Abstract
We aimed to assess the association between bariatric surgery and incident Dupuytren’s disease (DD) using propensity score-matched cohort studies among Swedish nationwide healthcare registries. Patients aged 30–79 years who underwent bariatric surgery 2006–2019 were matched on their propensity scores, up to two obese bariatric surgery-free (unexposed) patients. We applied Cox proportional hazard regression to calculate hazard ratios (HR) with 95% confidence intervals (CI) for the risk of DD overall, in subgroups of age, sex, bariatric surgery type and duration of follow-up. Among 34,959 bariatric surgery patients and 54,769 propensity score-matched obese patients, the risk of DD was increased in bariatric surgery patients compared with obese unexposed patients (HR 1.30, 95% CI 1.02–1.65), among women (HR 1.36; 1.00–1.84); those undergoing gastric bypass (HR 1.33; 1.04–1.71) and those with >5 years follow-up (HR 1.63; 1.14–2.34). Our results suggest that substantial weight loss is associated with an increased risk of DD in an obese population.
Introduction
The association between obesity and Dupuytren’s disease (DD) is highly biologically unusual, being one of only two diseases where obesity is found to be protective in the development of the disease (Majeed et al., 2021). Large epidemiological cohort studies have suggested that obesity is protective for DD (Gudmundsson et al., 2000; Hacquebord et al., 2017; Kuo et al., 2020). Furthermore, genome wide association and Mendelian randomization studies have also suggested a causally protective effect of increased body mass index (BMI) (Majeed et al., 2021; Major et al., 2019). However, other research has suggested an increased incidence of DD associated with diabetes mellitus and hyperlipidaemia, therefore complicating the relationship between DD, obesity and metabolic syndrome (Alser et al., 2020; Geoghegan et al., 2004; Hart and Hooper, 2005).
Weight loss is promoted in order to prevent sequelae of obesity, including diabetes mellitus, cardiovascular disease or cancer (WHO, 2020). Bariatric surgery is an effective treatment for obesity where lifestyle interventions alone have failed (Eaton et al., 2016). The majority of weight loss has been shown to occur within the first 2 years after surgery, with an established reduction in cardiovascular and inflammatory conditions, depression, cancer and mortality (Buchwald et al., 2004; Gill et al., 2019; Schauer et al., 2019).
While epidemiological and genetic studies of obesity and DD have been undertaken, the impact of weight loss following bariatric surgery among obese patients on incident DD has not been assessed to date. Modelling weight loss in epidemiological data is extremely challenging due to poor measurement, but bariatric surgery is frequently used as a surrogate as this is associated with immediate and sustained weight loss in patients. Therefore, we aimed to assess the impact of bariatric surgery on the incidence of DD in a large cohort of patients from national Swedish healthcare registries. Using propensity score-matching, we compared new-onset DD in obese patients who had undergone bariatric surgery with matched individuals who had not undergone bariatric surgery.
Methods
Data sources
We conducted a propensity score-matched sequential cohort study using data from the national Swedish healthcare registries, including the Patient Registry, Causes of Death Registry, Prescribed Drug Registry, Cancer Registry and the Scandinavian Obesity Surgery Registry (SOReg) (Brooke et al., 2017; Ludvigsson et al., 2011; SOReg, 2021). All individuals born or permanently residing in Sweden are assigned a 10-digit personal code that is used for identification in healthcare registries and allowed for linkage (Ludvigsson et al., 2009). The quality of the data on surgery has been externally validated within the Swedish patient registry, and excellent validity of bariatric surgery in the Swedish Patient Registry and SOReg has also been confirmed when compared against medical records (Ludvigsson et al., 2011; Tao et al., 2016). The Patient Registry was therefore used to identify bariatric surgery patients and SOReg to obtain details on the type of surgery codes and BMI measurements.
Study population
We identified all individuals diagnosed with obesity (International Classification of Disease (ICD) version 10 code E66, ICD version 9 278 A/B, ICD version 8 287,0, ICD version 7 277,99) or bariatric surgery aged 30–79 years at any time between January 2006 and December 2019 in the Swedish Patient Registry (WHO, 2016). We excluded patients with a record of previous bariatric surgery in order to only include those undergoing primary weight-reducing surgery, DD diagnosis or surgery for DD prior to cohort entry (Figure 1).
