Abstract
Objective:
This meta-analysis examined the relationship between excess body weight or body mass index (BMI) and risk of thyroid cancer. METHODS: PubMed®, MEDLINE®, EMBASE™ and Academic Search™ Premier databases were searched to identify cohort studies examining the effect of being overweight or obese on the risk of thyroid cancer. RESULTS: The inclusion criteria were met by seven cohort studies (total number of thyroid cancer cases, 5154). The pooled relative risk (RR) of thyroid cancer was 1.13 (95% confidence interval [CI] 1.04, 1.22) for overweight. Obesity was also linked with increased thyroid cancer risk in males and females, the strength of the association increasing with increasing BMI. The combined RR of thyroid cancer was 1.18 (95% CI 1.11, 1.25) for excess body weight (overweight and obesity combined). Being overweight was associated with a significant increase in thyroid cancer risk among non-Asians, but not among Asians. Overweight, obesity and excess body weight were all associated with papillary thyroid carcinoma risk. CONCLUSIONS: The association between overweight/ obesity/excess body weight and thyroid cancer risk was confirmed.
Introduction
The incidence of thyroid cancer has been growing throughout the world during recent decades. Thyroid cancer accounts for only ∼1% of solid organ malignancies (9% in women) and ∼0.5% of all cancer deaths, but is the most common endocrine-related malignancy.1– 3 Although thyroid cancer is more common overall in females, men are twice as likely as women to die from this cancer. 4 Early prevention of risk factors for thyroid cancer is therefore important.
The prevalence of overweight and obesity is increasing markedly, worldwide. There is convincing evidence of a positive association between overweight/obesity and increased risk of several malignancies including oesophageal adenocarcinoma, colorectal cancer and postmenopausal breast cancer.5,6 However, data provided by recent epidemiological studies are not sufficiently specific and detailed to determine the association between body mass index (BMI) and thyroid cancer risk,5,6 and the results of several earlier observational studies have been conflicting or inconclusive.7 – 9 The aim of the present meta-analysis was to examine the relationship between excess body weight or BMI and the risk of thyroid cancer in relevant published cohort studies.
Materials and methods
Study Methods
The methods used for this meta-analysis were in accordance with those proposed by the Meta-Analysis of Observational Studies in Epidemiology group. 10
Search Strategy
A systematic search of articles listed in electronic databases (PubMed®, MEDLINE®, EMBASE™ and Academic Search™ Premier), published between January 1980 and May 2012, was conducted using the following key words: thyroid AND (cancer OR carcinoma OR adenocarcinoma OR tumour OR neoplasm) AND (overweight OR obesity OR adiposis OR body mass index OR BMI OR Quetelet index). The list of articles was reviewed independently by two authors (Z.G.Z. and X.G.G.). In addition, a manual review of the references from primary or review articles was performed to identify any additional relevant studies. Articles in all languages were considered.
Study Selection
A study was included in this meta-analysis if it met the following criteria: (i) it was a cohort study; (ii) it reported an explicit description of BMI or excess body weight; (iii) data were available. A study was excluded if it met the following criteria: (i) it was conducted without the approval of the local ethics committee; (ii) it duplicated another study; (iii) data were incomplete. Unpublished reports or abstracts were not considered. After obtaining full reports of candidate studies, the same reviewers independently assessed each study for eligibility for inclusion in the meta-analysis. Differences between the two reviewers were resolved by discussion, and agreement on the final dataset was reached by consensus.
Data Abstraction
Data were extracted independently by two of the authors (Z.G.Z. and X.G.G.). All studies were assessed for quality using an extended tool initially developed by the United States Preventive Services Task Force (USPSTF). 11 The following characteristics were assessed: (i) initial assembly of comparable groups; (ii) maintenance of comparable groups; (iii) important differential loss to follow-up; (iv) measurements; (v) clear definition of interventions; (vi) all important outcomes considered; (vii) adjustment for potential confounders for cohort studies. Studies of high quality should meet all criteria. For each study, the following data were extracted: first author's last name; publication year; country where the study was performed; cohort size; study population; cancer diagnosis method; explicit description of BMI or obesity.
