide polymorphisms (SNP), in particular, have been reported to alter the promoter activity and subsequent gene expression in preterm premature rupture of membranes. 19 The SNPs influence breast cancer prognosis 20 and decrease lung cancer risk. 21 However, whether MMP-8 SNP affects the serum MMP-8 concentration is not known. Thus, we were primarily interested in examining in the present hypothesis generating study of: i) whether the variation is associated with arterial disease; ii) whether the variation affects the serum MMP-8 concentration in arterial disease; and, iii) whether SNPs or disease explain the high serum MMP-8 concentration associated with arterial disease.
Materials and methods
Patients
The sample was previously described in detail. 17 Originally, the patients were collected in order to investigate a wide range of infection and inflammation associated parameters in arterial disease. Briefly, the population comprised 124 patients who underwent surgery because of symptomatic aorto-occlusive disease (n = 18), carotid artery stenosis (n = 65) or abdominal aortic aneurysm (n = 41) in the Department of Vascular Surgery, Helsinki University Central Hospital between 2002 and 2004. Pre-operative blood samples were collected from all patients before the induction of anesthesia. The vascular tissue samples were obtained during surgery. The mean age (±SD) and percentage of male patients were 67.55±9.7 years and 79.0% respectively.
Reference group
The samples of the reference group were provided by healthy blood donors (n = 100) collected by the Finnish Red Cross, Oulu, Finland, as previously described. 17 Any of the following clinical characteristics were not accepted for blood donation: diabetes mellitus; strong allergies; neurologic diseases; any acute infections; rheumatoid arthritis; recent oral, or any other, operations; cancer; chronic liver or kidney disease; Crohn's disease; tendency to bleed; or sexually transmitted diseases; as well as asthma or hypertension not in treatment balance. Subjects with the following cardiovascular diseases were also excluded: stroke, acute myocardial infarction, coronary artery disease, eye thrombus, angina pectoris, frequent arrhythmia, aorto-occlusive disease, phlebitis, or rheumatic fever. The donors were between 18 and 65 years old, and their haemoglobin levels were 135–195 and 125–175 g/l for males and females, respectively. The mean age (±SD) and percentage of male reference subjects were 58.41±4.4 years and 53.0% respectively. The only data available from reference group was age and gender. Therefore, performing the combined effect of genetic and non-genetic factors in the patients was impossible with respect to reference group.
All patients and healthy blood donors gave their informed consent. The Local Ethics Committee at Helsinki University Central Hospital and the Finnish Red Cross, Oulu, Finland approved the study protocol.
DNA extractions
Vascular tissue samples were collected during the surgical procedure, were snap–frozen in liquid nitrogen and stored at −70°C for later DNA extraction. DNA was extracted as previously described using QIAamp DNA mini kit (QIAGEN, Crawley, UK) tissue protocol according to the manufacturer's instructions. 22 For the reference group, buffy coats were separated and the cells were stored at −80°C until the DNA extraction done with the QIAamp DNA mini Kit (Qiagen) tissue protocol according to the manufacturer's instructions. After extraction, the DNA concentrations and purity were determined photometrically.
MMP-8 and TIMP-1 serum concentrations
Serum MMP-8 and TIMP-1 concentrations 17 were determined both in the patients with arterial disease and the reference groups using ELISA kit (GE Healthcare, Buckinghamshire, UK) according to the manufacturer's instructions. All samples were analyzed in duplicate. Intra-assay co-efficient of variations for MMP-8 was2.5% (n = 12) and inter-assay co-efficient of variations was 4.8% (n = 12). The detection limit for MMP-8 was 0.032 ng/ml and for TIMP-1 it was 1.25 ng/ml.