Study overview. (a) Each entry block represented one cohort. The cohorts contained all eligible exposed patients and their (1 to 2) PS-matched unexposed patients. (b) Detailed representation of each individual entry block. Matched exposed patients entered on the date of their surgery; matched unexposed patients entered on a random date within the entry block. We followed all patients for a maximum of 13 years after their completed run-in period of 1 year, until they had a record of DD or they were censored due to change in exposure status, death, loss to follow-up, occurrence of an exclusion criterion or end of study period.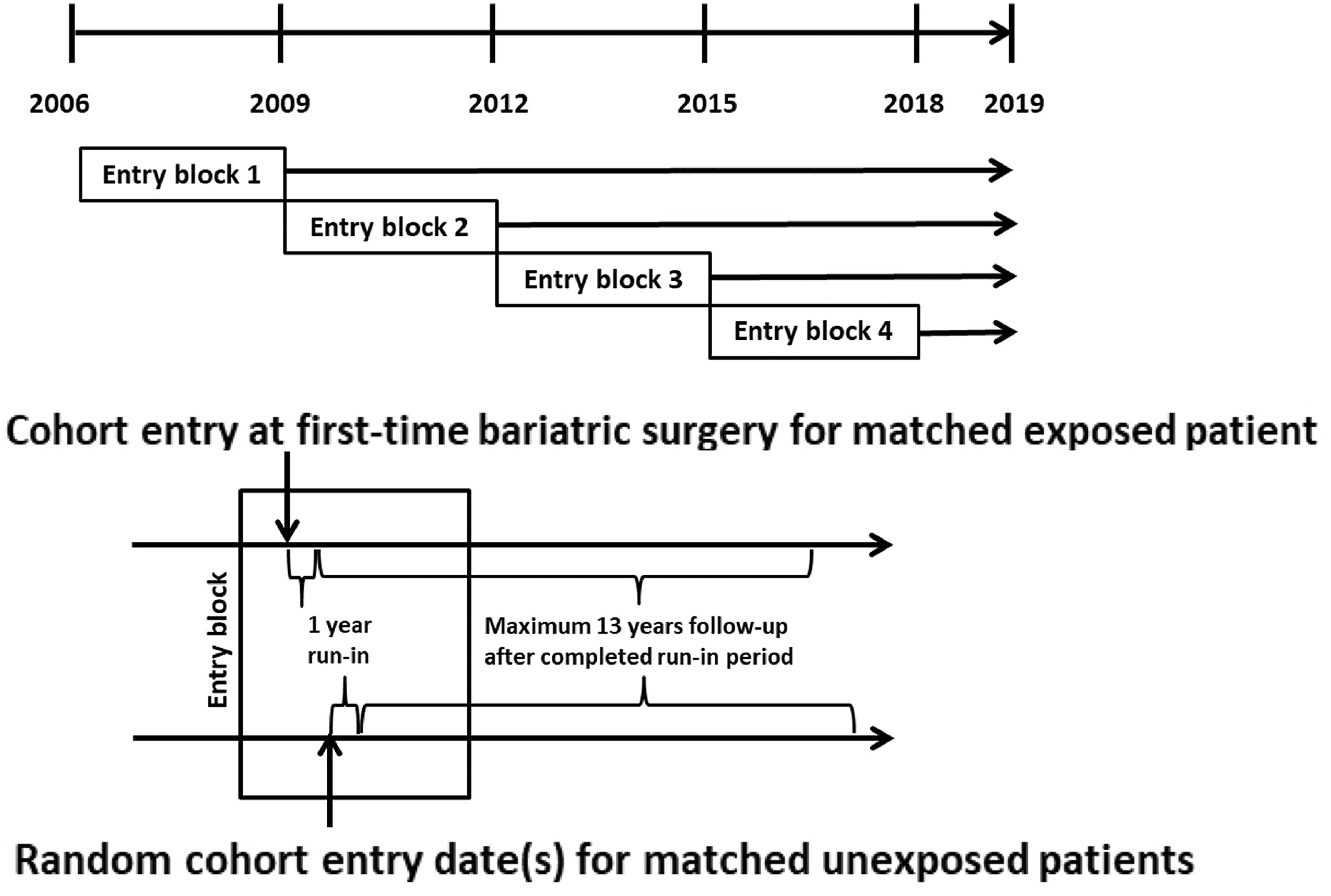
Study design
Exposures
Bariatric surgery was defined using NOMESCO (Nordic Medico-Statistical Committee) classification of surgical procedures codes (as of 1997: gastric bypass: JDF10–11; duodenal switch: JFD03–04; others: 93% sleeve gastrectomy according to SOReg, thus, we further referred to this group as sleeve gastrectomy: JDF00–01, JDF20–21, JDF96–97) (Nordic Co-operation, 2021). Patients were considered ‘exposed’ to bariatric surgery for their entire follow-up unless the surgical procedure was reversed to their original gastroenterological anatomy (NOMESCO code JFD23), in which case the follow-up ended. ‘Unexposed’ patients were defined as those identified with an ICD code for obesity but who had not undergone bariatric surgery. If an unexposed patient subsequently underwent bariatric surgery after entry into the cohort, they were censored at this stage and may have been eligible to enter the study as an exposed patient within the entry block design.
