Abstract
Yersinia pestis, a Gram-negative bacillus causing plague and Centers for Disease Control and Prevention (CDC) classified Category A pathogen, has high potential as a bioweapon. Lipopolysaccharide, a virulence factor for Y. pestis, binds to and activates A1 adenosine receptor (AR)s and, in animals, A1AR antagonists block induced acute lung injury (ALI) and increase survival following cecal ligation and perforation. In this study, rats were infected intratracheally with viable Y. pestis [CO99 (pCD1
Introduction
Yersinia pestis, the causative agent of plague, is a Gram-negative bacillus classified as a Category A pathogen by the Centers for Disease Control and Prevention (CDC), reflecting its high potential as a weapon of bioterrorism. There is significant concern that Y. pestis may be used as a bioweapon in warfare or a terrorist attack as it is widely available, can be mass produced, can be aerosolized for widespread dissemination, may spread rapidly beyond the initial site of the attack, and is both highly contagious and lethal.1–5 The World Health Organization has predicted that 50 kg of aerosolized Y. pestis released over a city of five million people would cause pneumonic plague in as many as 150,000 persons, of whom 36,000 would die.2,3 If persons exposed to the attack then fled, the outbreak could spread far beyond the initial site of dissemination. Pointedly, a government training exercise to assess reactions to an aerosolized Y. pestis attack found that disease would spread rapidly, with hospitals quickly overwhelmed and unable to care for sick patients. 4 A bioterrorist attack with aerosolized Y. pestis may occur when there is a limited supply of first-line antibiotics in the Strategic National Stockpile (SNS), as well as logistical concerns for providing these antibiotics in a timely manner to the vicinity of a Y. pestis attack. Owing to the virulence of Y. pestis, if treatment is delayed for more than 24 h after the onset of symptoms, the mortality rate of pneumonic plague approaches 100%.3,5 Equally important, strains of Y. pestis have been isolated in the natural environment that are resistant to multiple antibiotics. 6 Taken together, with the concern that multi-drug resistant strains of Y. pestis can be developed under laboratory conditions and that vaccines are not effective as post-exposure medical countermeasures (MCMs),.a critical need exists for novel therapeutic strategies to expand the window of opportunity for treating patients in the event of mass casualties following an aerosolized attack with Y. pestis.
An important virulence factor for Y. pestis is LPS (endotoxin), a glycolipid moiety and primary component of its bacterial outer wall that contributes to Y.pestis-induced acute lung injury (ALI) and septicemia in animals.7,8 In the lungs, LPS causes structural changes that result in disrupted alveolar septa, interstitial and alveolar edema, inflammatory cell infiltration, microvascular congestion and frank alveolar hemorrhage. 9 The pathology of ALI caused by an intravascular or intratracheal challenge with LPS resembles that with Y. pestis infection in animals and humans.10–12
It was previously reported that LPS binds to and activates A1 adenosine receptors on human pulmonary artery endothelial cells (PAECs) to induce the release of thromboxane and IL-6, immunomodulatory substances that are cytotoxic to endothelial cells.
13
In animals, antagonists of the A1 adenosine receptor (A
Materials and methods
Reagents and supplies
The Y. pestis strain CO99 pCD1
Bacterial growth and quantitation
For each experiment, CO99 from 80% glycerol frozen stocks was inoculated onto trypticase soy agar plates (TSA: 1% HIB, 1.5% agar, 0.2% D-galactose) and was grown for 72 h at 25 °C.17–19 Log-phase cultures were started by inoculating 8 ml of HIB with 5–10 white colonies of plated CO99 followed by shaking for 8 h at 25 °C. Then, 1.0 ml of the log-phase culture (OD600 0.2–0.4) was sub-cultured in 100 ml fresh HIB with overnight shaking at 25 °C.18,19 The CO99 inocula for infection were made by centrifuging these overnight stationary-phase cultures (OD600 > 1.0) for 10 min at 8000 g and then twice washing the bacterial pellet with 1 × PBS at 25 °C and centrifuging for 10 min at 8000 g.
Survival rates in rats after intratracheal infection with graded inocula of Y. pestis (strain CO99) at t = 0 h and left untreated through 6 d

CFU, colony forming unit.
Rat model of pneumonic plague
All procedures for surgery, infection, monitoring and euthanizing animals were performed according to relevant Health and Human Services (HHS) and National Institutes of Health (NIH) guidelines and only after prior approval by the Saint Louis Univer-sity Institutional Animal Care and Use Committee (IACUC). Male Sprague-Dawley rats (Harlan SPF, 250–350 g, Indianapolis, IN, USA) were housed in pairs upon arrival in positive-pressure isolation carrels within the animal care facility. After each of the two surgical preparations and intratracheal infection (i.t.) as described below, each rat had ad libitum access to food and water and was individually housed in a micro-isolator cage within a certified biosafety laboratory (BSL)-2+ laminar flow hood to prevent any cross-contamination among animals or contact between any animal and the laboratory environment.
Vascular catheterizations of the left carotid artery and right jugular vein were performed aseptically via a single 2 cm mid-ventral neck incision under 2% isoflurane/98% O2 anesthesia as described previously.18–22 Sterile PE-50 catheters filled with 10 U/ml heparinized NS were tunneled subdermally to exit at a single site on the dorsal neck to permit arterial blood draws and hemodynamics, as well as
The following morning, and 18–24 h after vascular catheterization, each rat was re-anesthetized as des-cribed above (2% isoflurane/98% O2) and positioned supine in a sterile surgical field. A 1.0 ml baseline carotid blood sample was obtained for the determination of arterial blood gases and quantitative culture, hematocrit and serum harvest. Each arterial sample was replaced with 3 ml of NS given by slow intra-arterial infusion. Only rats subsequently found to have normal blood gases and sterile baseline blood cultures before i.t. were included in the final data sets. Then, a parallel mid-ventral neck incision was made aseptically to expose the trachea. Each rat was infected by i.t. instillation of 1 × 108 CFU CO99 in 0.1 ml NS over 15–20 s during 5–6 normal inspirations, using a sterile 18 mm, 27-g needle directed caudally.18–20 To promote uniform dissemination of the inocula, one-third of each 0.1 ml inoculum was sequentially delivered with the rat in the supine, left decubitus, and right decubitus positions, while its body was inclined 20–30° cranially. Obser-vations for the next 1–2 min after needle withdrawal confirmed that inocula did not insufflate surrounding neck tissues. Uniform distribution of bacterial suspensions by this technique also was confirmed by instilling the trachea of other rats in preliminary studies with inocula containing Evans Blue dye (15 mg/ml) and inspecting their lungs 1 h later. The time of infection was designated as t = 0 and then the neck incision was closed, anesthesia was withdrawn and the animal roused within its cage as described above.
