Abstract
The regulation of interleukin (IL)-1 expression and secretion by salivary glands and macrophages in response to lipopolysaccharides (LPS) was compared. In wild-type mice, injection of LPS significantly decreased the volume of saliva stimulated by pilocarpine and increased its protein and amylase concentration. It did not modify the salivary concentration of IL-1β. The cytokine was expressed by submandibular acini and ducts. Macrophages also expressed IL-1β but at lower concentration than salivary glands. The pre-incubation of macrophages with LPS increased the phosphorylation of IκB and the expression of IL-1β. Adenosine triphosphate also promoted the secretion of the cytokine by these cells. These responses were absent in submandibular gland cells. These glands expressed CD14, TLR4 and MyD88. P2X7-KO mice secreted a lower volume of saliva which contained less proteins and amylase. In conclusion, IL-1β is constitutively expressed by submandibular glands and its secretion is not regulated by a P2X7 agonist. In these cells, LPS do not activate the nuclear factor-κB–pro-IL-1β axis in spite of the expression of the proteins involved in their recognition.
Introduction
Saliva is an exocrine secretion rich in various proteins probably less important for the digestion than for the protection of the mouth. Indeed, saliva contains not only enzymes like amylase, kallikrein or lipase but also various proteins such as peroxidase, defensins, histatins or cathelicidin. 1 These proteins increase the buffering capacity of saliva and have major antibacterial properties. Growth factors (like epidermal or nerve growth factors) are constituents of saliva: they contribute to the repair of wounded tissues and participate in the differentiation and maturation of specialized structures like taste buds. 2 The salivary secretion also contains cytokines like interleukin (IL)-1β or IL-6. 3 Interleukin-1β is a pro-inflammatory cytokine mostly expressed and secreted by macrophages. 4 The stimulation by lipopolysaccharides (LPS) of the Toll-like receptor 4 (TLR4) of these cells promotes the expression of pro-IL-1β, the inactive precursor of IL-1β. 5 The secretion of the cytokine requires its activation by a protease, caspase-1. The conversion and activation of procaspase-1 to caspase-1 is triggered by extracellular signals among which is ATP. 6 This nucleotide binds on plasma membrane purinergic receptors which were initially described and classified by Burnstock and Kennedy. 7 The P2Y receptors are metabotropic receptors coupled to guanylnucleotide regulatory proteins. The P2X receptors have two transmembrane domains and, after trimerisation, form a non-selective cation channel. Seven P2X receptors have been cloned and the P2X7 receptor, a receptor with low affinity for ATP, is the purinergic receptor coupled to IL-1β secretion in macrophages. 8 Experiments with macrophages which do not express this receptor fully support this conclusion. These cells were collected from transgenic mice (P2X7-KO mice); they strongly expressed the gene of pro- IL-1β when exposed to LPS but did not release IL-1β in response to ATP. The trigger of the coupling between the occupancy of P2X7 receptors and the release of IL-1β seems to be a decrease of the intracellular concentration of potassium. 9 Considering that salivary glands express IL-1β 10 and P2X7 receptors, 11 the purpose of our work was to determine whether the expression of IL-1β was regulated by LPS in salivary glands and if the P2X7 receptors could promote the release of the cytokine from submandibular gland cells like they do from macrophages.
Materials and Methods
Mice
The experiments were carried out on male C57Bl/6J P2X7R+/+ wild-type mice and P2X7R–/– null mice kindly supplied by Pfizer Inc. (Groton, CT, USA). Breeding of P2X7 knockout males with females was used to maintain the colony of receptor-deficient animals. Mice used in the experiments were between 20–25 g weight. The expression of P2X7 receptors in wild-type mice and absence in null mice was tested by immunoblot as previously described. 12
Materials
Ethylene glycol-bis-(β-aminoethyl ether)-N, N, N, N′-tetraacetic acid (EGTA), sodium pyruvate, sodium fumarate, sodium glutamate, N-[2-hydroxyethyl] piperazine-N′-[2-ethanesulfonic acid] (HEPES), N-α-benzoyl-
Preparation of a submandibular glands crude suspension
Four mice were sacrificed by increasing the CO2 concentration in accordance with the procedures of the Belgian Ministry of Agriculture under the supervision of the institutional ethical committee. The submandibular glands were immediately dissected and finely minced. They were incubated for 20 min at 37°C under constant shaking, in the presence of 1 mg collagenase P, in 10 ml HEPES-buffered saline (HBS) medium containing (m
Preparation of the ductal and acinar suspension
Acinar and ductal cells were separated from the submandibular glands crude suspension by the method of Dehaye and Turner. 13 Briefly, they were separated by centrifugation through an isotonic Percoll solution (40%), at 4000 g at 4°C for 10 min. The ducts, which remained on the top of the Percoll, and the acini, which migrated to the bottom, were recovered and washed three times with an isotonic saline solution.
Collection of saliva
The animals were treated in accordance with the guidelines of the National Institute for Dental Research (Bethesda, MD, USA). A 100 µg/ml saline solution of LPS was prepared to study the effect of LPS. After filtration, the solution was injected intraperitoneally into the mice (400 µg/kg), according to Yao et al. 14 Saliva was collected 6 h after the injection of LPS. The mice were anesthetized by injection of a mixture of ketamine (80 mg/kg) and xylazine (10 mg/kg) in the abdomen. They were placed under a light to avoid hypothermia. Pilocarpine HCl (1 mg/kg) was injected subcutaneously in the dorsolumbar region to stimulate salivation. The whole saliva was collected for 20 min in a glass micropipette by capillary flow and allowed to drop in a preweighed Eppendorf tube. At the end of the 20 min period, the amount of saliva was estimated by weighing the tube and the mice were sacrificed by increasing the CO2 concentration.
Isolation of peritoneal macrophages
Peritoneal macrophages were prepared as described by McCarron et al.
15
Three to five d before harvesting the cells, 2 ml thioglycollate solution (4% in sterile water) was injected intraperitoneally into the mice. After sacrifice of the animals, the peritoneal cavity was lavaged by 10 ml ice-cold phosphate buffered saline (PBS) containing 10 U/ml heparin. The abdomen was gently massaged before recovering the fluid. After centrifugation at 1500 g for 10 min at 4°C, the cells were resuspended in RPMI 1640 medium containing 20 m
Preparation of the cell extracts for the ELISA
The crude suspension of submandibular glands of four mice was prepared and the cells were resuspended in HBS medium in the presence of 1% (w/v) BSA and 0.5 m
Acinar and ductal cells were also prepared and, after separation and washing, they were centrifuged and resuspended in 1 ml ice-cold lysis buffer. They were then treated in the same way as the crude suspension or the peritoneal macrophages.