Outcome
We defined DD as the first recorded diagnosis of ICD-10 M72.0 or DD surgery (NOMESCO code of NDM09/19).
Entry block design
An illustration of the sequential block entry design is provided in Figure 1. We categorized exposed patients into one of four 3- to 4-year entry blocks according to the date of bariatric surgery, referred to as cohort entry (Figure 1(a)). Within each entry block, we additionally identified the eligible group of unexposed patients aged 30–79 years. These patients were assigned one or more random entry dates within the respective entry block (Figure 1(b)). Thus, patients could contribute one episode as an exposed patient but multiple episodes as an unexposed patient throughout the study period as their exposure to surgery changed.
Follow-up
Follow-up began on day 365 after cohort entry (i.e. run-in period) as no earlier effect of weight loss following bariatric surgery was clinically expected. We followed all patients in an ‘as-treated’ approach until the first occurrence of DD or censoring due to occurrence of an exclusion criteria, change of bariatric surgery exposure status, loss to follow-up, death or end of study period (December 2019).
Statistical methods
Propensity score
Baseline characteristics of bariatric surgery patients and unexposed patients with obesity (follow-up >365 days) before and after PS-matching.
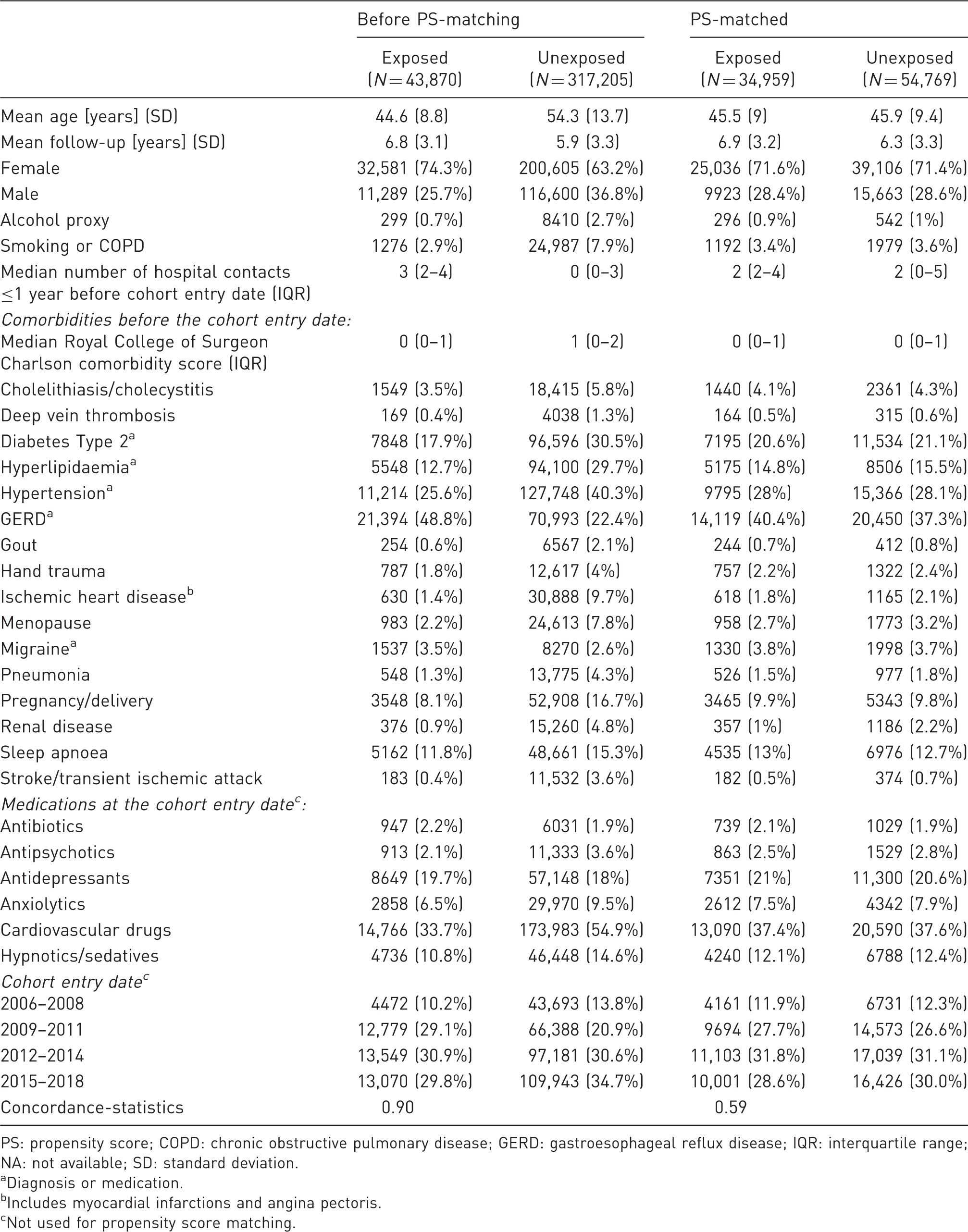
PS: propensity score; COPD: chronic obstructive pulmonary disease; GERD: gastroesophageal reflux disease; IQR: interquartile range; NA: not available; SD: standard deviation.