Genotyping
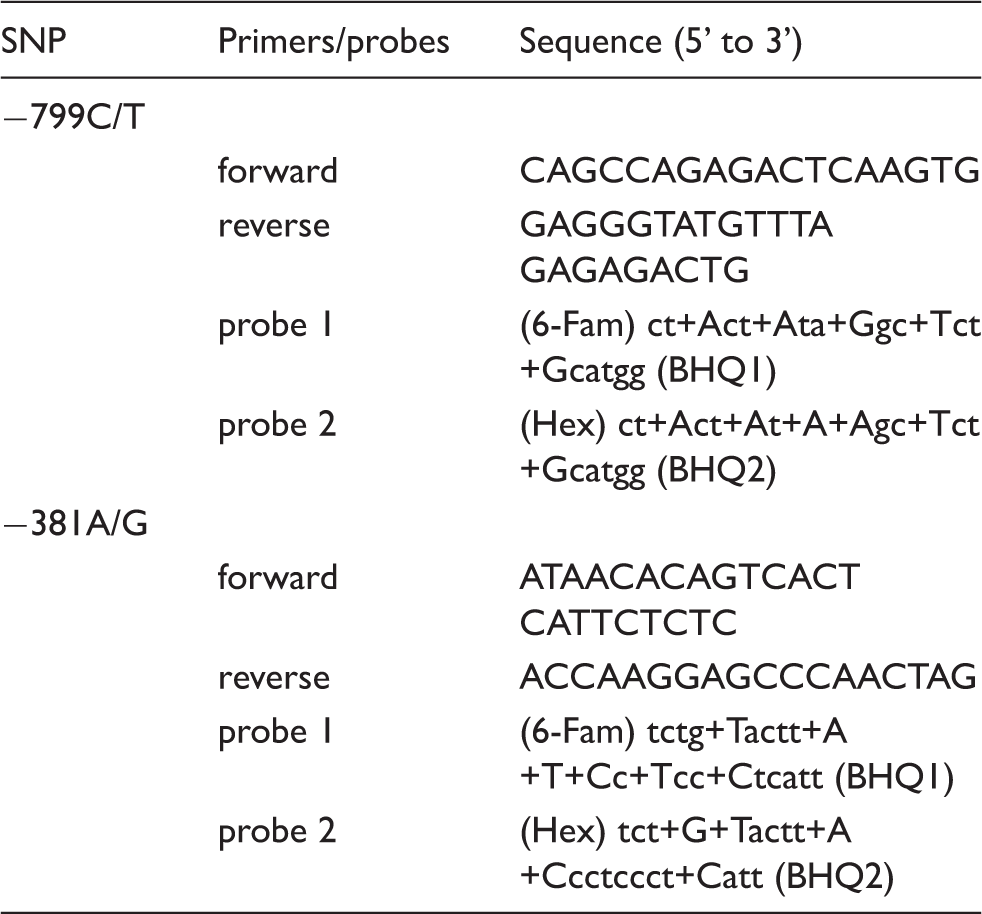
Primer and probe sequences used in the detection of MMP-8 SNPs
Statistical analyses
The genotype and allele frequencies were calculated using the direct genotype or gene counting method. The significance of the differences in the frequencies between the patients and the healthy blood donors wascompared using the Chi-square test producing odds ratio (OR) and 95% confidence intervals (95% CI).
The significances of the differences between the patients and the healthy blood donors in continuous variables, e.g. serum MMP-8 concentration were compared using the Mann-Whitney U test. Continuous data are presented as median [interquartile range (IQR) 25%–75%].
Logistic regression analyses were applied to study the potential association of the SNPs with the arterial disease. For this, four different models were performed. In the first models, only the allele data were used. In the multivariate models, in addition to the allele data, confounding factors, i.e. age, sex and serum MMP-8 and TIMP-1 concentrations, were added with forward stepwise method. Data are presented with the OR, 95% CI and P value.
Data were analyzed using the SPSS 15.0 statistical package (SPSS Inc., Chicago, IL, USA). For all statistical tests lower than 0.05, P-values were considered as statistically significant.
