Abstract
Recent studies have revealed that the immunological function of leukocytes is dependent on their cellular metabolism, and some researchers have advocated the beneficial effects of pioglitazone against sepsis in young mice, although bacterial infections are more prevalent in elderly hosts. Here, we investigated pioglitazone’s preventative effect against sepsis induced by intravenous injection of a lethal dose of Escherichia coli in elderly mice (50–60 wk old) and examined its immunological and metabolic effects on liver leukocytes. Pioglitazone improved bacterial elimination in the peripheral blood, lowered serum pro-inflammatory cytokines (TNF-α, IL-12, IFN-γ), and prevented septic death. It also enhanced bacterial elimination in the liver, by increasing the phagocytic and bactericidal activities of liver F4/80+CD11b+ recruited macrophages (Mφ), their CD206 expression and reactive oxygen species production. Quantitative PCR revealed that pioglitazone treatment enhanced gene expression of rate-limiting enzymes for glycolysis in hepatic CD11b+ cells (including neutrophils and recruited Mφ), and their improved phagocytic and bactericidal activities were abolished by glycolysis inhibiting reagents. These findings present the possibility that pioglitazone strengthens the phagocytic and bactericidal activities of liver recruited Mφ and that these immunological activities are closely associated with their glucose catabolism.
Introduction
The liver is the organ that controls metabolism and immunity, where hepatocytes and Kupffer cells are adjacently arranged. The hepatocytes mainly regulate glucose, lipid, and protein metabolisms. For example, following caloric intake, hepatocytes store excess energy by converting glucose into glycogen and triglyceride, and/or distribute it to peripheral tissues in the form of very low-density lipoprotein (VLDL). In the fasting state or during exercise, stored glycogen in hepatocytes is processed into glucose for supply to peripheral blood and tissues and, therefore, hepatocytes play a pivotal role in maintaining glucose homeostasis. 1 The Kupffer cells in the liver are the largest population of resident macrophages in the mammalian body and play an important role in eliminating pathogenic microbes.2,3 Many researchers have noted the heterogeneity of Kupffer cells, 4 and we recently demonstrated that they are mainly composed of liver resident F4/80+CD68+ Kupffer cells and F4/80+CD11b+ Kupffer cells/macrophages recruited from bone marrow (recruited Mφ).5,6 Importantly, they exhibit distinct properties and functions. Resident Kupffer cells are radio-resistant and exhibit great phagocytic and bactericidal activity, and recruited Mφ are radio-sensitive and have the capacity to produce a large amount of pro-inflammatory cytokines, such as TNF-α and IL-12, in bacterial infections or inflammations.6,7 IL-12 produced by recruited Mφ activates liver NK and NKT cells, 8 and they therefore play a major role in anti-tumor immunity. 6
We and others have found that immunological reactions in the liver might be affected by metabolic status.9,10 For example, we recently showed that liver NK/NKT cells of mice fed with a high fat and high cholesterol diet had increased tumoricidal activity against liver metastatic EL-4 cells due to increased IL-12 production from recruited Mφ, 9 resulting in improved survival rates. However, these mice were more susceptible to bacterial LPS-induced shock than control diet mice due to increased production of TNF-α from liver recruited Mφ, 9 suggesting that these mice would be more susceptible to Gram-negative bacterial infections. These results indicate that recruited Mφ may change their function depending upon metabolic conditions in the hosts. In addition, we previously reported that LPS-induced shock in mice was exacerbated in a TNF-α (from liver recruited Mφ) and age-dependent manner. 11
Peroxisome proliferator activated receptor (PPAR)-γ is a pivotal transcriptional factor in adipocyte differentiation and glucose homeostasis, and thiazolidinediones (TZDs) are potent insulin-sensitizing drugs that function by activating this receptor. 12 Among them, pioglitazone is the only TZD approved for the treatment of type 2 diabetes patients in Japan. In addition to effecting glucose control, the activation of PPAR-γ by TZDs contributes to the modulation of atherosclerosis and inflammation. 13
Recently, several groups of researchers have demonstrated that pioglitazone is effective in the prevention and treatment of sepsis in mice challenged with cecal ligation and puncture (CLP),14,15 and it has been suggested that pioglitazone could be used for the treatment of septic patients. 16 Despite the fact that pioglitazone is more widely used in middle-aged and senior patients than young adults, its immunological effects in elderly mice have not been investigated. In addition, elderly people have decreased immunity to pathogenic microbes. 17 These situations led us to investigate the mechanism of the effect of pioglitazone on the innate immune function of liver Kupffer cells/recruited Mφ in elderly mice infected with Escherichia coli (E. coli).
Materials and methods
The Ethics Committee of Animal Care and Experimentation, National Defense Medical College, Japan approved all requests for animals and the intended procedures for the present study (Permission number: 16020). All experiments were performed in accordance with relevant guidelines and regulations. Every effort was made to minimize suffering, and there were no unexpected deaths.
Mice and reagents
Young male (7 wk old) C57BL/6 (B6) mice were purchased from SLC Japan (Hamamatsu, Japan). They were housed less than ten per cage with free access to food and water. Animals were maintained at constant temperature and humidity (21–24°C and 40–55%, respectively) under a 12 h light/dark cycle. Some were used immediately as young mice and the others fed standard laboratory chow and kept under clean conditions until the age of 50–60 wk and used as elderly mice.
18
Their body masses were 20.5 ± 0.5 and 47.0 ± 3.5 g, respectively. E. coli strain B (
E. coli challenging and pretreatment with pioglitazone
Sepsis was induced in the elderly mice by intravenous injection of a lethal dose (8 × 108 CFU) of E. coli. This dose was not sufficient for sepsis induction in young mice, so we administered 1 × 109 CFU to them. Survival was monitored every 12 h for 5 d after bacterial injection, and mice showing signs of imminent death, such as decreased temperature and agonal breathing, were euthanized by intraperitoneal injection of pentobarbital (15 mg/kg).
Pre-treatment with pioglitazone (10 mg/ml) was performed by intraperitoneal injection 3 h before E. coli challenge (10 mg/kg). We administered the same volume of DMSO for control experiments. For inhibiting PPAR-γ in pioglitazone-treated mice, GW9662 (2 mg/kg) was administered 15 min before treatment with pioglitazone.
Determination of bacterial burden
Mice were intravenously injected with 6 × 108 CFU of E. coli, and blood from their vena cava and livers were harvested after 3 h. Blood was diluted ten-fold with PBS. Homogenates of livers were serially diluted 1 × 104 fold. A volume of 100 µl of these specimens was spread on BHI agar plates, and incubated for 12 h at 37°C for counting bacterial colonies.
Isolation of liver mononuclear cells (MNCs) and circulating neutrophils
Liver MNCs and circulating neutrophils were isolated 3 h after injection with pioglitazone as previously described,6,19 using HBSS containing 0.05% collagenase (Wako, Osaka, Japan) and 6% dextran in PBS, respectively.
Cell sorting
Liver CD11b+ cells were enriched to greater than 70% purity by positive selection using the MACS system (Miltenyi Biotec, Bergisch, Germany), and negative fractions were used as CD11b- cells (containing less than 3% of CD11b+ cells). We used PE-conjugated anti-mouse CD11b mAb (affymetrix eBioscience, Central Expressway Santa Clara, CA) and anti-PE MicroBeads (Miltenyi Biotec).
