Abstract
Trace elements are required in minute quantities in the diet but play a vital role in a wide variety of functions, such as co-factors in antioxidant reactions and normal immune function to DNA and protein synthesis and skeletal and tissue remodelling and repair. Critically ill patients are at risk of trace element deficiency or excess, due to changes in intake, absorption, metabolism or excretion. Deficiency or excess can lead to a wide range of cellular and organ dysfunction that may be seen in patients with an acute or critical illness, including cardiomyopathy, impaired glucose tolerance and reduced oxygen delivery. In addition, various diseases, such as systemic inflammation and renal and intestinal failure, and intensive care treatments, such as parenteral nutrition, renal replacement therapy and diuretics, can increase the likelihood of deficient or excessive amounts of micronutrient levels. This narrative review discusses sources and normal physiology of trace element handling and how this may be impaired in critically ill patients. It then discusses various conditions seen in critically ill patients that may be caused or exacerbated by abnormal trace element status and the current evidence around whether supplementation is of benefit in particular critical illnesses.
Introduction
Critically ill patients are often unable to obtain adequate oral intake of nutrition to meet requirements for prolonged periods due to a variety of reasons including frequent interruptions to feeding, gastrointestinal dysfunction such as poor absorption or following surgery, being prevented from eating due to aspiration risk, and increased energy expenditure. Nutritional support to critically ill patients can be delivered via enteral or parenteral routes and in broad terms, nutrients can be subclassified into macronutrients and micronutrients. Macronutrients include substances required by humans in large quantities, 1 such as fats, proteins and carbohydrates, whereas micronutrients comprise vitamins and minerals, which are present in much smaller quantities and do not significantly contribute to caloric intake but are nonetheless integral to a wide range of cellular and systemic functions. Both enteral and parenteral preparations typically contain micronutrients. 2
Minerals can be further classified into macro-minerals, trace elements (TE) and ultra-trace elements, depending on the amount required or present in the organism. The definition of a TE varies between sources; some definitions are based on the amount required per day 3 (see Table 1); the World Health Organization (WHO) defines a TE as one whose concentration is less than 250 µg/g, 4 or around 20 g in the average adult.
Table to show classification of minerals by daily amount required. 3

At least 19 TEs in the human body have been identified. The WHO divides them into essential elements, such as iodine and zinc, probably essential elements, such as manganese and silicon, and potentially toxic elements, such as fluoride, lead, cadmium and mercury, which nevertheless may also have some essential functions at low levels. 4
This narrative review discusses how critically ill patients may have altered absorption, metabolism and elimination, which may affect the concentrations of essential TEs. Various syndromes seen in critical illnesses, such as systemic inflammation and multi-organ dysfunction syndrome, can lead to deficiency or excess. In addition, certain critical care treatments, such as renal replacement therapy (RRT) and diuretics, can further exacerbate TE derangement.
The review then discusses some of the clinical features of deficiency and toxicity of these TEs, which in critical illness can include life-threatening sequelae such as arrhythmias, heart failure, neuropathy or predisposition to severe infections, along with more insidious presentations such as endocrine abnormalities, alterations in enzyme function, poor immune function, oxidative stress and impaired wound healing. Finally, the review discusses some of the clinical recommendations concerning measuring and replacing TEs, and whether or when supplementation should occur in critically ill patients.
Macronutrients and vitamins are not within the scope of this article. It is not intended as a definitive guide. Iron is excluded from this review, as it has been extensively elsewhere. 5
Various terms used in this article have specific definitions. The European Society for Clinical Nutrition and Metabolism (ESPEN) define depletion of a TEs as a level below recommended amounts but not sufficient to cause clinical signs or symptoms, and deficiency as a level sufficient to cause clinical signs or symptoms. Similarly, the terms overdose and toxicity define blood levels above the recommended levels without or with clinical signs or symptoms respectively. 2
Provision and bioavailability of trace elements in critical illness
This section discusses the sources and the handling of TEs by the body. Critical illness can affect absorption, metabolism, distribution and elimination of TEs, increasing or reducing total body amounts.
Sources of trace elements
General population
Adequate intake of TEs generally occurs through consuming a healthy and varied diet (see Table 2). In some cases, particular foods contain significant amounts of a TE: one 100 g portion of oysters for example contains around five times the recommended daily intake (RDI) of zinc and copper for adult males, 6 and one average Brazil nut contains more than the RDI of selenium. 6
Absorption from sources other than diet may also contribute. Oil workers may be exposed to higher levels of manganese, zinc, chromium and cobalt,8,9 and individuals living near mines may have higher systemic levels of cobalt and copper. 10 Increased serum levels of chromium and cobalt have been found after hip replacements; 11 the latter may be sufficient to cause difficulties with memory and concentration, and breathlessness and heart failure. 12 And while chromium is found in a wide variety of foods, most dietary chromium is reported to be derived from the processing of food with stainless steel equipment. 13
Enteral and parenteral nutrition
Most critically ill patients require supplemental nutrition to augment or replace their oral intake Parenteral nutrition (PN) was first used in the 1960s and is now used in around 30% of patients in ICU. 14 TEs were first added to PN in 1979 by the Nutrition Advisory Group of the American Medical Association after many occasions of TE deficiencies, especially zinc, copper, manganese and chromium, were observed in patients. Selenium was added a few years later.15,16 However, administration of PN may provide supranormal or toxic levels of certain TEs15,16 if provided in quantities greater than that required. Even without the intentional addition, PN often contains measurable levels of TEs, including those that may be toxic or non-essential. 17 Contamination may increase the amount of chromium for example delivered to the patient by up to 100%, 18 and is associated with liver and renal failure. 18 There is ongoing debate about whether certain TEs, such as boron and silicon, should be added to PN 19 and their precise physiological role. Table 3 shows the ESPEN current recommendations for TEs in enteral nutrition (EN) and PN in patients who are critically ill, hypermetabolic or receiving RRT.
Recommended daily intake of various trace elements and recommended supplementation of parenteral and enteral nutrition.1,20 There is no current guideline for RDI for cobalt, since it is a major component of vitamin B12 which does have a recommended intake set by ESPEN. ESPEN advises that EN and PN do not require additional cobalt supplementation as long as vitamin B12 is supplemented. 2
Other potential sources in the critically ill
Iodine absorption is associated with iatrogenic causes such as contrast media use, topical iodine disinfectants and amiodarone. 2 Significant amounts of iodine can be absorbed transcutaneously from iodine-based skin disinfectants, although this may reduce with the move to using chlorhexidine. 21
Each molecule of amiodarone contains two iodine ions, (compared with thyroxine and triiodothyronine (four and three iodine ions respectively). Every 200 mg of amiodarone contains 75 mg of iodine, of which 10% (7.5 mg) of the iodine is released as free iodide daily. 22 Amiodarone is very lipophilic and accumulates in adipose tissue, cardiac and skeletal muscle, and the thyroid gland. With long-term treatment, there is a 40-fold increase in plasma iodide levels, 22 and a long elimination half-life, of up to 77 days. 23 Hyperthyroidism is therefore a common complication of amiodarone treatment, affecting 8% of patients after 3 years. 24 Hypothyroidism affects 6% of patients after 3 years. 24
Absorption
Normal physiology
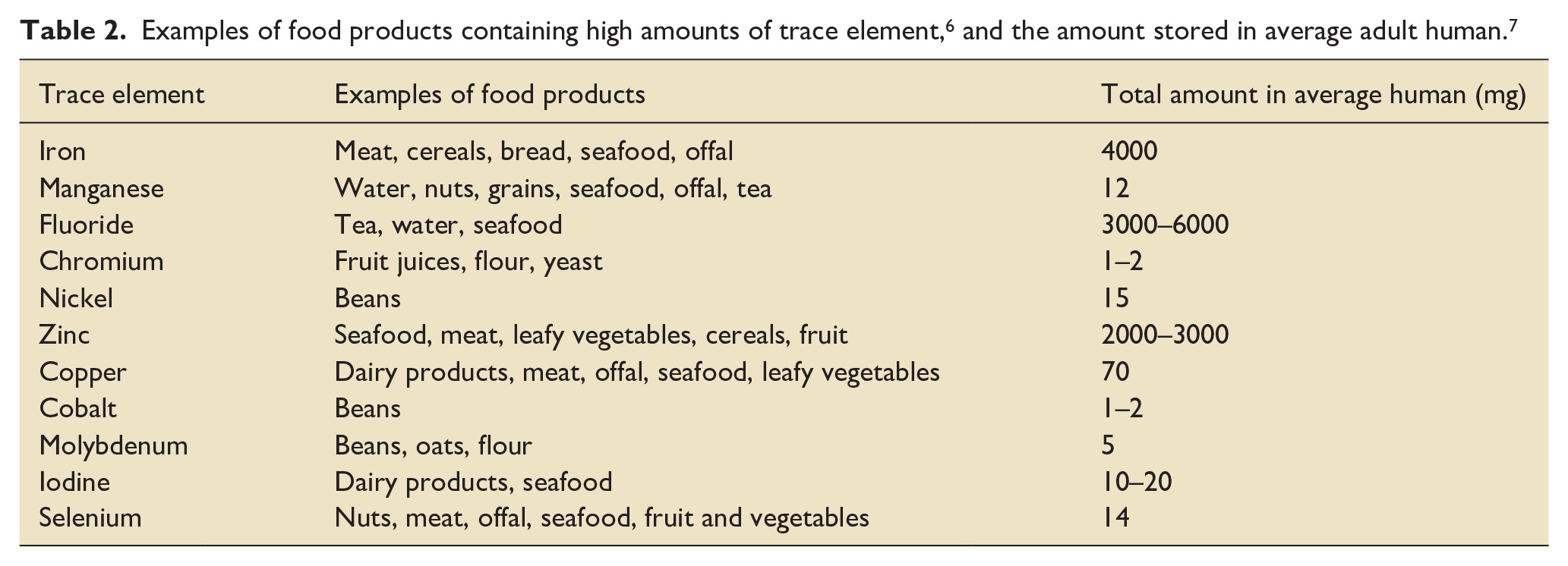
TEs are primarily absorbed in the proximal small bowel (see Figure 1), although absorption can occur in other parts of the gastrointestinal tract. Selenium can be absorbed in the colon and caecum as well as the duodenum, 25 and some absorption of copper 26 and fluoride occurs in the stomach.