Results
Genotype and allele distributions of MMP-8 SNPs among patients with arterial disease and healthy blood donors
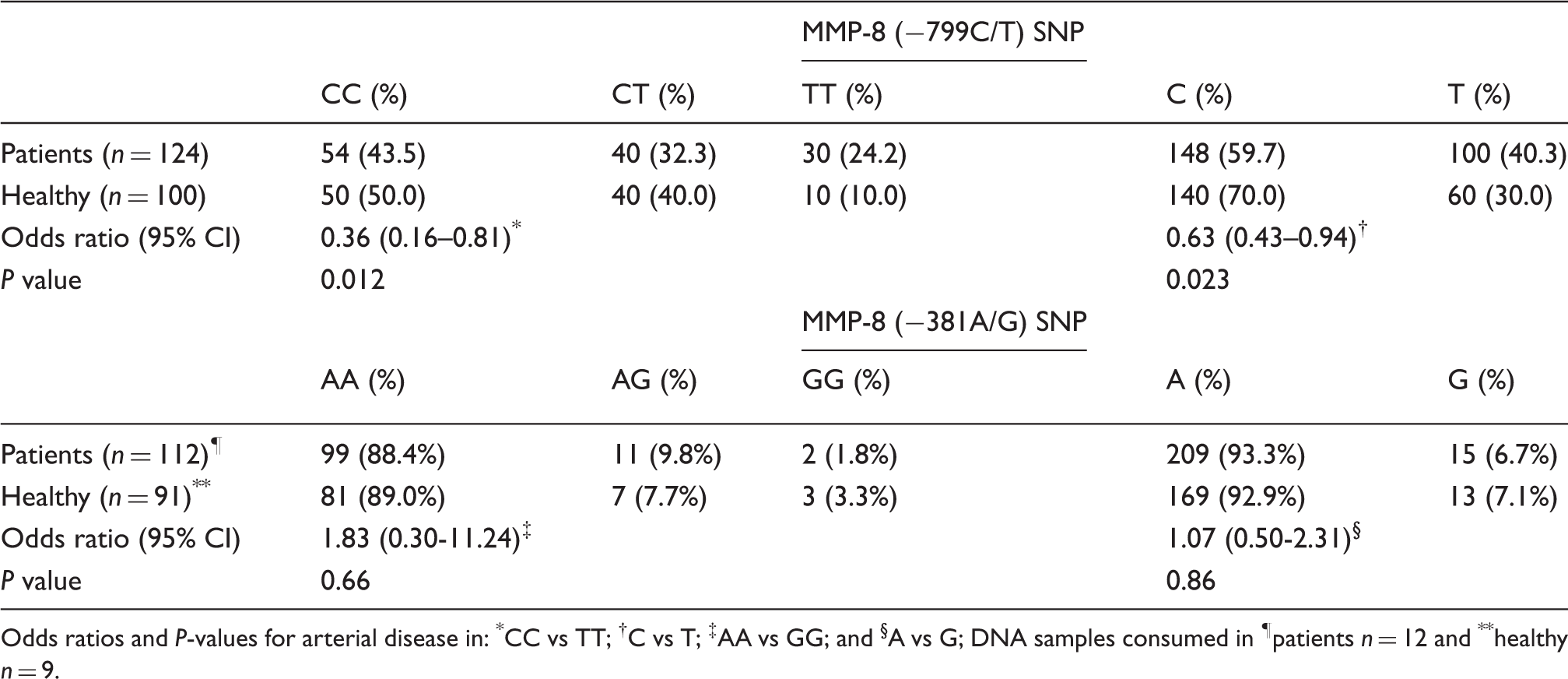
Odds ratios and P-values for arterial disease in: *CC vs TT; †C vs T; ‡AA vs GG; and §A vs G; DNA samples consumed in ¶patients n = 12 and **healthy n = 9.