Assessment of phagocytes for bacteria growth inhibition activity
The bacteria growth inhibition activity of magnetically sorted CD11b+ and CD11b- cells, or peripheral neutrophils was determined by incubating them (5 × 105 cells in 200 µl of RPMI1640 containing 10% FBS but not antibiotics) with viable E. coli (1 × 107) for 3 h. 19 As a control, the same number of E. coli were incubated without leukocytes in the medium. Then, aliquots of the cell suspension were diluted 10-fold with PBS, placed on BHI agar plates, and incubated at 37°C for 12 h. Then, the number of CFUs was counted. For determination of bactericidal activity, 0.5% Triton-X100 was added to the incubated cell suspension before dilution with PBS.
Assessment for intracellular killing activity by gentamicin protection assay
The intracellular killing assay was performed as previously described. 20
Assays for cytokines, C-reactive protein (CRP), aminotransferase (ALT), and glucose
TNF-α, IL-12 p40, and IFN-γ levels in the serum as well as IL-12 p40 and IL-10 levels in the culture medium were measured using the respective cytokine-specific ELISA kits (BD Bioscience, San Diego, CA).21,22 CRP was determined using ELISA Kit, Mouse (Kamiya Biomedical Company, Seattle, WA), while ALT and glucose were measured using a DRI-CHEM 3500V system (Fuji Film, Tokyo, Japan).
Flow cytometry and bacteria phagocytic activity analysis
The liver MNCs and circulating neutrophils were incubated with Fc-blocker (2.4 G2; BD PharMingen, San Diego, CA) to prevent any nonspecific binding. Then, they were stained with FITC-, biotin-, or Cy7-F4/80, PE- or Cy7-CD11b, PE-Ly6C, and Cy5-Gr-1 mAbs (affymetrix eBioscience). FITC-, biotin-, or Cy7-rat IgG2a, and PE-rat IgG2c mAbs (affymetrix eBioscience) were used as an isotype control for F4/80+ cells. Biotin-F4/80 and rat IgG2a mAbs were secondarily stained with APC (affymetrix eBioscience). Intracellular staining with PE-labeled CD206 (affymetrix eBioscience) or its isotype (rat IgG2a, affymetrix eBioscience) was conducted after surface staining and incubation with BD Perm/Wash solution (BD Bioscience) at 4°C for 20 min.
Bacteria phagocytic activity was assessed using pHrodo® E. coli (Thermo Fisher Scientific K.K., Yokohama, Japan), for which fluorescence increases as the pH of the surrounding environment become acidic, enabling detection of functional phagolysosomes in phagocytic cells. We cultured liver MNCs with pHrodo® E. coli (1 × 107 bacteria per 5 × 105 cells) for 1 h before surface staining. In some experiments, liver MNCs were pre-incubated with pioglitazone (10 µg/ml) in vitro for 12 h.
To determine engulfment of labeled E. coli, liver MNCs were either chilled on ice or incubated at 37°C, with FITC-E. coli (8 × 106 bacteria per 5 × 105 cells) for 1 h. After washing, they were stained with PE-CD11b and Cy7-F4/80 mAb. The results were determined by subtracting the percentages of positive populations for on ice from those for incubation at 37°C.
Reverse transcription and quantitative real-time PCR
Total RNAs were isolated from liver CD11b+ cells using RNeasy Mini kit 50 (QIAGEN, Valencia, CA), and their concentrations were determined with the ABI PRISM 7000 sequence detection system (Life Technologies Corporation, Carlsbad, CA). cDNAs were synthesized from 500 ng of total RNA by reverse transcription using a SuperScript® III First-Strand Synthesis device (Thermo Fisher Scientific K.K). Quantitative real-time RT-PCR was performed on a LightCycler 480 System (Roche, Mannheim, Germany) with SYBR Green PCR reagents (LightCycler® 480 SYBR Green I Master Version 12, Roche). The primers were designed by Takara Bio (Tokyo, Japan) and are listed in Supplemental Table 1. The data were normalized for the level of Rps18 expression in each sample.
Metabolic inhibition
For inhibition of glycolysis, high glucose (25 mM) DMEM (Thermo Fisher Scientific K.K.) containing 10 mM 2-deoxy-
Determination of ROS production
We used Fc OXYBURST® green reagent (Thermo Fisher Scientific K.K.) in the determination of ROS production from liver MNCs before staining their surface Ags.
To determine ROS production by phorbol 12-myristate 13-acetate (PMA)-stimulated liver phagocytes, freshly isolated liver MNCs were suspended with PBS (1 × 106 cells/500 µl in cold PBS), 10 µl of Fc OXYBURST® reagent was added, and then the suspension was incubated for 30 min with or without PMA.
To assess ROS production by liver phagocytes cultured with E. coli, the culture was suspended with PBS (1 × 106 cells/500 µl in cold PBS), 10 µl of Fc OXYBURST® reagent was added, and then incubated for 30 min.
Statistical analysis
Statistical analyses were performed using JMP version 13 (SAS Campus Drive, Cary, NC). The survival rates were compared using the Wilcoxon rank test, and other statistical analyses were performed using the Mann–Whitney U and F tests. Results are given as the mean ± SEM. Differences were considered to be significant at P < 0.05.
Results
Pioglitazone improved bacterial infection
To assess the effects of pioglitazone pretreatment on sepsis in elderly (50–60 wk of age) mice, we administered pioglitazone or the vehicle to the mice at 3 h before intravenously challenging them with a lethal dose (8 × 108 CFU) of E. coli. Decreasing blood glucose confirmed that this agent was effective at 3–4.5 h after administration (Supplemental Figure 1). Pre-treatment with pioglitazone significantly improved survival rates in the septic mice (Figure 1a). Pioglitazone had diminished viable E. coli counts in the peripheral blood as well as in the liver at 3 h after administration of E. coli (Figure 1b and c), and lowered serum pro-inflammatory cytokine levels (TNF-α, IL-12, and IFN-γ) (Figure 1d), regardless of pre-treatment with a PPAR-γ antagonist, GW9662. Pioglitazone also decreased serum ALT levels after E. coli infection, while serum CRP levels were not significantly different (Figure 1e). Pioglitazone pre-treatment had similar effects on young mice and tended to improve their survival rates, although the difference was not statistically significant (Supplemental Figure 2).
