Abstract
Background:
Critically ill patients are at risk of bleeding from stress ulcers. Comprehensive information regarding United Kingdom stress ulcer prophylaxis (SUP) practices are not available and may change over time. We aimed to describe SUP practices in 2020 and reevaluate the position in 2024.
Methods:
Critical care pharmacists provided observed SUP practice data for UK adult critical care units via an electronic repository in 2020 and 2024. One response was accepted from each critical care unit at each time point. Data collected included trigger criteria for commencing SUP, primary medication class used, primary SUP cessation criteria, and level of nutritional intake (if part of cessation criteria).
Results:
There were high response rates of 70.3% (2020) and 66.7% (2024) of registered UK adult critical care units. Few differences in primary SUP trigger criteria between 2020 and 2024 were seen, with small differences in the categories of ‘SUP not used’ (p = 0.002) and ‘Shock’ (p = 0.027) driving statistical significance (χ2(7, 454) = 16.76, p = 0.019). There was a significant change in the primary medication class used for SUP (H2 receptor antagonist 49.4% 2020, vs 0.4% 2024, proton pump inhibitor 44.7% 2020 vs 97.8% 2024; χ2(2, 458) = 159.62, p < 0.001). Primary SUP cessation criteria was ‘Patient fed’ (66.8% 2020, 64.6% 2024), with most describing this threshold as met when the patient receives full enteral feed (72.0% 2020, 78.8% 2024).
Conclusion:
The UK has moved towards proton pump inhibitors as the primary SUP medication class. SUP is most frequently discontinued on establishment of enteral nutrition.
Introduction
Critically ill patients are at risk of clinically important gastrointestinal bleeding (GIB) events from stress ulcers. 1 Medicines modifying gastric acid effects are administered to patients as prophylaxis to reduce this risk, a practice recommended by international guidelines.2,3 The primary pharmacological agent used for stress ulcer prophylaxis (SUP) has changed over time. Initially high dose antacids were used, and as newer medicines have been made available the medicine of choice has changed. Sucralfate and H2-receptor antagonists (H2RA) initially replaced antacids, and then proton pump inhibitors (PPI) became widely used.4,5 Globally, SUP practice has favoured PPIs, however United Kingdom practice continued to be divided between H2RAs and PPIs. 6
A large pragmatic trial was conducted to compare SUP outcomes between H2RAs and PPIs. The PEPTIC trial randomised critical care units to a pharmacological SUP strategy of ‘PPI first line’ or ‘H2RA first line’ and looked for effects on outcomes such as clinically important bleeding events, Clostridioides difficile infection, and mortality. 7 Given the apparent split in dominant SUP practices within UK practice, 6 this trial was expected to provide important information with direct relevance to UK practice. An opportunity existed to comprehensively understand UK critical care SUP practice, and to explore any potential effect on practice seen following the publication of PEPTIC. We therefore aimed to document UK SUP practice and describe any changes to practice after the publication of PEPTIC.
Method
The study was registered as a service evaluation with the Oxford University Hospitals NHS Trust (5758/9370).
An electronic platform was used to capture data about usual practices surrounding SUP in UK critical care units (SurveyMonkey®). Participants were asked to contribute data regarding indications for SUP, medications used for SUP, and usual SUP cessation criteria, with a supplementary question to elucidate further descriptions for ‘enterally fed’ if this was relevant.
Links to the platform were distributed to critical care pharmacists via professional networks (UK Clinical Pharmacy Association, Critical Care Operational Delivery Networks) and personal professional contacts from 11th November 2019 until 16th January 2020 (2020 cohort), and 13th May 2024 until 4th September 2024 (2024 cohort).
Critical care pharmacists were targeted because they can provide summary information about medication usage they observe in their routine critical care practice.8,9
One response was accepted from each UK critical care unit. Duplicate entries were reconciled. Entries containing complete data were preferentially retained. Any remaining discrepancies were resolved directly with respondents as necessary.
The denominator was set as all adult critical care units participating in the 2019 Intensive Care National Audit & Research Centre (ICNARC) Case Mix Programme (for England, Wales, and Northern Ireland) and 2019 Scottish Intensive Care Society Audit Group (SICSAG) quality reports, excluding obstetric units and independent (non-NHS) units.10,11
Response statistics were reported back periodically via the same professional networks initially used for distribution. Regional coordinators promoted the study within their NHS region and liaised with participants where necessary.
