Abstract
We retrospectively reviewed our series of primary total wrist arthroplasty with the Freedom® prosthesis. The primary outcome measure was revision, and secondary measures included radiographic loosening, pain, complications, movement range, grip strength and patient-reported measures. We reviewed 12 implants in 11 patients (mean age 59 years, range 45–80) with a mean radiological and clinical follow-up of 2.7 and 3 years, respectively. One radial component failed to integrate and was revised at Day 84. Four carpal components demonstrated areas of lucency. There was a statistically significant reduction in pain, and total flexion–extension increased. Despite high patient satisfaction on a ten-point visual analogue scale score (mean 8.7 out of 10), the mean patient-rated wrist evaluation, Quick Disabilities of the Arm, Shoulder and Hand and Patient Evaluation Measure scores were 52, 55 and 53, respectively. The Freedom® implant reduced pain and preserved wrist movement in our patients; however, annual surveillance is recommended due to the high incidence of early carpal component lucency.
Introduction
Total wrist arthroplasty (TWA) offers an alternative to arthrodesis for end-stage arthritis of the wrist. Arthroplasty may be especially appreciated in patients who have previously undergone total wrist fusion (TWF) on the contralateral side or in systemic inflammatory conditions with multiple joint involvement. TWA designs have evolved since the first dual component implants, and fourth generation designs have been shown to reduce pain and improve function and grip strength while providing some movement (Berber et al., 2018). The Freedom® TWA system (Integra LifeSciences, Princeton, NJ, USA, and Smith & Nephew, London, UK) is an unconstrained prosthesis that replaces the fourth generation Universal 2 implant system designed by Adams (KMI, San Diego, CA, USA). The enhanced features of the Freedom® implant include improved anatomic geometry of the radial component, locking screws for the carpal component and an ultra-high-molecular-weight polyethylene (UHMWPE) carpal component insert that is highly cross-linked. The Freedom® UHMWPE therefore has enhanced wear characteristics compared with its predecessor. The insert attaches to a cobalt–chromium–molybdenum alloy carpal component that includes a hydroxyapatite-coated central peg and two locking screws. The titanium radial component comprises a polished concave base plate and a porous-coated stem. No medium- or long-term case series for the Freedom® implant have been published, and short-term outcome data appears to be restricted to a single published article by Rossello et al. (2022).
The purpose of this study was to evaluate the short-term radiographic and clinical outcomes of the Freedom® TWA system when used to treat inflammatory and non-inflammatory wrist arthritis, including an assessment of radiographic loosening, pain, complications, movement range, grip strength and patient-reported measures.
Methods
We retrospectively identified all patients who had undergone primary TWA with the Freedom® implant with greater than 2 years of follow-up. All cases were performed by the two senior authors (ST and MH) at our tertiary orthopaedic hand unit between 2018 and 2019 for a minimum follow-up of 2 years (Tang et al., 2019). Both surgeons have a Level 4 of expertise according to Tang and Giddins (2016). Preoperative data were collected by the senior authors ST or MH (or their senior fellow or registrar) during routine outpatient clinics, including documentation in consultation letters and patient-reported outcome measure (PROM) questionnaires. Postoperative data were collected at a dedicated research facility, including in-person and virtual consultations (MB and RW). Our primary outcome measure was revision. Our secondary outcome measures were radiographic loosening, pain (visual analogue scale (VAS) scores from 0 to 10, with 10 being the worst), complications, range of motion (wrist flexion and extension), grip strength and PROMs. The study was registered and performed according to the research requirements of Wrightington Hospital, and patient consent was not required.
Radiographic assessment was performed preoperatively and at 6 and 12 months postoperatively and annually thereafter. The COVID-19 pandemic interrupted patient follow-up, and a research facility visit permitted final postoperative assessments. Arthroplasties were assessed for periprosthetic loosening according to the Wrightington zonal system (Badge et al., 2016) (Figure 1). Each zone was assessed (MB, ST, AK), and lucencies of 1 mm or greater were recorded according to the technique described by Cobb and Beckenbaugh (1996).