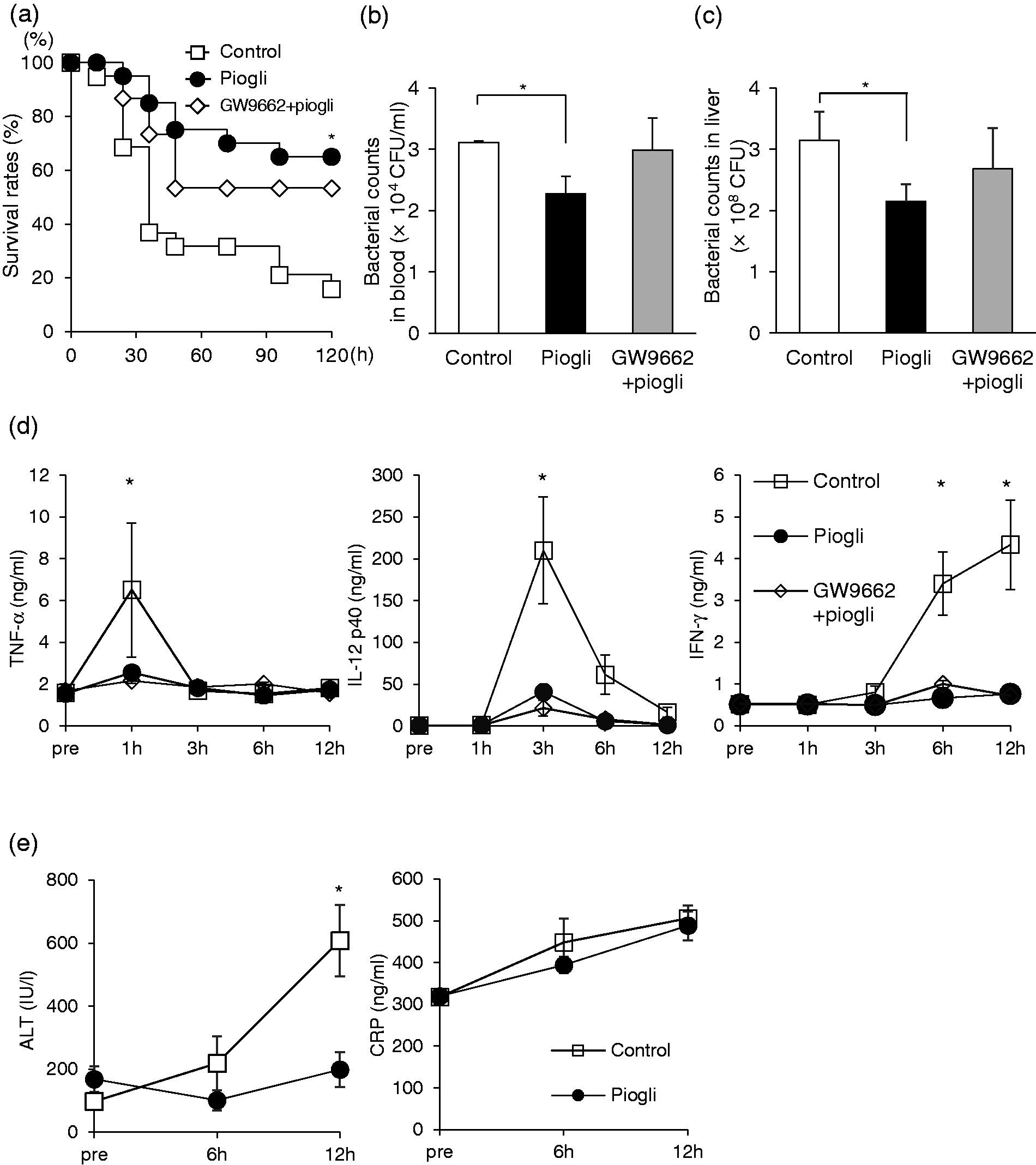
Pioglitazone prevents septic death in elderly mice by inhibiting bacteria growth and decreasing inflammatory cytokines. (a) Survival rates of elderly mice (50–60 wk of age) injected with lethal dose (8 × 108 CFU) of E. coli. Prior to bacterial challenge, they had been treated with pioglitazone (n = 20), GW9662 and pioglitazone (n = 15), or DMSO (n = 19). *P < 0.05 vs control. (b, c) Bacterial counts in blood (b) and liver homogenates (c) at 3 h after administration of sublethal dose (6 × 108 CFU) of E. coli (n = 4, respectively), in mice, which had been treated in the same manner as in (a). *P < 0.05 vs. control. (d) Serum TNF-α, IL-12 p40, and IFN-γ levels of mice challenged with sublethal dose (6 × 108 CFU) of E. coli. Data are means ± SEM, n = 5, at each data point. *P < 0.05 vs. control. (e) Serum ALT and CRP levels of control or pioglitazone-treated mice, challenged with sublethal dose (6 × 108 CFU) of E. coli. Data are means ± SEM from 5 mice in each group. *P < 0.05 vs. control.
Pioglitazone improves phagocytic activity of liver MNCs in elderly mice
Since several studies have found that most bacteria invading the body are accumulated and eliminated in the liver,23,24 we next examined the immunological effects of pioglitazone treatment on liver phagocytes. Intracellular killing assays (gentamicin assay) showed that pioglitazone treatment improved bacterial incorporation by liver MNCs in elderly mice (Figure 2a). But this effect of pioglitazone was blunted in vitro, because its addition to the culture of liver MNCs from normal mice did not increase their ingestion of pHrodo® E. coli (Figure 2b).
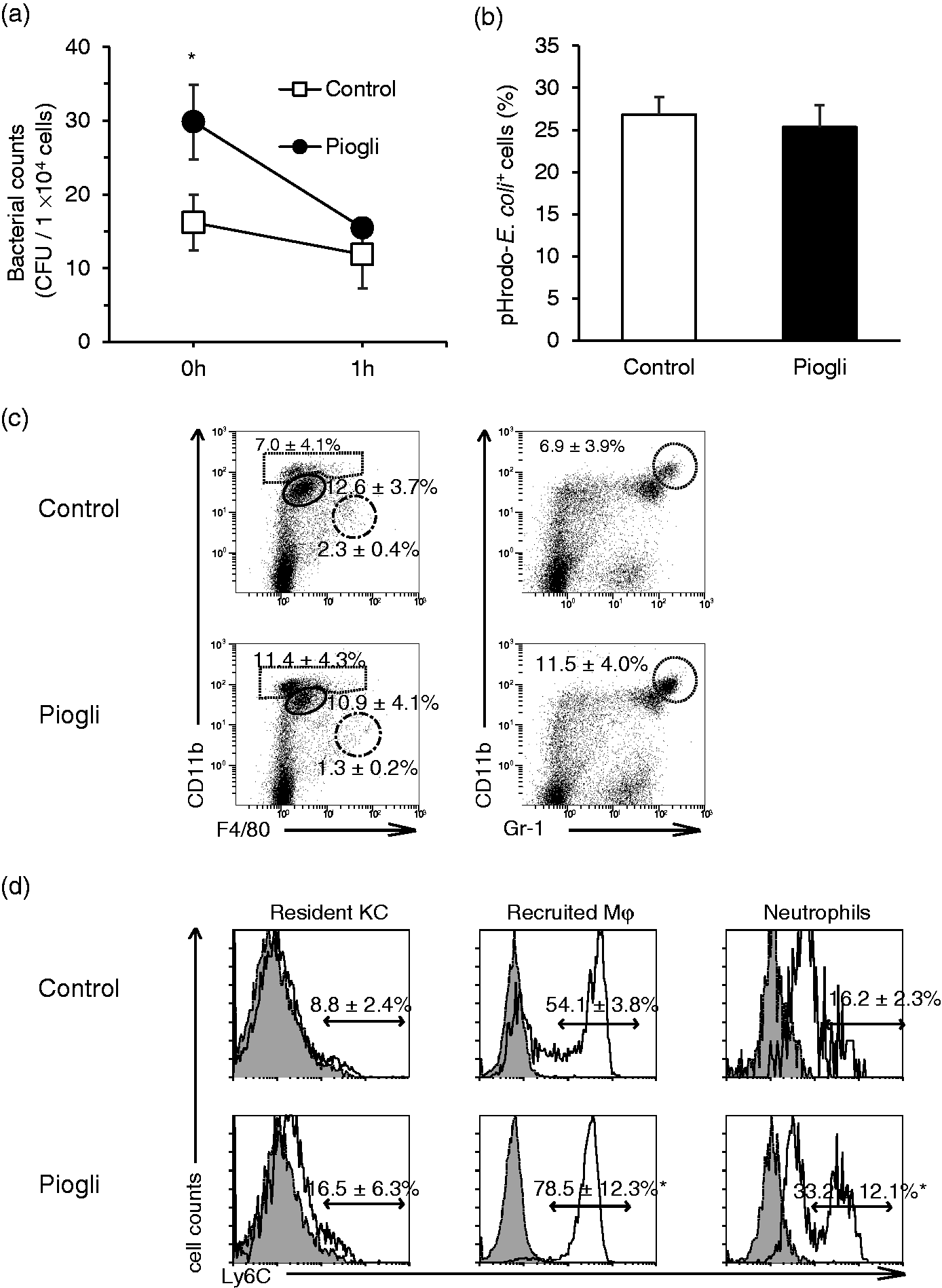
Pioglitazone improves phagocytic activity of liver MNCs in elderly mice. (a) Intracellular killing assays for liver MNCs from control or pioglitazone-treated (piogli) elderly mice. (b) Liver MNCs from normal mice were incubated with DMSO or pioglitazone for 12 h, before adding pHrodo® E. coli, for 1 h and analyzed for ingestion of pHrodo® E. coli. (c and d) Surface expressions of liver MNCs were analyzed by flow cytometry 3 h after administration of pioglitazone to elderly mice. (c) Populations of F4/80highCD11blow resident KC (dashed line), F4/80lowCD11bhigh recruited Mφ (solid line), CD11bhigh neutrophils (dotted line), are shown in the left panels, and the CD11bhighGr-1+ population (neutrophils) is shown in the right panels. (d) Ly6C expression (open curves) in resident KC, recruited Mφ, and neutrophils of control or pioglitazone-treated mice. Filled curves indicate the respective isotype controls. Data are means ± SEM for 4 mice in each population. *P < 0.05 vs. control.