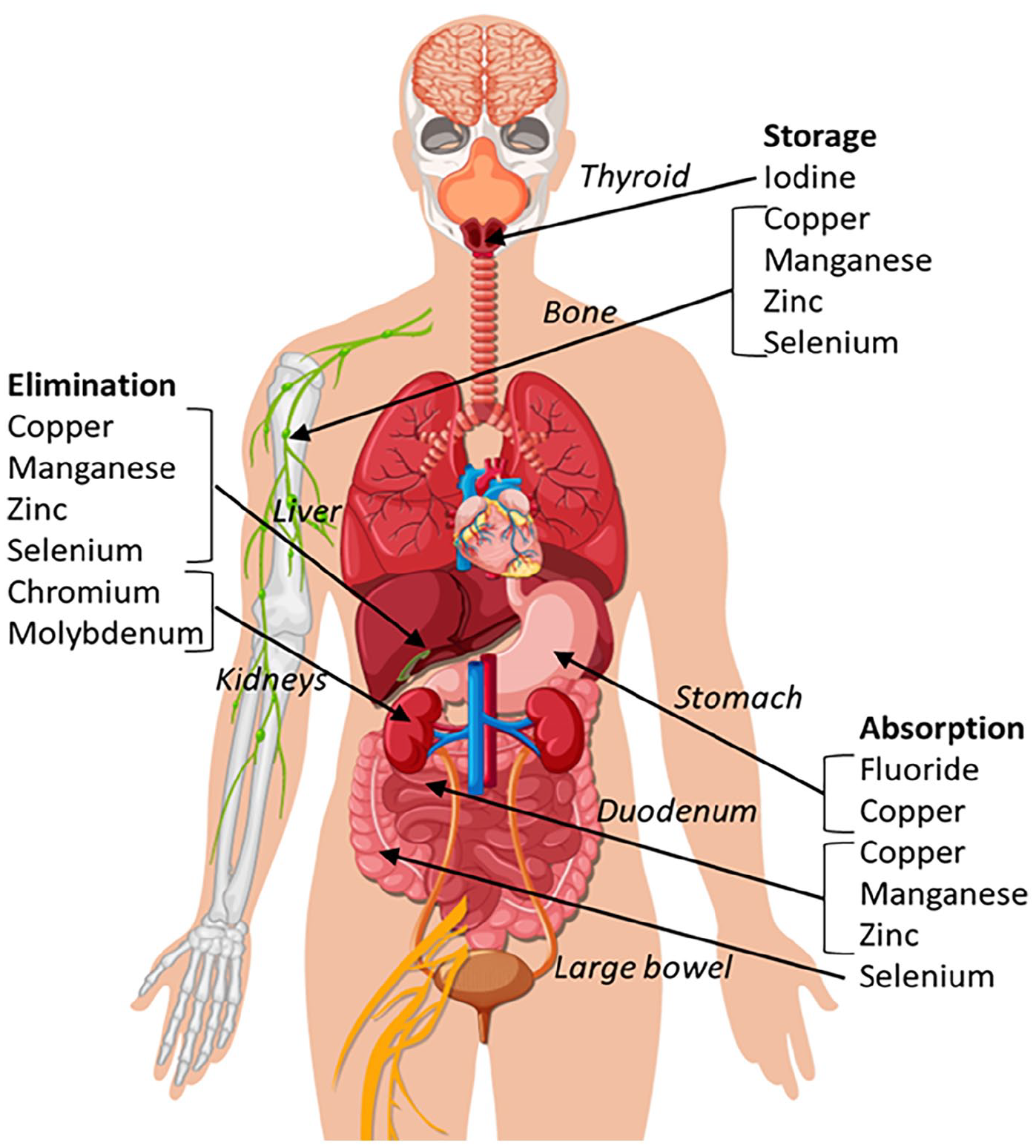
Commonest sites of absorption, storage and elimination of selected trace elements. See text for details.
The amount of TE absorbed varies significantly. Chromium absorption ranges from 0.4% to 2.5%, 27 whereas around 90% of dietary selenium 28 and molybdenum 2 is absorbed. Absorption can be affected by or dependent on the presence or concentrations of other substances. The presence of vitamin C, iron and zinc can reduce copper absorption. 29 Molybdenum acts as an antagonist to copper absorption; high serum molybdenum levels can be sufficient to cause copper deficiency. 30 Absorption of fluoride in the stomach is dependent on acidity of the gastric fluids – faster absorption occurs with a more acidic pH. 31 Cobalt and manganese absorption is inversely related to the degree of iron absorption due to competitive antagonism at the DMT1 transporter; 32 cobalt absorption is directly related to the degree of vitamin B12 absorption. Absorption occurs via different mechanisms (see Figure 2).
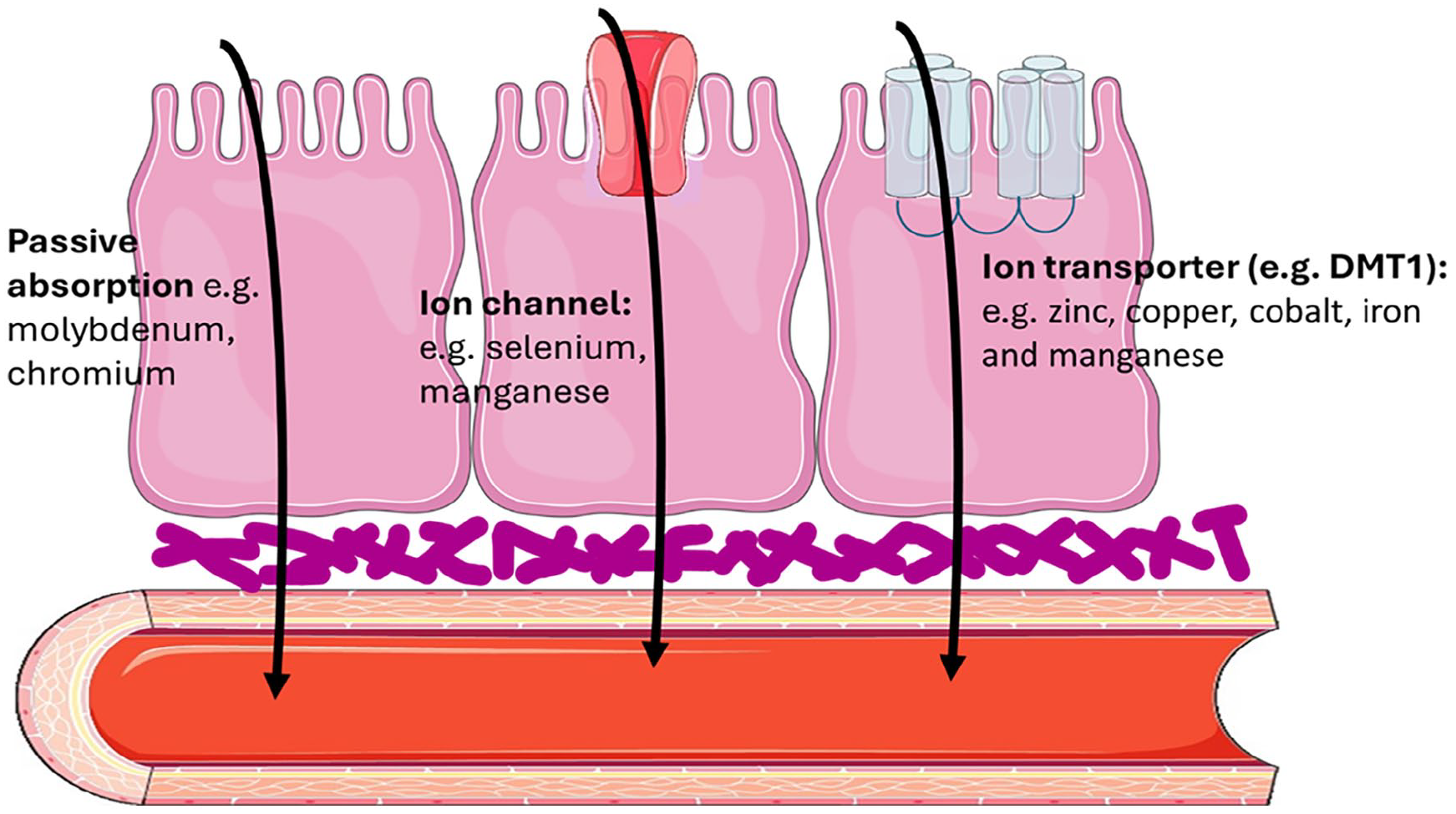
Suggested mechanisms of absorption of trace elements from the gastrointestinal tract through luminal cells into the circulation.