Post-infection monitoring and treatment intervention groups
Animals were assessed at t = 0, 1 h, 3 h, 6 h, 24 h, 48 h, 72 h, 96 h, 120 h and 144 h (6 d) or until death for survival, physical appearance, respiratory frequency (f), carotid arterial pulse rate (HR) and arterial blood pressure (AP), accompanied by 1.0 ml arterial blood draws for hematologic and microbiologic assays, again replaced with 3 ml of NS.18–22 Respiratory f (breaths/min) was assessed by counting chest movements for 1 min. Continuous or intermittent chart recordings of AP (mmHg) and HR (beats/min) were obtained with an MKIIIS Physiograph (International Biomedical, Houston, TX, USA) and small-displacement pressure transducers with independent analyses by two investigators. Mean arterial pressure (MAP) was calculated at key time points cited above as the sum of diastolic pressure plus one-third of the difference between systolic and diastolic pressures.18,22
Hematocrit (%) was determined on a 30-µl aliquot of each arterial blood sample to confirm that sequential bleedings did not reduce this value by more than 15% from t = 1 h.18,22 Arterial blood gases (ABGs) were measured on 100-µl aliquots using an IL-1600 Blood Gas Analyzer (Instrumentation Laboratories, Chicago, IL, USA) calibrated daily to ambient atmospheric pressures with a 1-m mercury barometer. Measured values for pHa, PaCO2 (mm-Hg), and PaO2 (mmHg) were used to calculate arterial HCO3
Infected rats were randomly assigned to one of the following treatment groups: no interventions (infection controls); cipro (10 mg/kg, b.i.d. × 3 d, IV) plus a continuous infusion of pharmacy grade sterile water (H2O) (0.5 ml/h × 8 h/d × 3 d); a continuous infusion of sterile H2O only (0.5 ml/h × 8h/d × 3 d); or cipro (10 mg/kg, b.i.d. × 3 d, IV) plus L-97-1 in sterile H2O (1, 5, 10, or 20 mg/kg/h) as a continuous
Post-mortem evaluations
In the 6 d survival studies, animals that survived 6 d (144 h) were necropsied; rats that died overnight were recorded as having survived until the previous observation period but were not necropsied. Also, in a 72 h time-killed study, infected rats that received no interventions were necropsied at 72 h. Rats were euthanized by isoflurane overdose (5%) and then underwent aseptic laparotomy and puncture of the diaphragm below the xiphoid process to allow an observed pneumothorax, followed immediately thereafter by dorsal aortic exsanguination.18,22
Right lung lobes were then aseptically excised, rinsed with sterile NS, and blotted dry on sterile gauze. Lung microvascular edema was estimated as tissue wet-to-dry ratios (W/D) by weighing a standard section of the right caudal lobe before and after drying at 60 °C to a constant weight for 3 d.18,22 Lung quantitative cultures were performed using freshly harvested right upper lung lobes homogenized in ice cold PBS with glass tissue grinders (10 mg fresh tissue/ml PBS, pH 7.4), serially diluted in PBS, and 50–100 μl plated as previously described.18–22 Lung bacterial burdens were calculated as CFU/g dry tissue. Likewise, standard segments of livers, spleens and kidneys were aseptically excised from all 6-d survivors and 72 h time-necropsied control infected animals. Using the same procedures described above for lung tissue, W/D ratios and quantitative cultures were determined for these organs.
Determination of ALI scores with histopathology
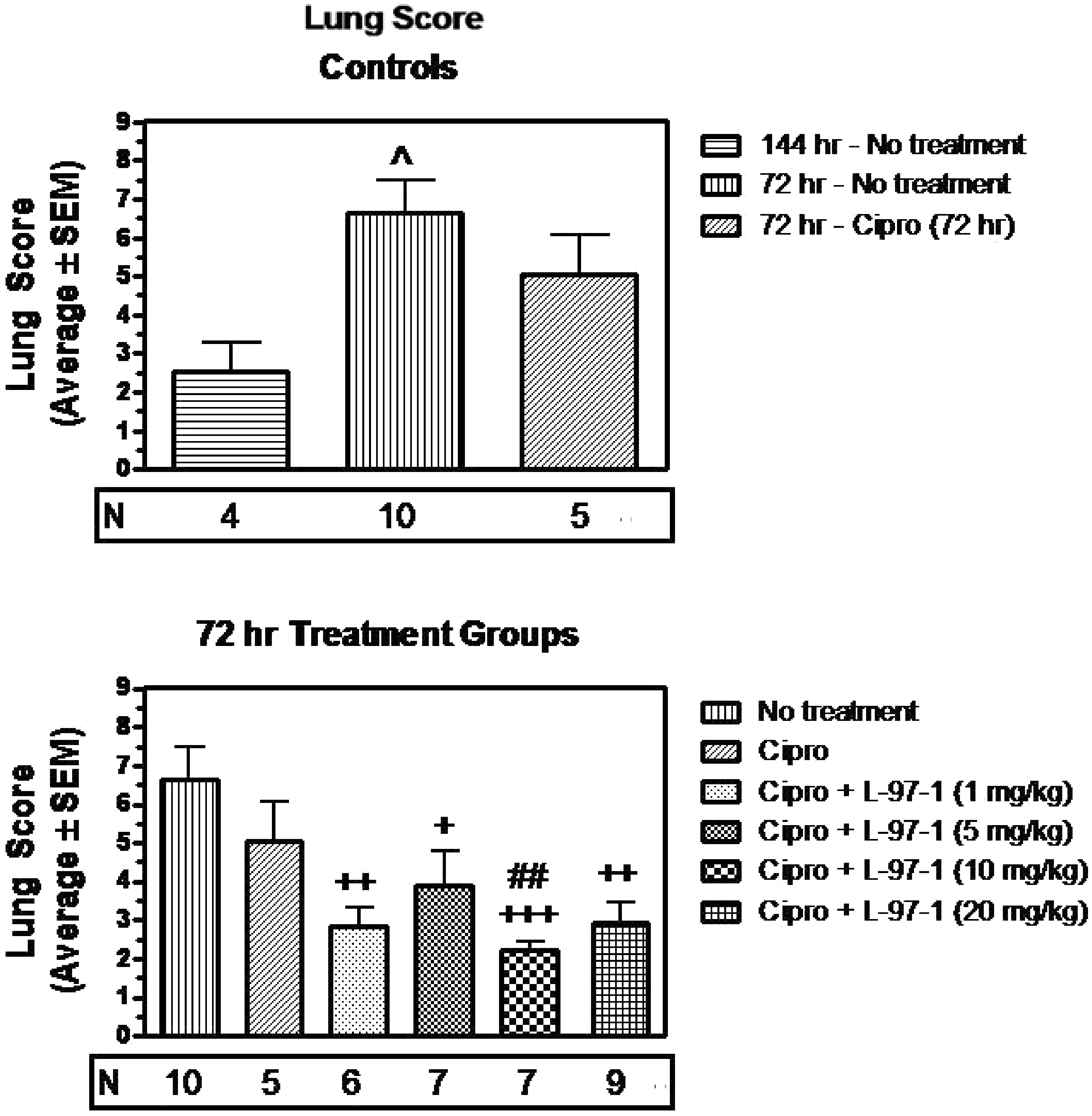
Entire left lung lobes were fixed in situ with phosphate-buffered 10% formalin at a transpulmonary inflation pressure of 20–22 cm H2O for 30 min.21,22 Then 2–3 mm thick midsagittal slices of each lobe were immersed in buffered formalin overnight at 5°C. Paraffin-embedded serial sagittal sections (6 µm) were stained with hematoxylin and eosin (H & E) for routine evaluation of histopathology. Using double-blind scoring methods, each H & E-stained section was evaluated at 40X, 100X and 400X using a scale of 0 (normal), 1 (mild), 2 (moderate), or 3 (severe) to grade parenchymal injuries.18,21 Acute lung injuries scored were: (a) interstitial and/or alveolar edema; (b) interstitial and/or alveolar hemorrhage; and, (c) interstitial and alveolar leukocytic infiltration indices (LII). For the first two injury types, the entire sagittal section was scanned at these multiple magnifications to assign a global severity index to each lung. In practice, at least ten random high-powered fields (HPFs) were assessed per sagittal section, starting at very low magnification (40X) and then magnifying these fields up to 400X, as needed, to denote a specific injury type, such as interstitial or alveolar edema or hemorrhage. For LII, a total of eleven HPFs for each lung section at 400X were independently selected by microscope stage coordinates and the median LII score of these eleven HPFs was reported. Total aggregate lung injury scores expressed as the sum of the individual lung injury scores (combined interstitial and alveolar scores) for edema, hemorrhage and LII were determined. Regions showing artifactual damage, such as pleural crushing, under-inflation, or compression atelectasis were excluded from analysis. Infected rats given no treatment were necropsied at 72 h (n = 10) and their lungs scored as described above. These animals served as treatment baseline controls for rats in those groups for which treatment began at 72 h post-i.t. infection and that were then necropsied at t = 144 h.