Data were analysed using Microsoft Excel 365 MSO v2407 (Microsoft Corporation 2024) to produce descriptive statistics and figures. Comparison of proportions in results between survey periods used the Chi-square test. Significance was defined as p < 0.05 and where detected, standardised residuals were calculated to identify factors associated with significant differences.
Results
Data were received for 235 adult critical care units for the 2020 cohort. There were 234 responses from the 333 eligible ICNARC/SICSAG registered units yielding a response rate of 70.3%. There was one additional response from a non-registered UK unit.
For the 2024 cohort, there were responses from 226 adult critical care units. There were 218 responses from the 327 eligible ICNARC/SICSAG units, giving a response rate of 66.7%. There were 8 additional responses from non-registered UK units.
Responses from non-registered UK critical care units were included in analyses. A cursory review of data from non-registered units did not identify any obvious indicators of practice differences from the registered cohort.
Mechanical ventilation, prior history of gastrointestinal bleeding, or being admitted to critical care (i.e. ‘all patients’) were the primary indications given for commencing SUP (Figure 1).
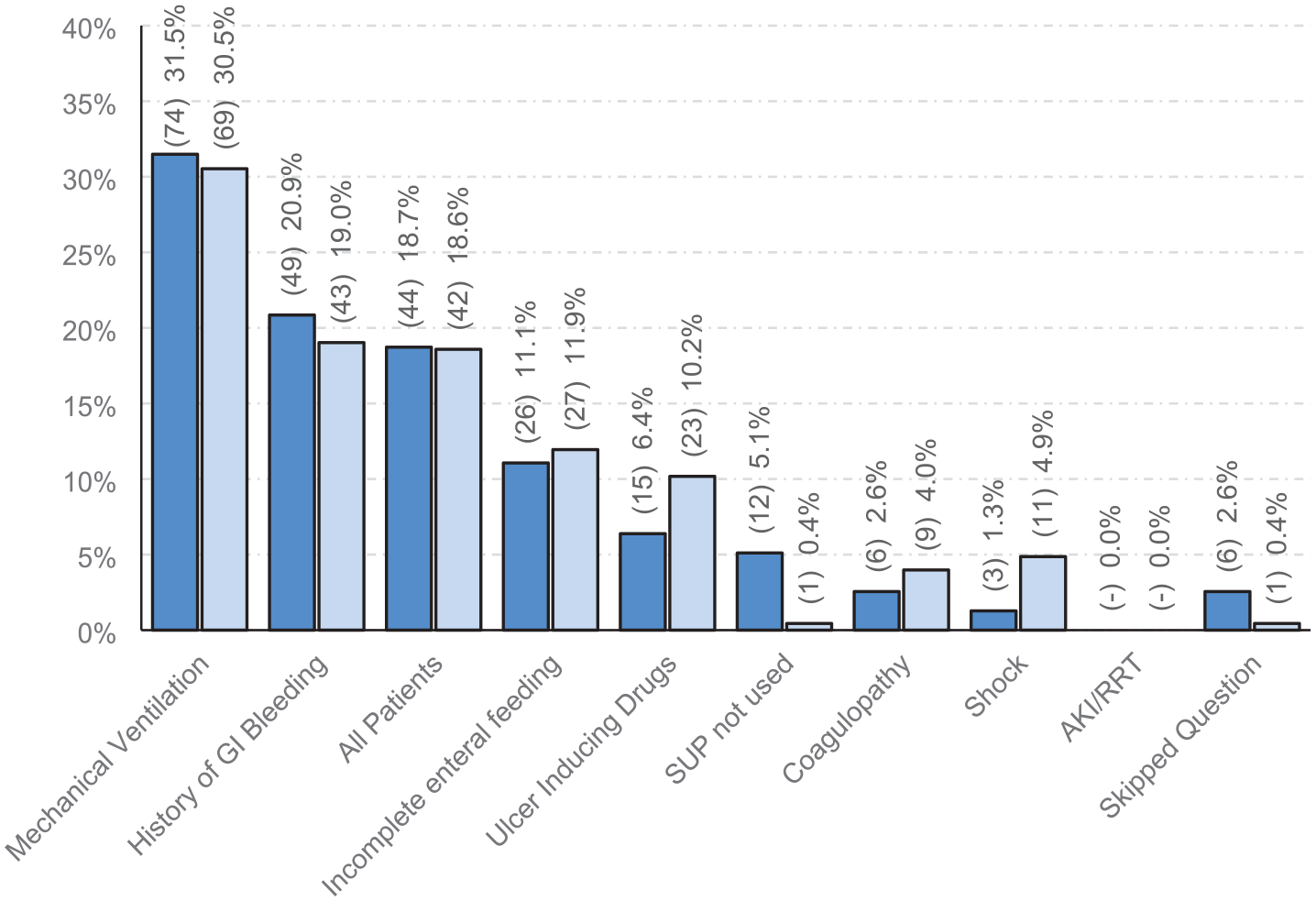
Primary trigger for commencing pharmacological stress ulcer prophylaxis in UK adult critical care units. Dark blue: 2020 cohort (n = 235). Light blue: 2024 cohort (n = 226). χ2(7, 454) = 16.76, p = 0.019 (null returns excluded). Adjusted residuals were significant for ‘SUP not used’, p = 0.002, and ‘Shock’, p = 0.027.
