Abstract
Intestinal macrophages (Mϕ) play significant roles in maintaining homeostasis by the efficient elimination of foreign particles in the large intestine. However, functional complement receptors have not been fully identified. In this study, we showed that a complement receptor of the Ig superfamily (CRIg, also known as Z39Ig), a receptor for complement fragments (C3b and iC3b), was expressed on a subset of intestinal Mϕ in murine and human large intestine. When abilities of uptake of antigens of murine CRIg+ Mϕ were examined, intestinal CRIg+ Mϕ displayed less endocytic and similar phagocytic abilities compared to resident peritoneal F4/80+CRIg− Mϕ and F4/80+CRIg+ Mϕ. Additionally, we found that a significant portion of C3b-dependent phagocytosis by large intestinal Mϕ involves CRIg, emphasizing the importance of efficient mechanisms to eliminate foreign particles in the large intestine. On the other hand, intestinal Mϕ from 2,4,6-trinitrobenzene sulfonic acid-treated mice had decreased CRIg expression but increased CD11b expression, implying some contribution to the removal of immune complexes. This study will shed new light on opsonization and phagocytosis by large intestinal Mϕ.
Introduction
The mucosa of the small and large intestines is the largest reservoir of tissue macrophages (Mϕ) in both humans and mice. 1 Although Mϕ possess various functions, the main task of intestinal Mϕ is thought to be the efficient elimination of foreign particles including microbes, apoptotic cells and foreign debris for maintenance of homeostasis while remaining tolerant to the harmful food antigens and the resident gut microflora.1,2 Because Mϕ subsets identified by cell surface markers are thought to display distinct functions dependent on their markers, 3 it is critical to characterize the surface phenotypes and functions of intestinal Mϕ to elucidate homeostatic mechanisms in normal and inflammatory disease conditions in the intestine. However, it has been reported that human intestinal Mϕ lack pattern recognition receptors such as CD14, TLR2 and TLR4, the Fc receptors for IgA and IgG and the complement receptors CR3 (CD11b/CD18) and CR4 (CD11c/CD18), 1 although the presence of these receptors has not been studied in detail on murine intestinal Mϕ.
A complement receptor of the Ig superfamily (CRIg, also described as Z39Ig) was recently identified as a novel receptor for complement fragments (C3b and iC3b) in murine and human tissues.4,5 Murine and human CRIg is also reported to function as a T cell negative regulator. 6 High expression of murine CRIg has been detected in several tissues, including the heart and liver, by northern blotting and immunohistochemistry using a polyclonal antibody.4,6 A FACS analysis has also shown that murine CRIg is highly expressed on Kupffer cells and a subpopulation of resident Mϕ in the peritoneum.4,5 Thus, it is presumed that murine and human CRIg+ cells are a unique subpopulation of resident tissue Mϕ and play significantroles in maintaining homeostasis by increasing complement-dependent phagocytosis and/or negative regulation of T cell activation. However, there have been no reports about the expression of CRIg in the murine and human intestine.
In this study, we found CRIg-expressing Mϕ in the murine and human large intestine. Furthermore, the phenotypes and phagocytic functions of these Mϕ were compared with those of Mϕ from the peritoneum and inflammatory colitis model.
Materials and methods
Human samples
Human specimens of normal large intestine were obtained from normal tissues of three patients with colon cancer who had their large intestine resected for pathological diagnosis. Informed consent was obtained from all donors in accordance with the requirements of the Human Investigation Committee of Kagoshima University.
Mice
Six- to eight-week-old female BALB/cN Sea mice werepurchased from KYUDO (Kumamoto, Japan) and maintained under specific pathogen-free conditions at the Frontier Science Research Centre, Kagoshima University. All animal experiments were done in accordance with the Ethical Guidelines for Animal Experiments, Kagoshima University and approved bythe Animal Ethics Committee for Animal Experimentation, Kagoshima University.
Antibodies
For human tissues, the biotinylated murine anti-human CRIg (Z39Ig) mAb (clone 15-b) 7 and the biotinylated murine anti-human CD68 mAb (Y1/82A) (BioLegend, San Diego, CA, USA) were used.
The following antibodies were used in mouse experiments: rat anti-mouse CRIg mAb (clone 8) was generated by immunizing rats as previously described. 8 If necessary, the anti-CRIg (clone 8) mAb was labeled with Alexa Fluor 647 according to the manufacturer’s instructions (Molecular Probes, Eugene, OR, USA). The blocking mAb against CRIg (clone 14G8) and isotype control mAb (anti-ragweed) 5 were kindly provided by Dr van Lookeren Campagne (Genentech). Fluorescein isothiocyanate-labeled anti-F4/80 mAb (F4/80-FITC) (BM8) and anti-F4/80-PE mAb (BM8) were purchased from BioLegend. Anti-CD11b (M1/70), anti-CD11b-PE (M1/70) and anti-CD11c-PE (HL3) mAbs were purchased from BD Biosciences (San Jose, CA, USA). Rabbit polyclonal antibody against CX3CR1 (H-70), rabbit anti-CCR2 mAb (E68) and rat anti-CD68 mAb (FA-11) were obtained from Santa Cruz Biotechnology (Santa Cruz, CA, USA), Abcam (Tokyo, Japan) and AbD Serotec (Oxford, UK), respectively. Donkey polyclonal antibody to rabbit IgG-H&L-F(ab)2 fragment (PE) were obtained from Abcam.
Induction of colitis
Colitis was induced in female BALB/c mice as previously described. 9 Briefly, mice were fasted for 1 d, anesthetized and then injected intrarectally with 2,4,6-trinitrobenzene sulfonic acid (TNBS) (Sigma-Aldrich, St. Louis, MO, USA) in 45% ethanol at a dose of 50 mg/kg. Control mice received 45% ethanol alone. Diarrhea, loss of body weight and survival were monitored daily. Four days after the injection of TNBS or ethanol, mice were sacrificed and the large intestine was obtained and used for immunohistochemical analysis and isolation of lamina propria mononuclear cells (LPMCs). Induction of colitis was confirmed by histological analysis in which inflammatory infiltrate, loss of goblet cells and bowel wall thickening were examined in sections of large intestine stained with hematoxylin-eosin as previously described. 10
Isolation of resident peritoneal cells and LPMCs
Resident peritoneal cells were lavaged with PBS and used for analysis.