Determination of plasma [L-97-1]
To establish the kinetics of L-97-1 clearance from plasma in normal animals on this
Data presentation and statistical analysis
Values shown are group means ± standard errors (SEM). Survival curves were evaluated using Kaplan-Meier survival analysis and Mantel-Cox logrank tests for group comparisons in GraphPad Prism v. 5.0 (GraphPad Software Inc., San Diego, CA, USA). Repeatedly measured parameters [MAP, HR, (A-a)-PO2, etc.] were evaluated in SAS version 9.2 using mixed linear model analysis for differences within and among treatment groups over time. Cross-sectional tissue parameters including W/D weight ratios were transformed by ranks before being analyzed using ANOVA. Lung injury scores determined by the criteria described above were analyzed using Student’s t test for unpaired data. The level of significance was set at P < 0.05. To ensure the adequacy of sample sizes needed to achieve statistical significance among groups, group sizes of n = 10 rats each provided the power to detect a 20% difference in the proposed criteria at the P < 0.05 level when analyzed using the mixed linear model method.
Results
L-97-1 plus cipro started at 72 h post-infection improves 6-d survival vs cipro plus H2O
In this rat model of pneumonic plague, only 33% (5 out of 15) animals survived the 6-d protocol when left untreated (‘No Intervention’, Figure 1, Table 2). Among rats that received 3 d of Six-day survival curves in catheterized rats following intratracheal (i.t.) infection with Y. pestis (strain CO99, 1×108 CFU/animal) at t = 0 h. Study groups shown here include those described in the ‘Materials and methods’ that received treatments starting at t = 72 h consisting of: no interventions; ciprofloxacin (cipro; 10 mg/kg, b.i.d. × 3 d, IV) plus sterile water (H2O, 0.5 ml/h × 8 h/d × 3 d, IV); H2O (0.5 ml, b.i.d. × 3 d, IV) plus L-97-1 (10 mg/kg/h × 8 h/d × 3 d, IV); or cipro (10 mg/kg, b.i.d. × 3 d, IV) plus L-97-1 (1, 5, 10, or 20 mg/kg/h × 8 h/d × 3 d, IV). Effect of treatment with cipro and without L-97-1 at different time points following intratracheal (i.t.) infection with Y.pestis CO99pgm- 1 × 108 CFU on survival *Actual survival times of the two rats on either side of the median point in a treatment subgroup containing an even number of animals.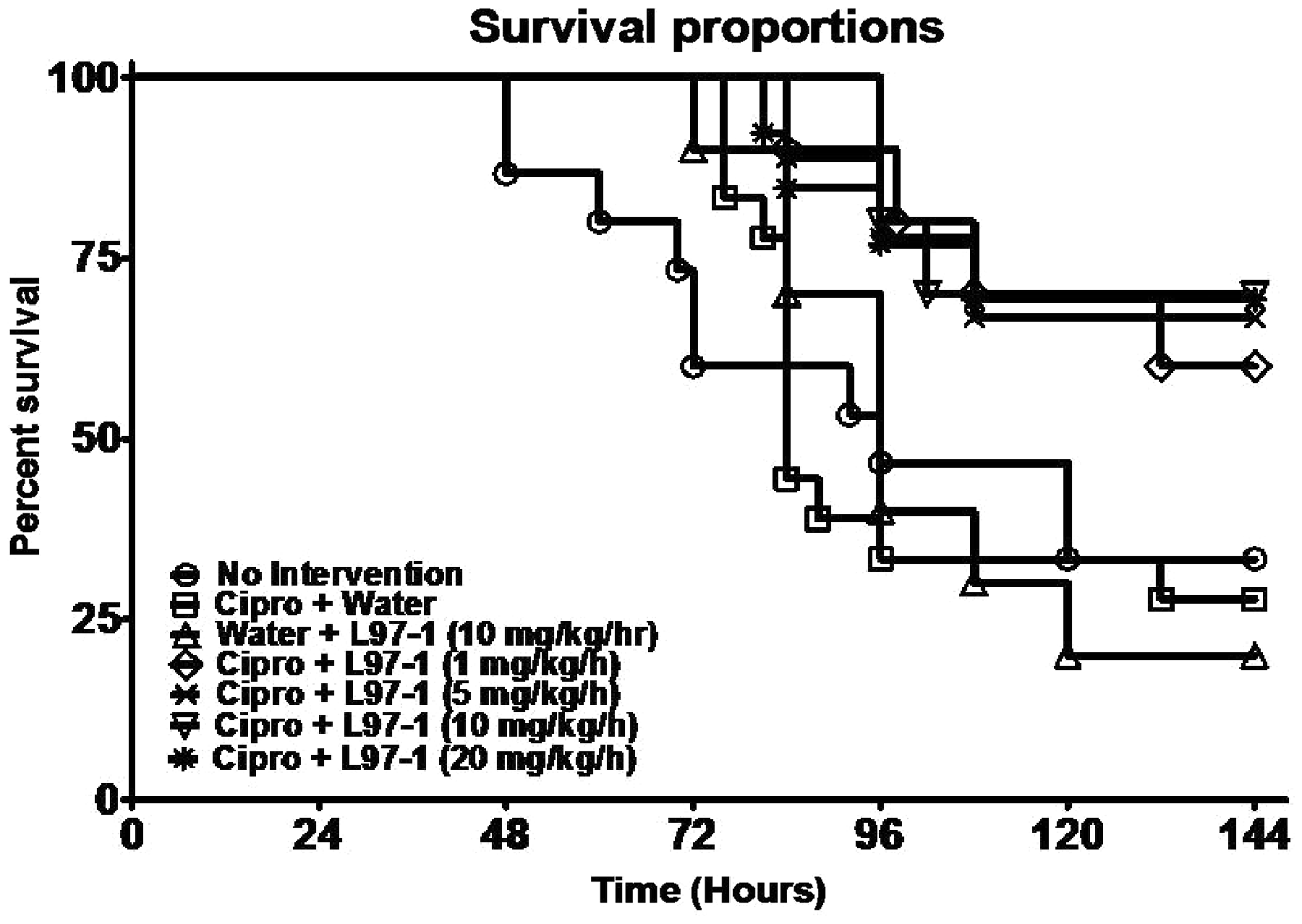
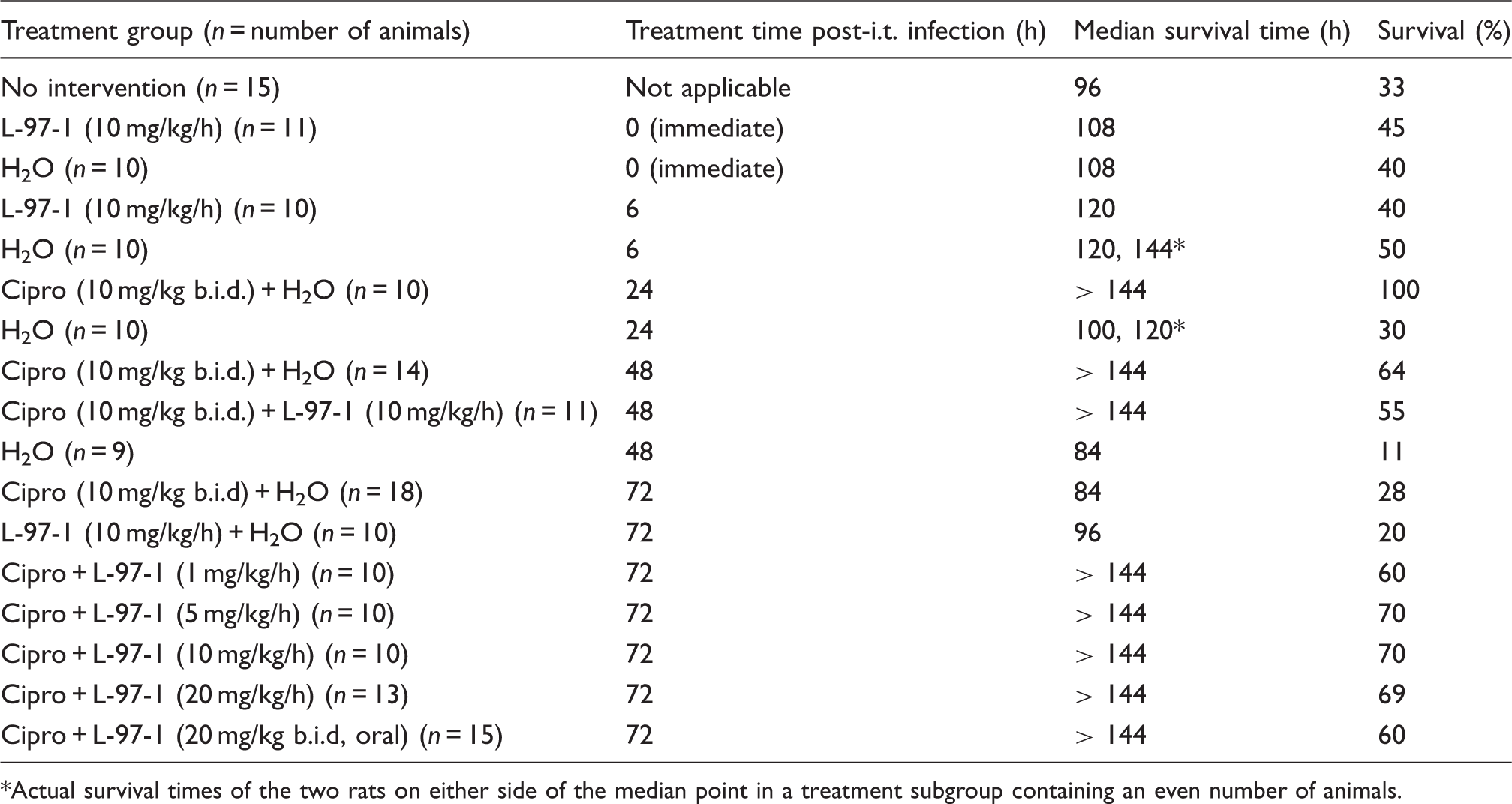