Exposure Definition
Body mass index is calculated as body weight in kg divided by the height in m2. BMI values were categorized as follows: normal range (18.5 ≤ BMI < 25 kg/m2); overweight (25 ≤ BMI < 30 kg/m2); obese (BMI ≥ 30 kg/m2); excess body weight (BMI ≥ 25 kg/m2). These groupings differed somewhat from the BMI categories used elsewhere in Asian populations: normal range (18.5 ≤ BMI < 23 kg/m2); overweight (23 ≤ BMI < 25 kg/m2); obese (BMI ≥ 25 kg/m2); excess body weight (BMI ≥ 23 kg/m2).
Statistical Analyses
Statistical analyses were performed using Stata® software (version 8.2; Stata Corp., College Station, TX, USA). A P-value < 0.05 was considered statistically significant for relative risk (RR). The combined RR and 95% confidence interval (CI) were used for analysis. Reported RRs and their corresponding SEs from individual studies were transformed into their logarithms, and the method of DerSimonian and Laird was used to quantify the significance of the effects of overweight/obesity/excess body weight on the risk of thyroid cancer under a random effects model. 12 Heterogeneity was checked using the χ2 and I2 tests. Several methods were used to evaluate the potential for publication bias. Visual inspection of asymmetry in funnel plots was performed. Begg's rank correlation method and Egger's weighted regression method were also used for statistical assessment of publication bias (P < 0.05 was considered representative of statistically significant publication bias).13,14
Results
The initial database search identified 680 papers. Of these, 11 papers describing cohort studies were identified for further evaluation after scanning the titles and abstract, and reading the entire article.8,9,15– 23 (Fig. 1). Four of the cohort studies did not report the available data or define body mass categories and therefore were excluded.15 – 18 Seven cohort studies, which included 5154 cases of thyroid cancer and a total studied population of 8 099411, were found to match the inclusion criteria.8,9,19– 23 All these studies were of high quality according to the USPSTF guidelines.
11
The major characteristics of the seven cohort studies are presented in Table 1, and their exposure and outcome definitions are presented in Table 2.
Flow diagram of quality of reporting of meta-analyses
Major characteristics of the seven cohort studies selected for a meta-analysis of the relationship between body mass index and risk of thyroid cancer

KNHIC, Korea National Health Insurance Corporation.
Exposure and outcome definitions of the seven cohort studies selected for a meta-analysis of the relationship between body mass index (BMI) and risk of thyroid cancer
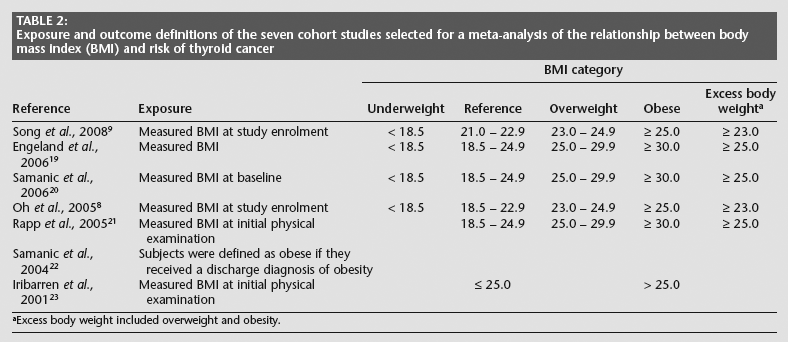
Excess body weight Included overweight and obesity.
The pooled results based on all studies demonstrated that there was a statistically significant association between BMI and thyroid cancer, and the strength of the association increased with increasing BMI (Table 3).
Statistical results for the meta-analysis of the seven selected cohort studies that analysed the relationship between body mass index and risk of thyroid cancer

Using the method of DerSimonian and Laird under the assumption of a random effects model. 12
Excess body weight included overweight and obesity.
RR, relative risk; CI, confidence interval; NS, not statistically significant (P ≥ 0.05).
A stratified analysis was undertaken according to sex. There was a statistically significant association between BMI and thyroid cancer risk for males (overweight, P = 0.007; obesity, P <0.001; excess body weight, P <0.001) and females (obesity, P = 0.002 excess body weight, P <0.001), except for the pooled RR for the overweight category for females. When stratifying for race, the positive association was similar among non-Asians and Asians, except for the pooled RR for the overweight category for Asians. The analysis demonstrated that overweight, obesity and excess body weight were all associated with an increased risk of papillary thyroid carcinoma.