Detection of IL-1β by ELISA
A 96-well plate was coated overnight with 1 µg/ml polyclonal anti-mouse IL-1β antibody at 4°C. After washing with Tris-Tween buffer (Tris-HCl 50 m
Amylase secretion
The samples of saliva were diluted five times before the assay. Aliquots of 10 µl were transferred into glass tubes and mixed with 240 µl of a solution containing 40 m
Analysis of RNA expression by RT-PCR
Primers used for the RT-PCR

To verify the accuracy of the cDNA obtained after RT-PCR for TLR4, MyD88 and CD14, a sequence analysis was realized. The amplified PCR products were purified and concentrated using the DNA Clean and ConcentratorTM-5 kit (Zymo Research, Orange, CA, USA). The nucleic acid concentration was determined using a NanoDrop 2000c spectrophotometer (Thermo Scientific, Waltham, MA, USA) and amplified cDNA was premixed with the specific primer (5 ng/µl cDNA containing 15 pmoles of primer). Samples were sent for sequence analysis to Eurofins MWG Operon (Ebersberg, Germany).
Immunoblot analysis of IκB phosphorylation
Macrophages or submandibular gland cells were incubated in the presence of 250 ng/ml LPS for different times. The cells were then lysed in ice-cold lysis buffer (NaCl 137 m
Determination of the [Ca2+]i
The cells were incubated at 25°C for at least 1 h in the presence of 3 µ
Determination of the [K+]i
The cells were incubated at 25°C for at least 1 h in HBT in the presence of 3 µ
Assay of kallikrein activity
Ducts and acini were homogenized in 10 m
Assay of protein concentration
The protein concentration was determined in duplicates with the bicinchoninic acid assay. Bovine serum albumin was used to establish a standard curve.
Statistical analysis
Results were analyzed with the Mann–Whitney non-parametric test. ***P < 0.001; **P < 0.01; *P < 0.05.
Results
Salivary concentrations of IL-1β in control and LPS-injected mice