In order to evaluate the effect of pioglitazone on hepatic phagocyte populations, we counted the number of F4/80highCD11blow (CD68+) cells (resident KC), F4/80lowCD11bhigh (CD68-) cells (recruited Mφ), and Gr-1+CD11bhigh cells (neutrophils) using flow cytometry. However, no significant difference was detected among the three populations with pioglitazone treatment (Figure 2c). An increase in neutrophils observed in young mice (Supplemental Figure 3a) was blunted in elderly mice, and resident KC remained in the minority despite treatment with pioglitazone in elderly mice. Of note, pioglitazone increased the Ly6Chigh population of recruited KC and neutrophils both in young (Supplemental Figure 3b) and elderly mice (presumably newly recruited from bone marrow) (Figure 2d). As the size of the Gr-1highCD11bhigh cell population (Figure 2c, right panels) was almost identical to that of the CD11bhigh population (Figure 2c, left panels), we could regard CD11bhigh cells as a Gr-1+CD11bhigh cells (neutrophils).
Pioglitazone increases phagocytic activity of liver recruited Mφ
Flow cytometric analysis using pHrodo® E. coli showed that phagocytic activity of recruited Mφ (but not of neutrophils) increased in the pioglitazone-treated mice with or without additional administration of GW9662 (Figure 3a). No significant difference in phagocytic activity of resident KC was observed between control and pioglitazone-treated elderly mice (data not shown). Pioglitazone treatment also increased uptake of FITC E. coli by liver recruited Mφ (Figure 3b). Consistent with the increased phagocytic activity, pioglitazone increased expression of CD206 in the liver recruited Mφ after incubation for 12 h, which may have been involved in their phagocytic function (Figure 3c).
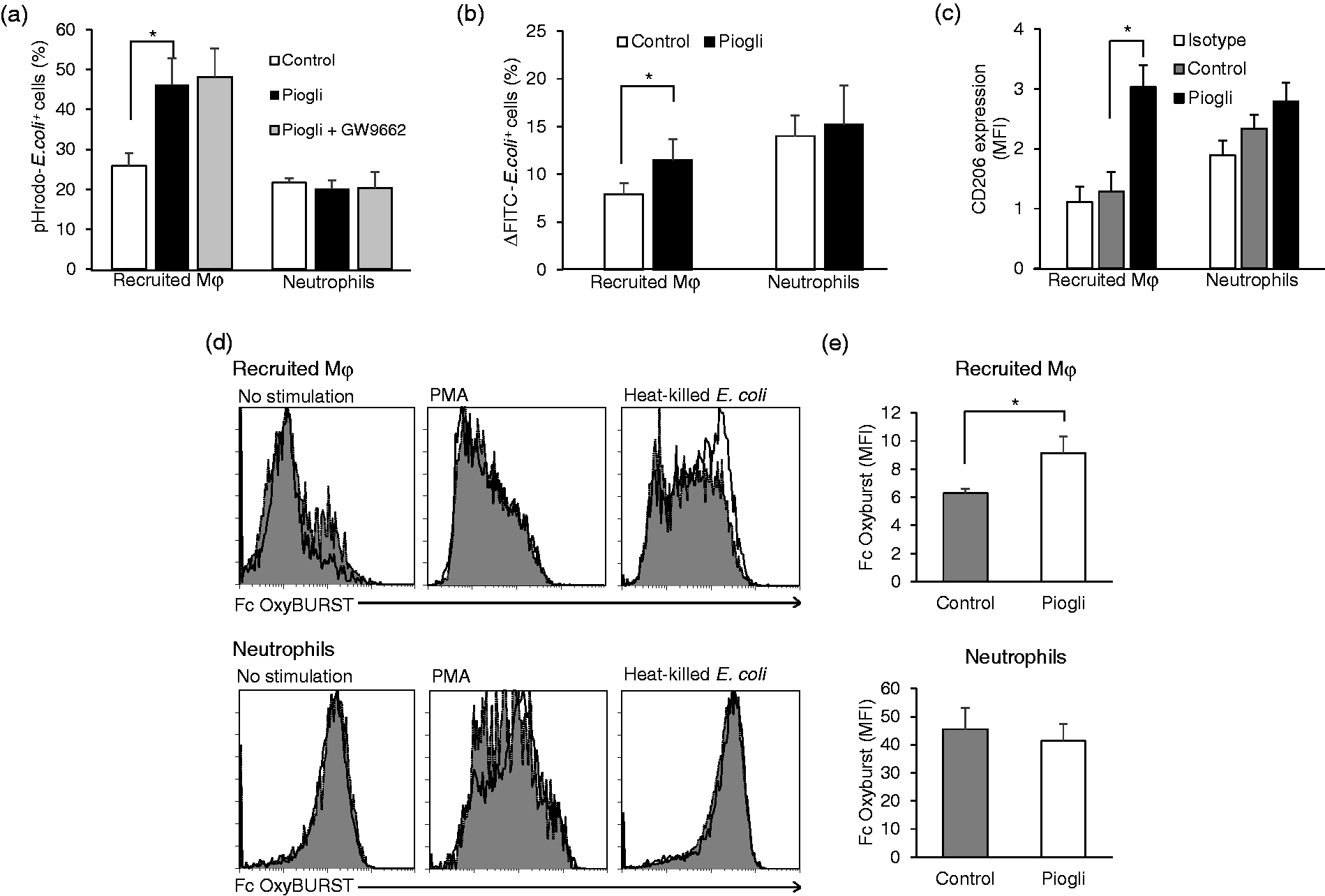
Pioglitazone increases phagocytic activity of liver recruited Mφ. (a) Phagocytic activity of liver recruited Mφ and neutrophils in elderly mice for control, pioglitazone-treatment, and GW9662 plus pioglitazone treatment. Liver MNCs were obtained from the control mice, those with pioglitazone treatment and those receiving GW9662 plus pioglitazone treatment, and then cultured with pHrodo® E. coli for 1 h. We determined pHrodo® E. coli-positive recruited Mφ and neutrophils by flow cytometry. (b) Engulfment of FITC-E. coli by liver recruited Mφ and neutrophils in control and pioglitazone-treated elderly mice. The results were determined by subtracting the percentages of positive populations for on ice from those for incubation at 37°C (Supplemental Figure 5). (c) CD206 expression after pioglitazone treatment. Liver MNCs were obtained from elderly mice 3 h after pioglitazone or vehicle treatment, and then incubated at 37°C for 2 h to determine intracellular CD206 expression in the recruited Mφ and neutrophils. Data are shown as MFI, and isotype controls are shown on the left. (d and e) ROS production by liver recruited Mφ and neutrophils. Liver MNCs were harvested from elderly mice 3 h after administering pioglitazone. Non-stimulated (left panels), PMA-stimulated (middle panels), or heat-killed E. coli-stimulated (right panels) MNCs were incubated with Fc OxyBURST® reagent, followed by cell surface staining. Recruited Mφ and neutrophils were analyzed for Fc OxyBURST® staining by flow cytometry. (d) Representative histograms for Fc OxyBURST® staining in recruited Mφ (upper panels) and neutrophils (lower panels). The dotted curves are for control mice, and the solid curves for pioglitazone-treated mice. (e) MFI for Fc OxyBURST® staining in recruited Mφ (upper graph) and neutrophils (lower graph) cultured with heat-killed E. coli. Data are means ± SEM from 4 mice in each group. *P < 0.05 vs. control.