Diagnosis or medication.
Includes myocardial infarctions and angina pectoris.
Not used for propensity score matching.
Descriptive analysis
Stratified by exposure, we assessed patient characteristics at cohort entry before and after propensity score-matching. Furthermore, we assessed censoring reasons of the cohorts before and after matching and described cumulative incidences of DD stratified by exposure.
Regression analysis
After combining all sequential cohorts into one study cohort, we compared covariate distribution between treatment groups before and after propensity score-matching through estimation of standardized mean differences and the concordance-statistic (c-statistic). This c-statistic indicates the level of covariate balance between study groups where 0.5 indicates perfect balance and 1.0 indicates maximal imbalance (Franklin et al., 2014). Using Cox proportional hazard analyses, we estimated hazard ratios (HR) with 95% confidence intervals (CI) for the incidence of DD with bariatric surgery, compared with unexposed patients.
We performed subgroup analyses by sex, age (30–54 years, 55–79 years), bariatric surgery type (sleeve gastrectomy, gastric bypass, duodenal switch). For all subgroup analyses we re-matched within subgroups. The proportional hazard (PH) assumption was tested using the Martingale residual method, which did not hold in the overall analysis. Thus, we performed subgroup analyses by median follow-up (>1–5 years, >5–13 years). Since patients may contribute several episodes and matching may lead to correlated patient clusters, we estimated robust sandwich estimates for the covariate matrix (results remained unchanged).
As a sensitivity analysis as propensity score methodology is still considered novel by some, we also conducted all analyses using multivariable Cox regression in the unmatched cohort, adjusting for all covariates, included in the propensity scores.
Results
Demographics
