Abstract
Replacing human hand function with prostheses goes far beyond only recreating muscle movement with feedforward motor control. Natural sensory feedback is pivotal for fine dexterous control and finding both engineering and surgical solutions to replace this complex biological function is imperative to achieve prosthetic hand function that matches the human hand. This review outlines the nature of the problems underlying sensory restitution, the engineering methods that attempt to address this deficit and the surgical techniques that have been developed to integrate advanced neural interfaces with biological systems. Currently, there is no single solution to restore sensory feedback. Rather, encouraging animal models and early human studies have demonstrated that some elements of sensation can be restored to improve prosthetic control. However, these techniques are limited to highly specialized institutions and much further work is required to reproduce the results achieved, with the goal of increasing availability of advanced closed loop prostheses that allow sensory feedback to inform more precise feedforward control movements and increase functionality.
Introduction
After losing a limb, patients not only face the grieving process of adjusting to their permanently altered life, but also the reality that existing prostheses fall far short of their expectations. The shortcomings of clinically available myoelectric prostheses are exemplified by hand prostheses that only provide simple grasping or wrist rotation actions (Calado et al., 2019; Engdahl and Gates, 2021). These prosthetic movements are controlled by bulk contractions of the residual forearm musculature in a non-intuitive manner, that is difficult to master and frequently inconsistent. While rudimentary functions can be performed, such as stabilizing a bottle while the contralateral unaffected hand performs the dominant task of removing the cap, patients with limb loss quickly learn to compensate without using their prosthesis (Espinosa and Nathan-Roberts, 2019). When asked, patients comment that their prostheses do not feel like a part of them and, together with reduced function, this may lead to device abandonment (Brack and Amalu, 2021; Smail et al., 2020).
While patients using passive and body-powered prostheses cited lack of comfort as the primary factor for abandonment, those using myoelectric prostheses cited both the lack of comfort and the lack of function as equal factors for abandonment (Smail et al., 2020). Engineering advancements in signal processing and implantable systems have provided some improvement in the feedforward dexterity of devices, yet research participants are still not consistently fluent with their movements (Yamamoto et al., 2019). This is not surprising when appreciating that 90% of upper limb peripheral nerve axons convey sensory information, which is not transmitted in any meaningful way by clinically available devices (Gesslbauer et al., 2017). As a result, there is increasingly active research into the integration of sensory feedback into prosthetic devices, with the specific goal of increasing patient embodiment of the devices by providing patients a greater sense of control and ownership of the prosthesis through more realistic sensory feedback, which improves fluency of movements (Zbinden et al., 2022).
What makes sensory feedback so challenging
As the nerves in the upper limb originating from nerve roots C5 to T1 proceed distally, they organize themselves in fascicles that branch off the nerve bundle to efferently innervate muscles and afferently innervate skin, tendons and bones. There are many differences in axons in the peripheral nerves (i.e. myelination, diameter, conduction speed), which correspond with the target innervation site, such as a motor unit in a muscle or various receptors of the sensory nervous system. These sensory receptors are classified as mechanoreceptors that sense touch and pressure, thermoreceptors that sense changes in temperature, and nociceptors that sense pain.
The optimal sensory feedback loop would consist of mechanized receptors on the surface of the prosthetic limb that can transform the external stimuli into electrical signals. In system with implanted neural electrodes, these encoded stimulation signals could then be used to stimulate the selective parts of the mixed nerve bundle that provide sensory information to the respective part of the brain. An ideal stimulator would have both high fibre type selectivity by controlling the type of stimuli perceived (constant pressure, vibratory, thermal) by targeting specific fibre groups (slow-adapting, fast-adapting, free nerve endings) and high spatial selectivity by decreasing the number of off-target induced action potentials, which provides higher resolution of stimulation. Improvements in location- and firing rate-matching lead to a decreased amount of time that patients spend training to interpret the signals they are receiving from the prosthesis (Makin et al., 2017). This is a surgical and engineering challenge at the neural interface that cannot be outsourced to the ability of the nervous system to change perceived location (Ortiz-Catalan et al., 2020) or quality(Ortiz-Catalan et al., 2019).