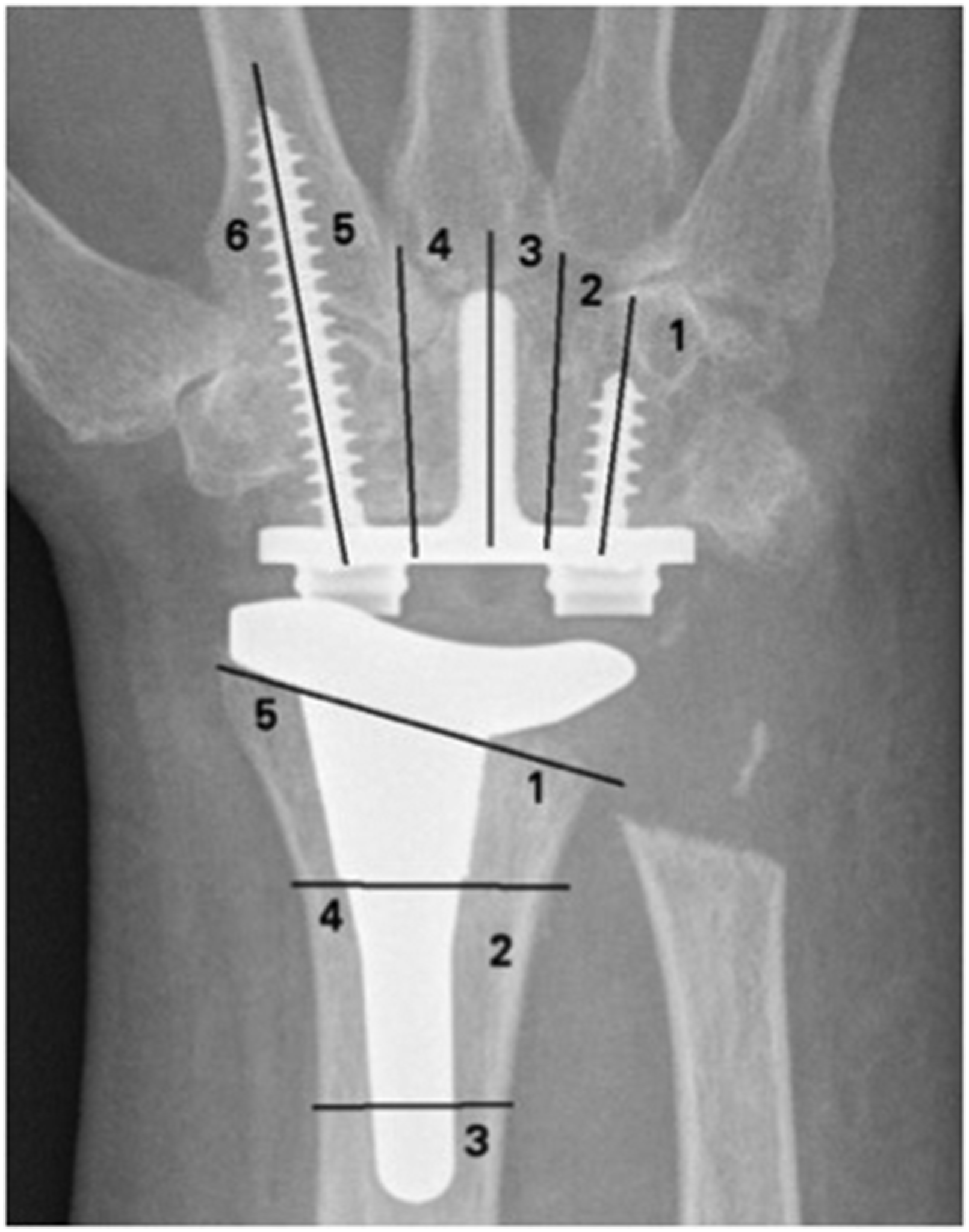
Posteroanterior wrist radiograph demonstrating the zonal method for assessing lucency for total wrist arthroplasty (Universal 2 implant shown) (Badge et al., 2016) (Copyright permission was obtained from The British Editorial Society of Bone & Joint Surgery).