The use of H2RAs and PPIs were balanced in the 2020 cohort, there was a clear dominance of PPI use in the 2024 cohort (Figure 2).
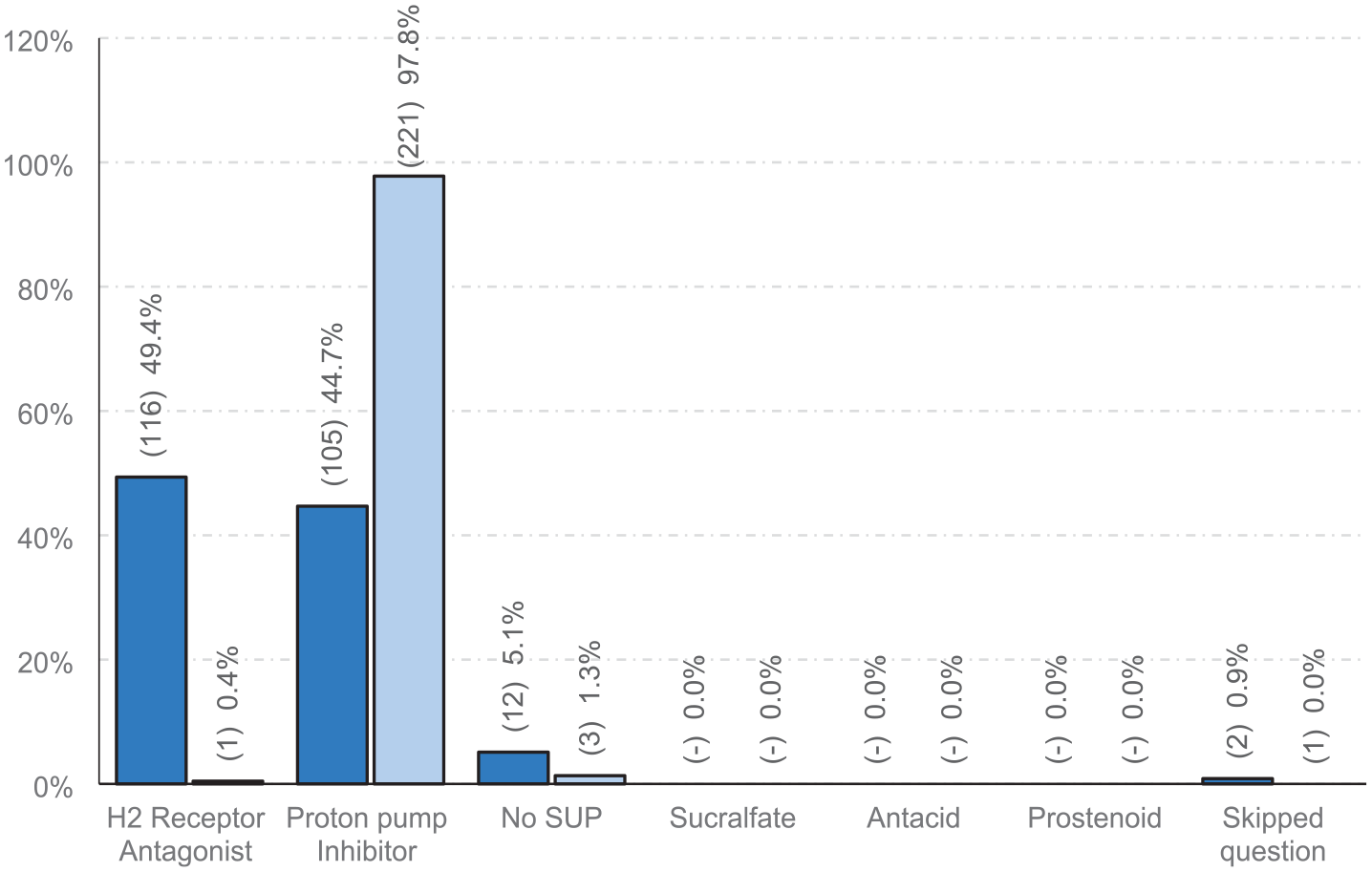
Primary medication class used for stress ulcer prophylaxis in UK adult critical care units. Dark blue: 2020 cohort (n = 235). Light blue: 2024 cohort (n = 226). χ2(2, 458) = 159.62, p < 0.001 (null returns excluded). Adjusted residuals were significant for ‘H2 Receptor Antagonist’, p < 0.001, ‘Proton Pump Inhibitor’, p < 0.001, and ‘No SUP’, p = 0.022.