For LPMC isolation, we first removed all visible attached tissue, such as fat and blood vessels that attach to, and surround, the surface of the large intestine to minimize the contamination of LPMCs with cells such as mesentery-associated cells 11 and peritoneal cells. After the removal of these tissues, the large intestine was cut into pieces and LPMCs were isolated as previously described. 12 In brief, the pieces of the large intestines were washed with cold calcium- and magnesium-free Hank’s balanced salt solution (HBSS). To remove the epithelium, the pieces were incubated in HBSS/EDTA for 30 min at 37°C with manual shaking. After washing, the pieces were digested with 200 Mandl units/ml of collagenase D (Roche Diagnostics, Mannheim, Germany) and 10 µg/ml of DNase I (Roche Diagnostics) in Iscove’s Modified Dulbecco’s Medium (IMDM) supplemented with 10% FBS for 1 h at 37°C with manual shaking. The cells were filtered through a 77 µm cell strainer and washed twice with calcium- and magnesium-free HBSS. Cells were then resuspended in 30% Percoll (GE Health Care, Uppsala, Sweden), layered on 70% Percoll and centrifuged for 20 min at 1100 g. The LPMCs were obtained from the interface fraction, washed twice with PBS and used for analysis.
Immunohistochemical staining
Six micrometer-thick serial sections from human large intestine were cut with a cryostat, mounted on silanized slides and fixed in acetone for 1 h. Endogenous peroxidase was blocked by 0.3% H2O2 for 10 min at room temperature (20°C). To block nonspecific binding, the sections were treated with Protein Block Serum-Free (Dako, Carpinteria, CA, USA) for 45 min at room temperature. The sections were then treated with primary mAbs or isotype-matched control mAb at 4°C overnight (20 h) and with horseradish peroxidase-conjugated streptavidin (ZYMED, South San Francisco, CA, USA) for 1 h at room temperature. Visualization was performed with NovaRED (Vector Laboratories, Burlingame, CA, USA).
To detect the co-localization of CRIg with CD68 in the murine large intestine, double immunostaining was performed as previously described.
13
Briefly, the first stain was performed with anti-CRIg and horseradish peroxidase-conjugated goat anti-rat IgG (Nichirei, Tokyo, Japan) followed by visualization with cerium-diaminobenzidine (DAB; Dako) as described above. Sections were then incubated with 0.1 M glycine-HCl (pH 2.7) for 1 min to remove antibodies. For the second staining, sections were washed with TBST (20 m
Flow cytometric analysis
After washing cells with PBS containing 2 m
Functional analysis
The ability to take up dextran was examined as previously described.
14
Briefly, 1 × 105 cells were incubated with FITC-dextran (MW 40000; Molecular Probes) at 1 mg/ml in IMDM with 10% FBS for 45 min at 37°C. After extensively washing cells with cold PBS containing 2 m
The ability to take up microspheres coated with BSA was assessed as previously described.
15
Briefly, 2 µm red fluorescent (580/605) FluoSpheres carboxylate-modified microspheres (Molecular Probes) were coated with BSA according to the manufacturer’s instructions. Microspheres were incubated with cells in IMDM media without FBS at a ratio of 75 microspheres/cell for 1 h at 37°C. After extensive washing of cells with cold PBS containing 2 m
Functional analysis of CRIg was performed as follows. Red fluorescent (580/605) FluoSpheres carboxylate-modified microspheres have been shown to be opsonized with C3b and have been used as targets of phagocytosis. 16 We prepared C3-opsonized microspheres in a similar manner. Mouse C3 was purified as previously described 17 and used for opsonization of FluoSpheres carboxylate-modified microspheres according to the manufacturer’s instructions. The production of C3b from C3 on microspheres was performed by incubating C3-opsonized microspheres with activated cobra venom factor for 3 h at 37°C as previously described. 17 These C3-opsonized or C3b-opsonized microspheres were used for a phagocytosis assay following the experimental procedure described previously. 5 Briefly, cells were incubated with 50 µg/ml of anti-CRIg (14G8) 5 or isotype-matched control Ab (anti-ragweed) 5 for 10 min on ice to block CRIg, followed by the addition of C3-opsonized or C3b-opsonized microspheres. After incubation for 1 h at 37°C, phagocytosis was stopped by placing the cells on ice, and the cell surface markers and extent of CRIg-mediated phagocytosis were monitored by FACS as described above. The substantial C3b-dependent uptake of microspheres was determined by comparing the amount of uptake of C3b-opsonized microspheres with that of C3-opsonized microspheres; the mean fluorescence intensity (MFI) of C3-opsonized microspheres was subtracted from that of C3b-opsonized microspheres.18,19
Statistical analysis
Statistical analyses were performed using the nonparametric Mann–Whitney U-test. A value of P < 0.05 was considered statistically significant.
Results
The presence of CRIg+ Mϕ in murine and human large intestine
In agreement with previous reports,4,5 our immunohistochemical and FACS analyses using anti-CRIg mAb detected murine CRIg+ cells in the heart, liver and peritoneum (data not shown). To further examine the distribution of murine CRIg+ cells, immunohistochemistry was performed on the small and large intestines. As shown in Figure 1(A), CRIg+ cells were not detected in the small intestine, although CD68+ cells were distributed throughout the tissue, whilst we found that CRIg+ cells were observed in the large intestine (Figure 1(B)). In addition, two-color immunostaining analysis showed that all CRIg+ cells were positive for CD68 and were distributed in the mucosal propria (Figure 1(B)). These findings support the notion that CRIg is a novel marker of intestinal Mϕ in the murine large intestine.