We then examined ROS production in cells, which is integral to microbial killing. 25 The Fc OxyBURST® green assay revealed increased fluorescence for liver recruited Mφ from pioglitazone-treated mice cultured with heat-killed E. coli (but not with PMA) (Figure 3d and e), which was not observed in neutrophils. Thus, ROS production was enhanced in pioglitazone-treated liver recruited Mφ stimulated with E. coli.
Immunological and metabolic alterations of liver CD11b+ phagocytes by pioglitazone treatment
For assessing the immunological and metabolic effects of pioglitazone treatment on liver phagocytes, we magnetically sorted CD11b+ cells (recruited Mφ and neutrophils) from liver MNCs from several elderly mice with or without pioglitazone treatment. By incubating these CD11b+ cells with living E. coli, we evaluated their inhibitory activities on bacteria growth. The results showed that pioglitazone pretreatment increased the phagocytic (Figure 4a) and bactericidal activities (Figure 4b) of magnetically sorted liver CD11b+ cells. On the other hand, no significant changes in phagocytic (Supplemental Figure 4a) or bactericidal (Supplemental Figure 4b) activities were observed in circulating neutrophils. When liver CD11b+ cells were cultured with dead E. coli, those from pioglitazone-treated mice produced greater amounts of IL-10 than those from the control mice. However, no difference was observed in IL-12p40 production (Figure 4c).
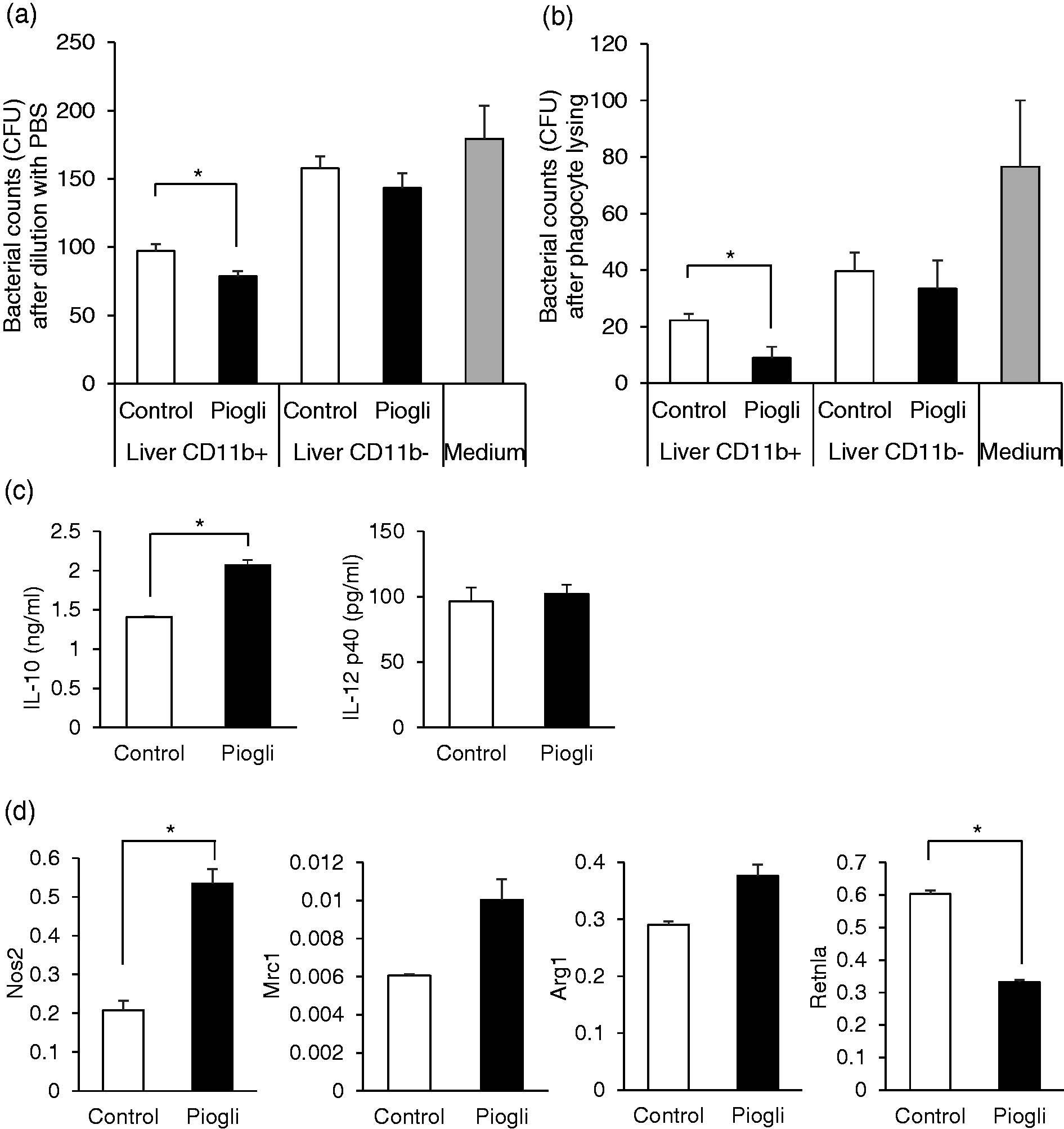
Pioglitazone alters immunological properties of liver CD11b+ phagocytes in elderly mice. (a, b) Bacteria growth inhibitory (a) and bactericidal (b) activity of liver CD11b+ and CD11b- cells obtained from several control or pioglitazone-treated elderly mice. Liver MNCs were obtained 3 h after pioglitazone treatment and then their CD11b+ or CD11b- cells were magnetically sorted. The sorted cells were incubated with 100 CFU of viable E. coli for 3 h. As a control, E. coli were incubated without leukocytes in the medium. After incubation, aliquots of the cell suspension were either diluted 10-fold with PBS (a) or diluted to10-fold with PBS after adding Triton X-100 (b), placed on BHI agar plates, and incubated at 37°C for 12 h. (c) Cytokine production by liver CD11b+ cells. Liver CD11b+ cells from control or pioglitazone-treated elderly mice were sorted, and their IL-12 p40 and IL-10 productions were determined by ELISA after culturing with heat-killed E. coli for 24 h. (d) mRNA expression of liver CD11b+ cells in control and pioglitazone-treated elderly mice. Sorted liver CD11b+ cells from elderly mice with or without pioglitazone treatment were examined for gene expression of Nos2, Mrc1, Arg1, and Retnla. Data are means ± SEM from four samples in each group. *P < 0.05 vs. control.