There was no evidence of publication bias as the funnel plot of the cohort studies that evaluated excess body weight (overweight and obesity) and thyroid cancer risk was not asymmetrical (Begg's rank correlation method, P = 0.45) (Fig. 2) and Egger's weighted regression method also confirmed the lack of publication bias (Egger's test, P = 0.56) (Fig. 3).
Begg's funnel plot for publication bias (with 95% pseudoconfidence limits) of the cohort studies that investigated excess body weight (overweight and obesity) and thyroid cancer risk (P = 0.45). OR, odds ratio; SE, standard error Egger's publication bias plot of the cohort studies that investigated excess body weight (overweight and obesity) and thyroid cancer risk (P = 0.56)

Discussion
Several studies and meta-analyses have shown that increased BMI is associated with an increased risk of several cancers in adults.6,24– 29 The present meta-analysis therefore pooled the results of previous studies that investigated the association between BMI and the risk of thyroid cancer.14– 20 The results described above extend previous observational findings by permitting additional evaluation of subgroups (e.g. by sex, race and pathological type). In accordance with the results of a previous study, 24 the present study demonstrated an association between BMI and thyroid cancer risk. The strength of the association increased with increasing BMI, in the pooled studies.
When stratifying for sex and race the results were similar, except that being overweight had no significant effect on the risk of thyroid cancer for either females or Asians. The following considerations may partly explain these results: (i) the use of different BMI reference values produced different prevalence estimates for the overweight category in the different populations; (ii) compared with Caucasians, Asians tend to have smaller body frames, and the mean BMI and the prevalence of excess body weight in Asian countries were once lower than those in Western countries;8,9,30–31 (iii) the prevalence and incidence of thyroid cancer vary among countries around the world,32,33 and the effect of BMI on the development of thyroid cancer in Asians might differ from that in non-Asians; (iv) although the difference was not statistically significant (RR 1.09, 95% CI 1.00, 1.20, P = 0.06) for overweight females, it almost reached the usual 0.05 level of significance and the result should be interpreted with caution.
The following mechanisms have been suggested for the effect of excess body weight on thyroid cancer. First, potential biological mechanisms for the association include increased levels of endogenous hormones (steroid hormones, adipokines, oestrogens, insulin and insulin-like growth factor-1) in overweight and obese individuals.7,24,28,34 Secondly, oxidative stress and the nuclear factor κB system are among the mechanisms or factors involved in the development of thyroid cancer.24,28,35,36 Thirdly, the volume of the thyroid increases with increasing BMI, and consequently it could harbour more cells at risk of mutation in persons with higher BMIs, leading to malignancy.37– 39 It has, however, been reported that elevated recreational physical activity could potentially affect thyroid cancer incidence rates. 40 Other lifestyle variables, including consumption of fruit, raw vegetables and tea, have also been found to be associated with a reduced risk of malignant thyroid tumours.41,42
One of the most important advantages of this meta-analysis was that it was restricted to cohort studies, which are less prone to recall and interviewer biases that may affect case–control studies of associations between BMI and the risk of thyroid cancer. However, this meta-analysis does have several limitations and potential biases. First, although no evidence of publication bias was observed, with nonsignificant Begg's and Egger's test results, other biases from observational studies might have distorted the results. To minimize this problem, every effort was made to collect suitable studies, and explicit methods were designed for study evaluation and data extraction. Secondly, some potential biases might exist in this analysis because of the unfavourable geographical distribution of the seven cohort studies (two studies were performed in Korea). Thirdly, anthropometric factors other than increased BMI (e.g. waist-to-hip ratio, waist circumference) might be better measures of adiposity in relation to thyroid cancer risk. 24 Fourthly, anthropometric measurements at the initial physical examination could not be used to assess changes during follow-up. Finally, this analysis did not take the underweight BMI category into account, in the interpretation of the results.
In conclusion, the findings of this meta-analysis suggested that overweight, obesity and excess body weight were all associated with an increased risk of thyroid cancer. However, in subgroup analyses based on sex and race, there was no statistically significant association between overweight and cancer risk, either in women or in Asians. The mechanisms that link excess body weight with thyroid cancer risk should be further evaluated.
Footnotes
Conflicts of interest: The authors had no conflicts of interest to declare in relation to this article.