The diversity and high density of sensory receptors in the skin complicate the reconstruction of a sensory feedback system during upper limb amputation. Current sensory interfacing devices cannot replicate the small size or large number of independent channels of information flow that exist in normal nerves. However, it is not certain that an exact biomimetic replica is needed, and the problem is determining how much sensory information is necessary to enable amputees to function with their prosthetic limbs. The amount of sensory information that is transduced to the patient’s nervous system is heavily dependent on the method of interface with the nervous system, and the common trade-off in choosing an interface is fibre type selectivity, spatial selectivity, invasiveness and longevity. To improve modality matching, the aim is to increase selectivity and resolution as much as possible without damaging nervous tissue (harmlessness), and preserving function (biostability), over a long period of time (Günter et al., 2019).
Methods of sensory restoration
The various methods of sensory feedback can broadly be separated into categories of invasiveness: extradermal and subdermal. The interested reader will find comprehensive reviews for all the design aspects of these extradermal and subdermal interfaces (Larson and Meng, 2020; Stephens-Fripp et al., 2018); however, this review will analyse each technique while providing examples of clinical or experimental implementations.
Extradermal
Extradermal methods are composed of mechanotactile, vibrotactile or electrotactile activation of nerves underneath the skin. Mechanotactile feedback is provided by an external device that provides direct contact to the skin in the form of localized pressure or skin stretch (Battaglia et al., 2019). Vibrotactile feedback consists of wearable devices with small motors or linear resonant actuators vibrating at various frequencies on the skin (Miyahara and Kato, 2021). Electrotactile feedback is achieved through transcutaneous electrical nerve stimulation (TENS), where current is delivered by electrodes through the skin, initiating an action potential in the underlying nerves. However, patients themselves do not like electrotactile feedback as the wrong intensity can produce painful stimulation.
Because of bulkier sizing, mechanotactile stimulators had previously not been preferred to vibrotactile or electrotactile stimulators. However, advances in wearable technology that incorporates electromyographic (EMG) recording and mechanotactile feedback into a lightweight fabric socket has demonstrated the superiority of proportional haptic feedback during prosthetic grasping (Borkowska et al., 2022). The combination of mechanotactile stimulation for spatial resolution and vibrotactile stimulation for intensity mapping (grip force) has been implemented with superior feedback compared with either mode of stimulation by itself (Huang et al., 2017). Mechanotactile stimulators have shown superior resolution to vibratory stimulators due to the highly localized receptive fields of skin deformation mechanoreceptors (Merkel discs and Meissner corpuscles) compared with the broader receptive fields of vibratory mechanoreceptors in the skin (Pacinian corpuscles) (Antfolk et al., 2013).
Subdermal
Subdermal methods of stimulation can also be classified into two further levels of invasiveness: extraneural and intrafascicular. A comparison of currently used subdermal electrodes can be seen in Figure 1. Extraneural electrodes sit along the surface of the nerve and transmit current into the nerve through the epineurium. The most common form of extraneural interface is a nerve cuff that wraps around the nerve (Naples et al., 1988). Electrodes are embedded within polyimide or silicon-based polymer sheets and secured around the nerve using plastic tubing, suture or polymer-based clips. Nerve cuffs are the least penetrative and damaging of the direct nerve interface, but mechanical mismatch between the surface electrode and the neural tissue can still cause fibrotic tissue growth (Grill et al., 2009). To address this mechanical mismatch, there have been attempts to use fully polymeric devices that have more similar mechanical characteristics to nerves (Cuttaz et al., 2019). Cuff electrodes are currently the most used neural interface and have shown long-term biostability for over a decade (Christie et al., 2017).