PCR analysis showed that liver CD11b+ cells obtained from pioglitazone-treated mice exhibited greater mRNA expression of NOS2 (M1 Mφ marker) than those from control mice. Regarding M2 Mφ markers, expression of the Arg1 gene was not significantly different, while that of Retnla was decreased (Figure 4d). Mrc-1 gene expression tended to be enhanced but this was not significant. These results of cytokine production and gene expression analyses suggest that liver recruited Mφ are activated but their functional state cannot be classified as either M1 or M2.
We next examined the gene expression of encoding enzymes related to metabolism in the liver CD11b+ cells of pioglitazone-treated mice. We found that mRNA expression of regulatory enzymes of glycolysis (phosphofructokinase, pyruvate kinase, and hexokinase-1) was increased (Figure 5). The OXPHOS-encoding gene Ndufs1 was also up-regulated, while the expressions of other related genes were similar to those for the control. However, expressions of the Β-oxidation-encoding genes Acadm and Cpt2 were lower (Figure 5).
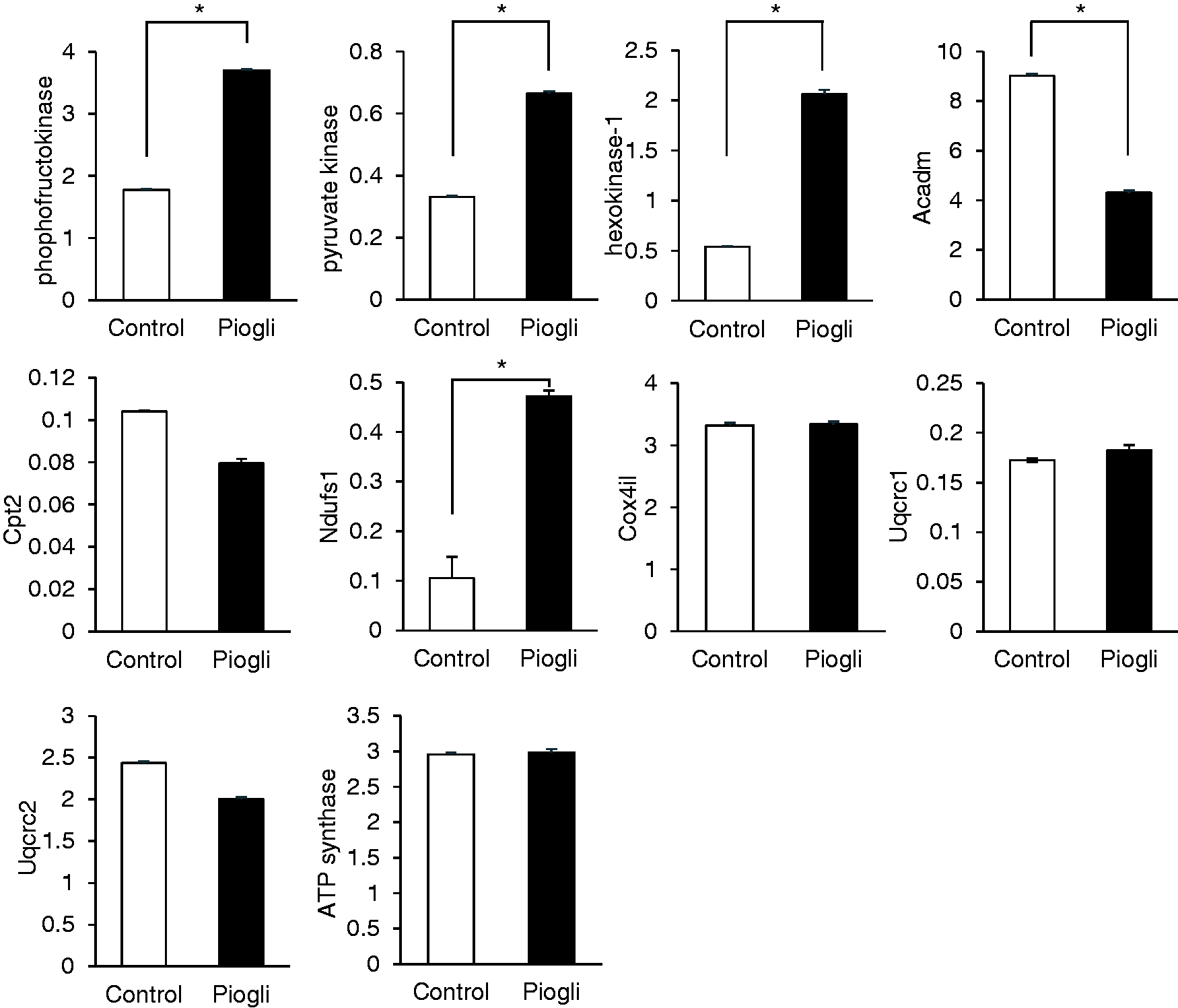
Gene expression of liver CD11b+ cells involved in cellular metabolism. Liver CD11b+ cells from elderly mice, obtained from 3 h after pioglitazone treatment, were examined for mRNA expression in glycolysis (phosphofructokinase, pyruvate kinase, and hexokinase-1), β-oxidation (Acadm and Cpt2), and OXPHOS (Ndufs1, Cox4il, Uqcrc1, Uqcrc2, and ATP synthase) pathways. Data are means ± SEM from four samples in each group. *P < 0.05 vs. control.