Wild-type mice were injected with saline alone or with 10 µg LPS. After 6 h, the mice were anesthetized and injected subcutaneously with pilocarpine (1 mg/kg). The muscarinic agonist stimulated the salivary secretions both in saline- and LPS-injected animals (Fig. 1, upper panel). The volume of saliva collected for 20 min from saline-injected mice (393 ± 11 mg) was significantly larger than from LPS-treated mice (250 ± 19 mg; n = 4; P < 0.05). The saliva from saline-injected animals contained 665 ± 164 µg/ml protein and the activity of amylase averaged 709 ± 35 µmoles maltose/ml (n = 4). The injection of LPS non-significantly increased the protein content of the saliva (1009 ± 111 µg/ml; n = 4; P = 0.54) but significantly increased the activity of amylase (818 ± 8 µmoles maltose/ml; n = 4, P = 0.026 when compared to control). IL-1β was detected in saliva from both groups of animals. Its concentration averaged 264 ± 33 pg/ml in saline-injected animals (n = 3) and decreased by 40% to 152 ± 8 pg/ml in LPS-treated animals (n = 4; P = 0.058). Similar experiments were performed with KO mice. As shown in Figure 1, KO-mice injected with saline secreted 255 ± 56 mg saliva (n = 4, P = 0.2 when compared to WT mice). The saliva of these KO mice had a non-significantly lower concentration of protein (443 ± 56 µg/ml; n = 5; P = 0.41 when compared to wild-type mice), amylase activity (577 µmoles maltose/ml saliva; n = 5; P = 0.057) and IL-1β (188 ± 36 pg/ml; n = 5; P = 0.14). The injection of LPS to KO mice had no effect on the volume of saliva (277 ± 10 mg; n = 4; P = 0.48 when compared to control KO mice) or the salivary concentration of protein (727 ± 130 µg/ml, P = 0.064) and significantly increased the secretion of amylase (713 ± 16 µmoles maltose/ml; P = 0.029). The secretion of IL-1β was very variable among the four tested mice but its average (138 ± 28 pg/ml) was not different from control KO mice (P = 0.73).
Composition of the saliva collected from mice injected with saline or LPS. Wild-type and P2X7-KO mice were injected intraperitoneally with saline (open bars) or with 10 µg LPS. Six h later, the animals were anesthetized and injected subcutaneously with 1 mg/kg pilocarpine. The saliva was collected for 20 min in a preweighed Eppendorf tube. The weight of the collected fluid and its concentration of proteins and IL-1β and its amylase activity were assayed as described in Materials and Methods. Results are mean ± SEM of (n) experiments.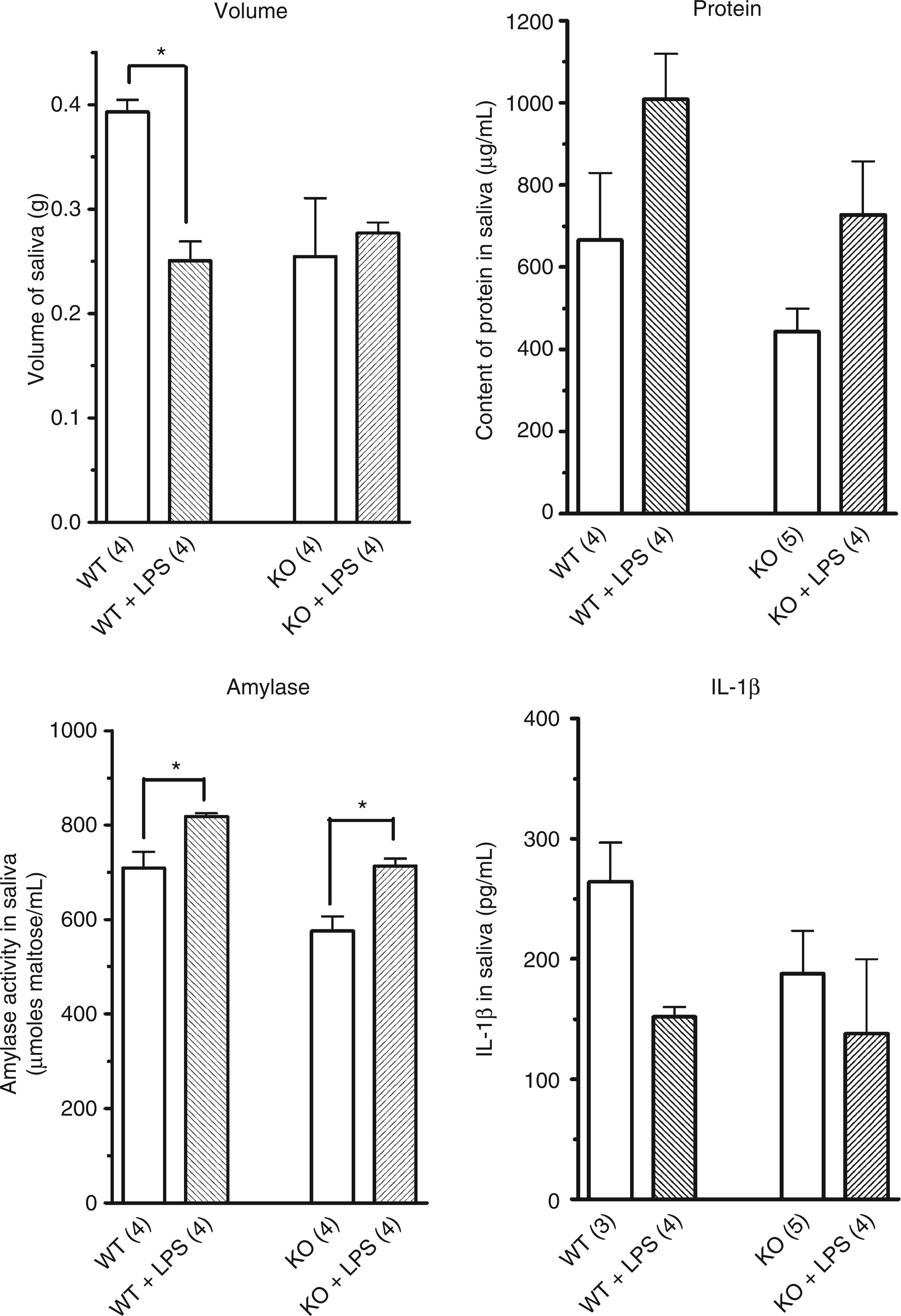