We examined the serum MMP-8 concentration in relation to SNP variations studied. We compared the serum MMP-8 concentrations separately between −799CC and −799TT genotypes and between −381AA and −381GG genotypes separately in the reference group and in the patients. In the patients, we found no significant differences (Table 3). Within the reference group, the subjects having the −799TT genotype (median = 5.10, IQR = 3.08–14.54) had higher MMP-8 serum concentrations when compared with the subjects whohad the −799CC genotype (median = 3.26, IQR = 1.93–6.36) (Mann-Whitney U Test, P = 0.047, Table 3). Similarly, in the whole study population, the subjects that had the −799TT genotype (median = 8.91, IQR = 6.25–19.60) had a higher MMP-8 serum concentration when compared with the subjects who had the −799CC genotype (median = 7.51, IQR = 3.16–12.23) (Mann-Whitney U Test, P = 0.025) (Figure 1). No other significant differences were noticed. We further analyzed the serum MMP-8 concentration within subgroups of the SNP variations. The patients had a significantly higher serum MMP-8 concentration when compared with the reference group, when analyses were made separately within the −799CT, CC genotypes, or C and T alleles, but not in the TT genotype (Table 3). Similar results were observed when analyses were made separately within the −381AA, AG genotypes, or A and G allele, but not in the GG genotype. However, no statistically significant differences in patients with the −799TT or the −381GG genotypes were observed (Table 3).
Analysis of serum MMP-8 concentrations and MMP-8 (−799C/T) SNP by box plot graph. The figure includes the whole study population (n = 224) as indicated. P-value indicated significance as compared between serum MMP-8 concentrations and MMP-8 (−799C/T) genotypes. Association of MMP-8 gene promoter SNP and serum MMP-8 concentration Values indicate median (IQR) (number