Altered absorption in critical illness
Enteral absorption of macronutrients may be reduced in critical illness, due to reduced oral intake, changes in gastrointestinal motility, dysfunction of absorption mechanisms and changes in mesenteric blood flow. 33 There are few studies investigating TE absorption, but malabsorption from the same mechanisms is likely to occur. Conversely, there is a risk of toxicity with TEs in PN as the normal regulatory mechanisms of the gastrointestinal tract are bypassed. Increased gastrointestinal permeability can occur in critical illness, 34 which may increase TE absorption.
Transport and storage
Normal physiology
In the circulation, TEs are principally bound to general transport proteins, such as albumin (chromium, copper, zinc, manganese), metallothionein (zinc, copper and selenium) and transferrin (chromium, manganese), while some are bound to specific proteins, such as copper (caeruloplasmin and transcuprein). Cobalt is transported incorporated into vitamin B12, accounting for its name cobalamin, a combination of cobalt and vitamin.
A large proportion of TEs are stored in the musculoskeletal system (see Figure 1). Almost two-thirds of the body’s copper 35 and over 85% of the body’s zinc 36 is located in the skeleton and muscle. The largest proportion of manganese is stored within bones (up to 40%), with the remaining proportion split between the brain, liver and pancreas. 37 Around 50% of the total content of selenium is stored within skeletal muscle with lower levels of serum selenium being more prevalent in the elderly population. 38 99% of stored fluoride is located in teeth and bones, where its half-life is several years, compared with a few hours in plasma. 39 Chromium is stored in the liver, spleen, soft tissue and bone. Around three-quarters of iodine is stored in the thyroid. 21
Altered transport and storage in critical illness
Inflammation results in release of cytokines through regulation of transcriptional factors, which causes altered function or concentrations of transport or binding proteins during acute severe illness. A rapid redistribution of zinc, iron, copper and selenium within the body therefore occurs due to decreased levels of the serum-binding proteins albumin, macroglobulin and transferrin, depending on the degree of inflammation. Serum levels can drop markedly in severe inflammation, decreasing by up to 90% of normal levels 40 (see Table 4). Low plasma levels may not necessarily imply whole body deficiency or depletion, and levels should therefore be interpreted with caution. 40
Relationship of the inflammatory response and circulating micronutrient concentrations. 40

Ferritin and ceruloplasmin levels are both acute phase reactants, so their levels, unlike a number of transport proteins, and corresponding levels of copper, may be increased in critical illness. 41 98% of circulating copper is bound to caeruloplasmin. 2
Elimination
Normal physiology
Excretion of most of the TEs occurs via the biliary and gastrointestinal tract (see Figure 1). Other TEs, such as chromium, cobalt and molybdenum, are primarily excreted via urine.42,43 Small amounts of TEs may also be lost through skin, hair and semen. 44
Altered excretion in critical illness
Renal or hepatobiliary dysfunction may result in reduced TE clearance, with a risk of chromium, 2 and manganese and copper accumulation, 16 respectively. Conversely, increased urinary, gastrointestinal or other losses may occur. In one study of urinary excretion of TEs in patients receiving PN after a traumatic injury, there was minimal loss of manganese, but selenium was excreted in higher amounts than was present in the PN, suggesting the risk of accumulation of manganese and deficiency of selenium. 45
RRT is a well-recognised cause of copper loss, along with other micronutrients, such as iron, zinc and selenium and amino acids and vitamins.46,47 The clearance of other TEs by RRT is poor, and plasma levels of chromium and manganese (and zinc in some studies) may therefore increase. 48
Variations in pharmacokinetics in critical illness may be significant. For example, as PN bypasses gastrointestinal malabsorption, 3 mg/day of zinc is considered sufficient to meet needs in most patients; however, those with increased gastrointestinal losses may require as much as 12 mg/day, 49 and 30–40 mg/day in patients with major burns, 49 due to huge exudative losses.
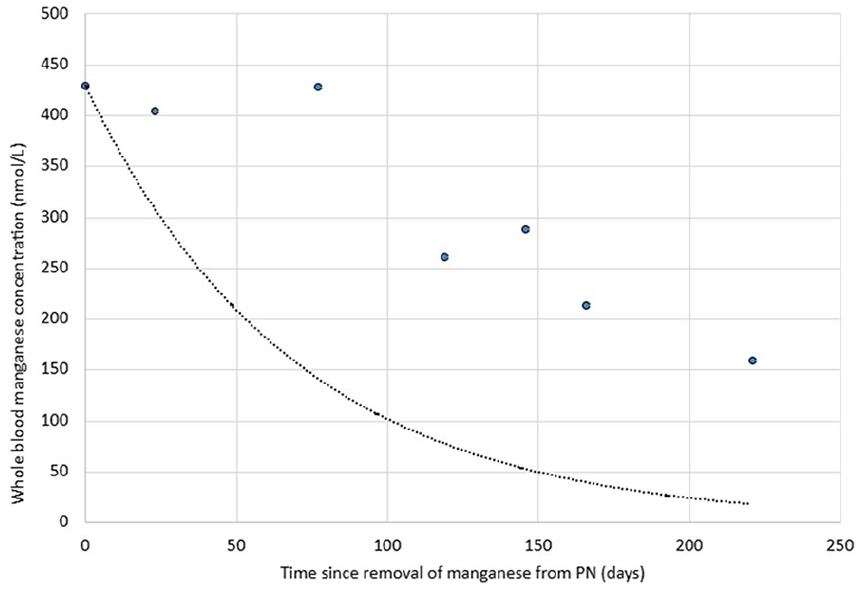
In the brain, manganese primarily accumulates in the basal ganglia and cerebral cortex; excess accumulation can therefore affect motor function and higher-order cognitive function. 50 Acute manganese toxicity with acute parkinsonism has been reported in a patient with poor acute hepatobiliary function while receiving PN. 51 The report suggested that elimination followed first-order kinetics, but that the elimination half-life was 153.3 days, over three times greater than in patients with normal liver function (see Figure 3).52,53
Trace element derangement in specific critical illnesses and treatments
Specific illnesses seen in acutely or critically ill patients and critical care treatments add to the risk of developing deficiencies (see Table 5).
Table to show conditions and treatments that may predispose to trace element abnormalities.