Expression of IL-1β by submandibular glands
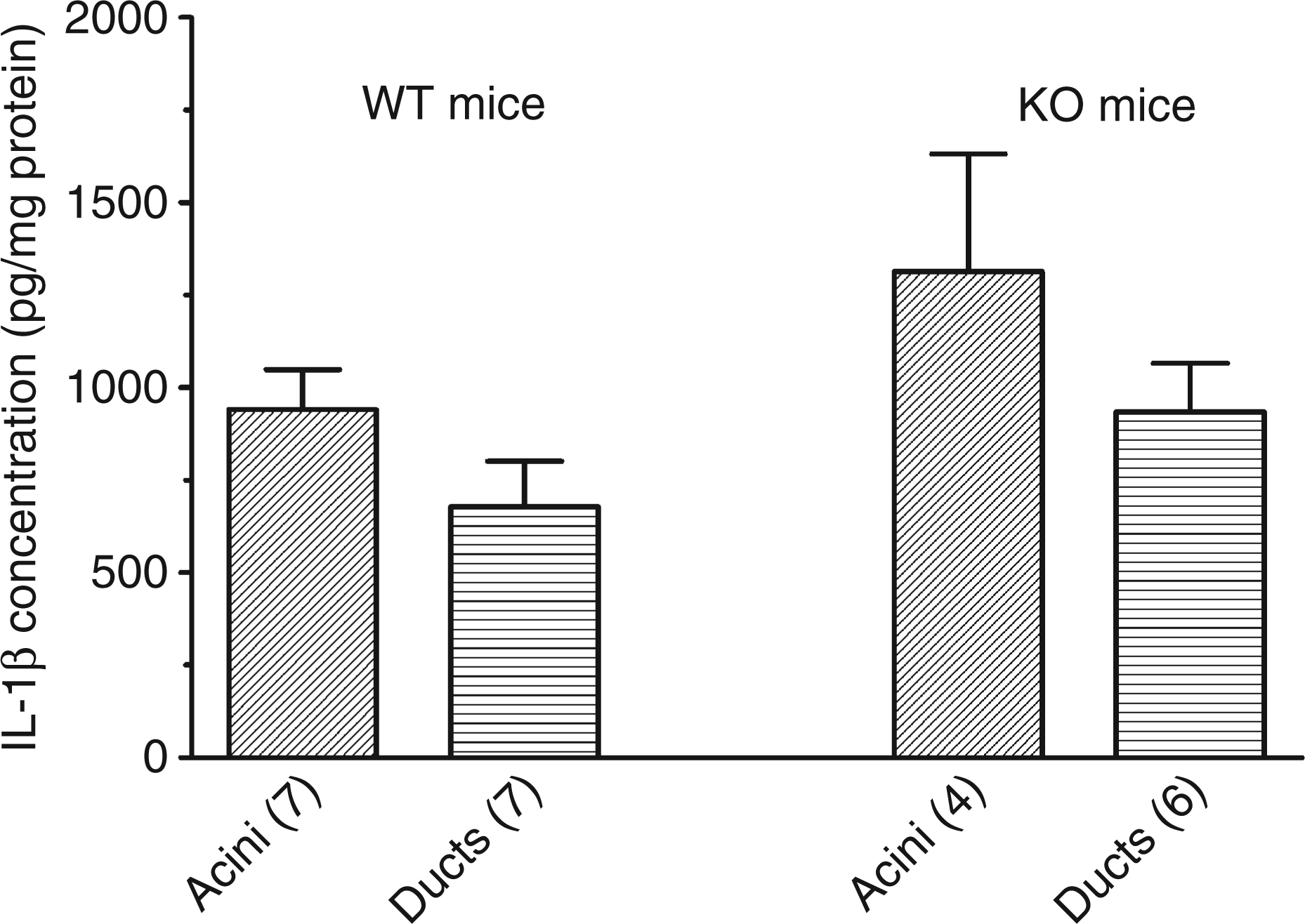
Previous results were obtained with whole saliva. In the next experiments, the submandibular glands from wild-type and KO mice were more extensively studied. Peritoneal macrophages from both mice were also tested for their IL-1β content. As shown in Figure 2, the level of IL-1β in macrophages pre-incubated in control conditions was very low (162 ± 27 pg/mg protein (n = 3) in cells from wild-type mice and 150 ± 48 pg/mg protein; n = 3) in cells from KO mice). The treatment of the macrophages for 2 h with 250 ng/ml LPS provoked a massive increase of the expression of IL-1β and the concentration of the cytokine reached in the cells from both mice more than 6000 pg/mg protein, the maximal concentration which could be assayed. Interleukin-1β was also expressed by submandibular glands and its level reached 390 ± 50 pg/mg protein (n = 4) in a crude cellular suspension isolated from submandibular glands of wild-type mice. This was significantly more than the basal level of IL-1β in macrophages from these mice (P = 0.002). The incubation of the cellular suspension from wild-type mice with LPS for 2 h did not modify the concentration of the cytokine (412 ± 51 pg/mg protein; n = 4). The expression of IL-1β by the submandibular glands was confirmed by RT-PCR. The mRNA of the cytokine was reverse transcribed and amplified in cellular extracts from submandibular glands of wild-type and KO mice. Immunoblots further confirmed the expression of the cytokine (data not shown). In cellular extracts from submandibular glands, a single band which migrated around 20 kDa and corresponding to IL-1β was observed. In submandibular glands from KO mice the concentration of IL-1β averaged 537 ± 95 pg/mg protein (n = 6) and 513 ± 139 pg/mg protein (n = 3) in cells incubated, respectively, in control conditions or in the presence of LPS. In a final experiment, the