of individuals). P-value for Mann-Whitney U test. Comparisons made between the healthy blood donors and the patients within the given genotype or allele. Comparisons made within healthy blood donors or patients. DNA samples consumed in healthy blood donors n = 91 and patients n = 112.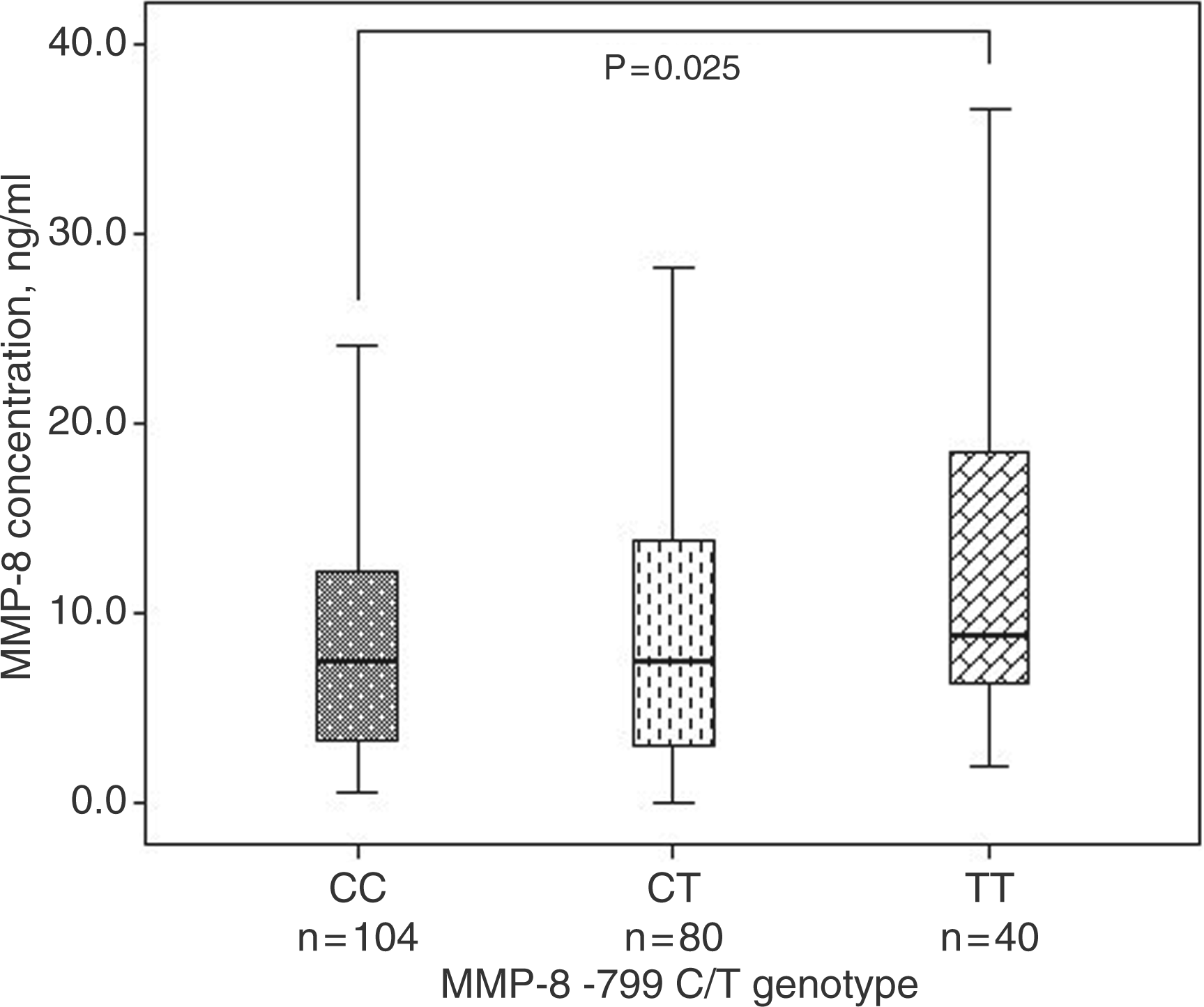
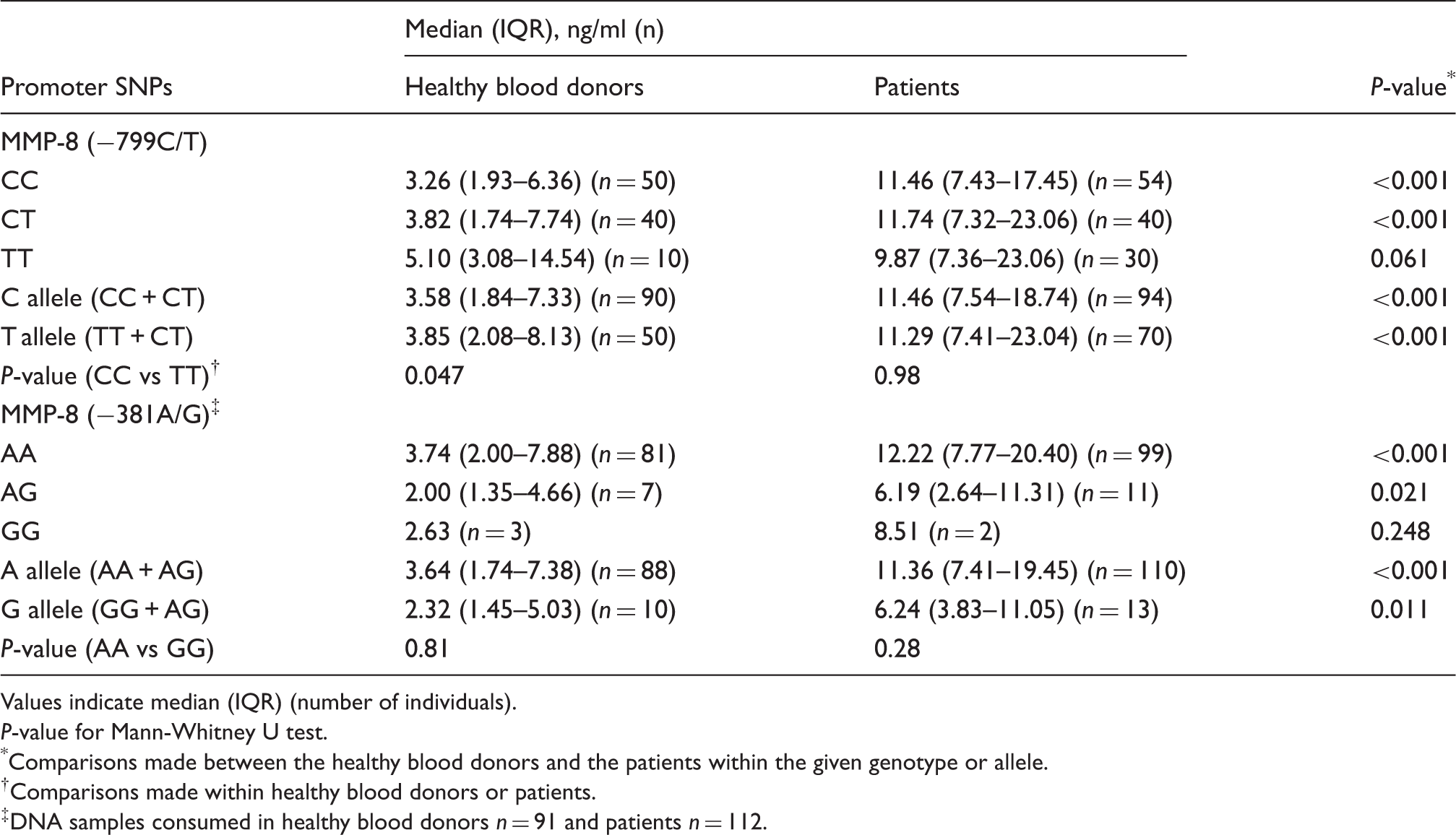
Analysis of arterial disease risk estimation *
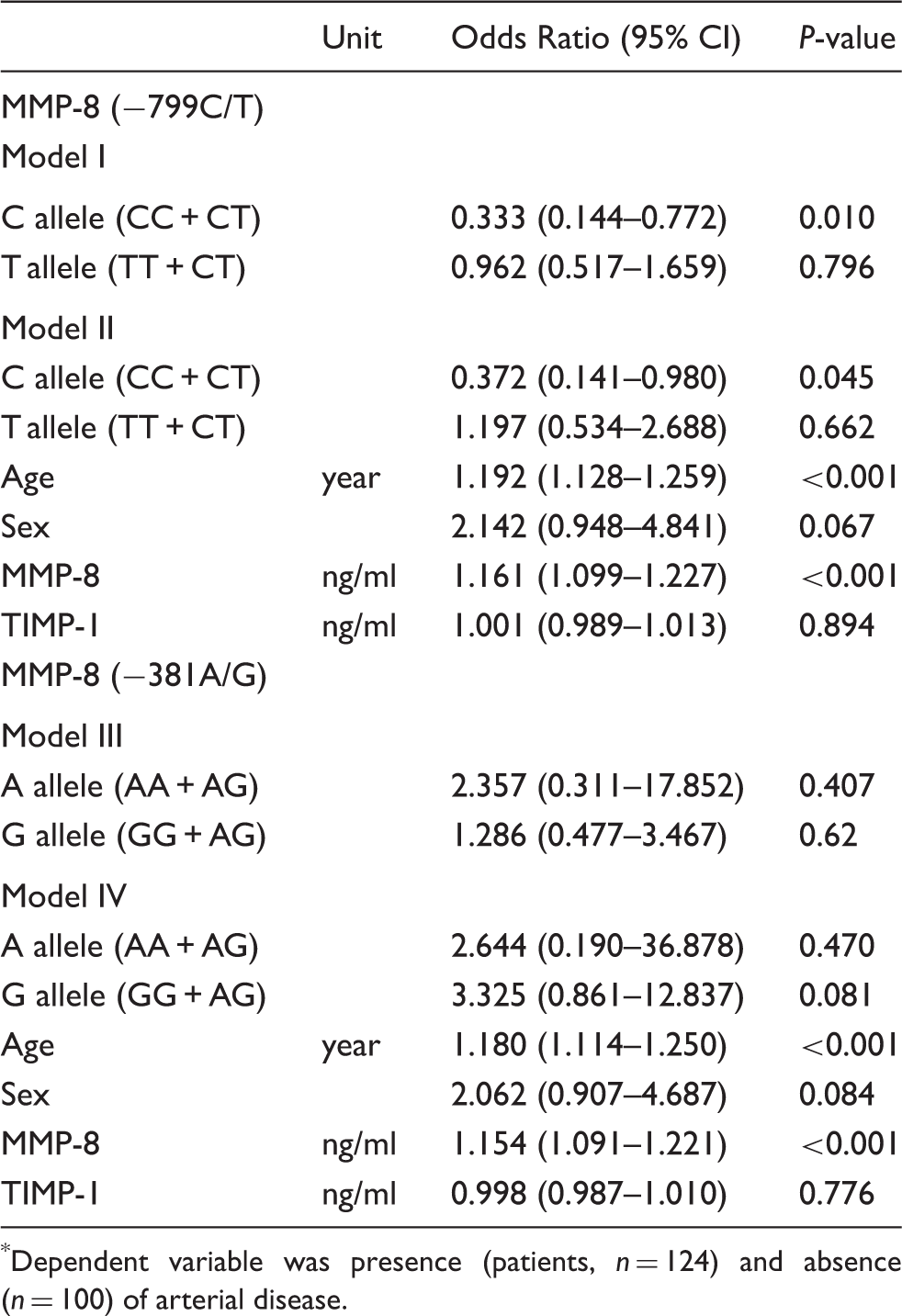
Dependent variable was presence (patients, n = 124) and absence (n = 100) of arterial disease.
Discussion
To the best of our knowledge, this is the first study inarterial disease that examines the functionally-important MMP-8 SNPs variations in relation to the serum MMP-8 concentrations. Our finding is the first to indicate that the C allele of the MMP-8 −799C/T exerts a protective effect against arterial disease that persisted after adjustment for age, gender and serum MMP-8 and TIMP-1 concentrations. This was supported by the univariate analysis and later confirmed by the multivariate analyses. Interestingly, we also showed, for the first time, that, in the reference group, −799TT genotype associated significantly with the increased serum MMP-8 concentrations. Similarly, in the whole study population, the −799TT genotype significantly associated with the increased serum MMP-8 concentrations. In patients, serum MMP-8 concentrations did not differ significantly between MMP-8 genotypes. In the recent studies, high serum MMP-8 concentration has been shown to associate with an increased risk for cardiovascular diseases 16 and arterial disease. 17 However, the reasons for these associations have remained unknown. Certain SNPs in the promoter region of the MMP-8 gene have been shown to exert a higher promoter activity and subsequent gene activation. 19 It is not fully known if these MMP-8 SNPs affect serum MMP-8 concentrations.