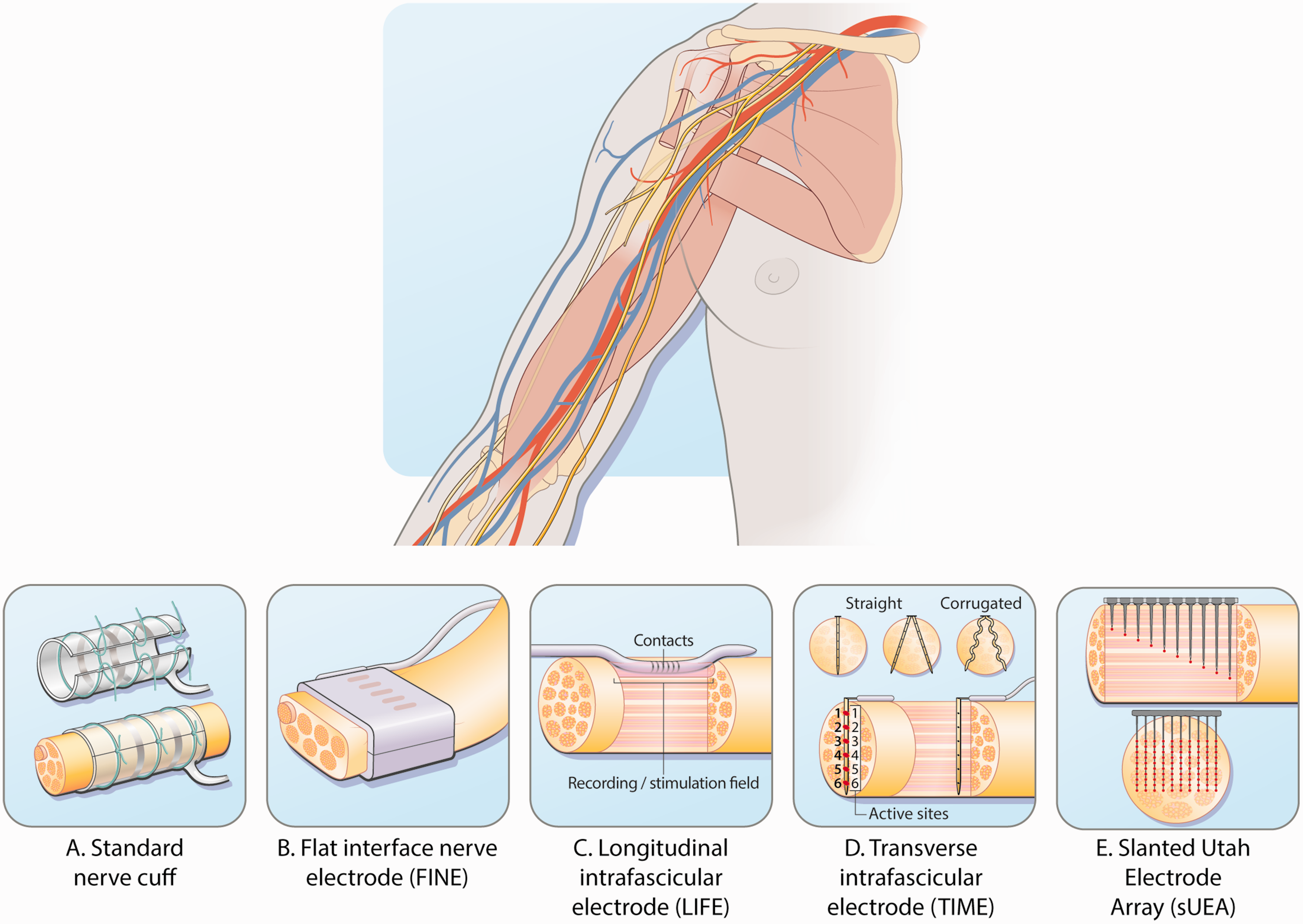
Comparison of implanted subdermal electrodes. (a) Nerve cuff. (b) Flat interface nerve electrode (FINE). (c) Longitudinal intrafascicular electrode (LIFE). (d) Transverse intrafascicular multiple electrode (TIME) and (e) Utah slanted electrode array (USEA).