Increased phagocytic and bactericidal activities of liver recruited Mφ due to pioglitazone treatment are dependent on glucose catabolism
In order to further examine whether the increased phagocytic and bactericidal activities of the liver recruited Mφ were associated with their enhanced glycolytic and OXPHOS activity, we cultured liver MNCs from pioglitazone-treated or control mice for 2 h in media together with the glycolysis inhibitor 2-DG or OXPHOS inhibitor oligomycin A. Inhibition of both glycolysis and OXPHOS sharply reduced incorporation of E. coli by liver phagocytes in the pioglitazone-treated mice (Figure 6a). However, similar trends in these metabolic inhibitions were also evident in the control mice (Figure 6a). Flow cytometric analysis using FITC-labeled or pHrodo® E. coli consistently showed that metabolic inhibition had decreased the phagocytic activity of liver recruited Mφ in pioglitazone-treated mice (Figure 6b and c). Consistent with the decreased phagocytic activity, both types of inhibition diminished the intracellular CD206 expression in liver recruited Mφ (Figure 6d) as well as ROS production in these cells when cultured with heat-killed E. coli (Figure 6e). In the control experiments, on the other hand, both types of metabolic inhibition decreased phagocytic activity for pHrodo® E. coli (Figure 6c) but not for FITC-labeled E. coli (Figure 6b). In addition, OXPHOS inhibition decreased CD206 expression and ROS production, while glycolysis inhibition did not (Figure 6d and e).
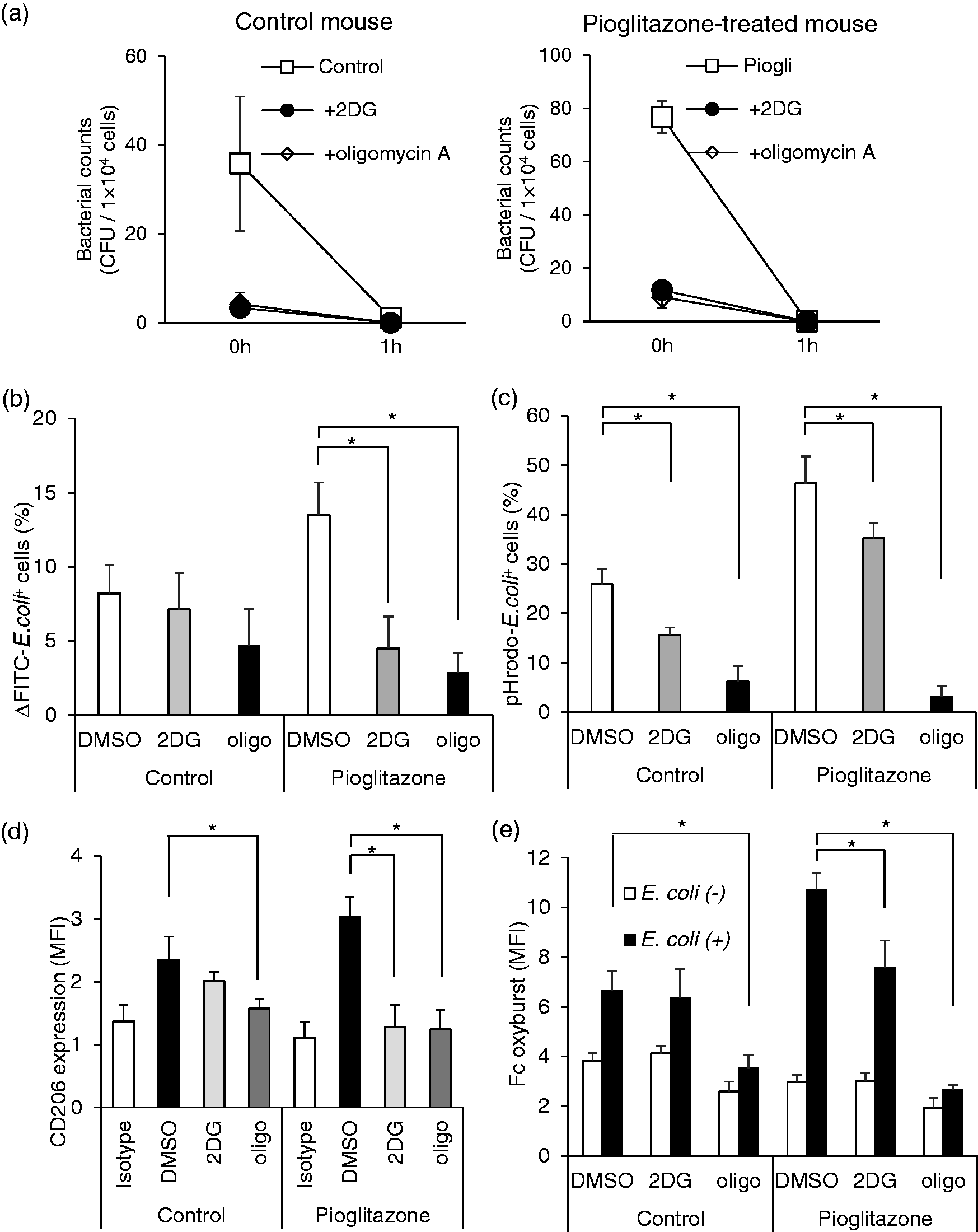
Inhibition of glucose catabolism reduces phagocytic and bactericidal activities in liver phagocytes from pioglitazone-treated mice. (a) Intracellular killing assay for liver MNCs in control and pioglitazone-treated elderly mice. Liver MNCs were pre-incubated for 2 h with DMSO, 2DG, or oligomycin A, and cultured with viable E. coli (1 × 107 bacteria per 5 × 105 cells) at 37°C for 45 min, before eliminating free bacteria by centrifugation and re-suspension in gentamicin solution (100 µg/ml). (b, c) Phagocytic activity of liver recruited Mφ in which glycolysis or OXPHOS were metabolically inhibited. (b) Liver MNCs from control and pioglitazone-treated elderly mice were preincubated for 2 h with 2DG, oligomycin A, or DMSO. They were either chilled on ice or incubated at 37°C with FITC-E. coli for 1 h, and positive recruited Mφ were determined by flow cytometry. The results were determined by subtracting the percentages of positive populations for on ice from those for incubation at 37°C. (c) Liver MNCs from control and pioglitazone-treated elderly mice were incubated with pHrodo® E. coli for 1 h after metabolic inhibition, and then positive recruited Mφ were determined. (d) Effect of metabolic inhibition on intracellular CD206 expression in liver recruited Mφ from control and pioglitazone-treated elderly mice. After incubating liver MNCs from the mice for 2 h with 2DG, oligomycin A, or DMSO, CD206 expression in recruited Mφ was analyzed. (e) Effect of metabolic inhibition on ROS produced by liver recruited Mφ in control and pioglitazone-treated elderly mice. Liver MNCs from the mice were preincubated for 2 h with 2DG, oligomycin A, or DMSO. They were cultured with or without heat-killed E. coli for 1 h, and then incubated with Fc OxyBURST® reagent, followed by cell surface staining. Liver recruited Mφ were analyzed for Fc OxyBURST® staining by flow cytometry. Data are means ± SEM from four samples in each group. *P < 0.05 vs. control.
Discussion
This study revealed that administration of pioglitazone improves the survival rate of elderly mice with lethal E. coli infection and decreases pro-inflammatory cytokines as well as bacterial growth in the peripheral blood. In addition, bacterial elimination in the liver was enhanced due to improved phagocytic function of liver recruited Mφ. As several studies have demonstrated that the liver is responsible for eliminating most of the E. coli invading the body,23,24 it seems obvious that the improved bacterial clearance in the liver and suppressed systemic inflammatory response due to pioglitazone would contribute to increasing the survival rate of mice with sepsis. In addition, since these effects of pioglitazone were only partially reversed by the administration of the PPAR-γ antagonist GW9662, they were not fully dependent on PPAR-γ.