Figure 2 shows the flowchart of data management. Of 43,870 exposed patients eligible for propensity score-matching, 34,959 (80%) were matched with up to two unexposed episodes resulting in a total of 89,728 matched episodes.
Flow-chart of the study composition.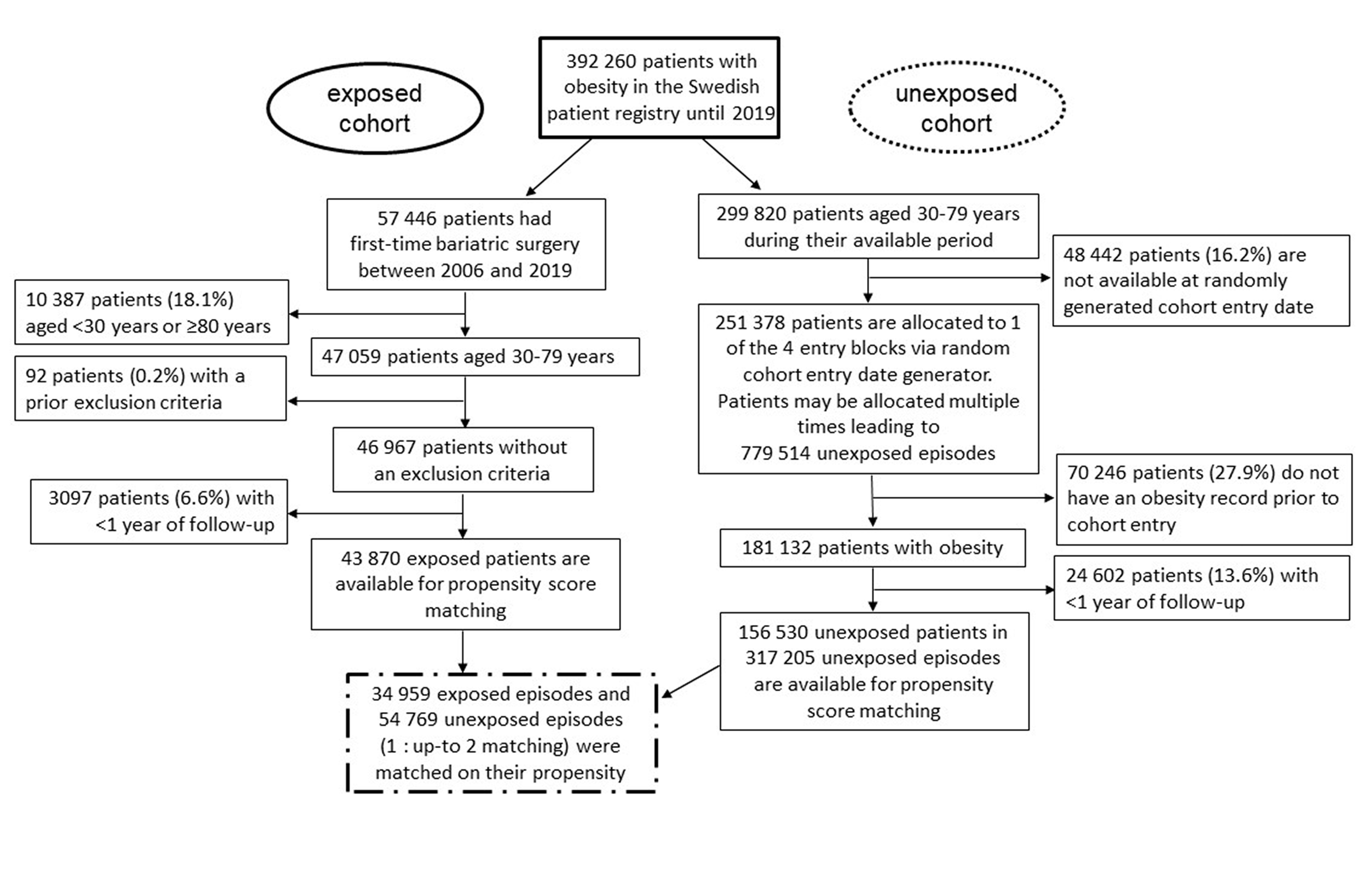
Before propensity score-matching, the average age of bariatric surgery patients was lower, they were more likely female, had more hospital contacts, were less frequently diagnosed with hypertension, type 2 diabetes or ischemic heart diseases, but more frequently diagnosed with gastroesophageal reflux disease (Table 1). Of those who underwent bariatric surgery, most patients underwent gastric bypass surgery (85%, Supplementary Table S1). Patients with duodenal switch had the highest mean BMI at surgery of 55.1 kg/m2. At 1, 2 and 5 years after surgery, BMIs were comparable between surgery types at around 30–33 kg/m2.
Supplementary Table S2 gives the details of follow-up and outcome. Before propensity score-matching, we observed 139 and 957 incident DD cases among exposed and unexposed patients, respectively. A total of 85% of DD cases were identified by a diagnosis code, 11% of cases were identified by a surgical code and 4% were identified by a combination of diagnosis and surgery (same day). In patients who had both a DD diagnosis and surgery code during the observation period, median time from diagnosis to surgery was 70 days (interquartile range 1–217 days).
After propensity score-matching, all patient characteristics of bariatric surgery patients and obese unexposed patients were highly similar (<10% of standardized mean differences between groups (Supplementary Figure S1), c-statistic of 0.59 (Table 1)). Moreover, censoring was comparable between groups after propensity score-matching.
Risk of DD
Results of the association of bariatric surgery and DD overall and in subgroups, propensity score-matched analysis.