Being the least penetrative, nerve cuffs also have the least success in stimulating axons that are deep within the nerve bundle (Figure 1(a)). Electrode quantity, geometry and orientation have been widely varied in attempts to target multiple parts of the nerve (Raspopovic et al., 2017). Cuff electrode configurations that are situated transversely across the nerve have shown better selectivity than configurations that place electrodes longitudinally down the nerve (Nielsen et al., 2011). In these transverse cuffs, bipolar and multipolar stimulation patterns have shown higher selectivity than monopolar stimulation in restoring sensory feedback by more accurately controlling the flow of current (Jantz et al., 2020). Recent efforts in increasing the selectivity of extraneural cuffs are employing complex, biomimetic stimulation techniques that target specific fibre types (Mastinu et al., 2020) and temporal interference techniques that target interior fibres using interfering high-frequency electrical fields (Sunshine et al., 2021).
Another method to increase selectivity in extraneural cuffs is the flat interface nerve electrode (FINE) that compresses the nerve, flattening the cross section (Figure 1(b)). This compression increases the surface area and brings central fibres closer to the periphery, allowing for greater spatial selectivity of individual axons (Tyler and Durand, 2002). Despite compression of the nerve, FINE cuffs show similar long-term implantation biocompatibility to the spiral cuff that allows for stimulated sensory feedback after 5 years from implantation (Graczyk et al., 2018; Tan et al., 2015).
A more invasive class of subdermal stimulation is the penetrating intrafascicular electrode. The longitudinal intrafascicular electrode (LIFE) is made of a Kevlar fibre with a single gold electrode that is embedded into the fascicle (Figure 1(c)). A more flexible version made from polyimide with an increased number of electrodes has been developed called the thin-film LIFE (tf-LIFE) (Overstreet et al., 2019). The needle insertion places the LIFE along the nerve fibres. Multiple LIFEs have been implanted into the same nerve bundle in a single intervention called the distributed intrafascicular multiple electrode (DIME) (Thota et al., 2015). While the invasiveness of the electrode does decrease the current necessary to provide sensory information, the longitudinal orientation localizes the spatial selectivity of the electrode to the region of the nerve directly underneath the electrode, so multiple electrodes become necessary, such as the DIME.
A more commonly used intrafascicular electrode in animal and some clinical trials is the transverse intrafascicular multiple electrode (TIME). The TIME is also a thin film of polyimide that is embedded with electrodes and then inserted transversely across the peripheral nerve cross-section (Figure 1(d)). This transverse orientation intuitively provides greater selectivity of internal axons for the TIME compared with that of cuffs or the LIFE (Badia et al., 2011), because its array of electrodes comes into direct contact with a greater number of internal fibres as it spans the cross section of the nerve bundle. This increase in selectivity is especially true when considering the use of multiple TIMEs in the same nerve bundle (Guiho et al., 2021). However, one primary concern with invasive interfascicular electrodes is the foreign body response (FBR) that can arise as macrophages, fibroblasts and extracellular matrix components begin to form a fibrotic capsule that remains embedded in the nervous tissue. Because of the small size and flexible materials of the TIME and LIFE, the FBR found in studies of these two implants has been notably small. The small FBR allows stable recordings to be obtained for up to 6 months, particularly with the TIME (D’Anna et al., 2019). Dexamethasone is often given in intrafascicular implants to slow down and reduce the overall FBR (de La Oliva et al., 2019).
Finally, Utah slanted electrode arrays (USEAs) are the most invasive intrafascicular electrodes that have been used in human trials. These electrode arrays are silicon-based wafers with spikes of different heights (Figure 1(e)). The spikes are thin and plated with platinum, followed by a coat of iridium oxide to increase bioconductivity (Wark et al., 2013). A high density of 25 electrodes/mm2 provides a total of 96 electrodes on the USEA, which is a much higher independent stimulation source than any previous kind of stimulator mentioned. The spikes of different lengths penetrate the nerve at different depths, which currently achieves the highest level of spatial selectivity. Because there are so many spikes going into the nerve, there is cause for concern regarding chronic biocompatibility and FBR (Carnicer-Lombarte et al., 2021). There are fewer human clinical trials using the USEA than those using the TIME; however, one patient had a fully functional USEA implant for 14 months, while the longest period a TIME has been implanted and functional is only 6 months (George et al., 2020; Petrini et al., 2019). While activation thresholds increased as time progressed owing to increased scarring, the peripheral nerve interfaces remained safely functional.
