Abstract
The purpose of this study was to assess the effect of dexmedetomidine as a local anaesthetic adjuvant on flap blood flow and patient satisfaction regarding postoperative analgesia anterolateral thigh free flap transfer for hand trauma. A total of 66 patients were randomized into two groups. One group was treated with intermittent 1.0 µg/mL dexmedetomidine + 0.125% ropivacaine bolus at 8 mL/2 h and the other with intermittent 0.2% ropivacaine bolus at 8 mL/2 h, administered via continuous infraclavicular brachial plexus block. Dexmedetomidine had no significant effect on the flap blood supply and had a good safety profile. Moreover, 1.0 µg/mL dexmedetomidine + 0.125% ropivacaine provided similar satisfactory postoperative analgesia as 0.2% ropivacaine (p > 0.05), reduced hand numbness and motor block, and improved patient satisfaction (p < 0.05). All the flaps in the dexmedetomidine + ropivacaine group and 30 of 31 flaps in the ropivacaine group survived. The addition of dexmedetomidine to the local anaesthetic infusion appears to be a safe method of improving postoperative symptoms.
Level of evidence:
I
Keywords
Introduction
The anterolateral thigh (ALT) flap is an effective method for repairing large skin and soft tissue defects (Atilgan et al., 2023). However, postoperative pain may be associated with increased sympathetic tone, vasospasm and microsurgical collapse, to the detriment of surgical success (Ruan et al., 2018). Therefore, implementing effective postoperative analgesia is very important for patients who are receiving ALT-free flaps.
Continuous infraclavicular brachial plexus block (CIBPB), a peripheral nerve blockade technique, not only has a precise postoperative analgesic effect on the hand injury but also carries few adverse effects, such as nausea, vomiting, drowsiness and respiratory depression (Ilfeld, 2017). However, CIBPB still has adverse effects on the symptoms of numbness and discomfort in the affected limbs and the inhibition of motor function, these effects being closely related to the concentration of local anaesthetic. These disadvantages strongly affect patient satisfaction with postoperative analgesia. The goal of postoperative analgesia is to minimize the inhibition of sensory and motor functions while maintaining effective analgesia.
Dexmedetomidine, a highly selective α-2 receptor agonist, conveys a synergistic nerve blocking effect when used in combination with ropivacaine, which manifests as an enhanced analgesic effect and prolonged action of the local anaesthetic (El-Boghdadly et al., 2017; Vorobeichik et al., 2017). Therefore, the pharmacological effect of dexmedetomidine combined with CIBPB can effectively reduce the concentration of local anaesthetics (Abdallah et al., 2016; Hu et al., 2017). However, flap transfer surgery is particularly dependent on flap blood flow, so the possible effects of dexmedetomidine on blood flow to the flap must be considered before its use.
A previous study showed that the contractile or dilative effect of dexmedetomidine on peripheral blood vessels depends on the dose administered. This action can occur at low doses and may lead to dilation of peripheral blood vessels, while high doses may lead to vasoconstriction (Kamibayashi and Maze, 2000). Therefore, in theory, application of local anaesthetic with low doses of dexmedetomidine should not have a negative effect on flap blood vessels or blood flow. However, due to a lack of relevant studies on the application of dexmedetomidine for postoperative analgesia after flap transfer, the effect of dexmedetomidine on flap blood flow is unclear.
In this study, we assessed the effect of dexmedetomidine as a local anaesthetic adjuvant on flap blood flow and patient satisfaction regarding postoperative analgesia anterolateral thigh free flap transfer for hand trauma. These results may provide a reference for the clinical formulation of appropriate postoperative analgesia regimens for hand injury patients undergoing ALT-free flap surgery.
Methods
The study was conducted at Ningbo No. 6 Hospital, Ningbo, Zhejiang Province, China between November 2017 and September 2023 and was approved by the hospital’s Ethical Review Board. Written informed consent was obtained from all patients participating in the trial in accordance with the code of the Declaration of Helsinki.