Range of motion was measured using a goniometer, and grip strength was assessed using the JAMAR hydraulic hand dynamometer (Sammons Preston, Rolyan, Bolingbrook, IL, USA). Three PROM scoring systems were used: the Patient-Rated Wrist Evaluation (PRWE) (MacDermid et al., 1998), the Quick Disabilities of the Arm, Shoulder and Hand (QuickDASH) (Beaton et al., 2005) and the Patient Evaluation Measure (PEM) (Macey and Burke, 1995). In addition, we asked patients to rate their overall satisfaction (VAS 0 to 10, with 10 being completely satisfied) and whether or not they would have the same operation again (yes or no). Assessments were completed by senior fellows in hand surgery; with nine of the 12 postoperative evaluations (radiographic, clinical) performed at the same consultation.
Surgical technique and rehabilitation
The surgical technique was based on the manufacturer’s recommendations and similar to that described for the Universal system by Menon and Doyle (1998). A dorsal longitudinal midline incision permits elevation of full thickness skin flaps. The capsule is exposed after elevation of retinacular flaps through the third extensor compartment (providing exposure from the first through to the fifth compartments). An inverted T-shaped capsulotomy provides carpal access. The lunate and the triquetrum are excised. Bone preparation is via jigs, which are assembled over a guide wire in the capitate and third metacarpal for carpal preparation, and a guide wire positioned in the distal radius for radial preparation. Formal arthrodesis of the distal carpal row to the metacarpals was not undertaken. The intercarpal spaces are packed with cancellous bone obtained from the bone cuts. A trial reduction assesses movement range, impingement and stability. Definitive components are secured without cement. The radial component is secured through metaphyseal press fit fixation, and the carpal component is secured with two locking screws. The capsule is sutured to the distal radius via cortical drill holes. The retinacular flap is closed above the extensor tendons while leaving the extensor pollicis longus superficial. A plaster of Paris backslab is used for up to 2 weeks. Range of motion is started as soon as pain permits, and strengthening usually begins after 6 weeks. Patients are advised not to load the wrist beyond 2 kg for the lifetime of the prosthesis.
Statistical analysis
Continuous data are presented as means with ranges. Paired t-tests were performed when comparing preoperative and postoperative continuous variables. A p-value of <0.05 was considered to be statistically significant.
Results
We identified 13 implants in 12 patients during the study period. One patient had bilateral implants. One patient died of unrelated causes 12 months postoperatively and was excluded. Two cases had incomplete postoperative data but contributed to our primary outcome measure of revision and are included in our final series of 12 implants in 11 patients (Table 1). There were eight women and three men with a mean age at surgery of 59.3 years (range 45–80). The mean radiological follow-up time was 2.7 years (range 1.4–3.7), and the mean clinical follow-up time was 3 years (range 2.1–3.8). The majority of PROM questionnaires were completed at the most recent clinical review; however, two cases completed their questionnaires at home and returned their responses by post.
Characteristics and results of Freedom® total wrist prosthesis cases.
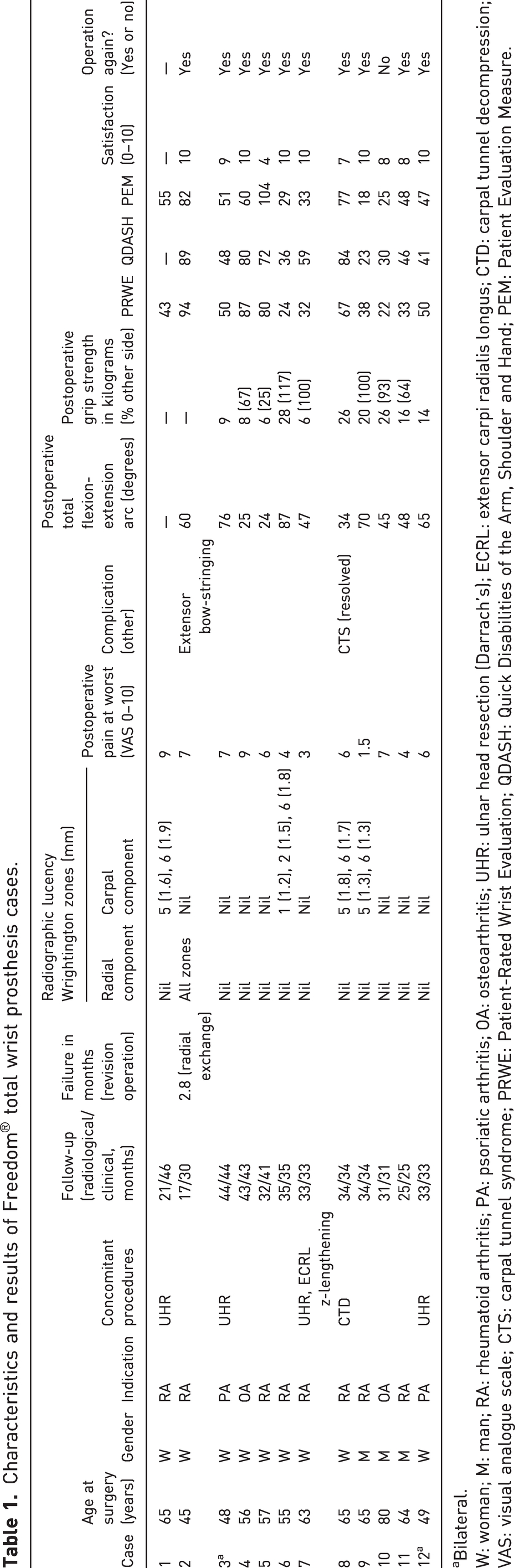
Bilateral.