The main reason for discontinuing pharmacological SUP was receipt of enteral nutrition (EN) in both cohorts, some patients have SUP discontinued on discharge from critical care, and a minority of units reported that SUP was not stopped (Figure 3).
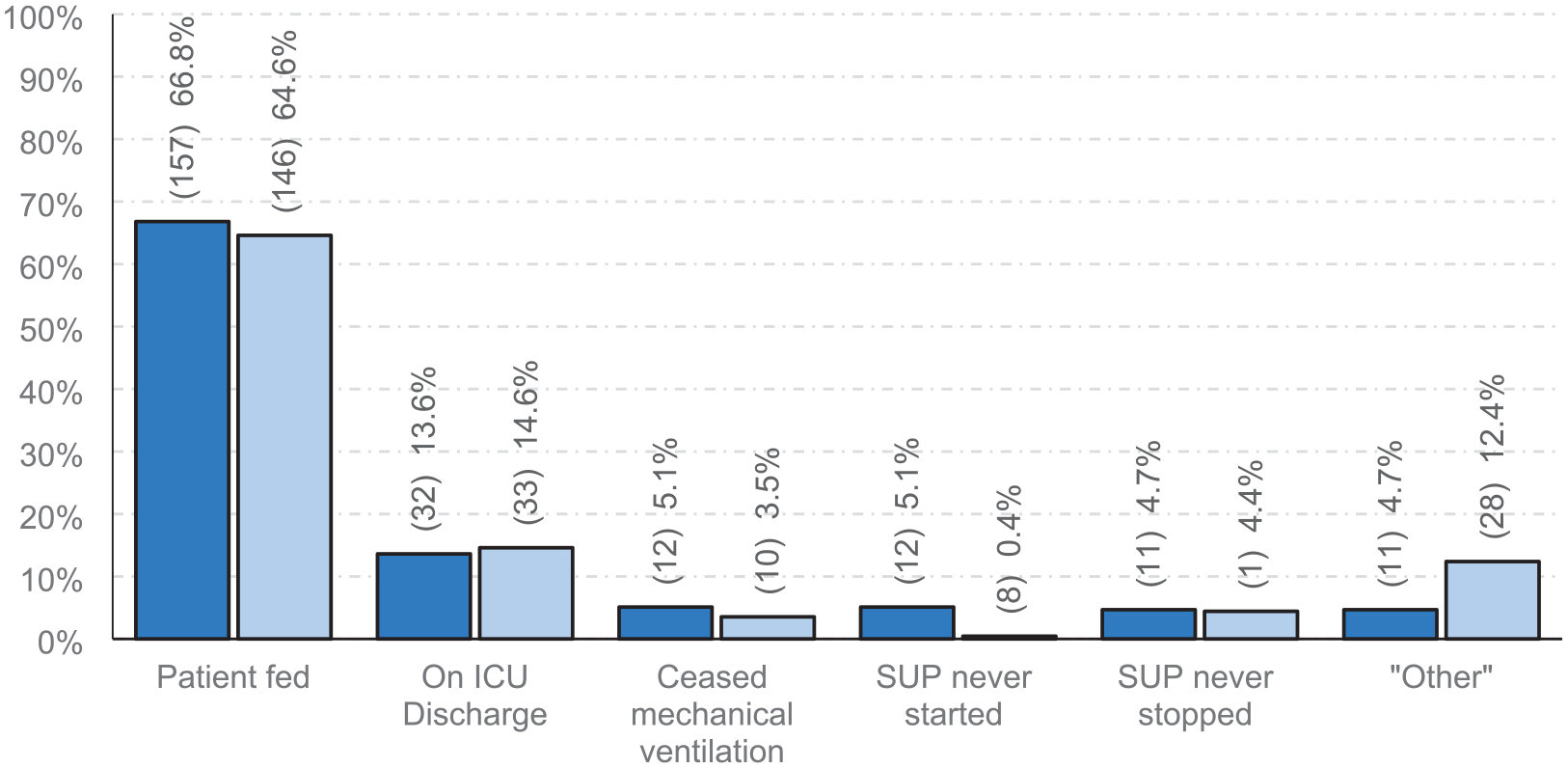
Primary criteria used for ceasing stress ulcer prophylaxis in UK adult critical care units. Dark blue: 2020 cohort (n = 235). Light blue: 2024 cohort (n = 226). χ2(5, 461) = 17.81, p = 0.003. Adjusted residuals were significant for ‘SUP never started’, p = 0.002, and ‘Other’, p = 0.003.