crude cellular suspension from wild-type mice was centrifuged through a Percoll gradient in order to separate ducts and acini. The quality of the purification was assessed by measuring the activity of kallikrein, a protease secreted by ductal cells. The activity of kallikrein expressed per milligram of cellular proteins was 19-fold higher in the ‘ductal fraction’ than in the ‘acinar fraction’ (data not shown) confirming the efficacy of the Percoll gradient to separate ducts from acini. As shown in Figure 3, there was no difference in the concentration of IL-1β in ducts (676 ± 123 pg/mg protein; n = 7) versus acini (941 ± 108 pg/mg protein; n = 7) in wild-type mice or in KO mice (940 ± 130 pg/mg protein, n = 6 in ducts and 1313 ± 318 pg/mg protein, n = 4 in acini).
Effect of LPS treatment on the concentration of IL-1β in macrophages and submandibular cells. Macrophages and cells from submandibular glands of wild-type and P2X7-KO mice were prepared and incubated for 2 h in control conditions or in the presence of 250 ng/ml LPS. The protein and the IL-1β of the various cellular preparations were assayed. Results are mean ± SEM of (n) experiments. Concentration of IL-1β in acinar and ductal suspension from submandibular glands. Ductal and acinar suspensions from submandibular glands of wild-type and P2X7-KO mice were prepared and their IL-1β content assayed. Results are mean ± SEM of (n) experiments.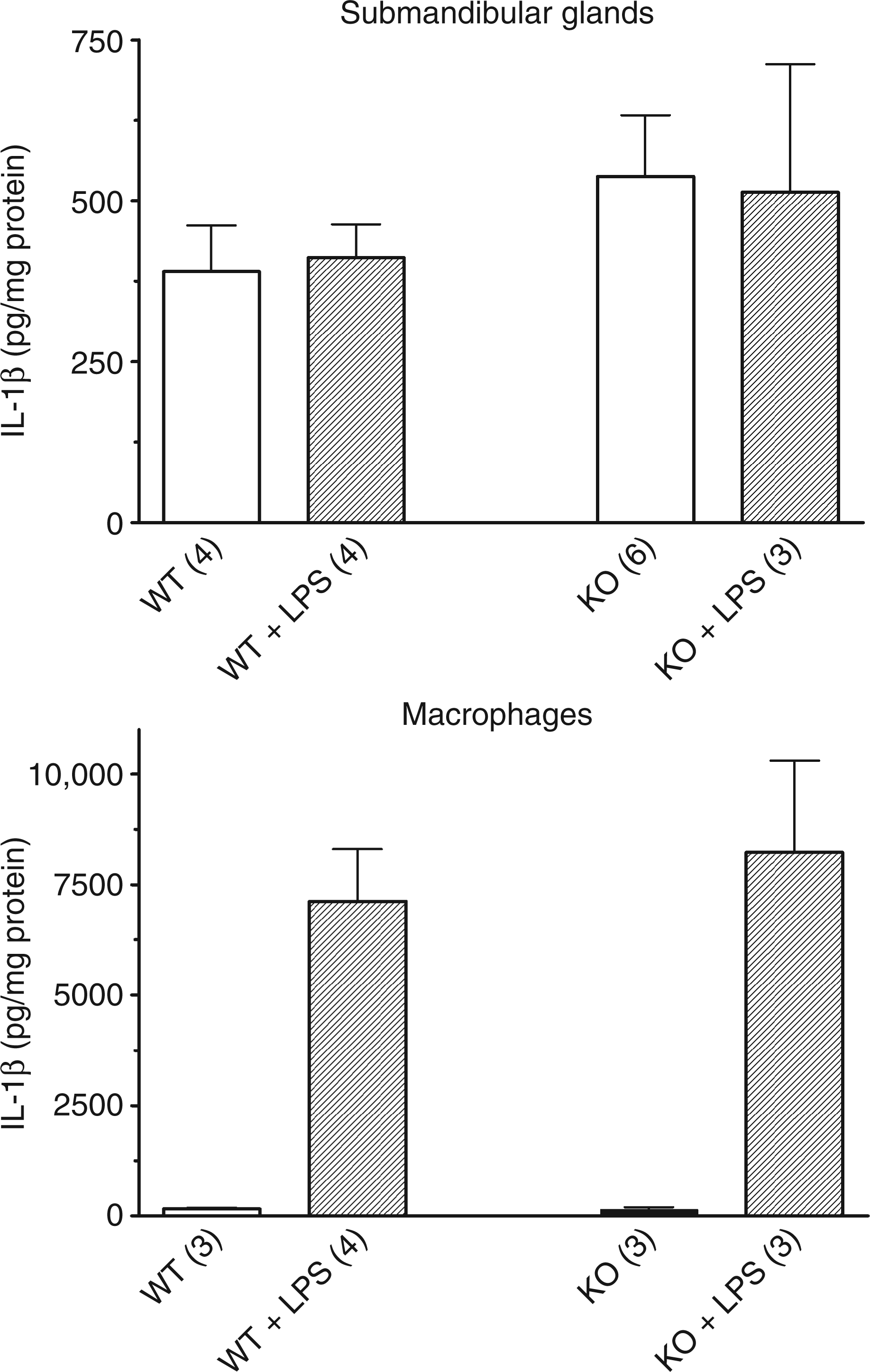
Response of submandibular glands to purinergic agonists
Adenosine triphosphate at a 1 m Effect of ATP and carbachol on the intracellular concentration of calcium of a cellular suspension from submandibular glands of wild-type and P2X7-KO mice. A crude cellular suspension from submandibular glands of wild-type and P2X7-KO mice was prepared and loaded with Fura2. The cells were incubated at 25°C in the presence of 1 m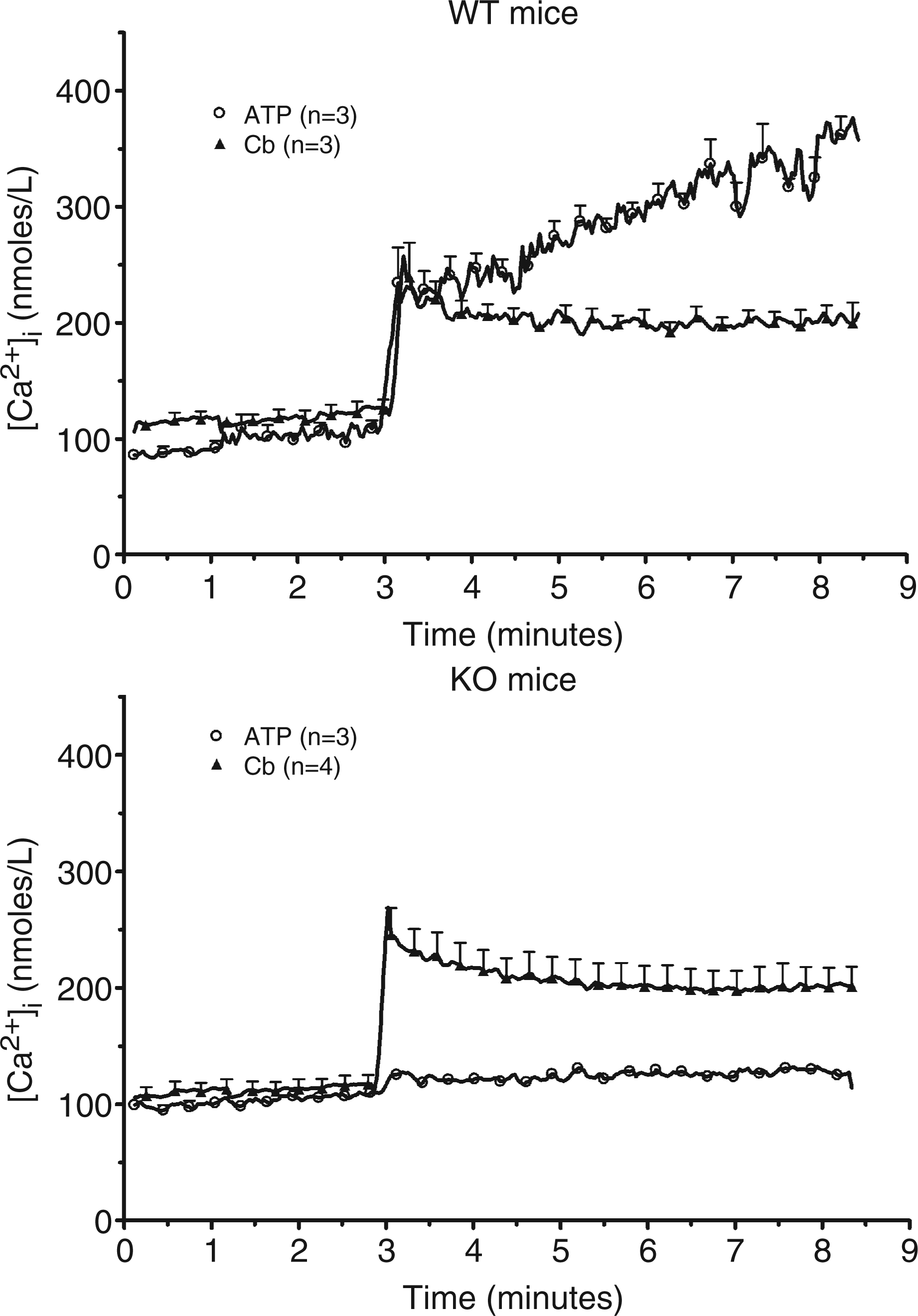