CRIg+ Mϕ are distributed in murine and human large intestine but not in murine small intestine. (A)Immunohistochemistry was performed on murine small intestine using anti-CD68 or anti-CRIg mAb, followed by the visualization with NovaRED. CD68+ Mϕ were detected but CRIg+ cells were not seen in the small intestine. Theoriginal magnification was x100. (B) Immunohistochemistry using anti-CD68 or anti-CRIg mAb on serial sections of murine large intestine showed the presence of CD68+ Mϕ and CRIg+ cells (upper panels). Double immunostaining indicated the co-localization ofCRIg with CD68 in murine large intestine (lower panel). Black arrow, CD68 single positive cells; white arrow, double positive cellsfor CD68 and CRIg. The original magnification was x 200 for single immunostaining and x 400 for double immunostaining. (C)Immunohistochemical staining of serial sections of human large intestine detected CD68+ Mϕ (black arrow head) and CRIg+ Mϕ(white arrow head). The original magnification was x 200.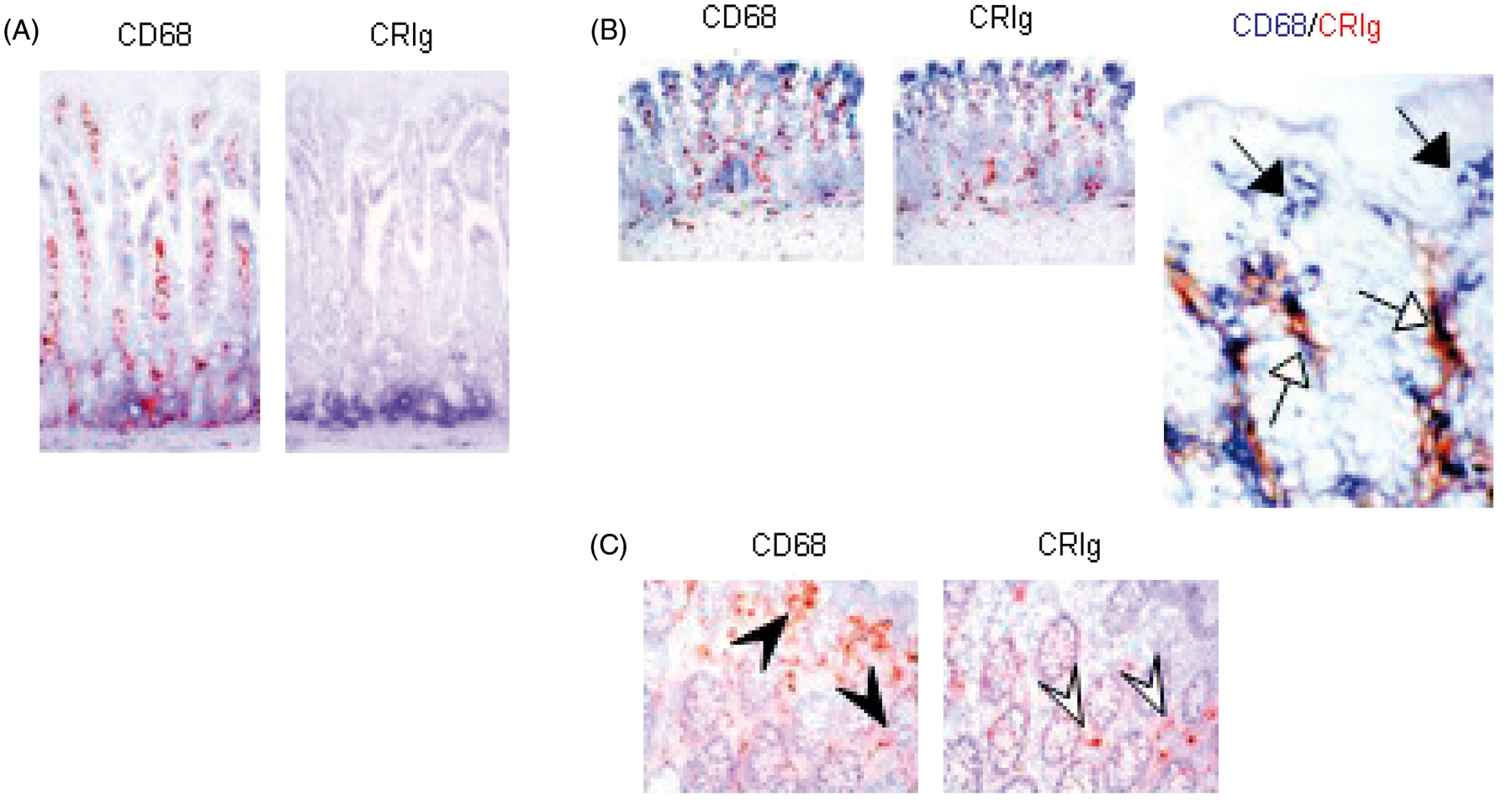
Similarly, in the human large intestine, CRIg+ Mϕ were mainly located in the mucosal propria. CD68+ Mϕ were distributed in the mucosal propria and particularly abundant in the surface portion (Figure 1(C)). These findings support the notion that CRIg is a novel marker of a subset of intestinal Mϕ in the human large intestine.
Different phenotypic profiles of murine CRIg+ Mϕ in normal large intestine compared with those in the peritoneum and large intestine with experimental inflammatory colitis
Phenotypic profiles of murine intestinal CRIg+ Mϕ were compared to those of resident peritoneal CRIg+ Mϕ.
As shown in Figure 2(A), the expression of complementreceptor was high for CD11b and negative for CD11c on both peritoneal F4/80+CRIg− Mϕ (39 ± 2%) and F4/80+CRIg+ Mϕ (25 ± 2%), in line with previous reports.
5
There was no cell surface expression of CX3CR1, a marker of resident monocytes, and CCR2, a marker of inflammatory monocytes, on either cell type.