When SUP was discontinued because of establishment of EN, most critical care units stopped when the patient attained full EN, with approximately one in four critical care units accepting lower exposure to EN as stopping criteria (Figure 4).
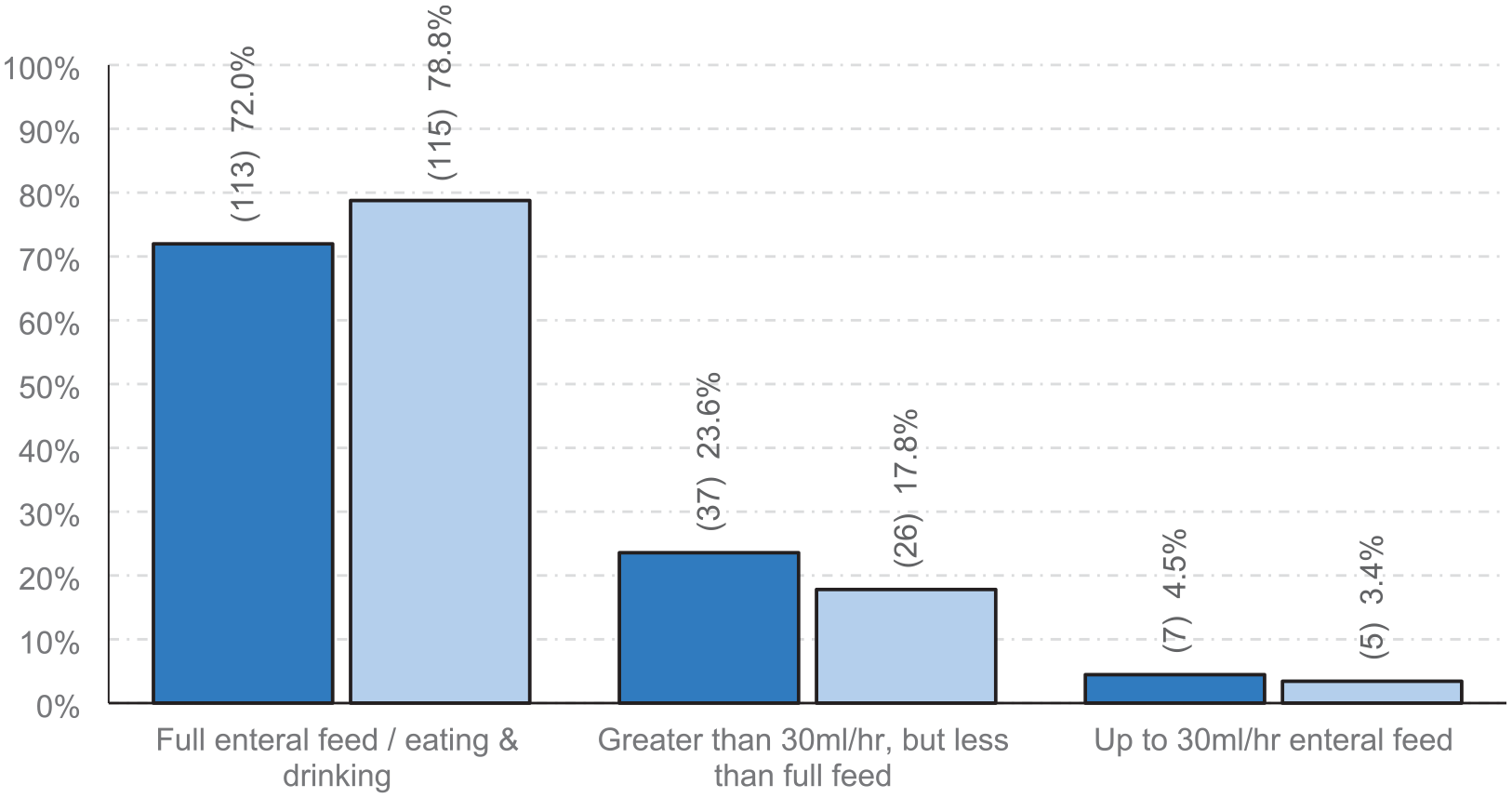
‘Patient fed’ criteria used for ceasing stress ulcer prophylaxis in UK adult critical care units. Dark blue: 2020 cohort (n = 157). Light blue: 2024 cohort (n = 146). χ2(2, 303) = 1.87, p = 0.392.