Submandibular cells from wild-type mice were next loaded with PBFI, a fluorescent probe sensitive to potassium. In basal conditions, the fluorescence of PBFI slightly decreased with time (Fig. 5, upper panel). The addition of 1 m Effect of ATP and carbachol on the intracellular concentration of potassium of a cellular suspension from submandibular glands of wild-type mice. A crude cellular suspension from submandibular glands of wild-type mice was prepared and loaded with PBFI. The cells were incubated at 25°C in the presence of 1 m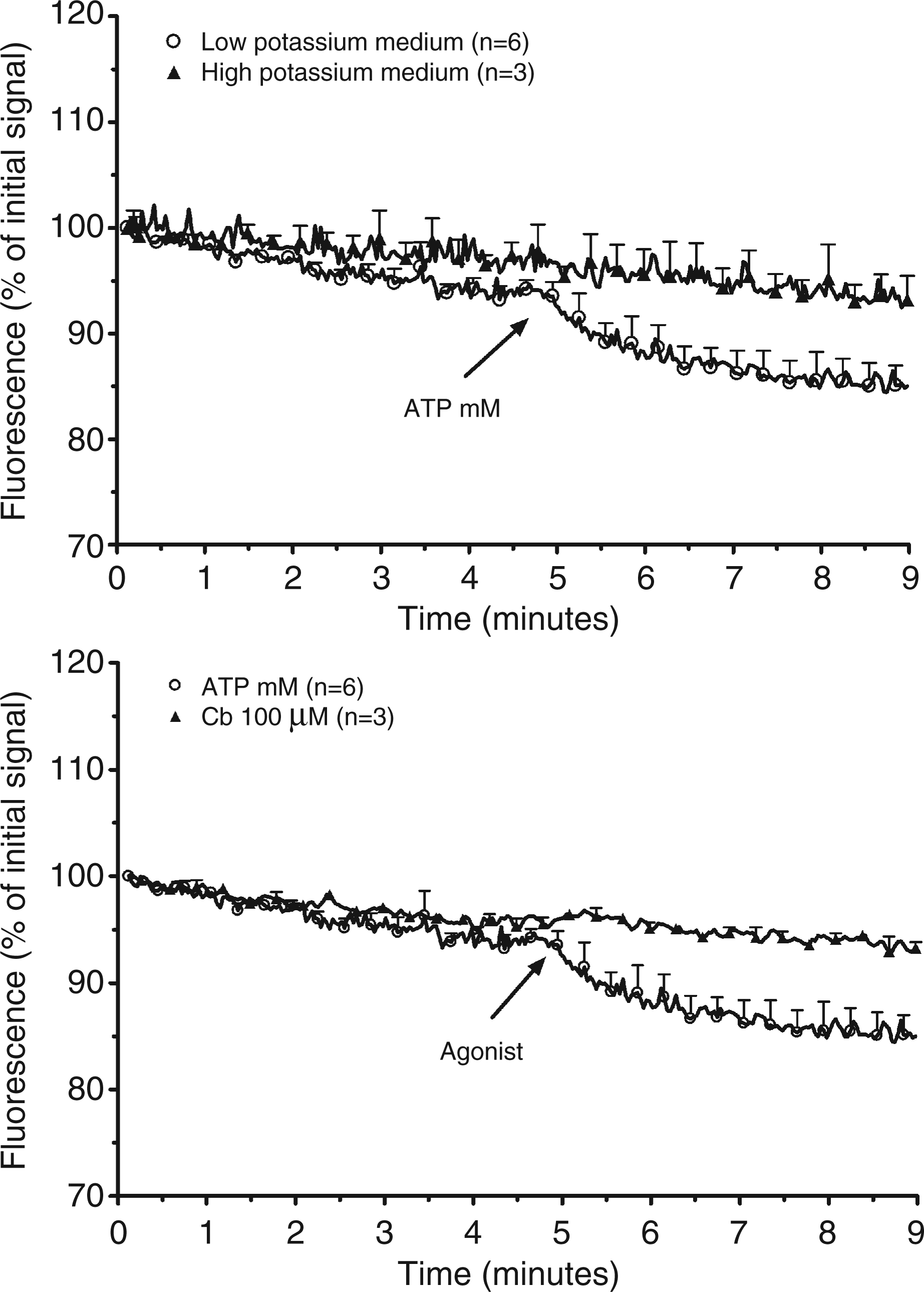
Study of the phosphorylation of IκB
In macrophages, LPS increases the expression of pro-IL-1β by activating NF-κB. The activation and nuclear translocation of NF-κB are triggered by the TLR4 signalling pathway leading to the phosphorylation, the ubiquitination and the degradation of IκB-α, a negative regulator of NF-κB. Immunoblotting was performed to analyze IκB-α phosphorylation. In macrophages, the incubation with LPS resulted in rapid phosphorylation of Ser32 of IκB-α, which peaked at 5 min (Fig. 6). The level of IκB-α in these cells was inversely correlated with its phosphorylation. In submandibular gland cells, LPS did not modify the phosphorylation or the level of IκB-α.
Immunoblot analysis of the phosphorylation of IκB. The phosphorylation of IκB in macrophages (M) and submandibular gland cells (S) was compared by immunoblotting of lysates made at the indicated time points after LPS treatment.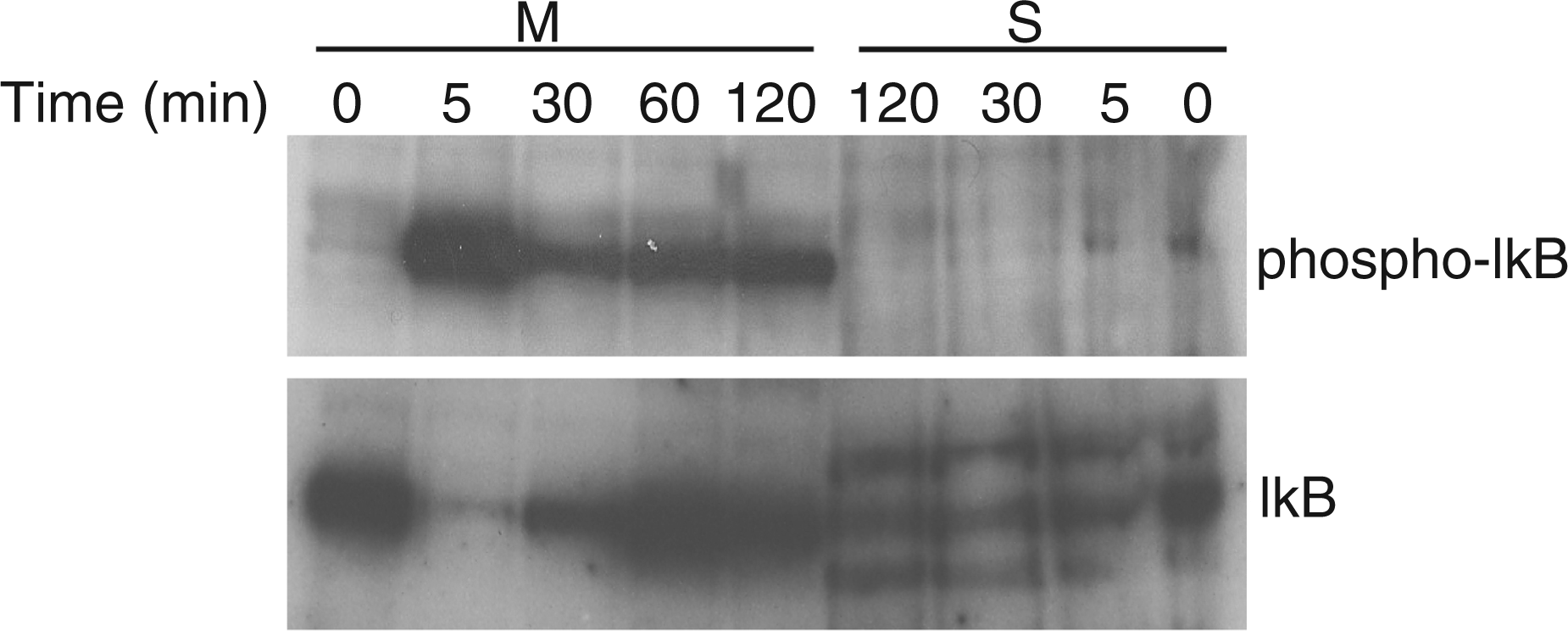
Expression of CD14, TLR4 and MyD88 in submandibular glands
The lack of effect of LPS treatment on the activation of NF-κB and on the expression of IL-1β in submandibular glands, but not in macrophages, prompted us to study the expression of some proteins involved in the response to LPS in both cell populations. CD14 binds the complex formed by LPS and the LPS-binding protein and activates TLR4. This protein interacts with MyD88, the protein which couples TLRs to intracellular pathways. The expression of CD14, TLR4 and MyD88 was investigated by RT-PCR. As shown in Figure 7, the mRNA of the three proteins was present both in macrophages and in submandibular glands. The identity of the mRNAs was confirmed by sequencing the amplified PCR fragments (data not shown).