W: woman; M: man; RA: rheumatoid arthritis; PA: psoriatic arthritis; OA: osteoarthritis; UHR: ulnar head resection (Darrach’s); ECRL: extensor carpi radialis longus; CTD: carpal tunnel decompression; VAS: visual analogue scale; CTS: carpal tunnel syndrome; PRWE: Patient-Rated Wrist Evaluation; QDASH: Quick Disabilities of the Arm, Shoulder and Hand; PEM: Patient Evaluation Measure.
Indications included rheumatoid arthritis in eight wrists, osteoarthritis in two and psoriatic arthritis in two (single patient with bilateral implants). Five implants were combined with concomitant procedures, including ulnar head resection in four cases. One case required release and repair of the extensor carpi radialis brevis tendon to permit implant placement. One further patient (a 63-year-old female) underwent Freedom® TWA as a treatment for failed (painful) pyrocarbon wrist interposition arthroplasty (Amandys®, Tornier SAS-Bioprofile, Grenoble, France) originally placed for failed proximal row carpectomy 15 months previously. The patient subsequently developed four zones of carpal component lucency (central peg and radial screw, Figure 2), worse PROM outcomes (PRWE 87/93, QuickDASH 80/89, PEM 86/95) and pain scores (VAS 10 compared with 8.5 preoperatively) when compared with the primary cohort. The implant was explanted and revised to a total wrist arthrodesis at 39 months, which improved her pain. This patient also developed carpal tunnel syndrome 5 months after TWA (on a background of previous carpal tunnel decompression), which settled following revision carpal tunnel decompression at 8 months. This case has not been included in the analysis of primary wrist arthroplasty, but is included for completeness to advise readers of our department’s complete experience with the Freedom® system.