Delaying the start of treatments until t = 72 h after infection, cipro plus H2O resulted in a 6-d survival rate of 28% (5 out of 18), not different from either the 20% (2 out of 10) survival rate for L-97-1 alone (10 mg/kg/h × 8 h/d) or the ‘No Intervention’ group cited above (Table 2). However, 3 d of treating with cipro plus L-97-1 beginning at t = 72 h dramatically and significantly increased 6-d survival (60–70%) vs cipro plus H2O (28%) (P = 0.02, logrank test) (Table 2, Figure 1). Among these 72-h delayed treatment groups, median survival times for cipro plus L-97-1 at 1, 5, 10, or 20 mg/kg/h all increased to > 144 h vs the median survival time of 84 h for cipro plus H2O, and 96 h after no intervention.
Given these striking results for 6-d survival among infected animals treated with cipro plus L-97-1 beginning at t = 72h, additional infected animals received either cipro plus H2O or cipro plus 10 mg/kg/h L-97-1 starting at t = 72 h for 3 d as above, before their catheters were sealed on day 6 and the animals monitored for an additional 22 d. Among these additional groups, we found the 28-d survival rates for cipro plus L-97-1 to be 78% (7 out of 9) vs 38% (3 out of 8) for cipro plus H2O (P = 0.12, logrank test). In addition to these
When only L-97-1 was administered (10 mg/kg/h × 8h/d × 3 d) starting immediately after infection at t = 0, the 6-d survival of 45% (5 out of 11) resembled the 40% (4 out of 10) for H2O alone (0.5 mL/h × 8h/d × 3 d) (Table 2). Likewise, beginning the same treatments at t = 6 h resulted in a 6-d survival of 40% (4/10) for L-97-1 alone vs 50% (5 out of 10) for H2O alone.
L-97-1 plus cipro started at t = 72 h post-intratracheal (i.t.) infection with Y. pestis improves lung injury scores vs cipro plus H2O in 6-d survivors
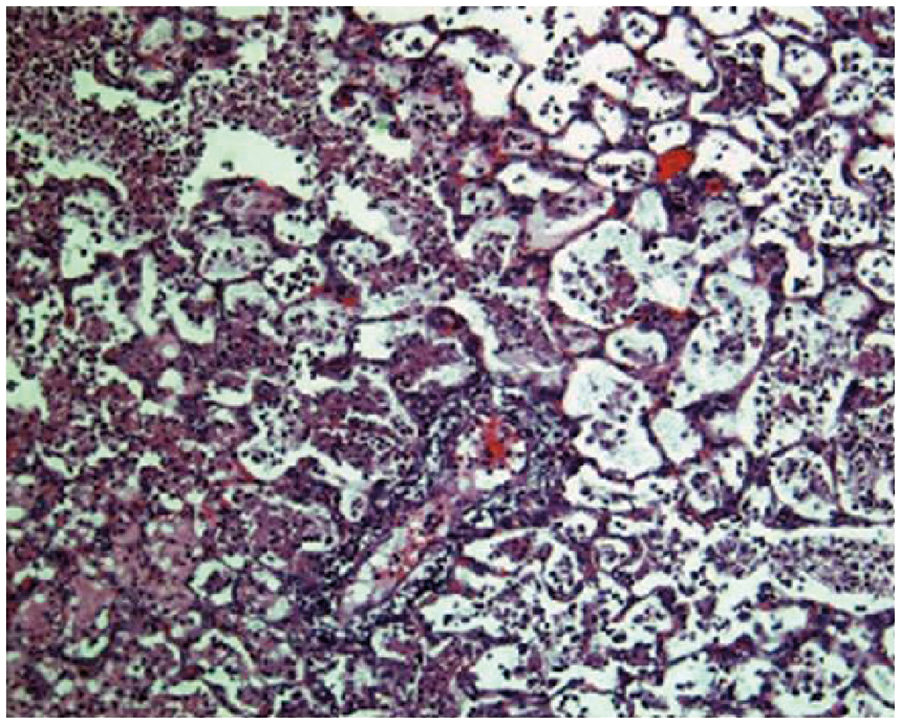
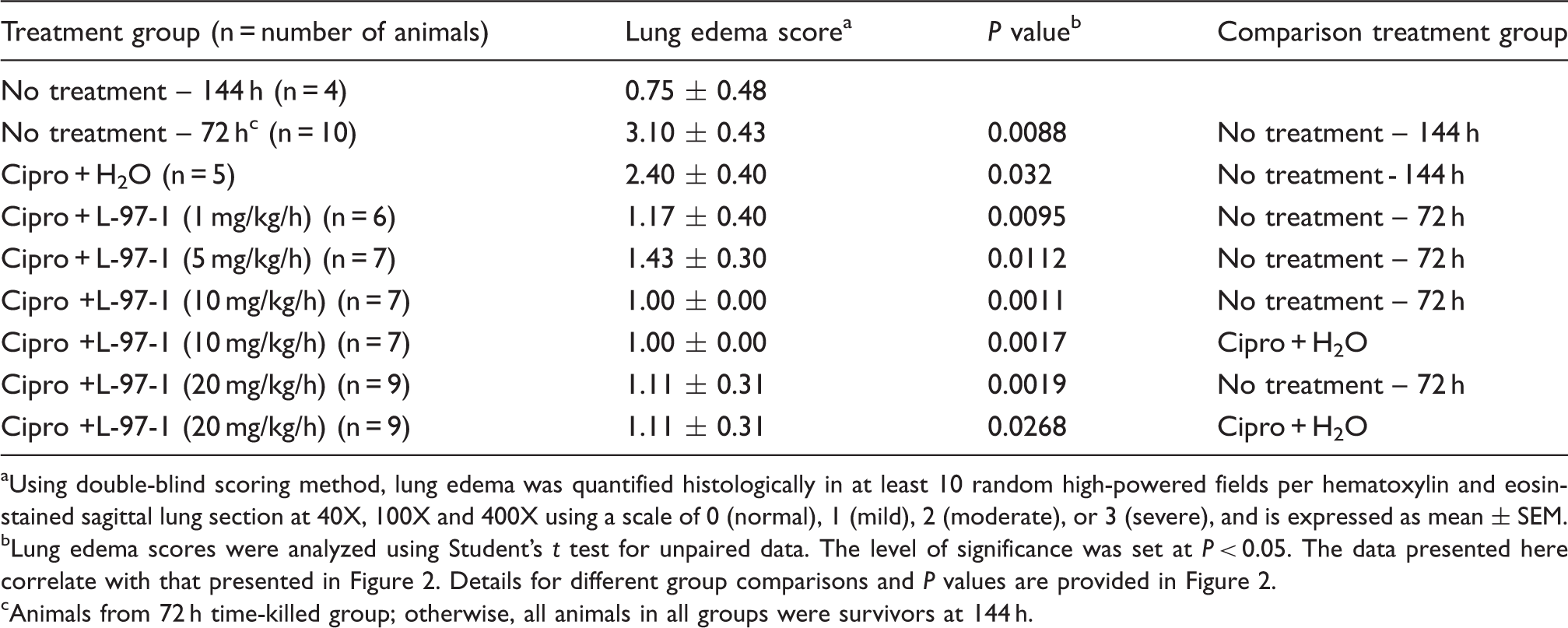
Within 72 h of infection with 1×108 CO99 and without interventions, histologically verified lung edema (Table 3, Figure 2), LII (Table 4, Figure 3) and aggregate lung injury scores (Table 5, Figure 4) were all significantly increased vs infected animals in the no intervention group that survived to 6 d (P < 0.05). In such infected but untreated animals studied at t = 72 h, alveoli were filled with mixed leukocytic infiltrates of neutrophils, lymphocytes and macrophages, as well as occasional hemorrhagic foci and fibrinous extracellular exudate (Figure 5).