Cell surface expression profiles are different among murine resident peritoneal CRIg+ Mϕ and intestinal CRIg+ Mϕ from the large intestine of untreated mice and TNBS-treated mice. (A) CRIg expression on resident peritoneal Mϕ. Left upper panel is a scatter plot of F4/80 + CRIg+ Mϕ. Histograms represent cell surface expression of the described antigen on CRIg− Mϕ and CRIg+ Mϕ. Shaded areas represent control Ab-staining, solid linesrepresent specific staining. Fluorescence-activated cell sorting analysis of resident peritoneal Mϕ showed that F4/80 + CRIg− Mϕ and F4/80 + CRIg+ Mϕ expressed similar levels of CD11b but were negative for CD11c, CX3CR1 and CCR2. Dataare representative of three independent experiments. Left lower graph is MFI of CRIg expression on F4/80 + CRIg+ Mϕ (R2) compared with F4/80 + CRIg− Mϕ (R1). Data are presented as the mean ± SEM. (B) CRIg expression onintestinal Mϕ. Left panel, a scatter plot of cells stained with control mAb; middle panel, a scatter plot of CRIg+ cells; right graph, MFI of CRIg+ cells (R2) compared with cells stained with control mAb (R1). Data were obtained by analyzing intestinal Mϕ isolated from a pool of four to eight large intestines in each experiment. Scatter plots are representative of eight independent experiments. Data are presented as the mean ± SEM. * P < 0.05 against R2 in (A). (C) FACS analysis of intestinal CRIg+ Mϕ showed that intestinal CRIg+ Mϕ were F4/80lowCD11b-CD11c-CX3CR1lowCCR2low. Shaded areas represent control Ab staining, solid lines represent specific staining. Data are representative of three independent experiments. (D) CRIg expression on intestinal Mϕ isolated from the large intestine of TNBS-treated mice. Left panel, scatter plot of cells stained with control mAb (R1);middle panel, scatter plot of CRIg+ cells (R2); right graph, MFI of R1 and R2 regions. Data were obtained by the analysis of intestinal Mϕ isolated from a pool of four to eight large intestines in each experiment. Scatter plots are representative of five independent experiments. Values are the mean ± SEM. **, P < 0.01 against R2 in (B). (E) Fluorescence-activated cell sorting analysis of intestinal CRIg+ Mϕ from TNBS-treated mice showed that CD11b was upregulated on the cell surface of intestinal CRIg+ Mϕ. Shaded areas represent control Ab staining, solid lines represent specific staining. Data are representative of three independent experiments.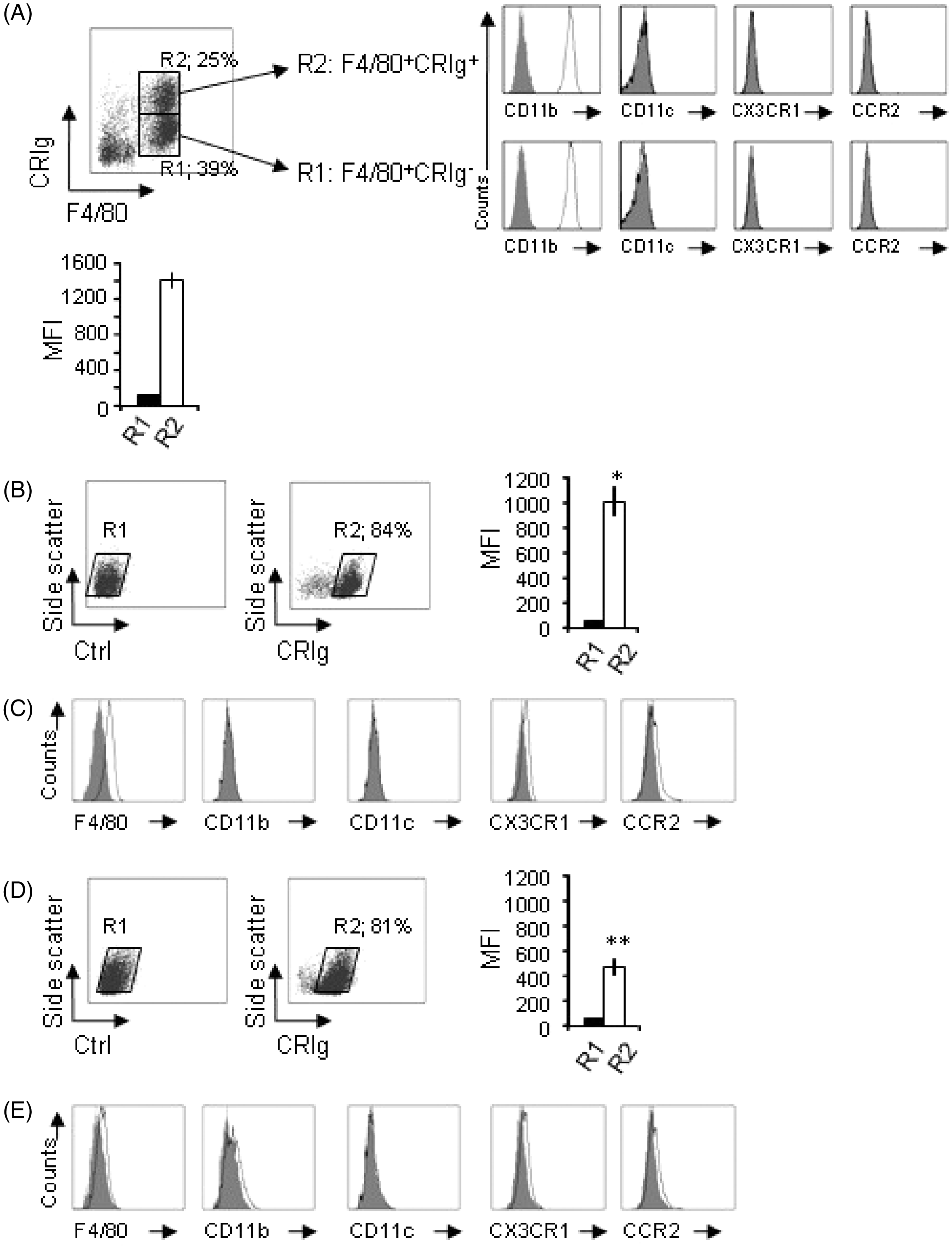
Flow cytometric analysis showed that 84 ± 11% of intestinal Mϕ expressed CRIg on their cell surface (Figure 2(B)). In addition, we found that the expression level of CRIg on the cell surface was significantly lower than that on resident peritoneal CRIg+ Mϕ. These intestinal CRIg+ Mϕ were F4/80lowCD11bnegCD11cnegCX3CR1lowCCR2low (Figure 2(C)), differing from the cells in the peritoneum which were F4/80highCD11bhighCD11cnegCX3CR1negCCR2neg.