In total, 66 patients were enrolled who were scheduled to undergo ALT-free flap repair of skin and soft tissue defects on the hand and agreed to have an infraclavicular catheter placed for postoperative analgesia. The inclusion criteria were age 20–50 years, American Society of Anaesthesiologists (ASA) grade I–II, and able to understand and cooperate during operation under nerve block and postoperative evaluation. The exclusion criteria were contraindications to peripheral nerve block (study drug allergy, coagulopathy or local infection), refusal of the nerve block procedure, severe cardiovascular disease, atrioventricular block or bradycardia, severe obesity or body mass index (BMI) >35 kg/m2, recent chronic use of opioids, a history of phrenic paralysis or phrenic nerve injury, moderate or severe pulmonary disease, chest deformity (pectus carinatum or pectus excavatum), or severe renal or hepatic failure.
Randomization and blinding
The 66 patients who met the enrolment criteria were randomized into two groups using the double-blind random number table method (see the CONSORT checklist in the supplementary material). The DR group was treated with an intermittent 1.0 µg/mL dexmedetomidine + 0.125% ropivacaine (Naropin; AstraZeneca AB) bolus at 8 mL/2 h. The R group was treated with an intermittent 0.2% ropivacaine bolus at 8 mL/2 h. All drugs were delivered using a PCA pump (LY-E; Zhangjiagang Longyi Medical Equipment Co., Ltd., Jiangsu, China). The patients and other anaesthetists were blinded to the infusion regimens.
Anaesthetic management
None of the patients received premedication. The patients were continuously monitored by electrocardiography, pulse oximetry and blood pressure measurements. Oxygen was applied at a flow rate of 3 L/min. After insertion of a venous cannula, 1 mg of midazolam was injected intravenously. The same anaesthetist performed the CIBPB and epidural anaesthesia in both groups. The patient was placed in the supine position, with a 5–10 cm high shoulder cushion on the affected side, and both hands were placed beside the body. After sterile skin preparation with povidone iodine and dermal injection with 2% lidocaine, a high-frequency linear array transducer probe (6–13 MHz; SonoSite M-Turbo, USA) protected by a sterile dressing was placed inferior to the clavicle, and the probe angle and scanning depth were adjusted to obtain a clear cross-sectional image of the infraclavicular brachial plexus. The insertion point was injected with 2% lidocaine. Using the in-plane technique, a 19 G needle from the continuous brachial plexus block package (StimuLong NanoLine; PAJUNK