The MMP-8 gene is located on chromosome 11 and codes for the MMP-8 protein. Three SNPs have been identified in this gene. 19 Two of these (−799C/T and −381A/G) are located in the MMP-8 promoter region and have putative functional significance, increasing the promoter activity and subsequent gene activation. 19 MMP-8 plays an important role in proteolytic degradation and remodeling of various extracellular matrix components in both physiologic and pathologic conditions, including tissue repair, angiogenesis, and tumor cell invasion and metastasis. MMP-8 can also, by processing the non-matrix bioactive molecules, modify the immune responses and participate in anti-inflammatory defensive processes.1,6–8,10,12
Under normal physiologic conditions, MMP-8 isexpressed at very low levels, whereas enhanced expression and activation has been found in various tumors 23 – 26 and cardiovascular diseases.15,27 It has been shown that MMP-8 can be produced by endothelial cells, plasma cells, smooth muscle cells and macrophages in atherosclerotic plaques 3 , and elevated levels of serum MMP-8 are associated with atherosclerosis 16 and arterial disease. 17 In vitro experiments in breast cancer have demonstrated that the T allele of −799C/T is associated with higher promoter activity compared with the C allele 20 and in preterm premature rupture of membrane 19 . However, no association has been demonstrated between MMP-8 SNP and head and neck squamous cell cancer 28 , hepatocellular carcinoma 29 , bronchiectasis 30 or metabolic syndrome. 4 Compared with the reference group, our patient population had a low frequency of the −799C allele, indicating a high probability of increased promoter activity and subsequent gene activation. In previous studies, the frequency of the −799C allele in patients with bronchiectasis was 66.8% 30 and in metabolic syndrome was 66.5%. 4 In our study, the allele frequencies were of the same magnitude in patients (59.7%) and in the reference group (70.0%). On the other hand, patients with arterial disease had a high serum MMP-8 concentration compared with the reference group, when the analyses were made separately within the different genotypes or alleles. Therefore, we were unable to demonstrate a direct relationship between the serum MMP-8 concentrations and the MMP-8 genotypes in the patients with arterial diseases. Similar findings were found by Aquilante et al. 4 when they investigated the association of MMP-8 −799C/T and −381A/G and metabolic syndrome.
The MMP-8 gene is probably activated as a result of continuously activated inflammatory cascades.3,15,27 It has been previously unknown whether the increment in MMP-8 concentration is affected by promoter polymorphisms. In this regard, our report is the first to show the direct association between serum MMP-8 concentrations and the −799TT genotype in the reference group. The previously known putative functional significance19,20 of the SNPs studied can probably be extendable, i.e. the effects of the −799TT genotype in serum MMP-8 concentrations can only be seen in healthy subjects, where normal biology is still intact, but not in patients with arterial diseases where normal biology is disturbed and a high degree of inflammation is present. Arterial diseases are such as a complex group of diseases where one phenomenon cannot be solely explained by one risk factor, and several component or markers collectively affect the disease outcome. Arterial disease is strongly associated with inflammation. Certain inflammatory markers are always found to be high; MMP-8 is one of them.1,16,18 MMPs are a complex family where one proteolytic enzyme may activate or inactivate another in cascades. On the other hand, MMPs also exist in different forms, e.g. latent and active forms. 31 Thus, we suggest that the increased serum MMP-8 concentration in patients with arterial diseases could be caused by the strong inflammation cascades typical for the arterial diseases. Furthermore, investigating the association between the SNPs and the degree of MMP-8 activation could be informative in understanding the exact role of MMP-8 SNP in the etiology of arterial disease. Additionally, the association between the MMP-8 SNP and differences in serum MMP-8 concentrations in the reference group has captured the attention for further investigation in larger, case-control studies.
Study limitations
First, our study population was relatively small. Second, the study population was enrolled from hospitals and reference subjects were collected from the healthy blood donors. Thus, inherent selection bias cannot becompletely excluded. Third, as we could receive only age and gender data from the healthy blood donors, we were unable to perform the combined effect of genetic and non-genetic factors in the diseases with regards to reference group.
Conclusions
The C allele of MMP-8 −799C/T appeared protective against arterial disease. Compared with the reference group, serum MMP-8 concentrations were higher in patients with arterial diseases; the genetic variations in the promoter region may have an effect in the gene activations. In the reference group, an association between the MMP-8 −799C/T SNP and serum MMP-8 concentrations was observed. However, at the level of serum concentration studies, the genetic effect may be shadowed by the strong inflammatory response found in patients with arterial disease. Our results warrant further investigation in larger, population-based studies.
Funding
This study was funded by grants from the Academy of Finland [#1130408 for TS], the Sigrid Juselius Foundation (PJP), Finnish doctoral program in oral sciences (PP-P) and grants from the Helsinki University Central Hospital Research Foundation.
Conflict of interest statement
None declared.