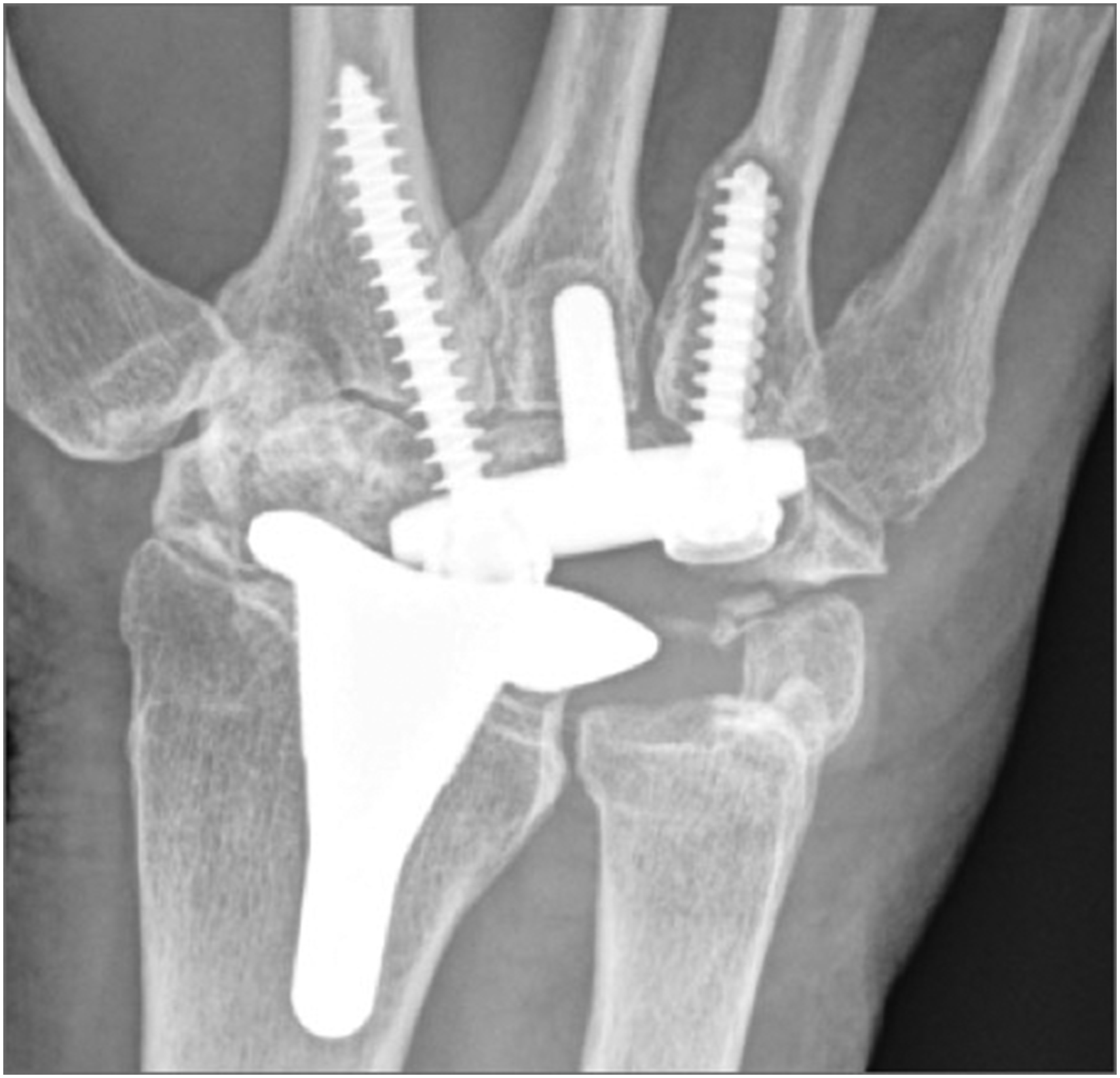
Posteroanterior wrist radiograph (35 months after surgery) demonstrating carpal component loosening of the ulnar screw and central peg (Zones 1–4).
Revisions and radiographic outcomes
A single case demonstrated complete failure of integration of the radial component and underwent radial component revision at Day 84 (Figure 3) (Case 2). The carpal component was well fixed and was retained. This patient had rheumatoid arthritis and a rare bone aplasia syndrome with a history of tibial fracture nonunion. Following radial component revision, no areas of radiographic lucency were observed on radiographs taken at 15 months, and the pain score at rest and for activities of daily living was 0 and 7, respectively. This patient’s lower limb issues have prevented travel for repeat radiographs, and subsequent follow-up was conducted through video consultation. Survivorship is therefore 11 of 12 implants at 3.4 years in this series. Eight implants demonstrated no areas of lucency of either component. Of the 12 carpal components, four demonstrated at least one zone of lucency, with the radial screw (scaphoid/triquetrum/second metacarpal) (Zones 5 and/or 6) most commonly affected (four implants), followed by the ulnar screw (hamate) (Zones 1 and 2) in one case. Of the 12 radial components, 11 demonstrated no areas of lucency.