GmbH, Geisingen, Germany) was inserted from the lateral long axis of the probe. Fifteen mL of 0.375% ropivacaine was injected when the tip was above the brachial plexus. The direction and angle were adjusted, the tip of the needle was placed between the posterior cord and the axillary artery, and a further 15 mL of 0.375% ropivacaine was injected (Figure 1(a)). Then, a 20 G perineural catheter was inserted through the needle and advanced to a depth of 1 cm beyond the needle tip between the posterior cord of the brachial plexus and the axillary artery (Figure 1(b)). The position of the catheter head was observed under ultrasound; if the catheter position was satisfactory and no blood was drawn, the catheter was fixed to the skin (Figure 1(c, d)).
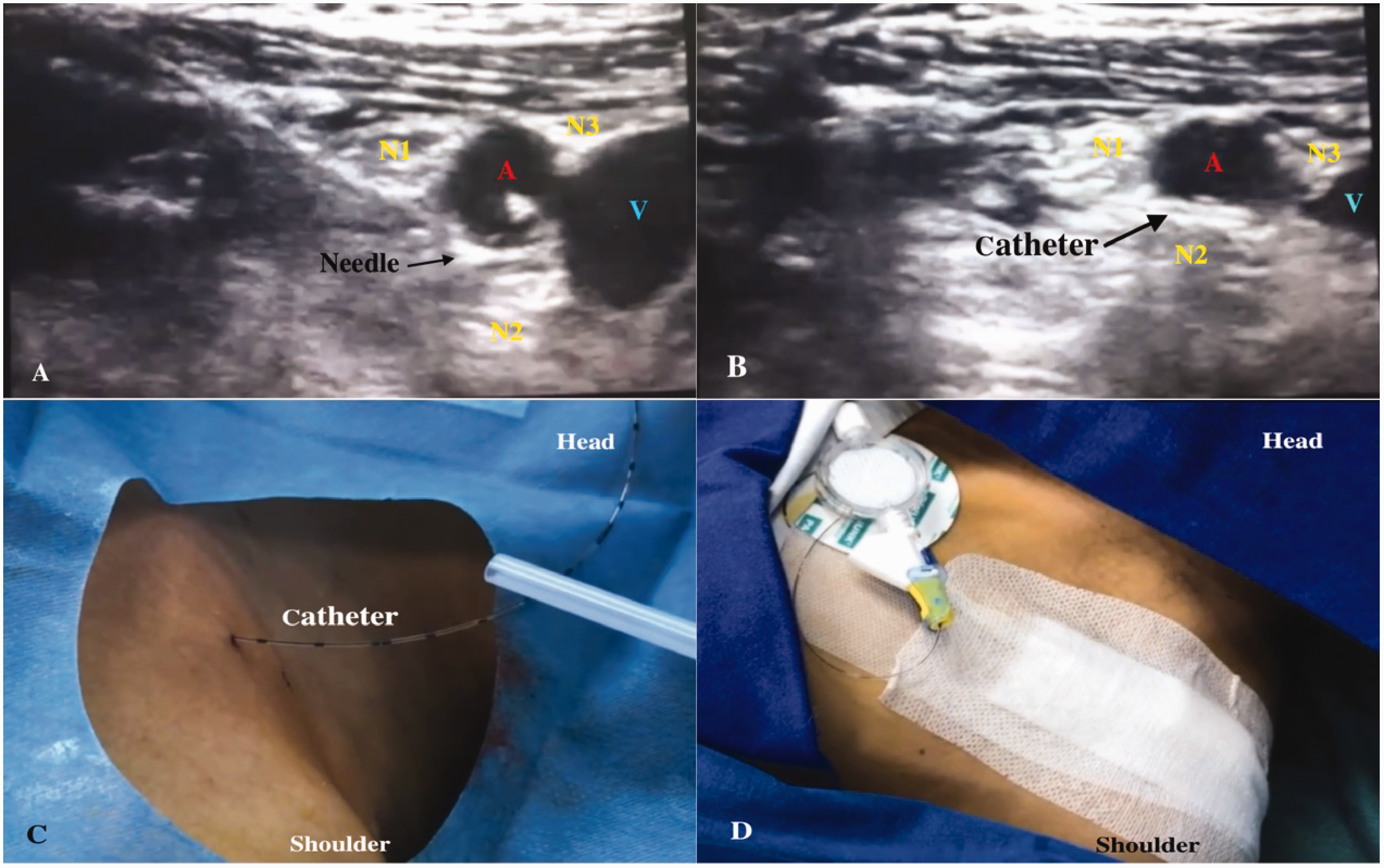
(a) Nerve block needle is placed around the brachial plexus and injected local anaesthetic. (b) A catheter was placed between posterior cord of brachial plexus and the axillary artery and (c, d) the catheter is secured to the skin surface. A: axillary artery; N1: lateral cord of brachial plexus; N2: posterior cord of brachial plexus; N3: medial cord of brachial plexus; V: axillary vein.
After the operation, a patient-controlled analgesia (PCA) pump was connected to the infraclavicular catheter. A programmed intermittent bolus (PIB) was used for postoperative analgesia. The PCA pump used contained 1.0 µg/mL dexmedetomidine +0.125% ropivacaine in the DR group and 0.2% ropivacaine in the R group. The PCA pump volume in each group was 200 mL and the intermittent bolus size was 8 mL/2 h.
The anaesthetic method for obtaining a free flap from the thigh was continuous epidural block into the L3–4 intervertebral space. After the operation, the epidural catheter was retained, and 0.2% ropivacaine was given for postoperative controlled epidural analgesia (PCEA); the background infusion rate was 4 mL/h, the dosage of PCEA was 2 mL and the locking time was 15 min. Parecoxib-Na (Dynastat®; Pfizer Co., Ltd.) 40 mg was intravenously injected (followed by 20 mg or 40 mg at 6–12-hour intervals as needed), and the PCEA pump was pressed to remediate the pain imparted by the hand and the site of the free thigh flap.
The patients were instructed to maintain a postoperative analgesic static visual analogue scale (VAS) score ≤4. The duration of postoperative analgesia in this study was 48 hours, after which the CIBPB and epidural catheter were removed.
Outcome assessment
A group of two trained anaesthetists and a hand surgeon blinded to group allocation carried out the outcome assessments and collected all of the outcome data.