Discussion
This study provides comprehensive information about SUP practices and patterns within UK adult critical care units. The response rate is high, giving confidence to the generalisability of these data to wider UK practice. Comparison of UK SUP practices between 2020 and 2024 identified a widespread change in SUP medication away from H2RA and towards PPIs. Other SUP practices remained relatively consistent between the periods.
A variety of reasons are reported as the primary trigger to commence SUP. Whilst there is a statistically significant difference between 2020 and 2024, this is driven by numerically small changes in the ‘SUP not used’ and ‘Shock’ categories. We did not specifically ask about the rationale behind trigger indications for prescribing SUP and so do not know what has prompted these changes. PEPTIC is the only major trial published between the two surveys, and this did not specifically study trigger criteria. PEPTIC was a cluster randomised clinical trial that compared the effect of preferentially using PPIs versus H2RAs for stress ulcer prophylaxis on in-hospital mortality in 50 critical care units (almost 27,000 patients). No statistically significant difference for in-hospital mortality was found (PPI 18.3%, H2RA 17.5%, p = 0.054), although post-hoc exploratory analyses showed heterogeneity in relation to illness severity. For the secondary outcome of clinically important gastrointestinal bleeding, PPIs conferred an overall lower risk than H2RA (PPI 1.3%, H2RA 1.8%, p = 0.009). Study medication was neither blinded nor fixed, cross over of study medication may have affected results.
It is notable that nearly a fifth of units in both 2020 and 2024 gave SUP to all patients, these units are unlikely to be applying an individual risk-based approach to SUP prescribing. New SUP specific guidelines strongly promote a risk-based approach, meaning the UK pattern may change in future as these guidelines become integrated into practice. 3
In 2007, 20% of UK critical care units used PPI first line for SUP, 12 this increased to 29% in the UK section of a 2015 international survey. 6 In our survey, the 2020 cohort shows a continued shift towards PPI as the first line SUP agent (44.7%), before almost universal use in the 2024 cohort (97.8%).