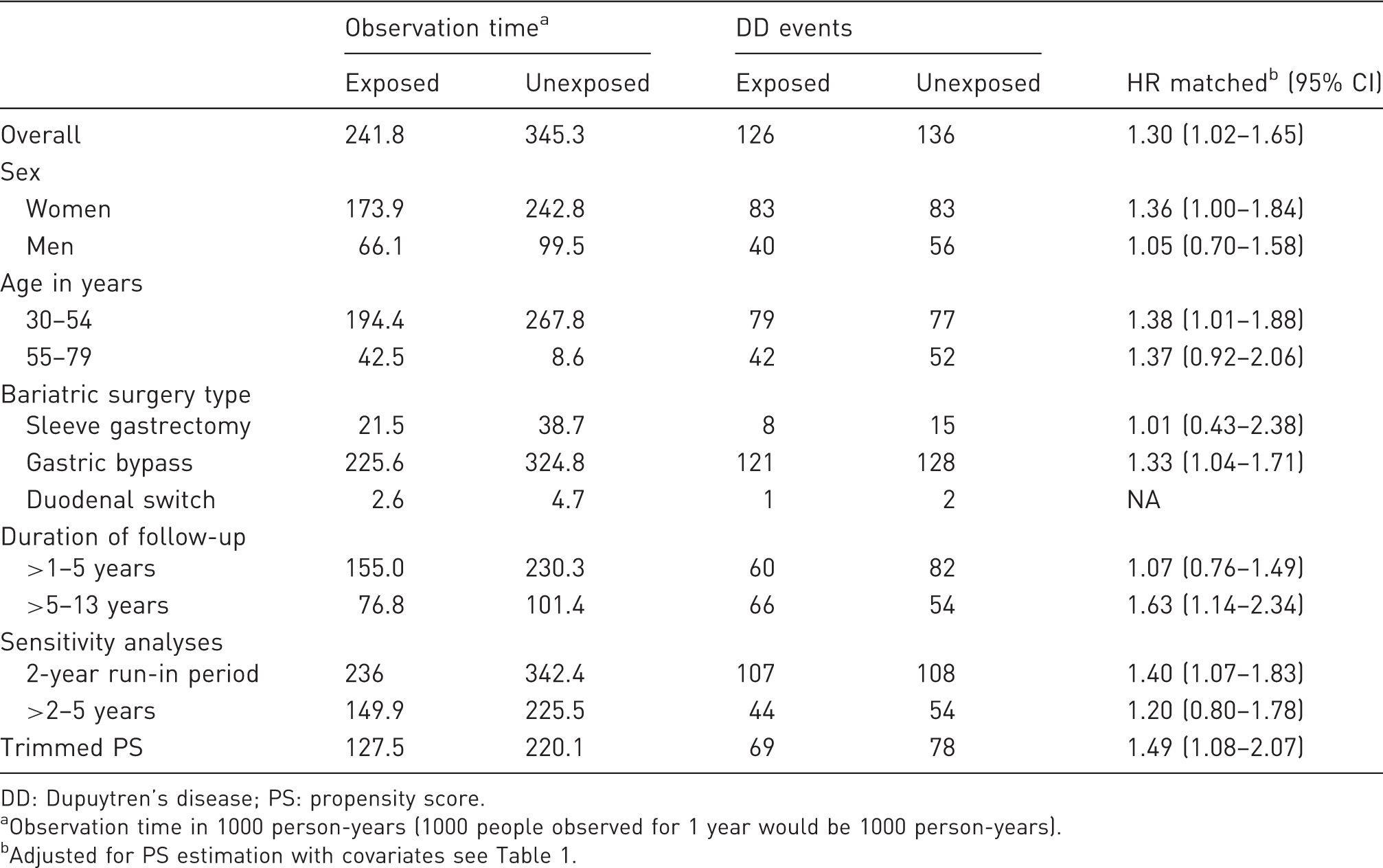
DD: Dupuytren’s disease; PS: propensity score.
Observation time in 1000 person-years (1000 people observed for 1 year would be 1000 person-years).
Adjusted for PS estimation with covariates see Table 1.
Cumulative incidence of DD in exposed and unexposed patients over time is shown in Figure 3. We observed an increased incidence of DD in exposed patients especially seen beyond 5 years of follow-up.
Cumulative incidences of Dupuytren’s disease (DD) in exposed patients to bariatric surgery and unexposed patients.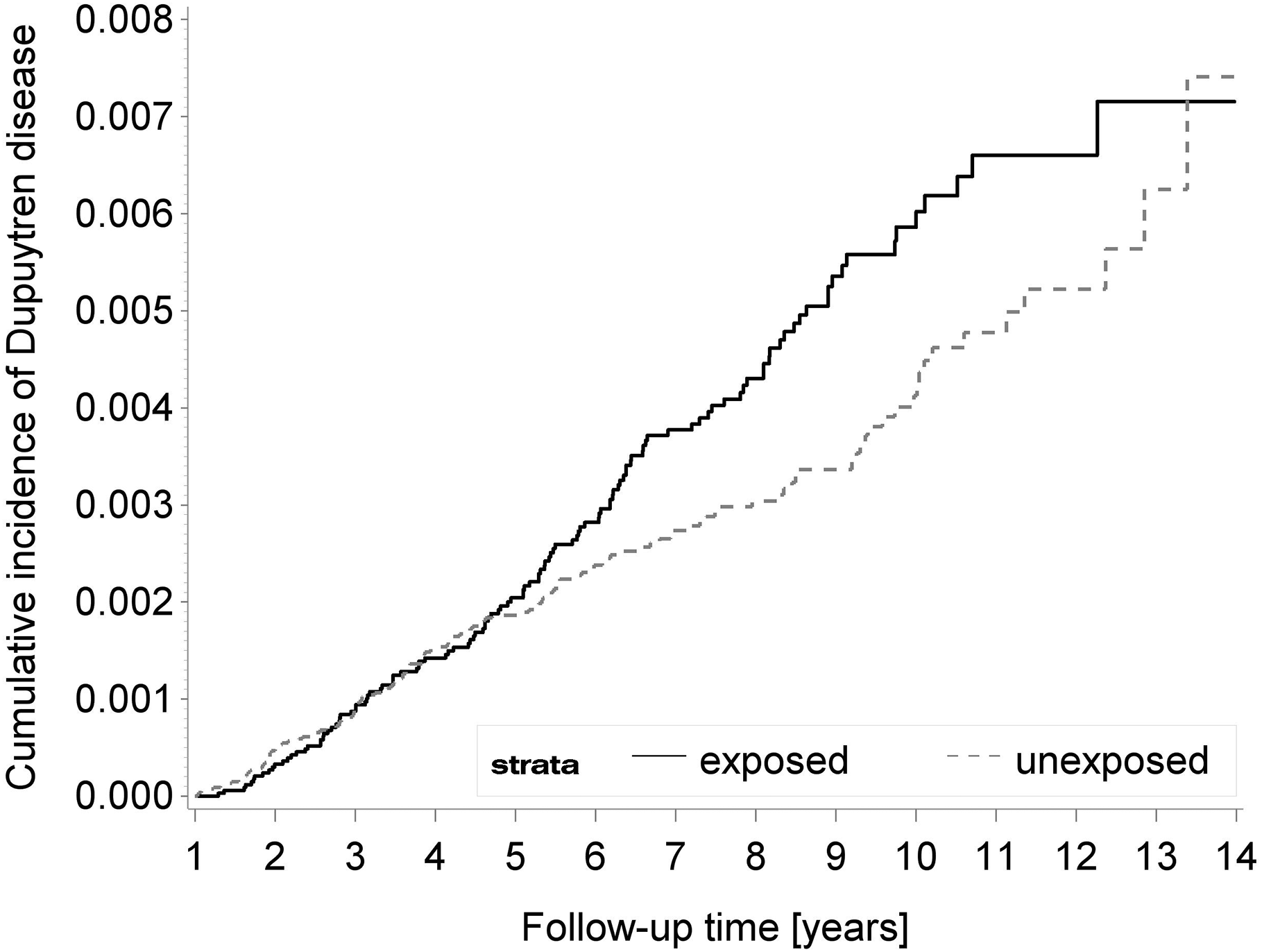
Discussion
In this large population-based, retrospective cohort study of 34,959 patients undergoing bariatric surgery in Sweden, we observed a 30% increased risk of incident DD following surgery to reduce BMI when matched to 54,769 unexposed episodes. A maximum follow-up time of 13 years within the study also enabled identification of an increased risk of DD as the difference in incidence increased over the long follow-up period available.
In our study, we showed that weight loss induced by bariatric surgery was associated with an increased incidence of DD, particularly after allowing time for weight loss to exert its metabolic effects. These results are consistent with previous studies that have shown obesity to be causally protective against DD (Majeed et al., 2021).
Two sensitivity analyses merit further discussion. First, we observed that the risk of DD was higher in women rather than men. We think that this slightly counter-intuitive result (as DD is well-known to be more common in men than women) is caused by two factors. First, 72% of our exposed cohort was female (Table 1). Second, the observation