RT-PCR for pro-IL-1β, TLR4, CD14 and MyD88 mRNA. The mRNA from submandibular glands (S) and macrophages (M) was retrotranscripted and a fragment of the cDNA of pro-IL-1β, TLR4, CD14 and MyD88 amplified with the primers described in Table 1. These results are representative of three experiments.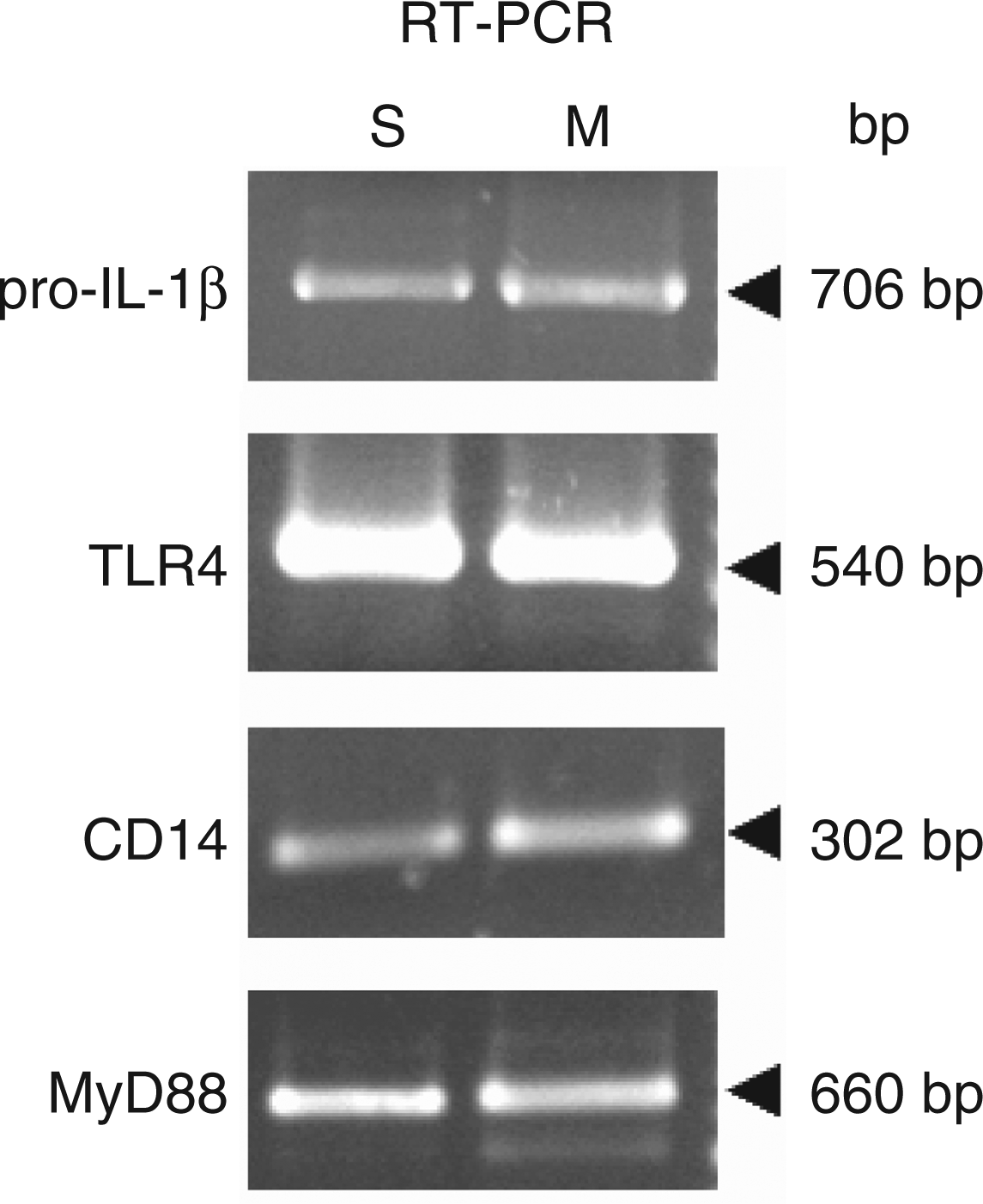
Discussion
The injection of LPS to wild-type mice decreased the volume of saliva. This result is in agreement with previous results showing that the intraperitoneal injection of LPS decreased by more than 50% the metacholine-induced salivary secretion in rat. 17 Conversely, it was reported that retrograde perfusion of LPS in salivary ducts increased the volume of saliva. 18 The different methodological approaches to deliver LPS might account for the discrepancies between these results. Indeed, the intraperitoneal injection of LPS provokes endotoxemia while the perfusion of the endotoxin inside the duct limits the diffusion of LPS and prevents most of its systemic effects. The two models also yield distinct results with respect to protein and amylase secretion. Our results show that saliva from LPS-injected mice contains more proteins and amylase than NaCl-injected animals. Taking into account the lower volume of saliva in LPS-injected animals, the treatment with the endotoxin had no effect on protein and amylase secretion. According to Correia et al., 18 LPS increased secretion from submandibular glands while Barta et al. 19 reported that LPS decreased amylase secretion from parotid acini. The pilocarpine-induced saliva collected from P2X7-KO mice did not differ significantly from control mice. This is consistent with our previous results showing that only the ionic composition of saliva (its potassium concentration) diverged between the two mice. 12 This result is also in agreement with Nakamoto et al., 20 who reported that the disruption of P2X7 receptors nearly abolished the nucleotide-evoked secretion from submandibular glands without affecting salivation in response to muscarinic agonists.