Case studies
Sensory feedback using cuff electrodes in neuromusculoskeletal prostheses (Figure 2(a))
As reported elsewhere, three patients with transhumeral amputations were implanted between 2013 and 2018 with an osseointegrated interface that allowed bidirectional communication through epimysial EMG electrodes for efferent motor control, and cuff electrodes around major nerve branches for afferent stimulation (Ortiz-Catalan et al., 2020). The patients ranged in age from 44 to 47 years, were all male and had lost their arms because of trauma or malignancy. Each cuff electrodes provided three stimulation contacts and were implanted in the median and/or ulnar nerves, with a maximum of two electrodes per patient (Ortiz-Catalan et al., 2014). These connected the users’ nerves, muscles and skeleton through a neuromusculoskeletal interface (Figure 2) (Ortiz-Catalan et al., 2020), taking advantage of osseointegration to enable safe, reliable and long-term mechanical and electrical connection percutaneously. Cuff electrodes have been used as part of this system because of their long-term stability (Christie et al., 2017), however any type of electrode could be connected through the osseointegrated implant. Earlier neurophysiological work had demonstrated that when short, square-wave pulse trains (0.2 ms pulses at 30 Hz for 1 s) were sent to the implanted cuff electrodes, patients were able to feel projected sensations across different areas of their missing hand (Ackerley et al., 2018). Notably, sensory feedback was applied via direct nerve stimulation using cuff electrodes during daily life, and functional improvements were observed in grip control, particularly when the weight of the test object weight was unknown to the patient (Mastinu et al., 2020).
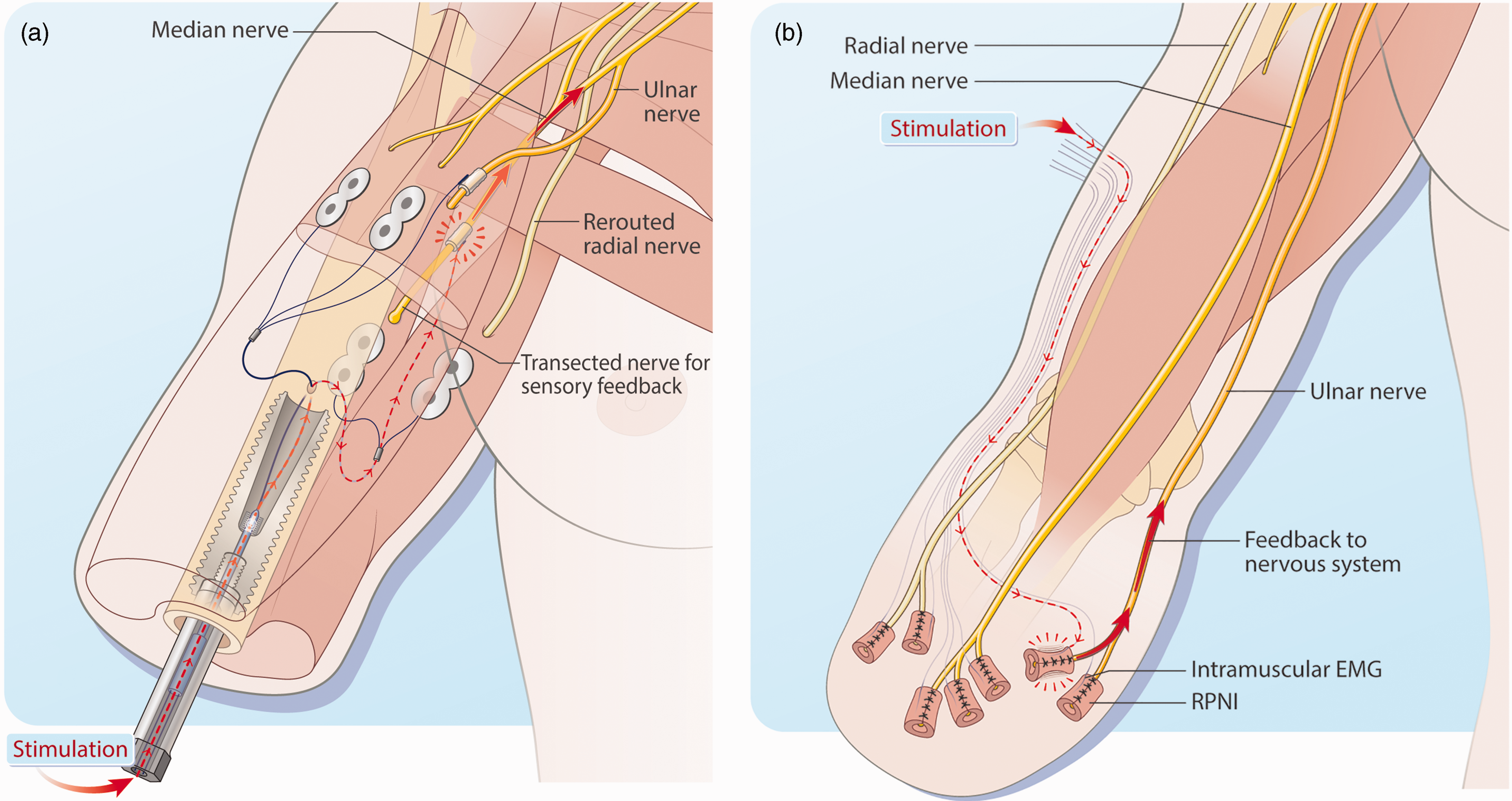
Case studies comparison – electrode implantation. (a) Cuff electrodes providing sensory feedback as part of a neuromusculoskeletal interface in which bidirectional communication is enabled via an osseointegrated implant and (b) Transcutaneous electromyographic (EMG) electrodes on regenerative peripheral nerve interfaces (RPNIs) providing sensory feedback.