The primary outcome was blood flow signals from the intrinsic artery of the flap: peak systolic blood flow velocity (Vs), diastolic blood flow velocity (Vd), mean blood flow velocity (Vm) [Vm = (Vs-VD *2)/3], resistance index (RI) [RI = (Vs-Vd)/Vs] and arterial diameter (AD); the average of the three measurements for each parameter was taken. These parameters were measured using ultrasound immediately (0) and at 12, 24 and 48 hours postoperatively. If the patient’s resting VAS score was ≤4 points, the affected limb lay flat on the side of the bed at a 30° angle to the body during measurements.
The secondary outcomes were patient satisfaction, numbness and motor block scores. Patient satisfaction with the analgesia was evaluated using a 5-point Likert scale (1 = very dissatisfied; 2 = dissatisfied; 3 = neutral; 4 = satisfied; 5 = very satisfied) (Jebb et al., 2021). Numbness was assessed by using a modified Barrow Neurological Institute (BNI) numbness score (I = no numbness; II = mild numbness, not bothersome; III = numbness, somewhat bothersome; IV = numbness, very bothersome) (Rogers et al., 2000). We asked the patient to evaluate the numbness in their hand, forearm and upper arm and recorded the highest numbness score for analysis. Motor blockade was evaluated using a modified Bromage scale for the upper extremities on a 3-point scale (grade 1 = able to flex the elbow and move the fingers; grade 2 = unable to flex the elbow but able to move the fingers; grade 3 = inability to move the elbow or fingers) (Kim et al., 2022; Sane et al., 2021).
The cumulative parecoxib-Na consumption and pain VAS score were recorded over the 48 hours after surgery as the third outcome. Postoperative pain was assessed using a 10-point VAS at 12, 24 and 48 hours after surgery.
Adverse reactions, such as haemodynamic indices (hypotension and bradycardia), local haematoma, local infection, nausea, vomiting, persistent upper limb paraesthesia or weakness, and respiratory depression, were recorded.
Statistical analysis
The sample size for this study was determined based on the satisfaction rates (the percentage of the sum of very satisfied and satisfied individuals) calculated from preliminary experiments, which indicated an 80% satisfaction rate for the DR group and a 40% satisfaction rate for the R group. A two-sided test with a significance level of α = 0.05 and a power of 80% (β = 0.2) was utilized, with an equal allocation ratio of 1:1 between the DR and R groups. Considering a 20% dropout rate, the sample size was 62 patients, with 31 participants in each group.
The normality of the data was tested using the Shapiro–Wilk test. The normally distributed continuous variables are presented as the means (SD, 95% CI) and were compared between two groups using Student’s t-test. The non-normally distributed continuous variables are presented as medians with interquartile ranges (IQRs) and were further analysed using the Mann–Whitney U or Kruskal–Wallis H test. The categorical variables are expressed as frequencies and percentages and were compared among groups using the chi-squared test. A two-sided p-value <0.05 was considered to indicate statistical significance.
Results
A total of 84 patients were screened for recruitment; however, 14 declined to participate and four did not meet the inclusion criteria. In total, 66 patients were randomly divided into two groups (33 in each group), and the primary outcome data of 60 patients were obtained (Figure 2). The catheter dislodged in four patients and two patients experienced catheter leakage; therefore, outcomes could not be collected for these six patients. There was no significant difference in demographics, injury, or flap characteristics or operation time between the two groups (p > 0.05) (Table 1).