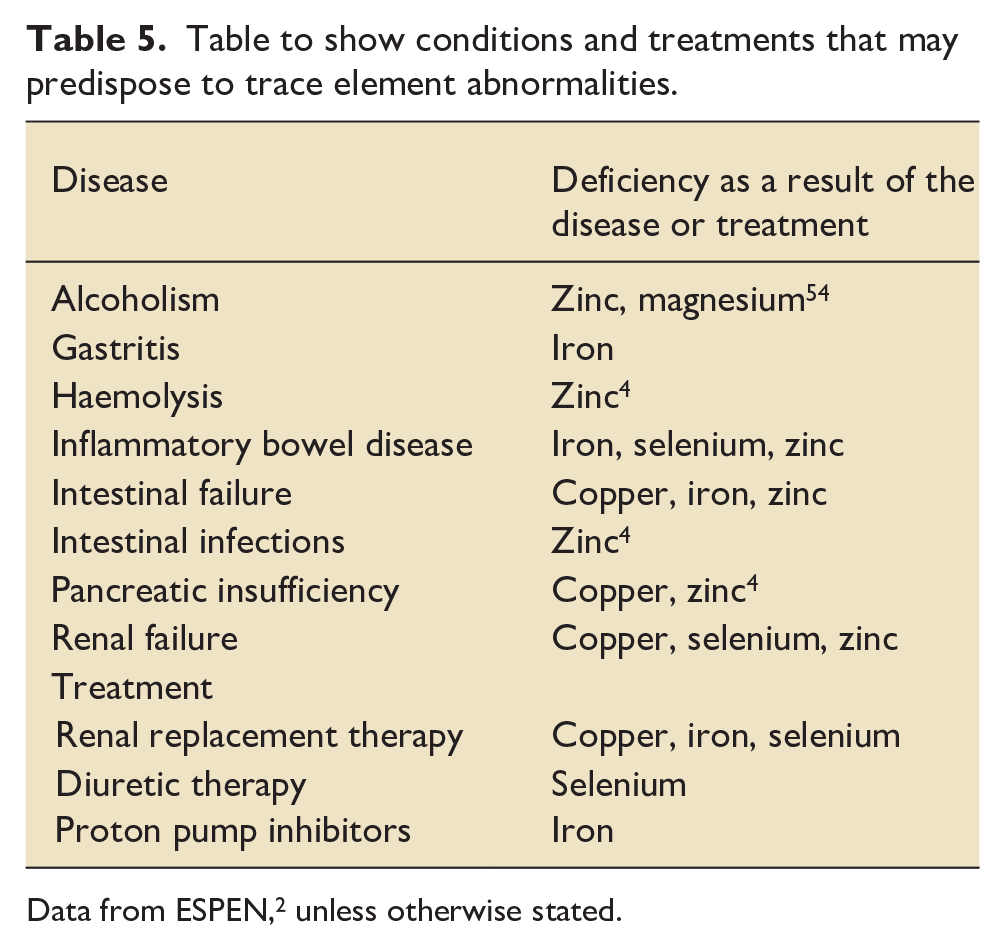
Data from ESPEN, 2 unless otherwise stated.
Physiological role of trace elements and features of derangement
Functions of trace elements in critical illness
TEs are involved in a huge range of enzymatic and protein activity and are vital to the normal function of the body. Zinc for example is thought to be a co-factor in more than 300 enzymes and be incorporated in around 3000 human proteins. 55 Copper, 56 selenium57,58 and molybdenum similarly are incorporated into a wide range of enzymes with diverse actions. The following section outlines TE functions especially important in critical illness.
Antioxidant effects
Oxidative stress-mediated cell damage, due to an increase in the production of reactive oxygen species (ROS) and depletion of antioxidants, is fundamental to the pathophysiology of various critical illnesses such as acute respiratory distress syndrome, ischaemia-reperfusion injury and multiorgan dysfunction in sepsis. Superoxide dismutase (SOD) is a ubiquitous family of enzymes that catalyse the reaction of potentially toxic excess superoxide radicals (O2−) into molecular oxygen (O2) and hydrogen peroxide (H2O2), which is subsequently further broken down to water (see equation (1)). Trace metals such as copper, zinc and manganese form an intrinsic part of the SOD enzyme.
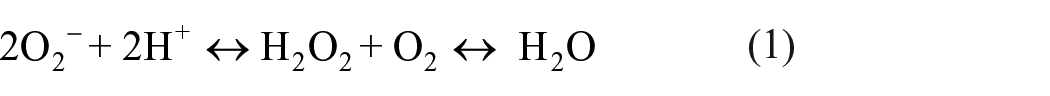
The enzymes catalysing the breakdown of hydrogen peroxide, such as catalase, glutathione peroxidase and cytochrome c peroxidase, require various TEs within their structure or for adequate function. Selenium is contained within glutathione peroxidase, while zinc and copper may have antagonistic actions on the activity of several of these enzymes.
TE derangement may have clinically apparent effects on the redox equilibrium. Chromium toxicity is associated with acute kidney injury, cardiomyopathy and liver failure, postulated to be via ROS-mediated oxidative stress. 59 Selenium deficiency is associated with cardiomyopathy. In one study, over 20% of patients with heart failure with reduced ejection fraction (HFrEF) had reduced selenium levels, 60 associated with increased mortality. Selenium supplementation has been shown to improve cardiac function. 61 The mechanism of cardiomyopathy with selenium deficiency is unclear, but has been postulated to be due to redox disequilibrium,60,62 or to inflammatory or immunological effects. 62
Metabolism and energy pathways
A number of TEs are known to have important functions within metabolic pathways that may be disrupted in acute or critical illnesses. Chromium enhances the action of insulin in peripheral tissues.63,64 Chromium deficiency is therefore associated with impaired glucose handling, hyperglycaemia and insulin resistance, which may be reversed by chromium supplementation (see Figure 4). Acute infection appears to reduce the availability of circulating chromium, which may contribute to the altered glucose metabolism in this setting. Chromium deficiency in critical illness has been suggested as one of the causes of hyperglycaemia, 2 and ESPEN therefore advise assessing serum chromium levels and the response of the glucose tolerance test to chromium supplementation in cases of severe insulin resistance. 2
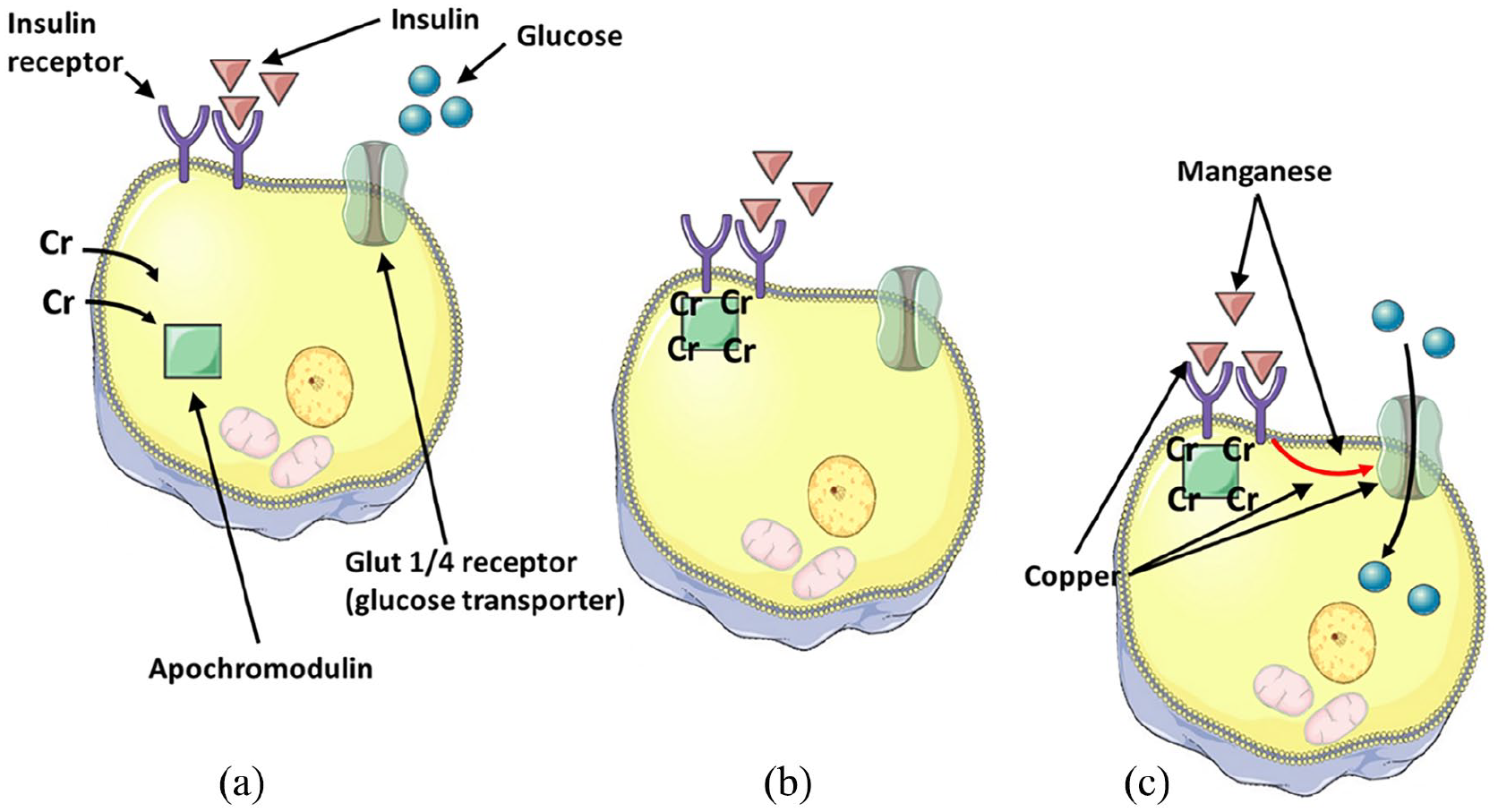
Proposed mechanism of action of intracellular chromium, copper and manganese. Binding of insulin to its receptor in response to glucose in the circulation allows chromium (Cr) to bind to inactive apochromodulin (a). Activated chromodulin binds to the insulin receptor (b), amplifying its effects of activating the glucose transporter Glut4 to transport glucose into the cell (c).63,64 Copper is thought to inhibit the breakdown of insulin, increase the activation of insulin receptors to increase glucose transport into cells, and increase the expression of glucose transporter type 1 (Glut1). 65 Manganese is thought to increase insulin production and secretion, and increase the activation of insulin receptors to increase glucose transport into cells. 65