Patients have continued to use their prostheses in daily life, and such daily use has resulted in positive personal and social changes in their lives, as indicated by patient interviews (Middleton and Ortiz-Catalan, 2020). Patients regarded the main factors in improved function to be the skeletal attachment (i.e. resolution of socket-related problems) and the more reliable and precise control provided by the implanted electrodes (i.e. resolution of surface electrodes instability) (Mastinu et al., 2019). Although sensory feedback was received positively, it was seen as the least important factor in comparison with function and comfort (Middleton and Ortiz-Catalan, 2020). Daily use of sensory feedback resulted in improved sensory acuity (Ortiz-Catalan et al., 2020). However, the nature and location of the perceived sensations did not change over the years (Ortiz-Catalan et al., 2020), even despite a visual–tactile perception mismatch, as the patients could observe that the location of the sensor in their prosthetic hand (thumb) did not exactly coincide with the location of their perceived sensations. The precise targeting of electrodes to elicit sensations in pre-defined locations remains a challenge, as does the low-resolution of the available neural interfaces.
Regenerative peripheral nerve interfaces role in sensory feedback (Figure 2(b))
The regenerative peripheral nerve interface (RPNI) is one novel approach that has the potential to provide both efferent control and afferent feedback from prosthetic devices. The RPNI has been shown in extensive animal studies and human trials to provide high fidelity motor control of prostheses (Irwin et al., 2016; Kung et al., 2014; Urbanchek et al., 2016; Vaskov et al., 2022; Vu et al., 2020). RPNIs are created by wrapping a free skeletal muscle graft around the end of divided peripheral nerves. Over time, the skeletal muscle becomes reinnervated creating a biological interface. Intramuscular bipolar electrodes can then be implanted in the RPNIs to record motor control signals. RPNIs have previously been shown to facilitate functional prosthetic control years after implantation and be effective in reducing neuroma and phantom limb pain (Hooper et al., 2020; Vu et al., 2020; Woo et al., 2016). In addition, RPNIs have the potential to provide afferent sensory feedback with the overall goal of optimizing a reliable bidirectional prosthetic interface.