time in females was 173.9 × 1,000 person-years, but for males it was 66.1 × 1000 person-years (Table 2). The effect of bariatric surgery on development of DD also takes several years to manifest and continues to increase over time (Figure 3). These factors combined also meant there was more statistical power to observe an effect in females compared with males. Second, the risk of DD did not differ between age groups. As with sex, this discrepancy may be caused by the increased number of younger people in the study. We note that after propensity score-matching, the mean age of the exposed and unexposed cohorts was 45.5 and 45.9 years, respectively (Table 1). Similarly, the observation time in the 30–54 age group was 194.4 × 1000 person-years, but for the older age group it was 42.5 × 1000 person-years (Table 2). These factors meant that while there were more absolute events in the younger age group, the risk of developing DD was similar between these two groups in the sensitivity analysis (HR 1.38 and 1.37, respectively; Table 2).
The reasons for a higher incidence of DD with decreasing obesity remains unclear. One potential mechanism, as previously discussed (Majeed et al., 2021), may be that adipose tissues in the palm suppress pathological myofibroblast activity, which may also account for the relative success of percutaneous aponeurotomy with lipofilling (PALF), a minimally invasive technique that in a randomized controlled trial showed equivalent outcomes to limited fasciectomy at 1-year follow-up (Kan et al., 2016). In addition, adipose-derived stem cells have also been shown to suppress fibroproliferation and reduce contraction of DD-derived myofibroblasts (Verhoekx et al., 2013).