Copper and manganese deficiency may also be associated with impaired glucose tolerance and insulin resistance 65 (see Figure 4). Higher levels of manganese are associated with a lower risk of type 2 diabetes. 66
Iodine is a key component of thyroid hormone and is required for myelination of the central nervous system. Selenium is fundamental to the structure and function of deiodinase (DIO) enzymes involved in the metabolism of thyroid hormones; the proposed mechanism of deiodination is that halogen bonding between the selenium of the deiodinase and the iodine of the thyroxine allows the swap of the hydrogen and iodine. 67 Selenium levels are often low in critical illness. Sick euthyroid syndrome, seen in critical illness, is characterised by low DIO1 and high DIO2 and 3, and therefore low plasma T3 (active) levels but high plasma rT3 (inactive) levels; this imbalance of enzyme activity is thought to be associated with the low selenium levels. 68
Cellular function
Various TEs in normal amounts are important in normal cellular function (see Figure 5). However, excess of a TE may have detrimental effects. Manganese excess is associated with mitochondrial damage, and various TEs, including iron, copper, zinc, selenium, chromium, cadmium, mercury and lead, influence and may disrupt membrane fluidity, integrity and function. 69
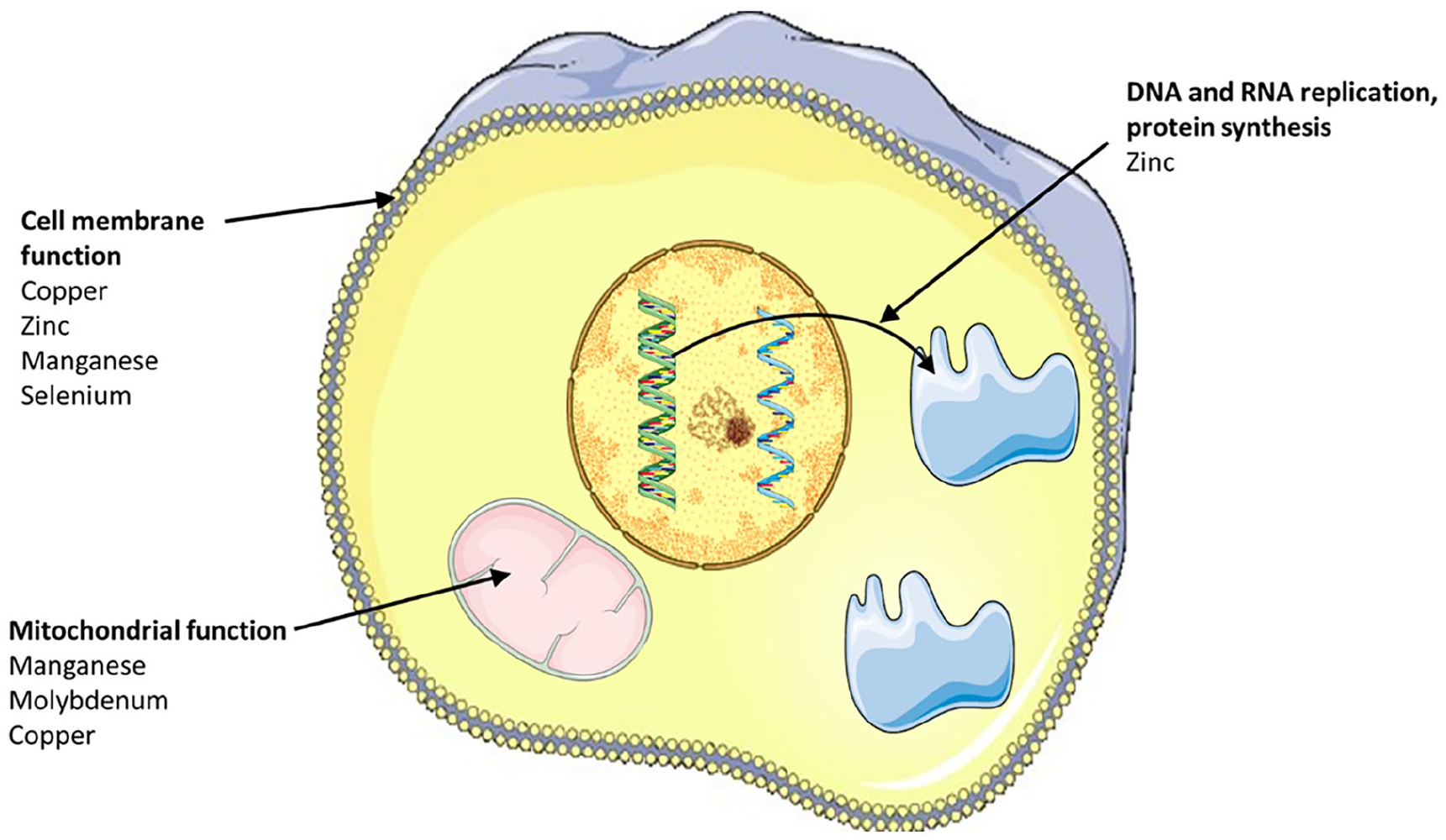
Putative roles and sites of action of selected trace elements in cellular function.
Cytotoxicity is believed to be the mechanism behind excessive cobalt causing a rapidly progressive but reversible cardiomyopathy; 70 it was originally observed in beer drinkers, due to the addition of cobalt to beer as a foaming agent. ESPEN suggests that testing for cobalt toxicity may be warranted if a patient develops cardiomyopathy. 2
Zinc deficiency can blunt senses of taste and smell. 71 In one study of patients with poor taste and smell function, 50% had low blood zinc levels, which improved with zinc supplementation. 72 Zinc deficiency is thought to be due to damage to chemoreceptors in the olfactory and taste nerves. 72
Manganese cytotoxicity can cause central nervous system (tinnitus, hearing loss, tremors and ataxia and parkinsonism) and psychological disturbances (such as delusions, irritability and changes in mood), since manganese can accumulate in certain areas of the brain. Cognitive decline in Alzheimer’s disease may also be linked to manganese accumulation within the frontal cortex. Magnetic resonance imaging of the brain has therefore been suggested in cases of suspected manganese accumulation. 2
Excess copper induces oxidative stress, DNA damage and reduced cell proliferation, leading to neurological features (behavioural and mood changes, abnormal muscle movements) and signs of heart, liver and renal impairment. Haemolysis can occur due to toxic damage to red blood cells. Excess copper can accumulate in various organs, including the brain, liver, heart and kidneys. Kayser-Fleischer rings, brown discolorations forming a ring in the outer cornea, are due to copper deposition in Descemet’s membrane of the cornea.
Immunity
TEs are required for the maturation, proliferation and activation of immune cells at different stages (see Table 6). 73 The need for zinc for adequate immunity in humans has been known since the 1960s. Populations living in areas or who had diets low in zinc tended to be of shorter stature and often died earlier, sometimes in their 20s, from recurrent infections. 71 Acute zinc deficiency causes a decrease in both innate and adaptive immunity (see Table 6); conversely, chronic deficiency increases inflammatory changes and may adversely affect the outcome of auto-immune diseases. 74
Abnormalities of innate and adaptive immune function caused by trace element deficiency or excess.