A 53-year-old woman underwent a transradial upper limb amputation to treat severe neuroma pain and phantom limb pain after necrotizing fasciitis and a partial hand amputation. At the time of elective distal transradial amputation, she underwent placement of one RPNI on the median nerve, two RPNIs on one ulnar nerve (one RPNI for each of two independent ulnar nerve fascicles) and one on her dorsal radial sensory nerve. All RPNIs were created as previously described (Kubiak et al., 2018). For newly created RPNIs, it takes approximately 3 months for revascularization and reinnervation to be complete. One year postoperatively, bipolar electrodes were implanted into each RPNI in compliance with the approved protocol. RPNI creation and electrode implantation can be done during the same operative procedure, as demonstrated in non-human primates (Irwin et al., 2016). Each bipolar electrode was implanted by creating a small 3- to 4-mm window in the muscle component of the RPNI. The electrode was inserted bluntly into the substance of the muscle and secured with an absorbable suture to reduce motion at the electrode–muscle interface. The wire was passed proximally in the subcutaneous plane, using a tendon passer, and brought out through the skin to a housing unit that was affixed to the skin with adhesive.
Sensory feedback experiments started 2 months after wire implantation and continued for 36 months. Stimulation thresholding and sensitivity trials were always conducted using a stimulation frequency of 20 Hz, an interphase interval of 10 µs and a pulse width of 100 or 200 µs. Results can be seen in Figure 3. Each month the patient was stimulated to determine stimulation perception thresholds and perceived locations of sensation for each RPNI over a 36-month period (27 total observations). At the stimulation threshold, she described their perceived area of sensation in the phantom limb, which remained relatively stable during this time (Figure 3(a)). Thresholds were most consistent for the median RPNI (M = 1.10, SD = 0.18 mA), followed by ulnar RPNI 1 (M = 0.95, SD = 0.20 mA), with the ulnar RPNI 2 having the highest variability over the collection period (M = 1.12, SD = 0.35 mA) (Figure 3(b)). Stimulation discomfort thresholds were only collected once for each RPNI (Median 6.74 mA; Ulnar 1: 3.88 mA; Ulnar 2: 9.77 mA) (Figure 3(b)). Cumulative normal distribution curves were then fitted to the data from these experiments (Figure 3(c)) to determine the Weber fraction, which measures an individual’s sensitivity to intensity changes for a given sensory stimulation. The patient was nearly half as sensitive to changes in intensity on the Median RPNI (Weber fraction: 0.13) compared with changes in intensity on the Ulnar 1 (Weber fraction: 0.088) or Ulnar 2 (Weber fraction: 0.087) RPNIs.