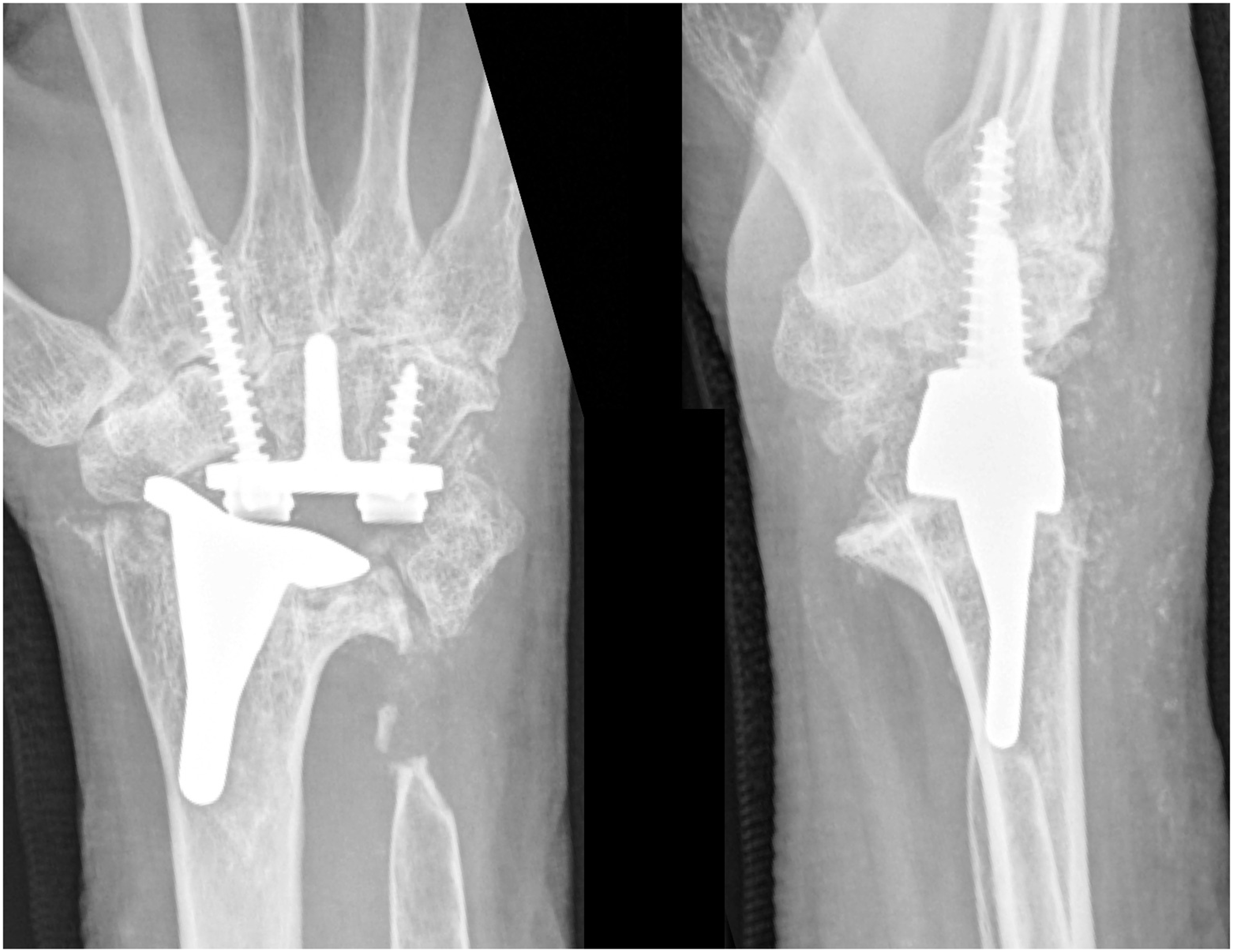
Posteroanterior and lateral wrist radiographs demonstrating complete failure of integration of the radial component (Case 2).
Wrist pain
The mean postoperative pain score (at worst) was 5.8 (range 1.5–9). Of the seven cases with preoperative pain scores, the mean preoperative score was 9.2 (range 8–10) and their mean postoperative score was 5.7 (range 3–9). When comparing these seven cases, the difference was statistically significant (p = 0.02), although this is based on low study numbers that can potentially skew data.
Complications
One case required repeat surgery for radial component revision (discussed above). One patient developed extensor tendon bow-stringing due to deficiency of the extensor retinaculum, and one patient developed recurrent carpal tunnel syndrome in the days following surgery that settled soon after without treatment. There were no cases of infection.