To examine the role of CRIg+ Mϕ in inflammatory conditions, a Crohn’s disease model was produced by TNBS administration, and intestinal Mϕ were isolated from the inflamed large intestine as was done with the normal large intestine. Under our conditions, a decrease in survival rate (50%), a decrease in body weight (82 ± 2.6%) and a shortening of colon length (5.2 ± 0.46 cm) were observed in mice on day 4 after TNBS administration. Histological analysis also confirmed the induction of colitis as described in the Materials and Methods (data not shown). As shown in Figure 2(D), the ratio of CRIg+ Mϕ was 81 ± 7%, which was similar to that of the normal large intestine. However, we found that the expression level of CRIg on the cell surface was significantly reduced on intestinal Mϕ from TNBS-administered mice compared to normal mice. This reduction was a sharp contrast to previous reports in which CRIg was highly expressed, even in the presence of inflammatory stimuli.5,7,20 In addition, CD11b expression was seen on the surface of CRIg+ Mϕ, whereas the expression levels of other markers on the cell surface were similar to those of CRIg+ Mϕ from the normal large intestine (Figure 2(E)).
Phenotypes of CRIg+ Mϕ were also analyzed in the large intestine from an ulcerative colitis model produced by administration of drinking water containing 5% dextran sulfate sodium salt. No remarkable change was observed for CRIg+ Mϕ. For example, the greatly decreased expression of CRIg and the upregulation of CD11b, which were observed in CRIg+ Mϕ of a Crohn’s disease model, were not seen (data not shown).
Endocytic and phagocytic abilities of murine CRIg+ Mϕ from the peritoneum or normal large intestine
The uptake of antigens has been reported to occur via endocytosis for soluble antigens and phagocytosis for particulates. 19 Thus, the endocytic and phagocytic abilities of CRIg+ Mϕ were examined using dextran and microspheres as fluid-phase and particulate markers, respectively. 19
A similar efficiency of dextran uptake was observed in peritoneal F4/80+CRIg− Mϕ and F4/80+CRIg+ Mϕ (Figure 3(A)). Intestinal CRIg+ Mϕ were also able to take up dextran (Figure 3(B)), although the level of uptake was less than that of resident peritoneal F4/80+CRIg+ Mϕ.
Intestinal CRIg+ Mϕ display endocytic ability but are less efficient than resident peritoneal F4/80 + CRIg− Mϕ and F4/80 + CRIg+ Mϕ. (A) Resident peritoneal Mϕ were incubated with or without FITC-dextran for 45 min at 37°C and then stained with anti-F4/80 and anti-CRIg. The amount of uptake was analyzed by FACS for F4/80 + CRIg− Mϕ and F4/80 + CRIg+ Mϕ. Shaded areas represent cells after incubation without dextran, solid lines represent cells after incubation with dextran. Histograms are representative of four independent experiments. Values are the mean ± SEM. (B) Uptake of dextran by intestinal CRIg+ Mϕ was analyzed by FACS as described in the legend of (A). The amount of uptake by intestinal CRIg+ Mϕ was significantly lower than that by F4/80 + CRIg− Mϕ and F4/80 + CRIg+ Mϕ. * P < 0.05 against R1 and R2 in (A).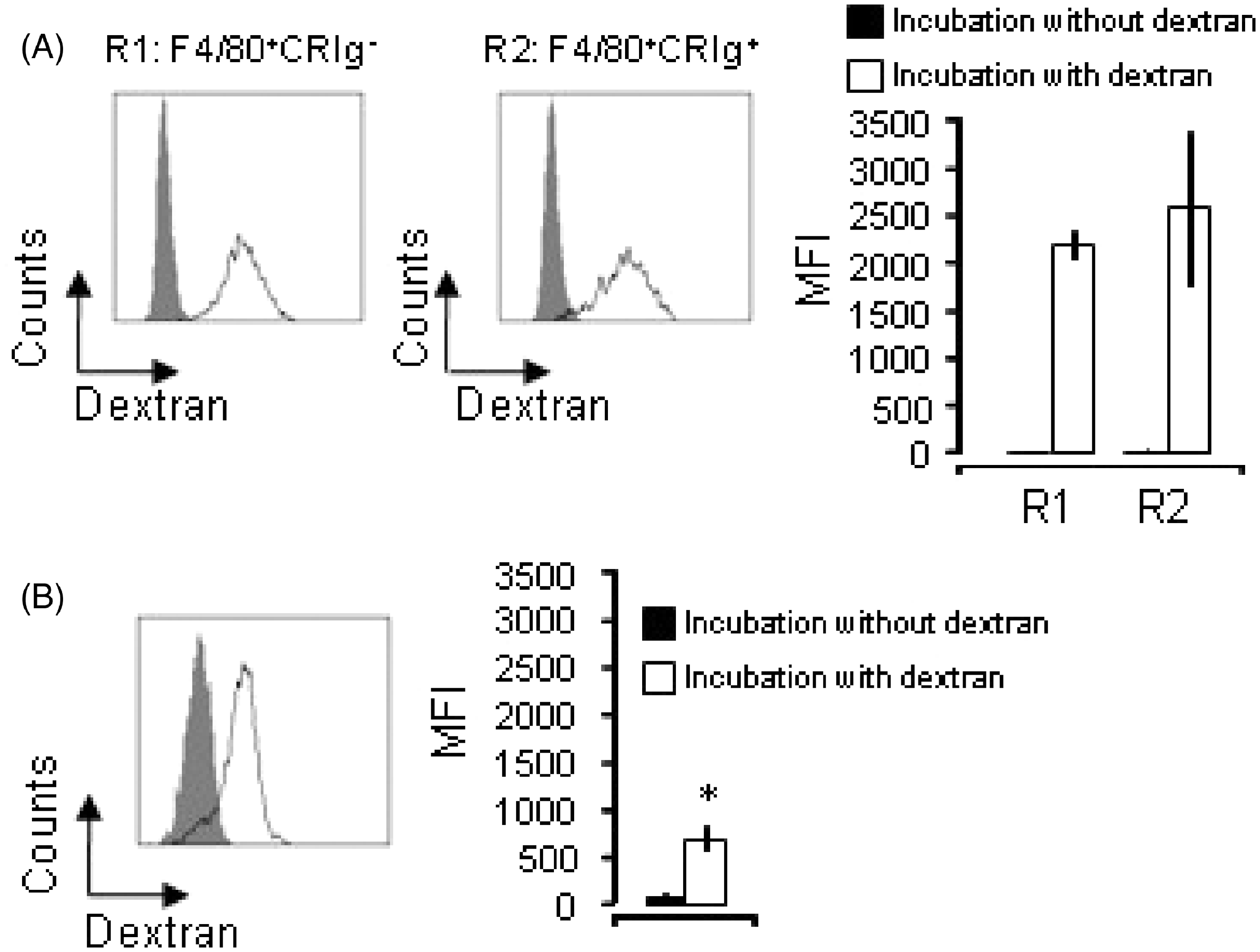
To analyze the phagocytic activity for particulates, the ability to take up BSA-coated microspheres, which depends on non-specific receptor pathways, was analyzed. As shown in Figure 4(A), the MFI of microsphere-positive cells were similar between peritoneal F4/80+CRIg− Mϕ and F4/80+CRIg+ Mϕ. In the case of intestinal CRIg+ Mϕ, the phagocytic activity for microspheres was as efficient as that of resident peritoneal F4/80+CRIg+ Mϕ, in contrast to the finding for endocytic activity (Figure 4(B)).