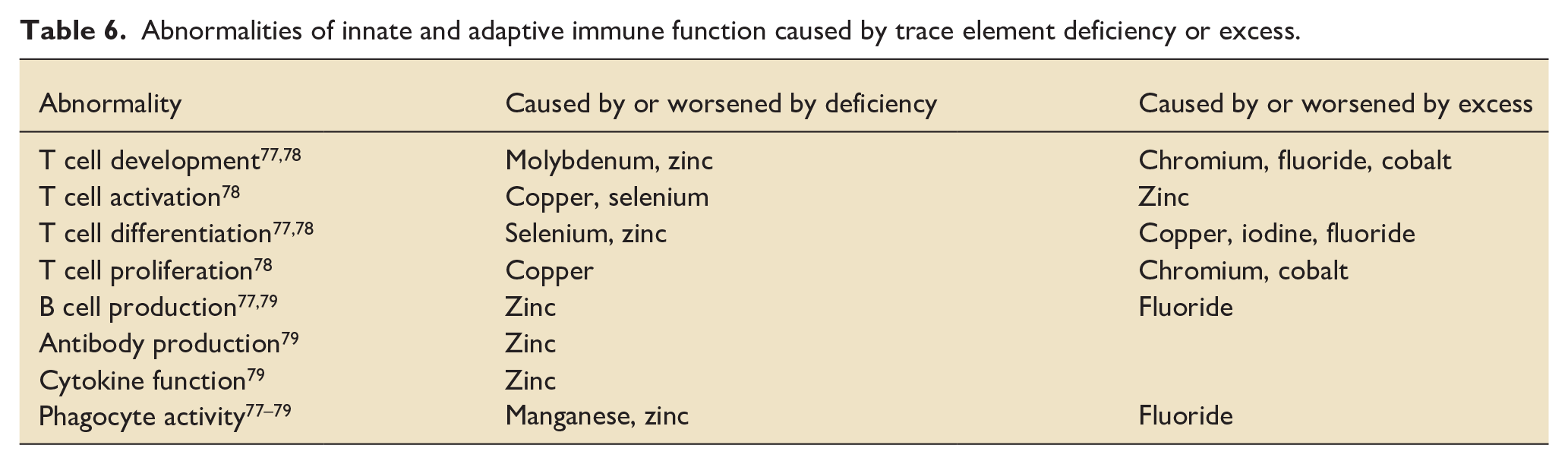
Adequate selenium levels are required for various stages of immune activation, differentiation and proliferation and function of both B- and T-lymphocytes (see Table 6). 75 In selenium deficiency, benign strains of Coxsackie and influenza viruses can become highly pathogenic. 75 Dietary supplementation may have a protective role against H1N1 influenza, polio and human immunodeficiency virus infections 76 and confer benefit in patients with HIV and influenza A viral infections and Mycobacterium tuberculosis infection. 75
Wound healing
Skin and soft tissue maintenance and repair involves four overlapping physiological processes (haemostasis, inflammation, proliferation and remodelling). 80 A number of these steps are critically dependent on specific TE activity. 81 Copper-containing enzymes, such as lysl oxidase, are required in a number of steps in the formation and cross-linking of collagen. Cellular growth and proliferation requires zinc, copper and selenium and several TEs are required for energy production and carbohydrate metabolism (e.g. chromium) to minimise protein catabolism.
Selenium also appears to be required for adequate wound healing, thought to be due to its antioxidant effects. Levels of selenium (along with copper and zinc) have been found to be higher at edges of wounds compared with elsewhere. 81 Supplementation of selenium after burns improves wound and graft healing. 82
Oxygen delivery and utilisation
A number of micronutrients are important in oxygen transport and delivery. Iron is required for erythropoiesis and as a component of the binding sites of oxygen in haemoglobin. Zinc lowers blood viscosity and resistance to flow, by increasing the deformability of red blood cells (RBCs). 83 Copper is integral to a wide range of enzymes involved in aerobic respiration. Conversely, fluoride adversely affects both the structure and function of RBCs, reducing the oxygen carriage capacity. 84
Clinical signs of acute deficiency and toxicity
Given the ubiquitous nature and function of TEs, acute deficiency or excess of particular TEs in critically ill patients can present in a wide range of features may increase the risk of developing or worsening a life-threatening acute illness (see Figures 6 and 7). Many of these clinical features are insidious and have a wide variety of other causes in critically ill patients; a high index of suspicion that they may be due to TE derangement is required.
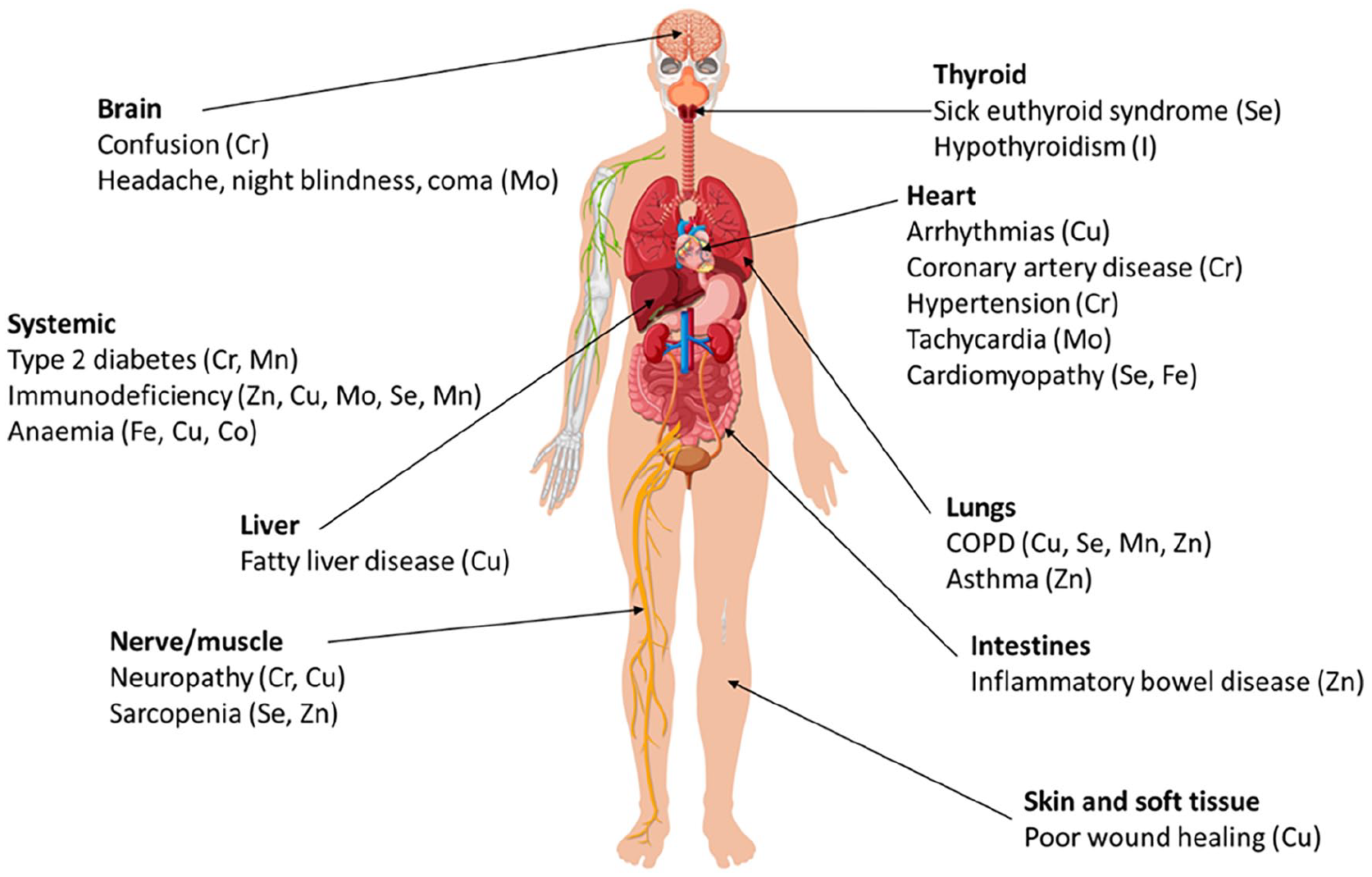
Conditions that may be due to or exacerbated by deficiency of selected trace elements.