Lung edema scored as described in the ‘Materials and methods’ for untreated animals killed by protocol 72 h following Y.pestis infection (strain CO99, 1×108 CFU/animal), as well as the 6-d survivors in Figure 1, in which no intervention occurred or treatments started at t = 72 h (see Figure 1 for additional details about treatments, doses, and routes of administration). Edema was scored to reflect fluid visible in either the interstitial or alveolar compartments, on a scale of none or normal (0), mild (1), moderate (2), or severe (3). ^ (P < 0.05), ^^ (P < 0.01) compared to Y. pestis, no treatment (144 h); + (P< 0.05), + + (P < 0.01) compared to Y. pestis, no treatment (72 hr); # (P <0.05), # # # (P < 0.001) compared to Y. pestis, ciprofloxacin (cipro; 72 h). Group-to-group comparisons were made using Student’s t test for unpaired data. Lung LII as described in the ‘Materials and methods’ for untreated animals killed by protocol 72 h following Y. pestis infection (strain CO99, 1×108 CFU/animal), and for the groups of 6-d survivors in Figure 1, in which no intervention occurred or treatments started at t = 72 h (see Figure 1 for additional details on treatments, doses, and routes of administration). The LII was scored as normal (0), mild (1), moderate (2), or severe (3), and reflected both the extent of alveolar and interstitial leukocyte involvement and the degree to which resident alveolar macrophages were displaced or outnumbered by neutrophils and lymphocytes not visible in the alveolar airspaces or lung interstitium of uninfected surgical controls. ^ (P < 0.05) compared to Y. pestis, no treatment (144 h); + (P< 0.05), + + + (P < 0.001) compared to Y. pestis, no treatment (72 h). Group-to-group comparisons were made using Student’s t test for unpaired data. Aggregate or overall lung injury score (alveolar and interstitial edema, hemorrhage, and LII scores) as described in the ‘Materials and methods’ for untreated animals killed by protocol 72 h following Y. pestis infection (strain CO99, 1×108 CFU/animal), and for groups of 6-d survivors in Figure 1, in which either no intervention occurred or treatments started at t = 72 h (see Figure 1 for additional details about treatments, doses, and routes of administration). A maximal score of 9 would reflect a ranking of ‘severe’ for that lung specimen across all categories of injuries. ^ (P < 0.05) compared to Y. pestis, no treatment (144 h); + (P< 0.05), + + (P < 0.01), + + + (P < 0.001) compared to Y. pestis, no treatment (72 h); # # # (P < 0.001) compared to Y. pestis, cipro (72 h). Group-to-groupcomparisons were made using Student’s t test for unpaired data. Representative lung tissue (hematoxylin and eosin, 100 X) from an untreated animal necropsied by protocol at t = 72 h after infection with viable Y. pestis (strain CO99, 1×108 CFU/animal). It was chosen randomly from among 10 untreated rats evaluated at 72 h, whose group lung injury scores are shown in the center of each of the upper histograms in Figures 2–4. Note that most alveoli are filled with neutrophils, lymphocytes, alveolar macrophages and fibrinous exudate. Such lungs reflected the presumptive baseline for infected animals whose various treatments began at t = 72 h. Effect of L-97-1 with and without cipro on lung edema at 6 d when treatment began at 72 h post-intratracheal (i.t.) infection with Y. pestis CO99pgm aUsing double-blind scoring method, lung edema was quantified histologically in at least 10 random high-powered fields per hematoxylin and eosin-stained sagittal lung section at 40X, 100X and 400X using a scale of 0 (normal), 1 (mild), 2 (moderate), or 3 (severe), and is expressed as mean ± SEM. bLung edema scores were analyzed using Student’s t test for unpaired data. The level of significance was set at P < 0.05. The data presented here correlate with that presented in Figure 2. Details for different group comparisons and P values are provided in Figure 2. cAnimals from 72 h time-killed group; otherwise, all animals in all groups were survivors at 144 h. Effect of L-97-1 with and without cipro on lung leukocyte infiltration index (LII) at 6 d when treatment starts at 72 h post-intratracheal (i.t.) infection with Y. pestis CO99pgm aUsing double-blind scoring method, LII was quantified histologically in a total of 11 high powered fields for each hematoxylin and eosin-stained sagittal lung section independently selected by microscope stage coordinates at 400X using a scale of 0 (normal), 1 (mild), 2 (moderate) or 3 (severe), and is expressed as mean ± SEM. bLung LII scores were analyzed using Student’s t test for unpaired data. The level of significance was set at P < 0.05. The data presented here correlate with that presented in Figure 3. Details for different group comparisons and P values are provided Figure 