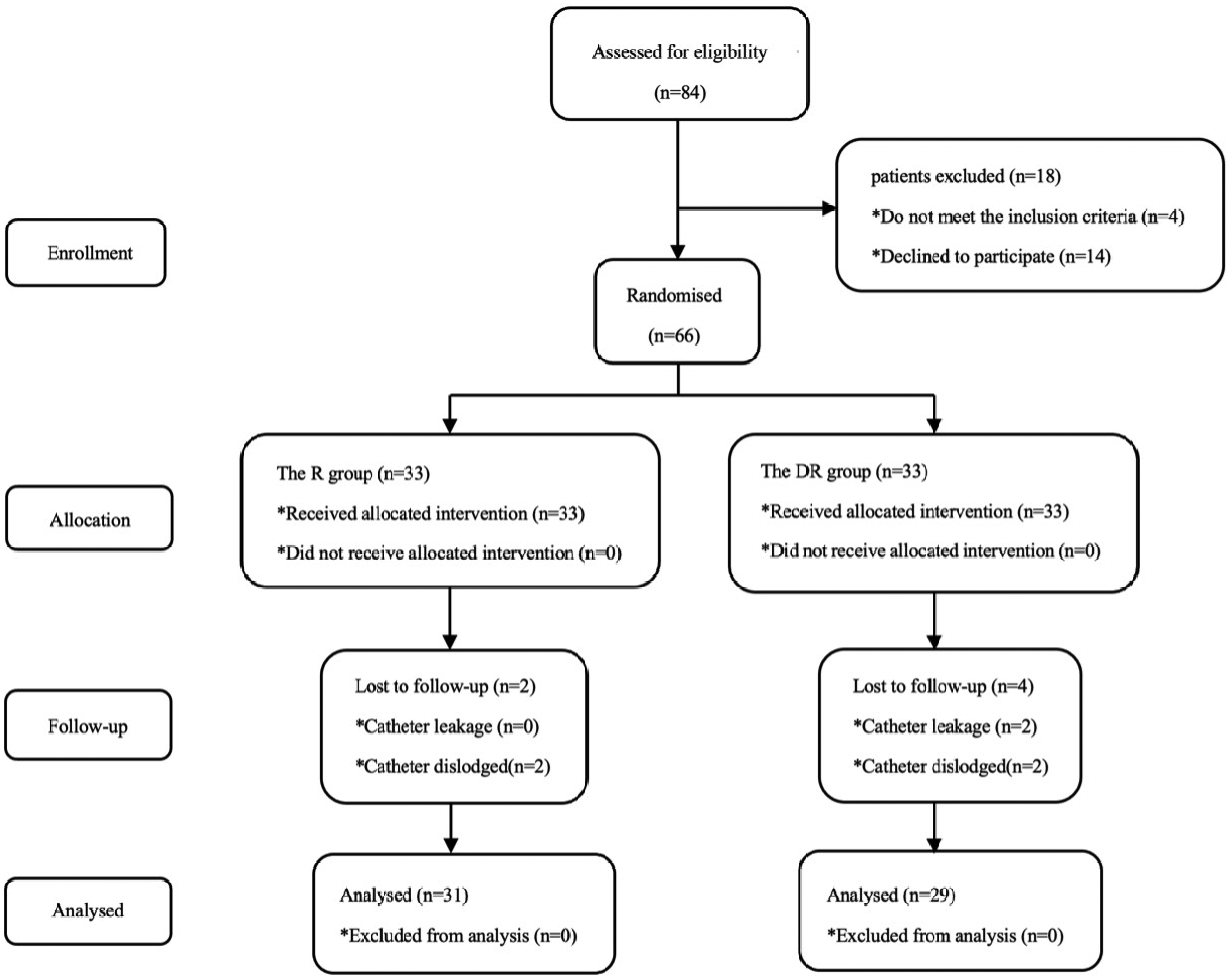
Flow diagram of the study.
Demographic and clinical characteristics of the patients, injuries and flaps.
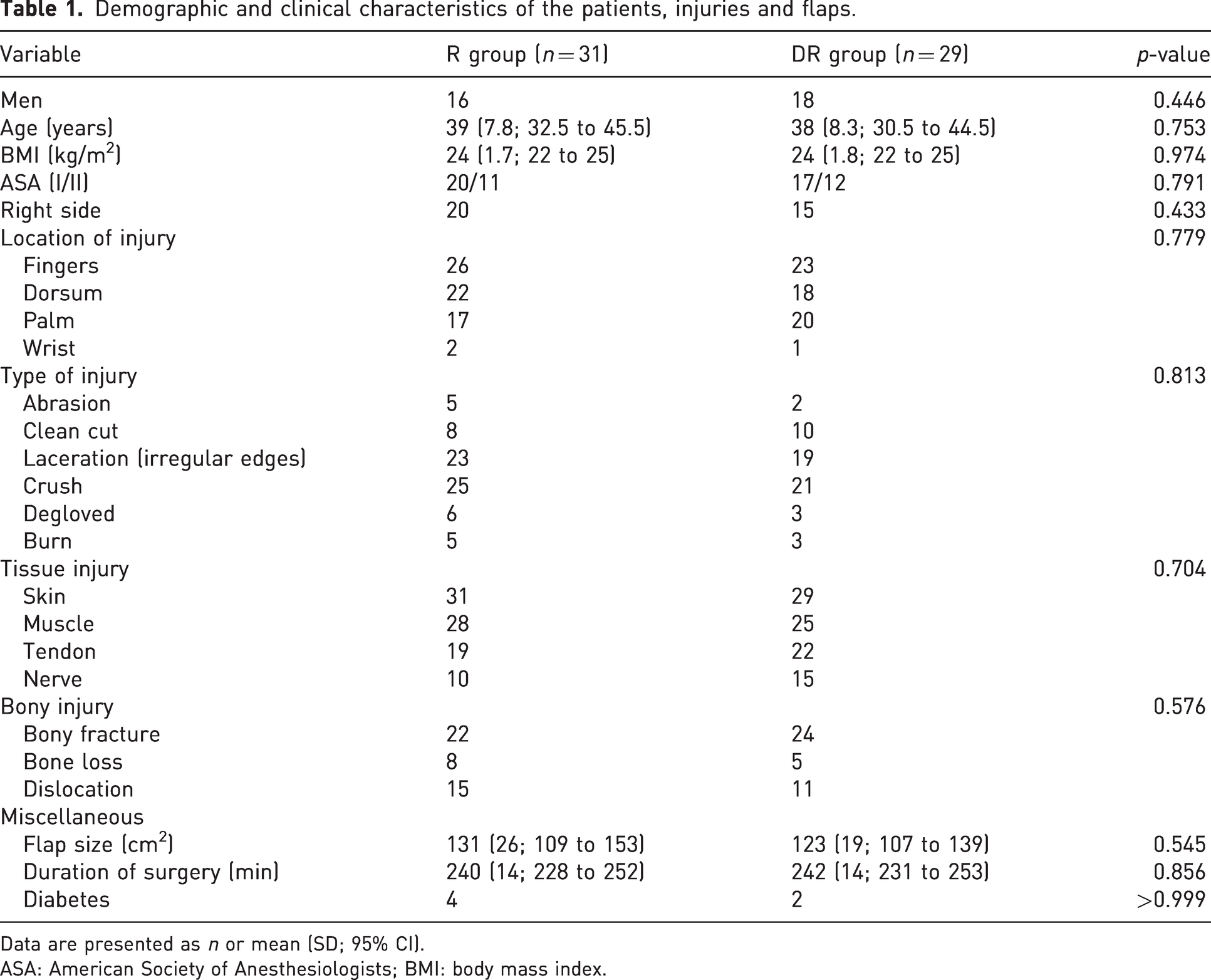
Data are presented as n or mean (SD; 95% CI).
ASA: American Society of Anesthesiologists; BMI: body mass index.
Blood flow signals from the intrinsic artery of the flap
There were no significant differences in the arterial diameter (AD), resistance index (RI) or average blood flow velocity (Vm) between the two groups at any of the postoperative time points (Table 2).
Comparison of flap blood flow parameters between the two groups after surgery.

Data are presented as mean (SD; 95% CI). Expand RI by a factor of 10 to facilitate data processing.
0 h: immediately postoperatively; AD, arterial diameter; RI, resistance index; Vm, mean blood flow velocity.
Postoperative satisfaction and analgesia
The satisfaction with postoperative analgesia in the DR group was significantly greater than that in the R group (p = 0.036) (Table 3). Group R had significantly higher modified Barrow Neurological Institute numbness scores at 24 and 48 hours after surgery than the DR group (Table S1 in the supplementary material). Group R also had a significantly greater modified Bromage scale for motor blockade than the DR group (Table S2). There were no differences in cumulative parecoxib-Na consumption or pain VAS between the two groups (p > 0.05) (Table 4).
Patient satisfaction with postoperative pain management.