Intestinal CRIg+ Mϕ have similar levels of phagocytic activity against BSA-coated microspheres compared to resident peritoneal F4/80 + CRIg− Mϕ and F4/80 + CRIg+ Mϕ at levels of MFI. (A) Resident peritoneal Mϕ were incubated with or without BSA-coated microspheres for 1 h at 37°C and then stained with anti-F4/80 and anti-CRIg. The amount of uptake by F4/80 + CRIg− Mϕ and F4/80 + CRIg+ Mϕ was analyzed by FACS. Shaded areas represent cells after incubation without microspheres. Solid lines represent cells after incubation with microspheres (%, a ratio of positive cells of total cells). Histograms are representative of four independent experiments. Values are the mean ± SEM. (B) Uptake of BSA-coated microspheres by intestinal CRIg+ Mϕ was analyzed by FACS as described in the legend of (A). The amount of uptake was similar among resident peritoneal F4/80 + CRIg− Mϕ, F4/80 + CRIg+ Mϕ and intestinal CRIg+ Mϕ at levels of MFI.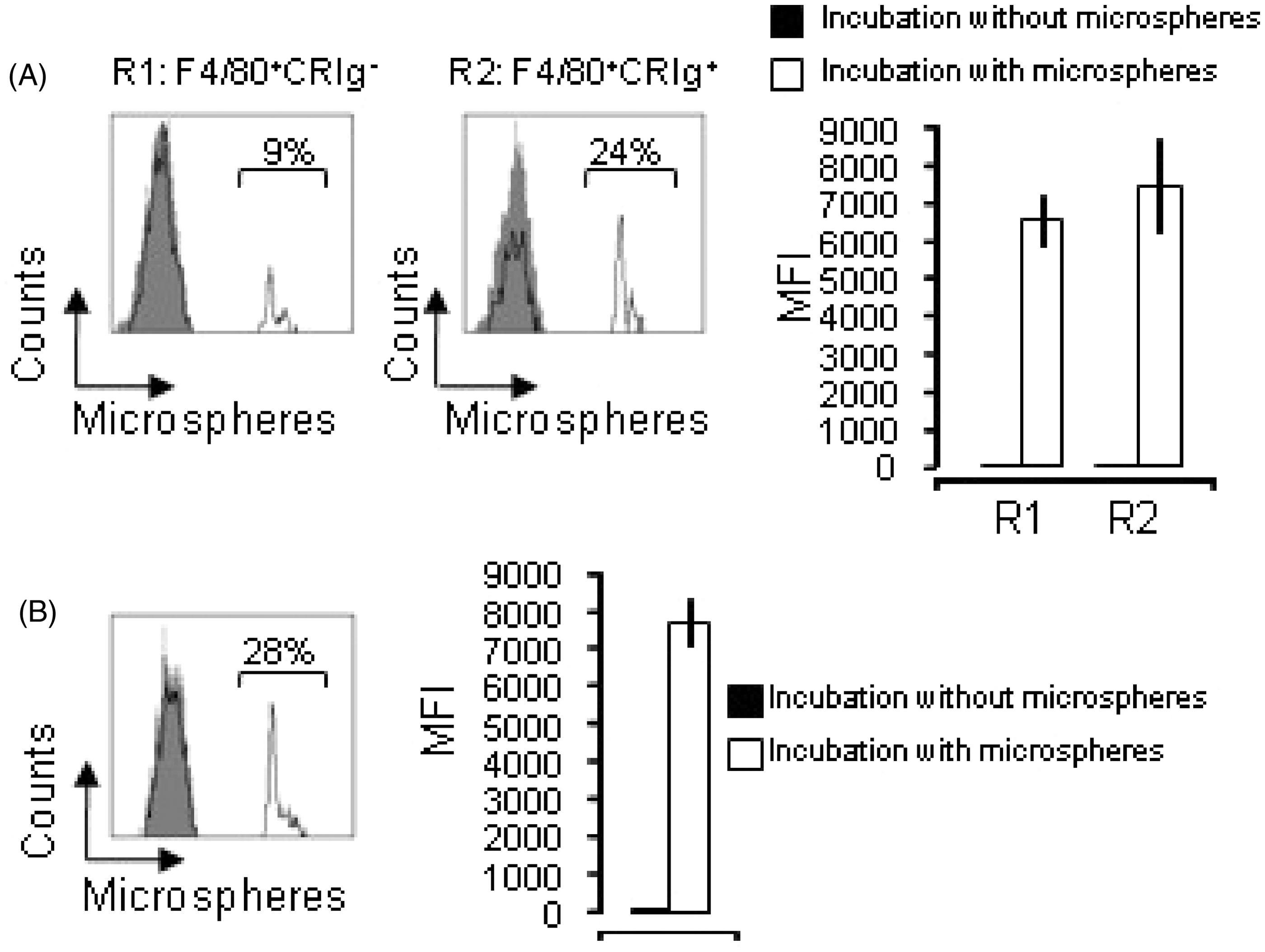
Complement receptor-mediated phagocytic ability of murine CRIg+ Mϕ from normal large intestine
To further examine whether the CRIg on intestinal Mϕ is a functional phagocytic receptor for C3b-opsonized targets, a phagocytosis assay was performed using C3-opsonized or C3b-opsonized microspheres in the presence or absence of blocking mAb against CRIg at the optimal concentration.