3. cAnimals from 72 h time-killed group; otherwise, all animals in all groups were survivors at 144 h. Effect of L-97-1 with and without cipro on aggregate lung injury score at 6 d when treatment began at 72 h post-intratracheal(i.t.) infection with Y. pestis CO99pgm aUsing double-blind scoring method lung, (interstitial plus alveolar) edema, hemorrhage and LII were quantified histologically in at least 10 random high powered fields per hematoxylin and eosin-stained sagittal lung section using a scale of 0 (normal), 1 (mild), 2 (moderate), or 3 (severe). Total aggregate lung injury scores expressed as the sum of the individual injury scores for lung edema, hemorrhage and LII were determined and are expressed as mean ± SEM. bAggregate lung injury scores were analyzed using Student’s t test for unpaired data. The level of significance was set at P < 0.05. The data presented here correlate with that presented in Figure 4. Details for different group comparisons and P values are provided in Figure 4. cAnimals from 72 h time-killed group; otherwise, all animals in all groups were survivors at 144 h.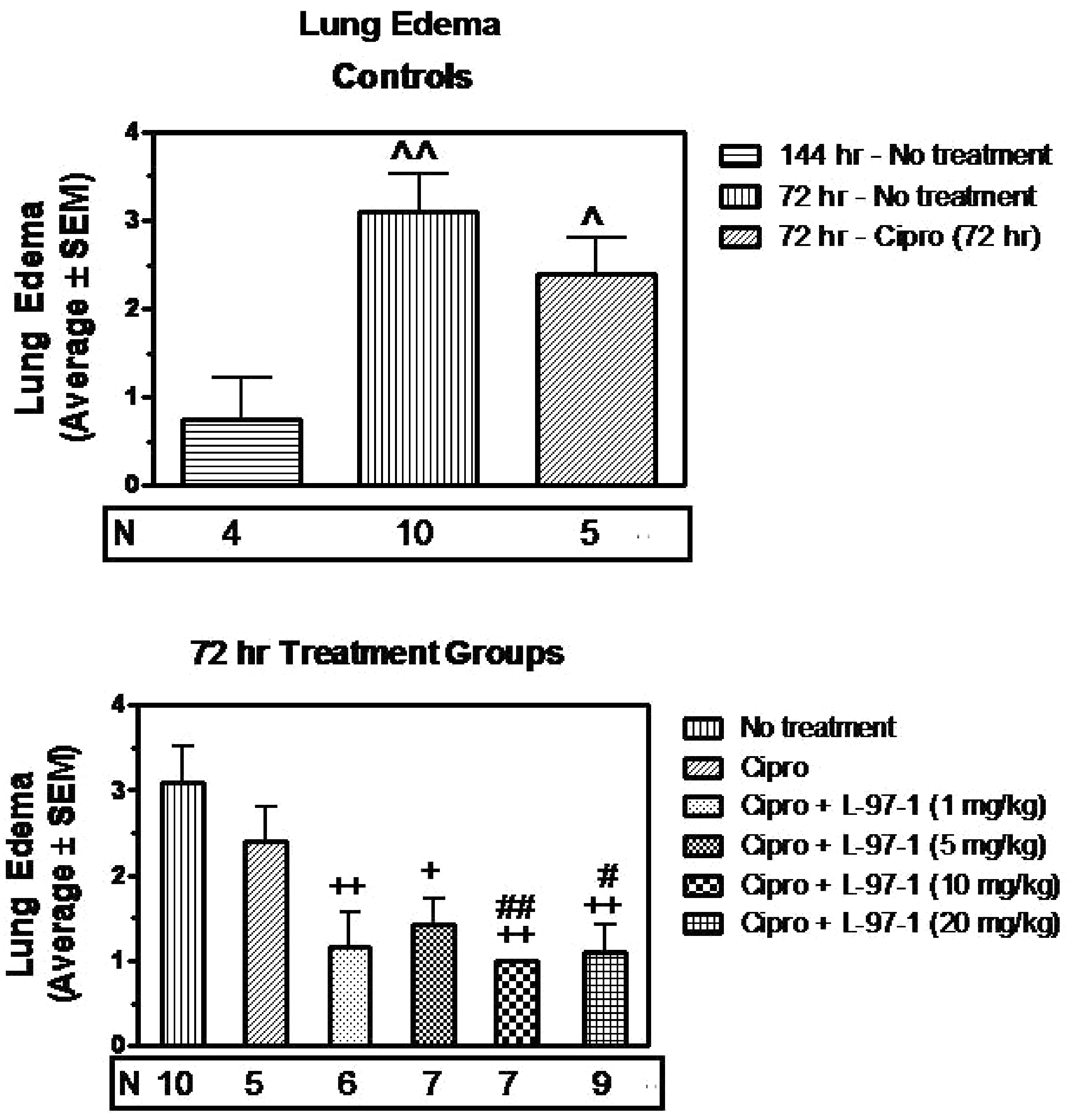
Effect of L-97-1 with and without cipro on physiologic measurements at 6 d when treatment began at 72 h post-intratracheal (i.t.) infection with Y. pestis CO99pgm
aMeasurements are from animals in 72 h time-killed group; otherwise, measurements are from survivors at 144 h.
Lung W/D weight ratios, (A-a)PO2, and MAPs were analyzed using mixed linear model analysis for differences within and among treatment groups over time.
*Indicates level of significance at P < 0.05 vs. no intervention group at t = 72 h.
W/D, wet-to-dry; MAP, mean arterial pressure.
These reduced lung injury scores among infected animals treated with cipro plus L-97-1 vs cipro plus H2O or untreated infection controls are readily apparent microscopically. In representative lung tissue from an infected rat that survived to t = 6 d after treatment for 3 d with cipro plus H2O starting at t = 72 h, the severity of alveolar and peribronchiolar edema and the amount of leukocyte-rich infiltrates (Figure 6) are hardly attenuated vs those noted for the untreated infection control (Figure 5). By contrast, in a representative lung field from an infected animal treated for 3 d with cipro plus L-97-1(10 mg/kg/h × 8h/d) starting at t = 72 h, there is only slight peribronchiolar edema, while the overwhelming majority of alveolar airspaces show no infiltrating leukocytes, extravasated erythrocytes, or fibrinous exudate (Figure 7).
Representative lung tissue (hematoxylin and eosin, 100 X) from a Y. pestis-infected animal that survived to t = 6 d, after being treated for 3 d with cipro 10 mg/kg, b.i.d., IV) plus sterile H2O (0.5 ml/h × 8 h, IV) starting at t = 72 h. The image was chosen randomly from those for only 5 such 6-d survivors among 18 rats in this treatment group (Table 2), and whose mean lung injury scores are shown as the ‘cipro’ group in Figures 2–4. Note the perivascular edema at the top and the extensive zone of leukocyte-rich infiltrates within alveolar airspaces, resembling lungs from an untreated rat at t = 72 h in Figure 5. Representative lung tissue (hematoxylin and eosin, 100 X) from a Y. pestis-infected animal that survived to t = 6 d, after being treated for 3 d with cipro 10 mg/kg, b.i.d., IV) plus L-97-1(10 mg/kg/h × 8 h/d, IV) starting at t = 72 h. The image was selected randomly from the 7 such 6-d survivors among 10 rats in this treatment group (Table 2), and whose mean lung injury scores are shown as the ‘cipro + L-97-1 (10 mg/kg)’ group in Figures 2–4. Note the clear air-filled alveolar airspaces and interstitium that were scored as ‘normal’ and considered indistinguishable from the lung tissue of uninfected surgical control animals, except for the slight cell-free peribronchiolar edema at left that was scored ‘mild’.
Physiologic measurements: lung wet/dry (W/D) weight ratios, (alveolar– arterial) partial pressure gradients for oxygen [(A – a) PO2]s, and mean arterial pressure (MAP)
Lung W/D weight ratios serve as an index of pulmonary microvascular permeability and were calculated for the 10 infected but untreated rats killed by protocol at t = 72 h, as well as for all untreated and treated 6-d survivors for the 72 h treated groups shown in Table 2 (Table 6). The lung W/D weight ratios among animals in the 72 h time killed group that received no treatment, 72 h no intervention group, was 4.77 ± 0.27 (mean ± SEM): a value very similar to those found in previous reports.20, 21 Among all survivors for the 72 h treated groups shown in Table 2, mean lung W/D ranged from 4.34 ± 0.58 to 4.97 ± 0.28; none of the treatment group means differed significantly from each other or from the value for the 72 h no intervention group (Table 6).