Our results confirmed that IL-1β was present in the saliva collected from mice previously injected with pilocarpine. RT-PCR and immunoblot analysis showed that the cytokine was constitutively expressed by submandibular glands. The basal concentration of IL-1β in submandibular glands was, on a protein basis, higher than in non-stimulated macrophages. Both acini and ducts similarly expressed IL-1β. Unexpectedly, the concentrations of IL-1β in both fractions was higher (2-fold) than the crude suspension. This apparent increase in IL-1β concentration following the purification of a cell population is not unique. Indeed Kauma et al. 21 reported that the isolation of trophoblats from placental villi strongly increased the expression of IL-1β. Another hypothesis would be that during the isolation procedure some antagonist interacting with IL-1β might be cleared. 22 Whatever the reason for this increase, it remains that both ducts and acini similarly express the cytokine. More importantly in basal conditions, the level of IL-1β per milligram of protein was higher in submandibular glands than in macrophages, cells which are major producers of IL-1β.
The exposure of macrophages to LPS increases their expression of IL-1β (this report). 4 A similar treatment of submandibular cells did not alter their expression of the cytokine and injection of LPS to the mice before collecting the saliva did not modify the concentration of the cytokine in saliva. Lipopolysaccharides activate TLR4 after interacting with CD14. MyD88 is the intracellular protein which mediates the activation of TRL4 leading to the activation of NF-κB and, among others, to an increase of IL-1β expression. 23 We showed that LPS induced rapid (within 5 min) phosphorylation of Ser32 of IκB-α and decreased the level of total IκB-α in macrophages. These data are consistent with previous reports that LPS induces IκB phosphorylation and subsequent degradation, thereby leading to NF-κB activation and IL-1β expression. In submandibular gland cells, IκB phosphorylation and degradation in response to LPS were not observed, explaining why the treatment of these cells with LPS did not increase the expression of the cytokine. The absence of activation of NF-κB in submandibular glands might be due to the lack of expression of some transducing proteins. To verify this hypothesis, we analyzed the expression of TLR4, CD14 and MyD88. The mRNA of these proteins could be amplified in extracts from macrophages and submandibular glands. This is in line with previous results reporting the expression of these proteins by salivary glands.24,25 The injection of LPS to C57Bl/6J mice increased the expression of four chemokines (CCL3, CCL5, CXCL2 and CXCL10) but not the expression of CCL2, CCL4, CCL9 and CXCL12. 26 This result confirms the coupling of these proteins to intracellular signalling pathways in submandibular glands. Our results are in line with the results of Uehara et al., 27 who observed that, in response to TLR4 activation, the salivary glands secrete antimicrobial peptides but not pro-inflammatory cytokines.
Macrophages and submandibular glands differ not only with respect to the expression of IL-1β but also to the regulation of its secretion. Adenosine triphosphate (via a P2X7 receptor) and nigericin promote the secretion of IL-1β from macrophages. This response is secondary to the decrease of the intracellular concentration of potassium. The modification of the ionic composition of the cytosol promotes the assembly of the inflammasome, the protein platform responsible for the activation of procaspase-1. 9 The incubation of submandibular glands with a high concentration of ATP had no effect on the release of IL-1β. Yet, the purinergic agonist could increase the intracellular concentration of calcium. The time-course of this response was distinct from the response to carbachol, a muscarinic agonist mimicking the parasympathetic activation of salivary glands. The coupling of the purinergic receptor to a non-selective cation channel was confirmed by the measurement of the intracellular concentration of potassium using PBFI, a potassium-sensitive fluorescent probe. 28 Exposure of submandibular cells to ATP provoked a decrease of the fluorescence of the dye. In spite of these ionic variations, ATP did not increase the secretion of IL-1β.
The concentration of IL-1β in macrophages and in submandibular glands was similar in P2X7-KO and wild-type mice. This suggests that the expression of IL-1β in these tissues is not regulated by P2X7 signalling. This is distinct from previous results showing that the expression of IL-1β was lower in brain and hippocampus of P2X7-KO mice when compared to wild-type mice.29,30 Considering that IL-1β increases its expression in an autocrine way, 4 the low expression level of the cytokine in KO mice might be secondary to the impaired secretion of the cytokine in this animal model.
The results presented in this work are at variance with recent results of Yao et al. 14 They reported that IL-1β was expressed by submandibular glands from C3H/HeN mice. The cytokine was located in secretory granules where it was processed by mK3, a kallikrein isoform present in secretory granules of granular convoluted cells. 31 This is rather unexpected considering that pro-interleukin is a leaderless protein which is located, at least in inflammatory cells, in the cytosol where it is eventually processed by the inflammasome before its secretion. Injection of LPS to C3H/HeN mice but not to C3H/HeJ mice, a mutant strain for TLR4 receptors, increased the expression of IL-1β. 14 These authors used not only another strain of mice (C3H/HeN versus C57Bl/6J in our work) but also LPS from other source. They stimulated the cells with LPS from E. coli O111-B4 and we used LPS from E. coli O55-B5. Koyama et al. 32 reported that the LPS from the two strains differently affected lung cells. These differences might account for the discrepancy between the results of Yao et al. 14 and our study.
Conclusions
We have shown that IL-1β is constitutively expressed by submandibular glands at a basal level higher than found in cells like macrophages confirming the importance of salivary glands in oral immunology. The expression of the cytokine is not affected by stimulation of the cells by LPS and its secretion is not regulated by purinergic agonists. IL-1β stimulates the secretion of growth factors by gingival fibroblasts which promote the proliferation of epithelial cells. 33 Its constitutive expression and secretion by salivary glands might contribute to the integrity of the oral mucosa.
Footnotes
Acknowledgements
M. El Ouaaliti is a Research Fellow from the FRIA. The authors thank Mr C. Zelbouni for his skilful technical help and Pr N. Verbruggen for giving access to the NanoDrop spectrophotometer.
Funding
This work was supported by grant 3.4.528.07.F from the Fonds National de la Recherche Scientifique to S. Pochet and J.P. Dehaye and by institutional grants.