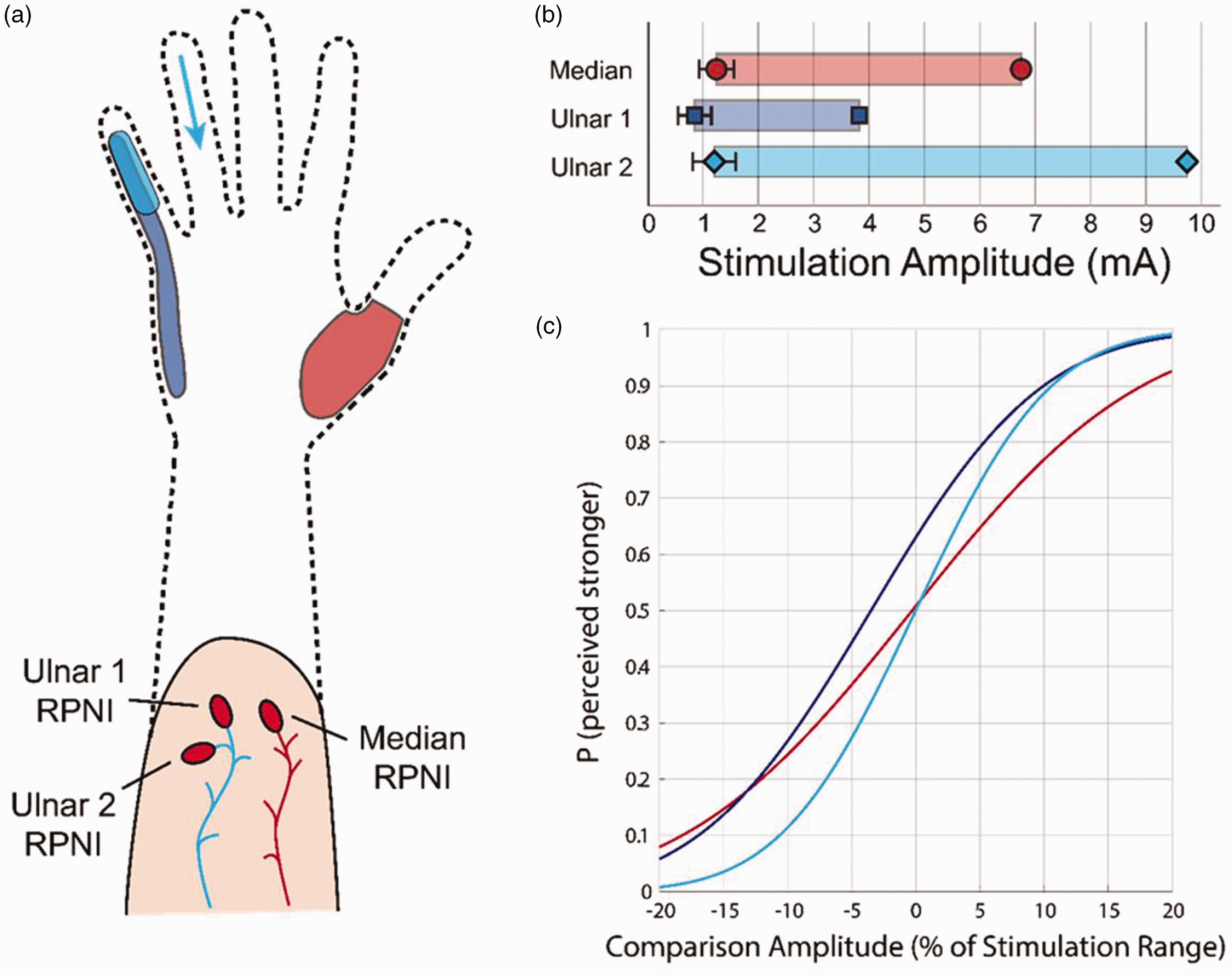
regenerative peripheral nerve interface (RPNI)-enabled sensation outcomes. (a) Locations of perceived location when P1’s RPNIs were stimulated. (b) Perception and discomfort thresholds for P1 on each RPNI and (c) Discrimination curves for each RPNI. The reference amplitude (0%) was different for each RPNI based on the average of their respective perception and discomfort thresholds.
Summary
Reconstructing upper limb loss remains a significant challenge. While engineers and surgeons have developed techniques for effective feedforward motor control, prosthetic movements still do not match that of an intact human hand. Various groups have now demonstrated in clinical trials of implanted electrode systems, that patients are able to detect some sensory feedback from the prosthetic limb resulting in better control. However, these are only research trials and have not yet entered widespread clinical practice. Alternate to implantable electrodes are sensorized RPNIs, which have the capacity to activate afferent pathways through biological means. These pioneering studies are enabling a realistic future where dexterous closed loop prosthetic control can effectively replace a lost limb.