These indistinguishable lung W/D weight ratios were anticipated from similar alterations in f and in arterial blood gases measured on all animals while they breathed room air, starting at t = 1 h post-infection and continuing until necropsied at t = 72 h or 144 h. From a baseline f at t = 1 h across all infected animals of 83 ± 3 breaths/min, virtually every animal became tachypneic by t = 24 h and reached a peak rate of 109 ± 5 breaths/min by t = 72 h. Thereafter, f subsided slowly among survivors toward a mean of 98 ± 4 breaths/min at t = 144 h, with rates not significantly different by treatments. Arterial blood gases measured at these same time points were used to calculate the (A – a) PO2, which averaged 11 ± 3 mmHg on room air at t = 1 h across all groups. Among the survivors that subsequently received no intervention, the (A – a) PO2 increased to 16.9 ± 9.5 mmHg by t = 72 h and to 29.8 ± 23.1 mmHg by t = 144 h. Among all 6-d survivors that received cipro plus either L-97-1 or H2O starting at t = 72 h, their group mean (A – a) PO2 values did not significantly differ from each other, from the value for the 144 h no intervention group, or from the value for the 72 h no intervention group (Table 6).
L-97-1 plus cipro started at 72 h post-infection improves MAP vs cipro plus H2O in 6-d survivors
At t = 1 h, MAP and HR averaged 141 ± 5 mmHg and 462 ± 12 beats/min, respectively, among all 86 animals in the seven 72 h groups in Table 2 that had been infected 1 h earlier but were then resting quietly in their cages breathing room air. Over the next 71 h, MAP declined to 141 ± 8 mmHg and HR to 420 ± 11 beats/min among these animals, and were similar to values reported for the early stages of sepsis in other studies using conscious rats.18–21 Over the next 3 d, HR did not change appreciably, but MAP continued to decline in animals that received only cipro plus H2O or H2O treatments, falling to 91 ± 20 by t = 144 h in animals receiving no intervention (n = 5), and to 90 ± 18 in rats given cipro plus H2O (n = 5) (P < 0.05 for both vs the value for the no intervention group at 72 h) (Table 6). By contrast, MAP among all 6-d survivors that received 3 d treatment with cipro plus any dose of L-97-1 starting at t = 72 h, were not significantly different vs their respective means for MAP at t = 72 h or from the mean MAP of 134 ± 26 mm Hg for untreated, infected animals at 72 h, being: 119 ± 21 mmHg for cipro plus L-97-1 (1 mg/kg) (n = 6), 120 ± 21 mmHg for cipro plus L-97-1 (5 mg/kg) (n = 7); 112 ± 19 mmHg for cipro plus L-97-1 (10 mg/kg) (n = 5); and 120 ± 21 mmHg for cipro plus L-97-1 (20 mg/kg) (n = 4) (Table 6).
Bacterial culture data in the lung and blood and systemic dissemination of Y. pestis
Quantitative culture of lung homogenates yielded large numbers of CO99 from all 10 infected animals that received no intervention and were necropsied at 72 h post-infection, averaging 1.3 × 107 CFU/g dry lung tissue. For 5 of the 15 infected animals that survived to t = 144 h without intervention (Table 2), quantitative culture of lung homogenates yielded counts of CO99 averaging 9.2 × 104 CFU/g dry lung tissue. For the five 6-d survivors among 18 animals that received 3 d of cipro plus H2O starting at t = 72 h, lung culture counts for 4 rats averaged 2.8 × 103 CFU/g dry weight, and the lungs of 1 rat were negative for Y. pestis by culture. Combining 3 d of L-97-1 infusions with twice-daily cipro yielded equally impressive reductions in lung culture counts, as exemplified by the group treated for 3 d with L-97-1 at 20 mg/kg/h × 8 h/d. Among the nine 6-d survivors in that group, lung culture counts of CO99 for five rats averaged 3.0 × 103 CFU/g dry weight, while the lungs of the other four rats were negative for Y. pestis by streak-plate culture.
Arterial blood samples were occasionally positive for CO99 by quantitative streak-plating, but this occurred too infrequently to yield statistically useful data. However, standard segments of livers, spleens, and kidneys were aseptically excised from all 6-d survivors whose treatments are described in Figure 1 and Table 2 for the 72 h treatment groups, using the same procedures for quantitative culture described for lung tissue in the ‘Materials and methods’. Quantitative cultures of homogenates from livers, spleens and kidneys from all 10 infected animals that received no interventions and were necropsied at 72 h post-infection yielded large numbers of CO99 averaging 9.3 × 107, 2.1 × 108 and 1.7 × 104 CFU/g dry tissue, respectively, supporting significant dissemination to distant organs of Y. pestis infection from the lung following i.t. administration. None of these three tissues was found to be negative for CO99 by culture among animals given no intervention that survived to 6 d. However, 3 d of treatment with cipro plus H2O starting at t = 72 h consistently reduced Y. pestis counts in all three tissues, occasionally to zero. Combining L-97-1 infusions at any dose with cipro never increased the mean culture counts above those for cipro plus H2O. Indeed, at an L-97-1 dose of 20 mg/kg/h, only one of nine livers sampled at t = 144 h grew any CO99, and all nine spleens were negative for CO99 by streak-plate culture.
Plasma levels of L-97-1 following IV infusions into uninfected animals
Plasma L-97-1 levels (ng/ml) during a single continuous 8-h infusion (10 mg/kg/h, IV) in normal, uninfected rats
Data are presented as mean ± SEM, n = 4.
Peak and trough plasma levels (ng/ml) of L-97-1 during three consecutive daily 8-h infusions (10 mg/kg/h, IV) in normal, uninfected rats
Data are presented as mean ± SEM, n = 5.
Discussion
In this rat model of pneumonic plague induced by i.t. administration of Y. pestis CO99 (pCD1
Lipopolysaccharide is a glycolipid-rich moiety forming the major constituent of the outer wall of Gram-negative bacteria (GNB), and is a recognized virulence factor responsible for the waves of pro-inflammatory cytokines released within 48–72 h in animal models of Y. pestis-induced pneumonic plague.6–8,10,23–25 The systemic effects of circulating LPS, whether released from proliferating bacteria in distal organs like liver and spleen after hematogenous spread from the lungs, or by antibiotic-induced lysis of bacteria, may explain the ALI, multi-system organ failure and fatal shock clinically seen with GNB infections, including Y. pestis pneumonia.6,26,27
In the lungs, LPS causes disruption of the blood-air barrier, interstitial and alveolar edema, hemorrhage, and inflammatory cellular infiltration as noted for intrapulmonary Y. pestis infections in humans and animals.3,9–12,18,19,21,25,28–30 In animal models of pneumonic plague, histopathologic changes of ALI appeared within 48 h of inhalation and increased in severity by 72 h.25,29 The role of LPS in Y. pestis induced ALI and septic shock is supported by studies in rats made bacteremic by
It is possible that in the rat model of pneumonic plague described in these studies, and as is suggested by previous reports, LPS released from bacterial foci following blood-borne dissemination to multiple organs, contributes to the histopathologic changes of ALI evident at 72 h following inhalation of Y. pestis. In the current study, treatment with only cipro plus H2O that began at 24 h and 48 h following infection was effective and improved 6-d survival. However, at 72 h post-exposure to i.t. Y. pestis, cipro plus H2O was not effective and increased the mortality rate versus the untreated infected controls. When administered concurrently with cipro at 48 h post-exposure, L-97-1 did not improve survival versus cipro plus H2O. However, when administered with cipro starting 72 h post-exposure to Y. pestis, L-97-1 significantly increased 6-d survival. These findings suggest that the proliferation of intrapulmonary and disseminated bacteria within 72 h had reached sufficient mass to produce circulating levels of LPS that cause systemic effects. This is supported by the findings in a murine pneumonic plague model, where bacterial transcription profiles changed within the lung from day 2 to day 3 after infection with Y. pestis, confirming that expression of virulence factors, including LPS, increased with the duration of infection.31,32
The findings from this current study are consistent with reports by others demonstrating a peak in bacterial counts in the lung and distal organs, as well as peaks in plasma cytokine levels and severity of lung injury at 72 h post-infection with Y. pestis CO92 via an intrapulmonary (intranasal or aerosol) route.10,25 It has been suggested by others that this pro-inflammatory response observed at 72 h post-infection may be due, in part, to the effects of circulating LPS. 24 Equally important, the reduced survival rate seen in the 72 h cipro plus H2O treated group versus untreated, infected animals in the current study, may be caused by lysis of bacteria by cipro and the subsequent release and systemic effects of LPS. In this setting, as an adjunctive therapy to cipro, L-97-1 may be blocking the effects of LPS, as L-97-1 did not improve survival when administered alone, either immediately (t = 0 h), 6 h or 72 h post-i.t. Y. pestis exposure.