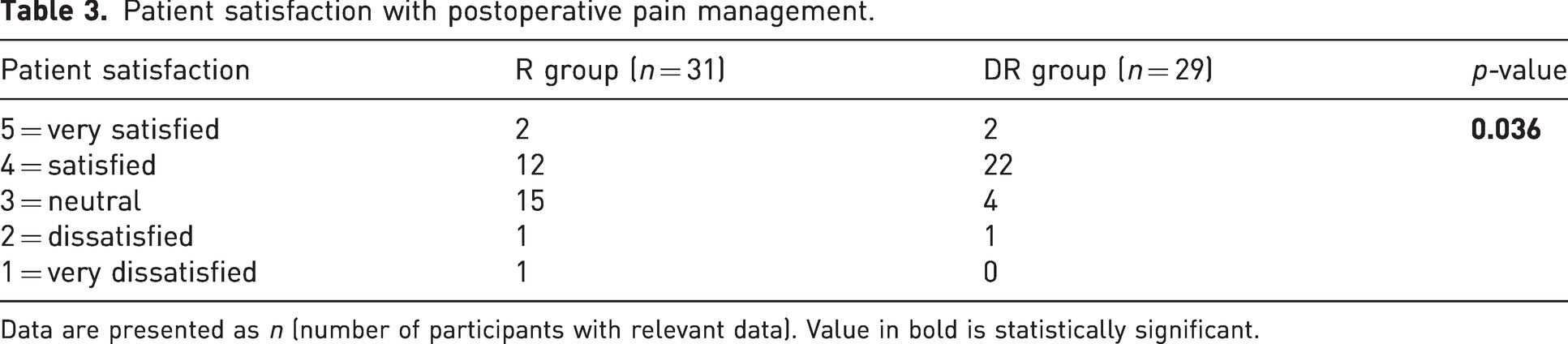
Data are presented as n (number of participants with relevant data). Value in bold is statistically significant.
VAS scores and cumulative parecoxib-Na consumption after surgery.
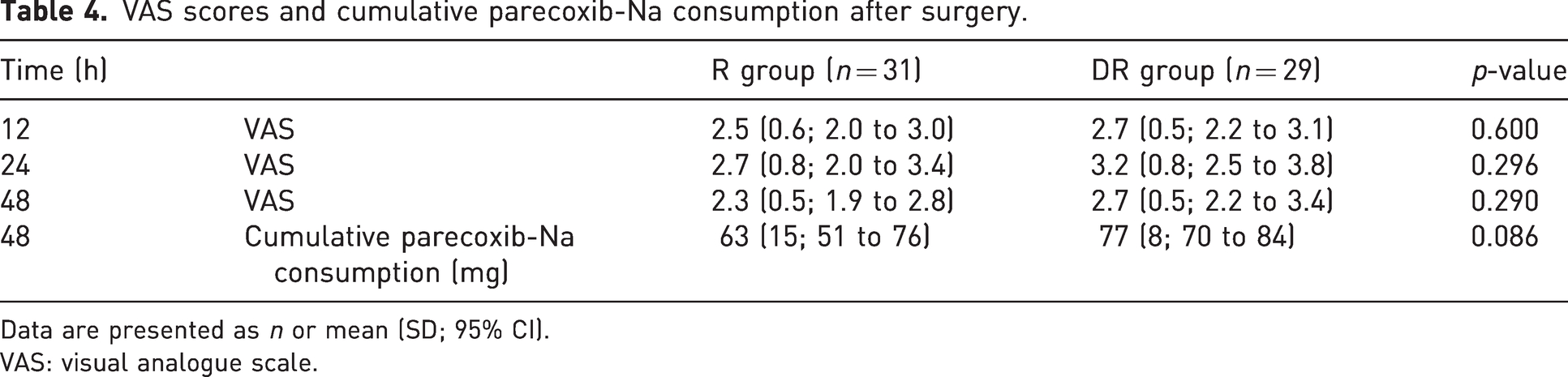
Data are presented as n or mean (SD; 95% CI).
VAS: visual analogue scale.
Adverse events
Two patients in the DR group developed bradycardia and hypotension, and one patient developed postoperative nausea and vomiting. In the R group, one patient developed bradycardia, and one patient developed postoperative nausea and vomiting. No other adverse events occurred. There were no significant differences in the incidence of adverse events between the two groups (p > 0.05).
Clinical outcomes
Within 48 hours after surgery, three of the 31 patients in the R group developed flap vascular crisis and underwent flap vascular exploration. One of the patients underwent three explorations and the vascular crisis was not resolved, so the patient was treated with free skin grafting. Of 29 patients in the DR group, two developed flap vascular crisis and underwent flap vascular exploration. The difference between the two groups was not statistically significant. After 2 weeks, 30 of the 31 flaps from patients in the R group and all the flaps in the DR group were alive.