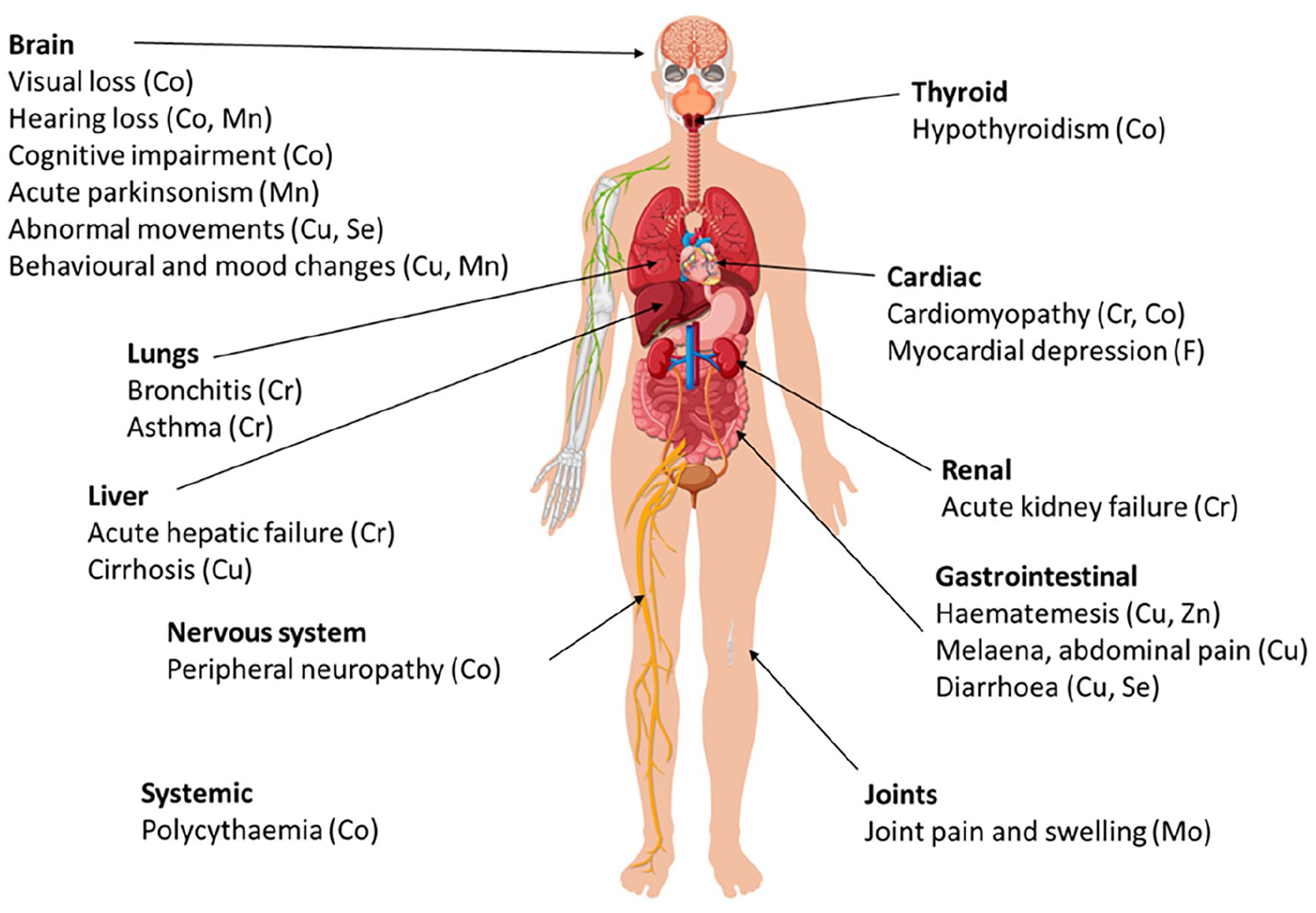
Conditions that may be due to or exacerbated by excess of selected trace elements.
Assessment of trace element status and supplementation
Accurate assessment of the total amounts of TEs in the body is difficult for a number of analytical and clinical reasons, in addition to changes in pharmacokinetics and the effects of inflammation in the critically ill. Whether and how to replace a deficiency may also be difficult to discern; ESPEN advise integrating clinical assessment and laboratory results with the degree of the concomitant inflammatory response; 2 a CRP level of greater than 20 mg/L is considered high enough to affect interpretation of TE levels. 2 Discussion with a biochemist or dietitian is also advised.
Measurement of trace element concentrations
Plasma levels of TEs may be unreliable and not accurately reflect total body levels. In addition, many of the currently available tests may be inaccurate; in one study, while the coefficient of variability was generally below 15%, it was up to 25%–50% with some samples. 85 Some blood collection bottles may be contaminated with TEs, such as manganese, aluminium and zinc; specimen collection in a TE-free tube is therefore recommended.
Recommendations for measuring particular TEs vary. Plasma levels for some, such as zinc, may be reliable. 2 For others, such as cobalt, fluoride, iodine, manganese and molybdenum, urine levels may be used to assess levels in the body. 2 Substantial amounts of certain TEs, such as manganese and selenium, are contained within erythrocytes, so whole blood estimations rather than plasma may be useful and may be less affected by inflammation.2,86,87 In some cases, clinical markers may also be useful; for example, thyroid function tests for iodine, magnetic resonance brain imaging for manganese, and the effect of chromium on glucose tolerance test has been advised. 2
Since copper is primarily transported bound to caeruloplasmin, an acute phase reactant, levels are often raised in critical illness. Measuring caeruloplasmin and CRP levels in addition to copper levels is recommended; low serum copper level with a raised CRP, or a low caeruloplasmin level, suggests copper deficiency (see below). 2 One study suggested that copper levels can therefore be adjusted for caeruloplasmin levels according to equation (2), similar to how serum calcium can be adjusted for albumin levels; 88 however, this is not universally adopted. 2
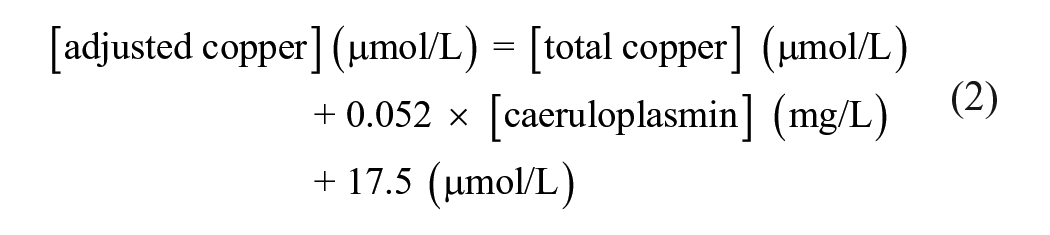
Trace element supplementation and clinical outcomes
Given the risk of TE deficiency in critical illness and the wide range of essential functions they perform, there is interest in whether supplementing TEs with doses higher than that required for maintenance (see Table 3) improves outcomes. However, conclusive evidence is generally lacking.