5
As shown in Figure 5(A), intestinal CRIg+ Mϕ displayed highly efficient uptake of C3b-opsonized microspheres when compared to C3-opsonized microspheres in the absence of CRIg-blocking mAb, indicating the presence of mechanisms enhancing the uptake of C3b-opsonized targets. In addition, the substantial C3b-dependent uptake was found to be reduced by approximately 30% by blocking anti-CRIg mAb (Figure 5(B)). On the other hand, the amount of C3b-dependent uptake was reduced by approximately 50% in resident peritoneal CRIg+ Mϕ, in agreement with a previous report (data not shown).
5
Intestinal CRIg+ Mϕ display CRIg-mediated phagocytic activity against C3b-opsonized microspheres. (A) Intestinal Mϕ were incubated with C3-opsonized or C3b-opsonized microspheres in the presence or absence of blocking anti-CRIg mAb for 1 h at 37°C, followed by FACS analysis to monitor the amount of uptake. In histograms, light-shaded areas represent cells after incubation without microspheres (left panels), dark-shaded areas represent cells after incubation with C3- or C3b-microspheres in the presence of blocking anti-CRIg mAb (middle panels), solid line-areas represent cells after incubation with C3- or C3b-microspheres in the absence of blocking anti-CRIg mAb but in the presence of control mAb (right panels). Data are representative of more than three independent experiments. (B) A substantial C3b-dependent uptake of microspheres was determined as described in the Materials and Methods. The substantial uptake obtained in the presence of blocking anti-CRIg mAb was compared with that in the presence of control mAb, and the effect of blocking anti-CRIg mAb on the uptake was presented as a percentage of MFI against the uptake in the presence of control mAb. Values are the mean ± SEM. * P < 0.05.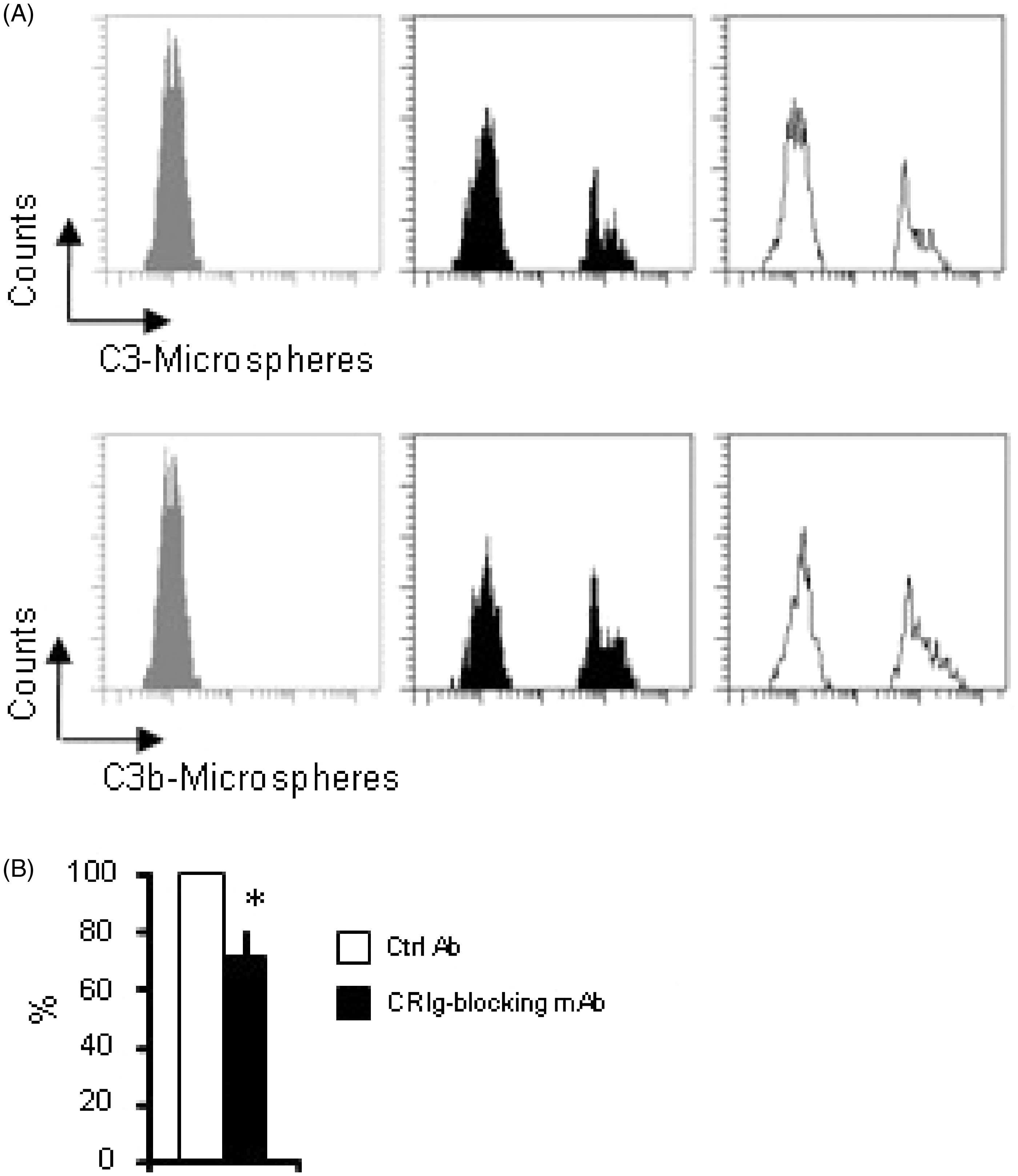
Discussion
Expression of CRIg was seen on a subpopulation of resident tissue Mϕ, consistent with previous reports that used murine and human tissues.4,7 In addition, we found CRIg expression on a major subset of murine and human Mϕ in the large intestine. Efficient endocytic and phagocytic abilities are characteristic of resident tissue Mϕ. 21 Indeed, intestinal CRIg+ Mϕ showed efficient endocytic and phagocytic abilities. Because commensal bacteria are numerous in the large intestine, it is questionable as to whether all intestinal CRIg+ Mϕ belong to resident intestinal Mϕ. A portion of these Mϕ may be infiltrating Mϕ from the circulation that, after recruitment, may acquire CRIg expression. It has been reported that an increase in number of Mϕ in the large intestine in a Crohn’s disease model is a result of infiltrated Mϕ 22 ; however, our data show that these Mϕ had decreased expression levels of CRIg compared to those in the normal large intestine, suggesting that CRIg expression is not a marker of infiltrating Mϕ. Although this study supports that CRIg is expressed on resident intestinal Mϕ, further ontogenetic approaches of CRIg+ Mϕ should verify whether CRIg+ Mϕ are resident intestinal Mϕ.