Range of movement
The mean postoperative total flexion-extension arc was 53° (range 24–87) in 11 wrists. Eight wrists had preoperative data with a mean total arc of 44° (range 15–80) compared with a postoperative arc of 53° (range 25–87) (p = 0.24).
Grip strength
The mean postoperative grip strength in 10 cases was 16 kg (range 6–28). When excluding the bilateral case and the single case who underwent a recent contralateral Freedom® TWA (not included in this series on account of being less than 2 years since surgery), the mean percentage grip strength of the remaining eight cases was 81% (range 25–100) of the contralateral side. Preoperative grip strength was not assessed.
Functional (PROM) scores
Postoperative PROM scores were completed in all patients at final clinical follow-up. One patient completed the PRWE and PEM questionnaires but not the QuickDASH. The mean scores (out of 100) were similar across all scoring systems: PRWE 52 (range 22–94), QuickDASH 55 (23–89) and PEM 53 (18–104). The single case with preoperative PROM scores demonstrated improvement across all scores (PRWE 72/50, QuickDASH 75/48, PEM 77/51).
Patient satisfaction
Regarding overall patient satisfaction (‘How satisfied are you with your wrist replacement?’), the mean score was 8.7 (range 4–10). When asked if they would have the same operation again, 10 answered yes and one answered no. The patient answering ‘no’ had rated their satisfaction as 8 and explained that they were happy with their wrist but believed that TWF would have been preferable, as no restrictions would have been placed on their level of activity.
Discussion
We carried out a retrospective study of patients who underwent TWA with the Freedom® prosthesis. We found that the procedure improved the pain and preserved motion with four cases of carpal lucency, although these were not symptomatic.
TWA improves function, pain and grip strength, although it may not necessarily outperform TWF according to a meta-analysis (Berber et al., 2018). Mean 10-year survival of first, second and third generation implants has been poor, with quoted incidences of 40%, 83% and 77% for the Universal 1, Biaxial and Meuli (8 years only), respectively (Boeckstyns, 2014; Yeoh and Tourret, 2015). Previous generation TWA performance failed to match that of mean accepted revision incidences for total hip and knee arthroplasty, with 94% and 97% survivorship at 15 years, respectively (NJR Editorial Board and contributors, 2021). Fourth generation implants include the Maestro, Motec, ReMotion and Universal 2, and a recent systematic review demonstrated improvements in pain scores and PROMs and superior survivorship when compared with earlier generations (Zijlker et al., 2021). The best survivorship was seen with the Motec implant, with 89% at 8 years and 86% at 10 years. The Universal 2 had the longest outcome data and demonstrated 78% survivorship at 15 years. The meta-analysis by Berber et al. (2018) compared TWA and TWF outcomes and concluded that fourth generation TWA implants had the lowest incidence of complications compared with earlier generations (range 0.1%–2.9%), but the risk of complications with TWA was higher than with TWF (Berber et al., 2018).
In our series, four out of 12 implants demonstrated at least one zone of carpal component lucency, which mandated the need for ongoing outpatient review. Lucency did not correlate with patient symptoms or function, and periprosthetic lucency for upper limb arthroplasty should be interpreted with caution, especially when changes are static. Non-weight bearing joints do not mandate revision for painless loosening in the way that lower limb arthroplasty may require. In our unit such appearances are monitored but are not deemed an indication for further intervention when symptoms are minimal or well controlled. Of particular relevance to those surgeons who may have implanted the Freedom® TWA and are considering patient discharge, we recommend long-term follow-up with a minimum annual surveillance radiograph. This follows our experience with Universal 2 TWA, where loosening was only noteworthy if found to be progressive or widespread (Figure 1) (Badge et al., 2016).