This study used nationwide data with known high data quality and established external validity, which ensured a large study sample with long and complete follow-up. Use of linked data within the registries also enabled a fuller picture of patient demographics to be generated and enabled a propensity score-matched study design. This led to the creation of well-balanced groups, and also balanced any censoring criteria. Moreover, the use of sensitivity analyses with increased control for confounding suggested that robust results were yielded. Finally, a matched unexposed patient started follow-up at the random cohort entry date within the same entry block as their matched exposed patient. Since this happened within the same entry block as the matched exposed patient (i.e. a maximum of 4 years apart of one another), bias was prevented through time trends in health care delivery, for example, through increasing frequency of bariatric surgery over time. This entry block design therefore helped to maximize comparability between matched patients.
Despite the rigorous methodology of this study, our results must be interpreted in the context of the following limitations. While patients with obesity unexposed to surgery were identified using the relevant ICD code, actual BMI measurements in unexposed patients were not available. Thus, there could be a potential baseline difference in BMI between the exposed and unexposed groups. However, we observed a higher prevalence of risk factors associated with obesity (e.g. type 2 diabetes, hypertension, cardiovascular disease) in the unexposed group before propensity score-matching, and all these risk factors were balanced after propensity score-matching. Thus, it is a credible assumption that BMI was also sufficiently balanced along with obesity-related conditions.
This study assessed the risk of DD following bariatric surgery in a large population-based registry. Our findings are consistent with previous epidemiological and genetic data demonstrating that obesity is protective in the patho-aetiology of DD (Hacquebord et al., 2017; Majeed et al., 2021; Major et al., 2019), a most unusual biological phenomenon. The latency of risk increase of DD after bariatric surgery may suggest that slowly adapting metabolic changes may be part of the mechanism of DD emergence. Future research should focus upon the interplay of DD with metabolic syndrome, in order to identify potential biological mechanisms and therapeutic targets.
Supplemental Material
sj-pdf-1-jhs-10.1177_17531934211062023 - Supplemental material for The association of bariatric surgery and Dupuytren’s disease: a propensity score-matched cohort study
Supplemental material, sj-pdf-1-jhs-10.1177_17531934211062023 for The association of bariatric surgery and Dupuytren’s disease: a propensity score-matched cohort study by Theresa Burkard, Jennifer C. E. Lane, Dag Holmberg, Anders Thorell, Andrea M Burden and Dominic Furniss in Journal of Hand Surgery (European Volume)
Supplemental Material
sj-pdf-2-jhs-10.1177_17531934211062023 - Supplemental material for The association of bariatric surgery and Dupuytren’s disease: a propensity score-matched cohort study
Supplemental material, sj-pdf-2-jhs-10.1177_17531934211062023 for The association of bariatric surgery and Dupuytren’s disease: a propensity score-matched cohort study by Theresa Burkard, Jennifer C. E. Lane, Dag Holmberg, Anders Thorell, Andrea M Burden and Dominic Furniss in Journal of Hand Surgery (European Volume)
Supplemental Material
sj-pdf-3-jhs-10.1177_17531934211062023 - Supplemental material for The association of bariatric surgery and Dupuytren’s disease: a propensity score-matched cohort study
Supplemental material, sj-pdf-3-jhs-10.1177_17531934211062023 for The association of bariatric surgery and Dupuytren’s disease: a propensity score-matched cohort study by Theresa Burkard, Jennifer C. E. Lane, Dag Holmberg, Anders Thorell, Andrea M Burden and Dominic Furniss in Journal of Hand Surgery (European Volume)
Footnotes
Acknowledgements
We thank Dr Jesper Lagergren (Karolinksa Institutet, Stockholm, Sweden) for hosting Dr Theresa Burkard for a research stay at the Upper Gastrointestinal Surgery Group and making the data available for use. Furthermore, we thank Dr Giola Santoni for her technical support and the professorship of AMB is partially supported by PharmaSuisse and the ETH Foundation.
Theresa Burkard and Jennifer C. E. Lane contributed equally to this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Versus Arthritis [21605] (JL), Medical Research Council Doctoral Training Fellowship [MR/K501256/1] (JL) and the Oxford NIHR BRC (DF), the Swiss National Science Foundation [Project Number: IZSEZ0_193622] (TB), the Erling-Persson Foundation [Grant Number 140604] (AT).
Ethical approval
Regional Ethical Review Board in Stockholm, Sweden approval (registration number 2020-04112).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