In this study, we detected almost no F4/80 on the surface of intestinal Mϕ, whereas the surface expression of F4/80 was clearly detected in more than half of peritoneal Mϕ. Conversely, we clearly observed intracellular F4/80 expression in the large intestine using immunohistochemical analysis (data not shown). It has been suggested that the lack of CD14 expression on the cell membrane of intestinal Mϕ may be caused by transport interference from the endoplasmic reticulum to the Golgi apparatus. 23 A similar mechanism may be involved in the transport of F4/80. Although we carefully removed attachments such as vessels and adipose tissues when collecting the large intestine, several reports have not mentioned performing such removal. Thus, the difference in phenotypes of large intestinal Mϕ in previous reports may be attributed to contamination with Mϕ from tissue attached to the large intestine.
As intestinal CRIg+ Mϕ were less endocytic but similarly phagocytic compared to resident peritoneal CRIg+ Mϕ, phagocytosis is suggested to be more dominant than endocytosis in intestinal CRIg+ Mϕ. This would be a significant characteristic of intestinal CRIg+ Mϕ, because phagocytosis has been thought to be an important function of triggering the production of immunosuppressive cytokines from intestinal Mϕ, contributing to the establishment of immunosuppressive milieu in the large intestine. 1 In this regard, this phenotype of intestinal CRIg+ Mϕ is a sharp contrast to that of rat synovial A cells, in which endocytosis is more enhanced than phagocytosis compared to rat peritoneal Mϕ. 19 These differences would reflect functional adaptations of resident Mϕ in response to different tissue microenvironment. 19
Although complement components have been reported to be locally synthesized by intestinal Mϕ and epithelial cells, 24 functional complement receptors have not been fully identified in the large intestine. Here, we show that CRIg is one of the components responsible for the clearance of immune complexes. As intestinal CRIg+ Mϕ display an efficient phagocytosis that depends on non-specific receptor pathways, even particulates which are not, or inefficiently, opsonized would be phagocytosed by these Mϕ. Besides this inherent phenotype, acquiring functional CRIg would enhance phagocytic activity of intestinal CRIg+ Mϕ against C3b-opsonized targets. These characteristics would partly explain why intestinal Mϕ are capable of highly efficient clearance of foreign particles in the large intestine which is proximal to microbe-rich environment.
Complement receptors and Fc receptors generally act in concert to facilitate phagocytosis, but these receptors have not been identified in the normal large intestine. The present study showed that intestinal CRIg+ Mϕ had a significant level of C3b-dependent phagocytosis. Application of the optimal concentration of blocking anti-CRIg mAb resulted in a reduction of C3b binding to peritoneal CRIg+ Mϕ by approximately 50%, consistent with a previous report. 5 In resident peritoneal Mϕ, another part of C3b-dependent phagocytosis is dependent on CD11b. The use of this antibody resulted in an average reduction of C3b binding to intestinal CRIg+ Mϕ by 30%, in spite of the lack of expression of CR3 (CD11b/CD18) on this Mϕ. These data indicate the presence of functional CRIg on Mϕ in the large intestine. The insufficient inhibition of anti-CRIg mAb may be explained by the presence of other C3b-binding receptors on intestinal CRIg+ Mϕ. Otherwise, the CRIg protein on intestinal Mϕ might be different in the conformational modulation from that on peritoneal Mϕ and, consequently, blocking anti-CRIg mAb may bind with low affinity to this protein. It has been suggested that the constitutive recycling of CRIg and the endocytosis of ligands by resting Mϕ fits with a potential role during the initial phase of bacterial infection. 25 Thus, C3b-dependent phagocytosis by CRIg may contribute to the efficient elimination of commensal and/or pathological bacteria from the intestinal lumen to the mucosa and apoptotic cells in the intestine. It has been shown that administration of anti-CRIg mAb induced the secretion of IL-8 and matrix metalloproteinase (MMP)-9 from Mϕ cell lines, which was dependent on NF-κB activation. 20 The regulation of immunological tolerance and inflammation in the intestine by this pathway is intriguing.
Notably, Mϕ from a Crohn’s disease model showed CD11b expression in addition to decreased CRIg expression, while tissue from an ulcerative colitis model showed similar CRIg expression and no CD11b expression, which was similar to the expression patterns in normal intestinal Mϕ. The difference in the inflammatory milieu in these disease models may contribute to the differential expression of these complement receptors. There is considerable evidence that the host’s commensal microbiota are crucial for the development of inflammatory bowel diseases.26,27 As it has been reported that CRIg and CD11b show distinct binding properties and ligand specificity, 25 it is interesting to consider how these complement receptors could contribute to the removal of commensal microbiota in the pathogenesis of Crohn’s disease.
In conclusion, we identified for the first time that the C3b receptor is present on Mϕ in the normal large intestine; this study will shed new light on the opsonization and phagocytosis by large intestinal Mϕ.
Footnotes
Funding
This work was supported by a Grant-in-Aid for Young Scientists (B) from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan (no. 19790690).
Acknowledgements
We thank Dr van Lookeren Campagne (Genentech) for providing blocking mAb against CRIg (clone 14G8) and isotype control mAb (anti-ragweed).