The modest improvement in total flexion–extension arc following TWA in our series (44° to 53°) is not likely to be clinically meaningful. This finding complements published data for alternative implants: 65° for Universal 2, 10°–90° for Maestro, 61° for ReMotion and 112°–125° for Motec (Badge et al., 2016; Boeckstyns, 2019; Reigstad and Røkkum, 2018; Yeoh and Tourret, 2015; Zijlker et al., 2021). These series also demonstrate reduced pain, with total improvement in VAS scores ranging from 2.8 to 6, which is comparable with the 3.5-point improvement demonstrated in our current series. Grip strength reporting is variable in the literature, including the comparison of pre- and postoperative scores, comparison between operated and non-operated sides and others reporting absolute figures. Most authors reporting postoperative grip strength suggest improvements of 20–40% when compared with preoperative, with an absolute grip strength of 5–6 kg being most common. In our series the mean strength achieved was 16 kg and this represented 81% of the contralateral side. Low numbers preclude comment on the difference in grip strength observed in our series compared with published literature. Absolute grip strength values can be misleading due to the effects of disease in adjacent joints.
PROMs are reported in variable ways in the TWA outcome literature. There is no consensus regarding the most appropriate outcome measure (Marks, 2020), although the usefulness of the PEM score for conditions affecting the wrist (including arthroplasty) has been confirmed, which includes data from our unit (Dias et al., 2001; Singh et al., 2017). As with all areas of hand surgery, systemic inflammatory or degenerative conditions are rarely restricted to the joint or joints being addressed with a specific procedure. Our series does not have sufficient preoperative PROM data to confirm a functional improvement with the Freedom® implant, but the mean postoperative scores of PRWE 52, QuickDASH 55 and PEM 53 are similar to published data (Singh et al., 2017). Those series that have reported preoperative and postoperative PROM scores universally demonstrate improvements with alternative implant designs (Badge et al., 2016; Reigstad and Røkkum, 2018; Zijlker et al., 2021).
In the systematic review by Zijlker et al. (2021) there were no articles describing PROMs or survivorship of the Freedom® prosthesis available for inclusion. Rosello et al. (2022) subsequently published clinical outcomes in 12 patients undergoing Freedom® TWA and demonstrated significant improvements in pain scores, wrist range of motion, and Mayo Wrist and PRWE scores (Rossello et al., 2022). Their mean follow-up was 48 months (range 24–72), and there were no reported complications and no revisions. There was a single case of carpal component loosening without progression, which was accepted. There were adverse radiological features in four cases, and progressive malalignment in one case. In our series, the case of radial component failure of integration requiring early revision had a potential metabolic bone disorder and underwent impaction bone grafting and revision of the radial component. Radiological loosening has not occurred at most recent follow-up.
There are limitations in our study. This was a retrospective review of a small number of implants that reflects the totality of our experience with a contemporary implant design. Our study is limited by incomplete preoperative data, including grip strength and PROM scores. The distance between our tertiary centre and many of our patients’ home locations precluded complete radiological surveillance, for example, when patients were too unwell to travel, and as such clinical data are more complete than radiological data. Medium- and long-term outcome data will identify if the carpal component lucency is of critical importance, but the high prevalence identified on short-term follow-up necessitates close continued observation.
Current TWA implants are heterogeneous, with multiple design differences, and have so far failed to match the longevity or reliability of lower limb arthroplasty. Our experience and short-term data highlight concern regarding early performance, which will enhance preoperative informed consent and necessitate long-term surveillance.
Our series demonstrates a revision incidence of one in 12 wrists at a mean 3 years clinical follow-up. We emphasize that this is a small series, but the carpal lucency incidence of 33% is of concern. The Freedom® TWA is effective for pain relief in low-demand patients wanting to preserve wrist movement. Patient selection and a detailed preoperative discussion regarding the expected outcomes, existing data limitations and comparisons with alternative treatments, such as denervation and TWF, remain important. Those surgeons who have implanted the Freedom® TWA are advised not to discharge their patients but to maintain annual surveillance on account of the high incidence of carpal periprosthetic lucency in the short-term.
Footnotes
Acknowledgements
Alastair Konarski (specialist registrar ST8 in trauma and orthopaedic surgery) of Wrightington Hospital is thanked for their support in reviewing the radiographs.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sumedh Talwalkar was a consultant for Integra LifeSciences and is now a consultant for Smith & Nephew. The other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Wrightington, Wigan & Leigh NHS Foundation Trust does not require ethical approval for reporting individual cases or case series and this was therefore waived.
Informed consent
Written informed consent was not obtained because of the retrospective nature of this study, and therefore waived by Wrightington, Wigan & Leigh NHS Foundation Trust Research & Audit Department.