Discussion
In this study, the flap blood flow parameters did not significantly differ between the R group and the DR group. Correspondingly, there was no significant difference in flap survival between the two groups. These results suggest that postoperative analgesia via CIBPB and programmed intermittent bolus with dexmedetomidine as a local anaesthetic adjuvant has no adverse effect on flap blood supply. Previous research has shown that the effect of dexmedetomidine on the dilation or constriction of peripheral blood vessels is dose-dependent (Kamibayashi and Maze, 2000). This may explain why the small dose of dexmedetomidine in our study did not significantly affect the blood supply of the flap. However, determining whether the use of dexmedetomidine is beneficial to patients was another important aim of our study.
Reviewing the study results, we found that the postoperative analgesic effect of 0.2% ropivacaine could be achieved by administering 1 µg/mL dexmedetomidine + 0.125% ropivacaine through the programmed intermittent bolus method via CIBPB, and all patients received effective analgesia. By reducing the concentration of local anaesthetics, the analgesia satisfaction was improved and the numbness and motor block were effectively reduced. Some studies have shown that the prolonged motor block associated with local anaesthetic dosage may cause patient dissatisfaction (Fredrickson and Price, 2009; Fredrickson et al., 2010). In our study, numbness and motor block were reduced, which may be the main reason for the improved patient satisfaction. In addition, there was no significant difference in the consumption of parecoxib-Na between the groups.
Although dexmedetomidine may be beneficial to improve patient satisfaction, its adverse reactions also need to be considered. The results showed that dexmedetomidine-related adverse reactions, such as bradycardia and hypotension, were not significantly greater in the DR group than in the R group. However, previous studies have shown that the use of dexmedetomidine as an adjuvant in a single nerve block can cause a significant decrease in blood pressure and a slowed heart rate (Abdallah et al., 2016; Bharti et al., 2015; Lee et al., 2016; Singh et al., 2016). This may be because the dose per unit time of dexmedetomidine in a single nerve block is much greater than that in our study. Relevant research showed that more than 50–60 µg of dexmedetomidine in a single nerve block may cause hypotension and bradycardia (Vorobeichik et al., 2017). The dose of dexmedetomidine administered per unit time in our study (4 µg/h) was much lower than 50 µg. A smaller dosage should avoid the occurrence of adverse drug reactions to a great extent.
Two patients in the DR group developed hypotension and bradycardia but did not experience a significant decrease in flap blood flow. This is likely because dexmedetomidine may maintain flap blood flow without the significant effects of hypotension or bradycardia by reducing the vascular tension of the flap and inhibiting upper limb ischaemia–reperfusion injury (Chikuda and Sato, 2019; Yagmurdur et al., 2008; Yamaguchi et al., 2023). However, this hypothesis needs to be verified by further research.
Our study also has some limitations. First, due to the lack of accuracy of ultrasound detection, we could not measure the change in the artery diameter of the flap accurately. Second, the sample size was small, and the results can only provide a clinical reference. Third, the number of patients with diabetes and hypertension was low in the study population. The results of this study may not be applicable to that population. Fourth, patients in this study had different types, location and tissue of injury. This heterogeneity may affect the accuracy of the results. In addition, we only measured numbness below the upper arm, without emphasizing more accurate location. The mean size of the flaps was relatively small (120–130 cm2). These defects may make the results biased or not have wide applicability. Fifth, the mechanism by which dexmedetomidine affects peripheral nerves is still unclear. Animal experiments have shown that the analgesic effect of dexmedetomidine on peripheral nerves may be due to blockade of the hyperpolarization-activated cation (Ih) current, thus preventing the subsequent action potentials (Brummett et al., 2011).
In conclusion, dexmedetomidine, as an adjuvant in combination with ropivacaine, had no significant effect on the blood supply of the ALT free flaps during postoperative analgesia. Moreover, this method can effectively improve patient satisfaction and has no significant adverse effects, such as hypotension or bradycardia.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Zhejiang Ningbo Yinzhou district agricultural and social development science and technology project (reference no. 2018YZQ02029) and Zhejiang province medical and health science and technology projects (reference no. 2017KY141).
Ethical approval
The study was approved by the Ethical Review Board of Ningbo No. 6 Hospital (reference no. 2017010).
Informed consent
Written informed consent was obtained from all patients participating in the trial, in accordance with the code of the Declaration of Helsinki.
Supplemental material
Supplemental material for this article is available online.