Sepsis
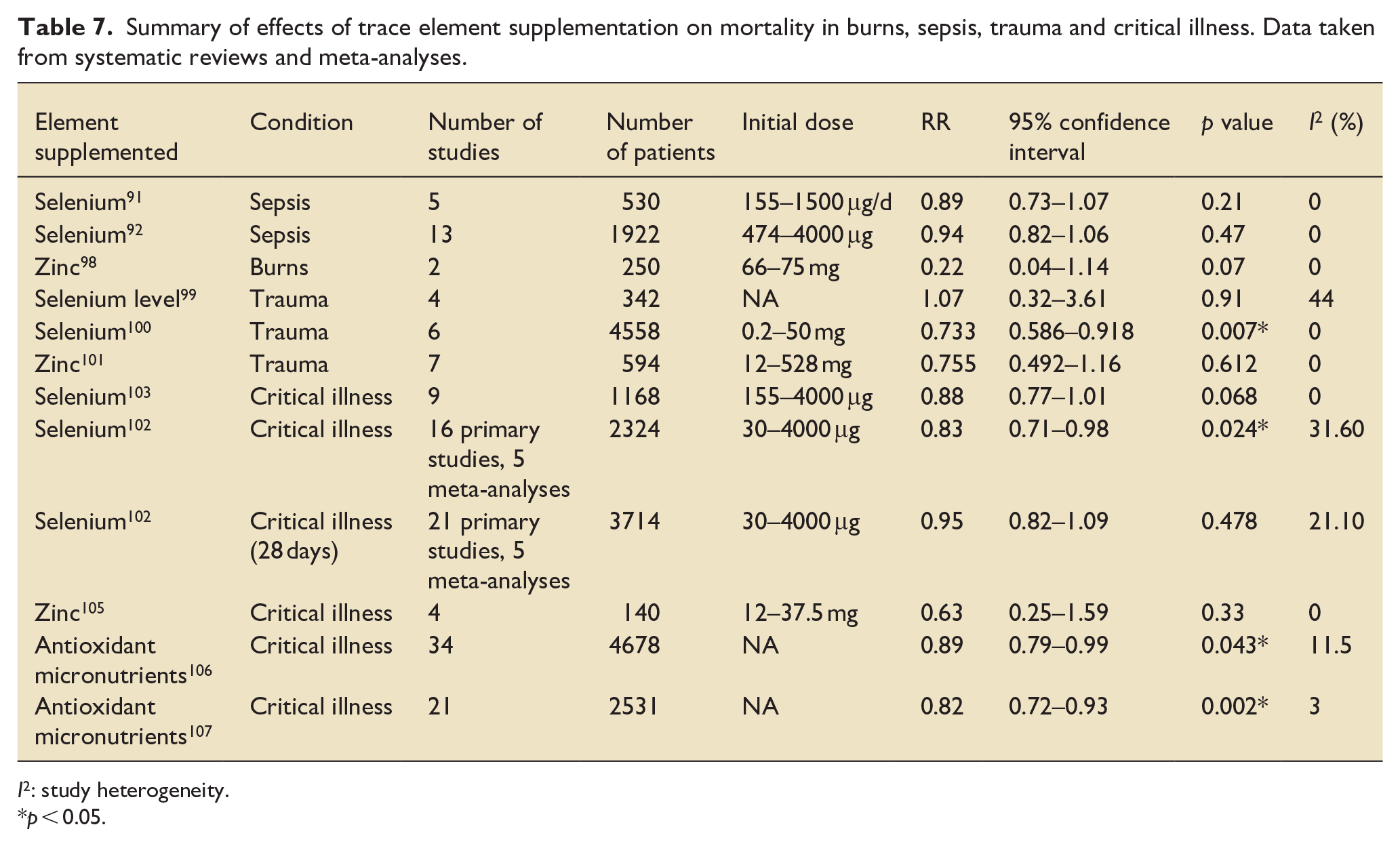
Low selenium levels in sepsis are associated with poor outcomes. In one study of patients with systemic inflammatory response syndrome (SIRS) or sepsis, a low selenium plasma level (0.7 µmol/L or lower) on admission was associated with a three-fold increase in the frequency of ventilator-associated pneumonia, organ failure and mortality; 89 the Selenium in Intensive Care (SIC) trial showed a reduction in 28-day mortality by around 25% with administration of a bolus of 1000 µg of sodium selenite following a continuous infusion of 1000 µg/day for 14 days. 90 However, one meta-analysis of selenium supplementation in sepsis failed to show any significant improvement in all-cause mortality (see Table 7), the incidence of hospital-acquired pneumonia or length of ICU stay. 91 A larger meta-analysis also failed to show an improvement in 28-day and 6-month mortality (see Table 7), the incidence of renal failure, secondary infection or duration of mechanical ventilation. However, it did appear to reduce the duration of vasopressor therapy, length of stay in ICU and hospital, and the incidence of ventilator-associated pneumonia, 92 although there was significant heterogeneity in study designs. In one RCT of zinc supplementation in sepsis in adults, supplementation was associated with a higher temperature, which the authors considered would augment the acute phase reaction, of potential benefit to the host. 93 There are no published systematic reviews on zinc supplementation; further evidence is required.
Summary of effects of trace element supplementation on mortality in burns, sepsis, trauma and critical illness. Data taken from systematic reviews and meta-analyses.
I2: study heterogeneity.
p < 0.05.
Burns
Patients with burn injuries are hypermetabolic; they can lose significant amounts of proteins and micronutrients in wound exudates, predisposing them to malnutrition, significant weight loss, in particular muscle mass, and associated complications. Severe burns lead to an intense oxidative stress as well as a substantial systemic inflammatory response, adding to the depletion of the endogenous antioxidant defences. Decreased levels of vitamins A, C and D and iron, copper, selenium and zinc have been found to negatively impact wound healing and immune function. 94
There has therefore been much interest in whether supplementation of TE, particularly antioxidant TEs (zinc, copper and selenium) and other antioxidants such as vitamins, is of benefit. In one systematic review, parenteral supplementation of combined TEs was associated with a significant decrease in the risk of infections, and possibly improving the length of stay, although optimal doses and regimes are not known. 95 A further systematic review showed that wound healing time and infection rates were generally reduced by the administration of antioxidant micronutrients (such as vitamins A, C and E and zinc, copper and selenium), although there was much study heterogeneity and further studies were recommended. 96 Other studies suggest that supplementation of antioxidant TEs can improve wound healing and reduce rates of infection including nosocomial pneumonia.82,97 Oral zinc supplementation in isolation may have beneficial effects on mortality but current studies are not conclusive (see Table 7). 95
Trauma
A meta-analysis of four studies showed that a low serum selenium level did not worsen mortality in trauma (see Table 7), the incidence of infectious complications, length of ICU stay or the hospital length of stay. 98 Despite this, a further meta-analysis of seven studies showed that selenium supplementation was associated with a lower mortality rate (see Table 7), a reduced ICU length of stay and a reduced hospital length of stay. However, selenium supplementation did not have an effect on the incidence of infectious complications. 99
A meta-analysis of seven studies of zinc supplementation after trauma found the opposite to selenium supplementation: zinc supplementation was associated with a lower risk of pneumonia, but did not influence the mortality rate (see Table 7) or the length of hospital stay. 100
Outcomes in critical illness
In a meta-analysis of seven ICU studies, patients with lower plasma or serum zinc or selenium levels had higher mortality rates and higher levels of organ dysfunction. 87 However, there is no conclusive evidence for high-dose selenium supplementation (e.g. 500–4000 µg) compared with routine maintenance supplementation (e.g. 50–200 µg; see Table 3). One systematic review of 17 meta-analyses suggested that while high-dose supplementation of selenium in critical illness may improve overall mortality and the incidence of acute renal failure, 101 there was no significant effect on 28-day mortality, and there was significant heterogeneity between the studies and uncertainty. A recent Cochrane review of nine RCTs (1168 participants) on high-dose selenium supplementation also showed no evidence of improvement in ICU mortality, but most trials were considered to be at high risk of bias and had heterogeneous study designs, limiting the reliability of the results. 102 The risk of developing an infection, number of days on a ventilator and length of ICU and hospital stay was not affected by routine high-dose selenium supplementation101–103 (see Table 7).
Similarly, in a systematic review of four papers investigating zinc supplementation, there was a trend towards a reduction in mortality and length of stay in ICU, but neither were significant. There was significant heterogeneity in study designs and dosing. Routine high-dose zinc supplementation in critically ill patients is therefore not recommended currently 104 (see Table 7).
Antioxidants in combination may be more effective than in isolation. Two systematic reviews have suggested that antioxidant supplementation (TEs selenium, zinc, copper and manganese and vitamins A, C and E) in the critically ill may reduce mortality105,106 (see Table 7) and length of mechanical ventilation. 106 However, these results are not conclusive; in one of these reviews, when only trials with a low risk of bias were included, no such benefit was evident. 105 In addition, a blinded, randomised controlled trial failed to show any benefit of glutamine and the antioxidants zinc and selenium in patients with multiorgan failure undergoing mechanical ventilation; glutamine may instead worsen mortality. 107
Conclusion
TEs are ubiquitous and vital for a wide range of physiological processes. Some are especially important in affecting the response to critical illness; conversely, critical illness affects the availability and activity of certain TEs.
Unfortunately, features of deficiency and toxicity are often insidious and non-specific particularly in critical illness, and assessment of TE status is often difficult in the context of critical illness and inflammation. Practitioners need to be aware of the wide range of signs and symptoms in critically ill patients that may be due to abnormal levels of TE. Low levels may require replacing, taking into account the clinical features and degree of inflammation; caution is needed if inflammation is high. Advice from a biochemist may be warranted to interpret and correct abnormal results. Current evidence does not suggest high-dose supplementation of any TE in critically ill patients, but instead maintaining normal physiological levels.
Footnotes
Acknowledgements
The authors are grateful for the expertise and knowledge of Dr C Livingstone, consultant chemical pathologist and Dr L Coppel in the production of this paper.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Edward Walter is on the editorial board of the Journal of the Intensive Care Society. None of the other authors declare a conflict of interest.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.