International perspectives on SUP recommendations have also changed over time. The Surviving Sepsis Campaign initially favoured H2RA in 2004, giving equivalence to H2RA or PPI in 2008, then favouring PPI in 2012.13 –15 This stance has reversed more recently with equivalence between H2RA and PPI suggested again in 2017, 16 and in the most recent 2021 version the strength of recommendations in support of SUP were downgraded from ‘strong’ to ‘weak’, with no mention made regarding preferred medication class. 2
The 2020 cohort provides data about UK SUP practice just prior to the publication of the PEPTIC. 7 In contrast to UK practice, the use of PPIs for SUP was highly dominant outside of the UK. 6 Whilst the results of PEPTIC had the potential to directly inform and affect UK SUP medication choice and practice, the withdrawal of all presentations of the H2RA ranitidine in 2020 will have had a major effect on practice independently of PEPTIC. 17 The withdrawal was prompted because ranitidine presentations were found to contain a probable carcinogen,18,19 and this left a significant gap in medicines availability. No other UK licenced parenteral H2RA was readily accessible, and it is perhaps not surprising that the critical care community turned to PPIs to fill the gap. Two large low risk of bias RCTs have reported that PPIs reduce clinically important GIB.20,21 The results of one of these, the Stress Ulcer Prophylaxis in the Intensive Care Unit trial (SUP-ICU) were available prior to the withdrawal of ranitidine. SUP-ICU reported no change in 90-day mortality when pantoprazole was compared with placebo for stress ulcer prophylaxis (RR 1.02, 95% CI 0.91–1.13, p = 0.76), and a reduced risk of clinically important GIB (RR 0.56, 95% CI 0.40–0.86). The Re-Evaluating the inhibition of Stress Erosions trial (REVISE) also reported a reduction in clinically important GIB with pantoprazole (HR 0.30, 95% CI 0.19–0.47, p < 0.001), with no effect on 90-day mortality (HR 0.94; 95% CI 0.85–1.04, p = 0.25). The results of REVISE were published after the majority of our second cohort had reported practice in 2024. Taken together, SUP-ICU and REVISE provide high certainty evidence that PPIs reduce clinically important GIB in critically ill patients. 22 However, there remains some concern over the potential for PPIs to adversely affect mortality, with a meta-analysis reporting moderate credibility evidence of increased mortality in sicker patients. 22
Our data show that enteral feed provision impacted on SUP use. Incomplete enteral feeding was considered a trigger for SUP in approximately 10% of units, and almost two-thirds of units reported that the main reason for stopping SUP was provision of EN – this was the most common reason cited for discontinuing SUP. EN is thought to have protective effects against clinically important GIB from stress ulcers, however direct data to robustly confirm this are still needed.3,23,24 There are associations between the provision of EN and lower risk of clinically important GIB, but there are no trials where GIB is investigated as a primary or secondary outcome where EN exposure has been randomised.3,24
Recent RCTs did not use EN targets to discontinue SUP. Instead stopping criteria were based on step down from critical care (SUP-ICU), 20 cessation of mechanical ventilation (REVISE), 21 or a maximum of 90 days. The most recent international guidelines suggest administering EN to reduce GIB, and additionally suggest not commencing SUP in critically ill patients with a low risk for clinically important GIB. 3
About a fifth of units in both cohorts reported that SUP was ceased on ICU discharge or not specifically ceased at all. Risks associated with newly initiated PPI therapy in ICU that are then continued without an appropriate indication have been recently highlighted by a large German population cohort study, where such patient exposure was associated significantly with higher rates of pneumonia, cardiovascular events, rehospitalisation, and 2-year mortality. 25 Deprescribing strategies are therefore required as a key care component to avoid unnecessary harm from inappropriate continuation of SUP or other medications after step down from critical care. 26 New international guidelines recommend stopping SUP when risk factors are no longer present and before transfer out of ICU. 3
Strengths of this study include the high response rate, and the use of critical care pharmacists to describe and summarise usual practice seen rather than reporting personal beliefs, although it is possible that some recorded personal beliefs instead. Data acquisition for the 2020 cohort was completed before PEPTIC was published, although a small amount of data for the 2024 cohort were gathered after REVISE was published. The original intention was to investigate possible changes in UK SUP practices brought about by the results of PEPTIC, which were not known at the time of inception. Instead, this study has largely captured the effect of the unexpected withdrawal of ranitidine, although remaining data points about primary trigger criteria, primary cessation criteria, and a broad description of what is meant by ‘patient fed’ if used in cessation criteria, are unlikely to have been affected.
Overall, indications for prescribing SUP have not changed notably from 2020 to 2024 and include an element of blanket prescribing of SUP in all admitted patients. There has been a significant shift towards using PPIs after the publication of PEPTIC, although the change likely results from the international withdrawal of ranitidine. Enteral feeding is the dominant stopping criteria in use despite a lack of direct evidence to inform this practice. In cases where SUP is stopped on ICU discharge or not stopped at all, robust deprescribing processes are needed to reduce the potential for longer term patient harm.
Footnotes
Acknowledgements
The authors would like to acknowledge Dr Sarah Carter of the United Kingdom Clinical Pharmacy Association for help and assistance in running the study.
Declaration of conflicting interests
Greg Barton MBE receives funding for his role as an Editor-in-Chief for Critical Illness (Pharmaceutical Press). Dr Richard Bourne is funded by a National Institute for Health and Care Research (NIHR) Senior Clinical and Practitioner Research Award (NIHR304524). The views expressed in this publication are those of the author(s) and not necessarily those of the NHS, the National Institute for Health and Care Research or the Department of Health and Social Care. All other authors have declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