Ferreira et al. have already reported the beneficial effects of pioglitazone in septic mice created by CLP. 26 They found that the intraperitoneal injection of pioglitazone not only enhanced CLP-induced migration of neutrophils into the peritoneal cavity to clear bacteria but also increased anti-inflammatory cytokine (IL-10) production in situ. 26 In the present study, we found that it increased the phagocytic and bactericidal activities of liver recruited Mφ as well as their IL-10 production when stimulated with E. coli. Flow cytometric analysis using FITC-E. coli and pHrodo® E. coli reproducibly revealed an obvious increase in the phagocytic activity of liver recruited Mφ but not of neutrophils due to pioglitazone, suggesting that recruited Mφ are mainly involved in the therapeutic effect of pioglitazone.
We previously reported that liver recruited Mφ were not so important for bacterial elimination because of their weak phagocytic activity. 5 Their role in bacterial infection was to produce pro-inflammatory cytokines such as TNF and IL-12, and elimination of bacteria from liver tissue was mainly performed by resident Kupffer cells. 5 In this study, however, the resident Kupffer cells only accounted for a few percent of liver MNCs in elderly mice and their phagocytic activity was not increased by pioglitazone. In addition, in our more recent study, liver recruited Mφ in LPS-tolerant mice exhibited remarkably increased phagocytic and bactericidal activities but decreased TNF and IL-12 production, and thereby marked resistance to E. coli infection was induced. 27 These findings prompted us to re-evaluate the phagocytic activity of liver recruited Mφ in pioglitazone-treated mice. The results suggest that the functional features of liver recruited Mφ in mice treated with pioglitazone were close to those of these cells in LPS-tolerant mice. Therefore, recruited Mφ appear to have flexibility in regulating their function depending on the conditions in the host. This preferential pioglitazone effect on recruited Mφ was unexpected at first, because we previously found that resveratrol (an antioxidant) increased resident Kupffer cells with phagocytic activity but reduced TNF-α-producing recruited Mφ in the liver. 28 However, we assume that pioglitazone and resveratrol have distinct metabolic effects on resident Kupffer cells and recruited Mφ, respectively. Consistent with this assumption, it was reported that resveratrol inhibited glucose uptake in adipose tissues but TZDs increased it. 29
We speculate that two mechanisms are possibly involved in the enhancement of the phagocytic activity of liver recruited Mφ due to pioglitazone treatment. One is enhanced intracellular CD206 expression and the other is increased cellular ROS production. CD206 (also known as mannose receptor C type 1, MRC1) recognizes microbial carbohydrates and becomes active in phagocytosis and Ag presentation. 30 Moreover, its enhanced expression in Mφ may be related to greater phagocytosis of E. coli. 31 ROS production is crucial for microbial killing in phagocytes, 25 as evidenced by susceptibility to bacterial infection in patients with chronic granulomatous disease (CGD). 32 In this regard, it was reported that pioglitazone restores mitochondrial oxidants as well as the bactericidal capacity of phagocytes in gp91phox-/- mice, a model of CGD. The peritoneal phagocytes of these mice were shown to have a defect in mitochondrial ROS production after PMA stimulation, probably due to lack of NADPH oxidase signaling. However, pioglitazone treatment increased ROS production in the mitochondria of phagocytes in gp91phox-/- mice and rendered them capable of killing Staphylococcus aureus, presumably because pioglitazone treatment could bypass the need for NADPH oxidase. 33 The enhanced ROS production in liver recruited Mφ due to pioglitazone treatment in our study is consistent with these results.
It is commonly recognized that TZDs increase insulin sensitivity and glucose uptake in peripheral tissues. 12 In previous studies assessing their metabolic effects on whole liver tissues, TZDs improved glycolysis but inhibited glyconeogenesis. 34 We found that liver recruited Mφ in pioglitazone-treated mice enhanced expressions of genes encoding rate-limiting enzymes involved in glycolysis (phosphofructokinase, pyruvate kinase, and hexokinase-1). 35 Furthermore, they ingested larger numbers of bacteria, which was abrogated by the inhibition of glycolysis or OXPHOS. These results suggest that pioglitazone might increase the phagocytic and bactericidal activities of liver recruited Mφ by regulating their metabolic catabolism, especially of glucose.
Consistent with the present study, we previously reported that in db/db mice, a model of diabetes in which leptin receptor activity is deficient, the phagocytic and bactericidal activity of peripheral blood neutrophils was decreased, but recovered with insulin treatment, 36 and thus increased phagocytic and bactericidal activity in neutrophils might be dependent on enhancement of glucose catabolism. Vender et al. reported that enhanced glucose incorporation might induce actin cytoskeletal changes and increase phagocytic activity for complement-opsonized zymozan (COZ) in LPS-stimulated macrophages, 37 and added that phagocytic activity for COZ was not affected by inhibition of OXPHOS. However, we found that inhibiting OXPHOS diminished the ingestion and killing of bacteria by liver recruited Mφ in the control and pioglitazone-treated mice. This issue will need further studies for clarification.
Finally, the number of neutrophils tended to be increased by pioglitazone treatment. They produced larger amounts of ROS than recruited Mφ from control and pioglitazone-treated mice. Therefore, neutrophils may additionally contribute to the improved antibacterial effect of pioglitazone.
In summary, pioglitazone enhances the bacteria phagocytic activity of liver recruited Mφ and regulates their production of pro-inflammatory and anti-inflammatory cytokines, thereby endowing elderly mice with resistance to bacterial infection. In addition, their phagocytic activity might be regulated by their glucose catabolism. Having the potential to regulate recruited Mφ, pioglitazone could be a candidate therapeutic modality for elderly patients with bacterial infections.
Supplemental Material
Supplemental Material1 - Supplemental material for Pioglitazone improves phagocytic activity of liver recruited macrophages in elderly mice possibly by promoting glucose catabolism
Supplemental material, Supplemental Material1 for Pioglitazone improves phagocytic activity of liver recruited macrophages in elderly mice possibly by promoting glucose catabolism by Masahiro Nakashima, Manabu Kinoshita, Hiroyuki Nakashima, Aya Kotani, Takuya Ishikiriyama, Shoichiro Kato, Sadayuki Hiroi and Shuhji Seki in Innate Immunity
Supplemental Material
Supplemental Material2 - Supplemental material for Pioglitazone improves phagocytic activity of liver recruited macrophages in elderly mice possibly by promoting glucose catabolism
Supplemental material, Supplemental Material2 for Pioglitazone improves phagocytic activity of liver recruited macrophages in elderly mice possibly by promoting glucose catabolism by Masahiro Nakashima, Manabu Kinoshita, Hiroyuki Nakashima, Aya Kotani, Takuya Ishikiriyama, Shoichiro Kato, Sadayuki Hiroi and Shuhji Seki in Innate Immunity
Footnotes
Acknowledgement
We thank Susumu Tominaga for his technical assistance.
Declaration of conflicting interests
The author(s) have declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Masahiro Nakashima has received a JSPS KAKENHI Grant (Grant Number 17K08611). Manabu Kinoshita (advanced research program entitled “CBRN Defense”) and Hiroyuki Nakashima (special program entitled “Host defense by liver lymphocytes and myeloid cells”) have received Grants from the National Defense Medical College. The other authors have no funding to declare.
Supplemental material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