Adenosine receptors are G protein-coupled receptors, with A1, A2a, A2b and A3 subtypes defined to date by pharmacologic and molecular biologic approaches and identified in a number of different species, including humans.
33
The A1AR couples via a pertussis toxin-sensitive inhibitory G protein to multiple effector systems, including: adenylyl cyclase; phospholipases A2 and C, K
When considered together, several reports suggest that the activation of A1ARs in the lung may contribute to LPS-induced ALI. LPS from GNB binds to and activates A1ARs on human PAECs to induce the release of thromboxane and IL-6, substances that are cytotoxic to endothelial cells. 13 Activation of A1ARs on endothelial cells increases their permeability, while their activation on alveolar type 2 cells decreases lung fluid clearance, actions on both cell types that promote pulmonary edema.9,41–43 In one report, the high-affinity A1AR antagonist 1,3-dipropyl-8-cyclopentylxanthine (DPCPX) restored alveolar fluid clearance by 80%. 43 Thus, by blocking activation of A1ARs, an A1AR antagonist may provide positive inotropic support, maintain pulmonary and renal blood flows, and attenuate LPS-induced ALI.
Support that the A1AR is an important molecular target for LPS comes from both in vivo and in vitro studies. It was reported previously that selective A1AR antagonists similar to L-97-1 block LPS-induced ALI in animals. 9 In addition, efficacy for L-97-1 as an anti-sepsis treatment was shown in a rat model of CLP, an animal model of polymicrobial septicemia, endo-toxemia and ALI. 14 In this CLP model of septicemia, L-97-1 increased 7-d survival rates from 17% for untreated septic controls and 25% in animals given antibiotics alone, to 30–40% with L-97-1 alone, and to 50–70% with L-97-1 plus antibiotics.
Additional information suggesting that the A1AR may be an important molecular target for LPS comes from in vitro pharmacology assays for the A1AR, including radioligand competition binding and functional assays. In in vitro competition binding studies, LPS from US Pharmacopeia (Rockville, MD, USA) had an affinity (Ki) of 2.4 µg/ml for rat A1ARs stably expressed in Sf9 cells and labeled with [3H]-DPCPX (unpublished data, Constance N. Wilson). Previously, it was reported that in competition binding studies using membranes prepared from human PAECs, LPS from Escherichia coli, Salmonella typhimurium, Klebsiella pneumoniae, and Pseudomonas aeruginosa displaced the binding of another high affinity A1AR antagonist radioligand, [125I] BWA844U (IC50 = 195 ng/ml, 290 ng/ml, 602 ng/ml, and 693 ng/ml respectively). 13
In an in vitro pharmacological functional assay, e.g. a [35S] GTPγS GTP binding assay to measure A1AR agonist-mediated activation of G proteins, using membranes prepared from Chinese hamster ovary (CHO) cells stably expressing the rat A1AR, both LPS and N6-cyclopentyladenosine (CPA), a selective A1AR agonist, activated G proteins. This activation by both LPS and CPA was significantly blocked by the A1AR antagonist, DPCPX, as well as by pertussis toxin, an inhibitor of Gi/o protein (unpublished data, Constance N. Wilson). These results suggest that LPS functions as an A1AR agonist to activate A1ARs coupled to a Gi/o protein. These results considered with a previous report that LPS activation of A1ARs on human PAECs induces the release of cytotoxic substances, thromboxane and IL-6, suggest that LPS induces ALI by activation of A1ARs. Collectively, the results from the in vitro studies taken with those from the in vivo studies that A1AR antagonists block LPS-induced and Y. pestis-induced ALI and increase survival in two different animal models of sepsis, CLP-induced polymicrobial sepsis and Y. pestis-induced Gram-negative sepsis as reported in the current study and in previous reports, suggest that the A1AR is an important molecular target for LPS.
In the current study, plasma levels of L-97-1 for a single 8-h infusion of L-97-1 (10 mg/kg/h, IV) in normal, uninfected animals peaked at a concentration of 3.66 ± 1.06 µg/ml at the 4-h midpoint of the infusion. During the 3-d 8 h infusion, trough plasma levels of L-97-1 never declined below 0.38 µg/ml. These plasma levels correlate with L-97-1 concentrations of 6 μ
In addition to the improved survival and reduced ALI achieved by using L-97-1 as an adjunctive therapy to antibiotics in a rat CLP sepsis model and in this study of pneumonic plague, L-97-1 has shown efficacy in an animal model of asthma, where it blocked allergic airway responses, hyper-reactivity and inflammation.44,45 Others have reported that A1AR antagonists block ischemia-reperfusion injury of the heart, lung, liver, and pancreas. 33 It is worth noting, however, that as opposed to a pro-inflammatory effect of A1AR activation, other reports have suggested an anti-inflammatory, protective effect for the A1AR. 33 Based on the findings in one of these reports, the use of A1AR antagonists in sepsis is not supported. In that report, the survival rate of A1AR knockout (KO) mice with CLP-induced sepsis was lower than that for the wild-type mice with CLP. In the same report, wild-type mice with CLP-induced sepsis treated with the A1AR antagonist, DPCPX, had a lower survival rate than the CLP controls.
There are a number of possible explanations to clarify the differences between these studies in mice with CLP sepsis versus the current studies in rats. Firstly, genetically manipulated animals may have compensatory expression of other proteins that alter the phenotypes of cells and organs. These altered phenotypes may play a significant role in the organ injuries seen in these KO models. Secondly, although at low concentrations DPCPX has high affinity for the A1AR and serves as an A1AR antagonist, at higher concentrations, DPCPX may function as an inverse agonist for the A1AR. 46 In addition, at higher concentrations, DPCPX is not selective for the A1AR and may block the anti-inflammatory effects of the Gs coupled A2bAR. 33 A detailed discussion of the pro-inflammatory versus the anti-inflammatory effects of A1AR activation is provided in a recent publication. 33
In summary, the findings in the current study support efficacy for L-97-1 as a post-exposure MCM to cipro for the treatment of Y. pestis-induced sepsis. The mechanism of action for the beneficial effects of L-97-1 as an adjunctive therapy to cipro on survival, ALI and shock in the rat model of pneumonic plague in the current study is not known at this time. Based on reports that LPS is an important virulence factor for Y. pestis, that LPS binds to and activates A1ARs, and that A1AR antagonists block LPS-induced ALI and improve survival following CLP, it is possible that L-97-1 is blocking activation of A1ARs by LPS to produce ALI and septic shock in this animal model of Y. pestis-induced sepsis. The role of LPS in Y. pestis-induced ALI and septic shock in this rat model of pneumonic plague, as well as other possible mechanisms of action to explain the efficacy of L-97-1 as an adjunctive therapy to cipro in this animal model of sepsis, are areas for future investigations. Furthermore, it is important to note that the host response to a more virulent strain of Y. pestis, such as the parent wildtype CO92 strain from which the CO99 pCD1
Footnotes
Acknowledgements
The authors are indebted to Dr Susan C. Straley at the University of Kentucky School of Medicine for her gift of the CO99 pCD1
Conflict of interest
Constance N. Wilson is the Chief Scientific Officer of Endacea, Inc. (Research Triangle Park, NC, USA). Endacea is developing L-97-1 as an anti-sepsis therapeutic.
