Abstract
Introduction:
Antiplatelet and anticoagulation therapies are frequently required in acutely unwell patients and confer an increased propensity for procedural bleeding. During percutaneous tracheostomy insertion, the decision to stop these therapies is left to clinical decision-makers. This meta-analysis summarises the risk of bleeding associated with antiplatelet and anticoagulation therapy during percutaneous tracheostomy insertion.
Method:
We conducted a systematic review and meta-analysis of studies which reported intraoperative bleeding during percutaneous tracheostomy while on single antiplatelet therapy (SAPT) or dual antiplatelet therapy (DAPT), as well as therapeutic anticoagulation (TAC). Groups were compared against those with prophylactic or no anticoagulation. Studies were pooled using random effects via the inverse variance method.
Results:
Four databases found 22 eligible studies, of which 14 studies presented data for meta-analysis representing 3,485 percutaneous tracheostomy insertion procedures. These included six studies that reported intraoperative bleeding outcome SAPT, six for DAPT and five for TAC. Overall, methodological quality was poor. No significant association was found for SAPT (Odds ratio 1.58; 95% confidence interval 0.72–4.41); P = 0.25; I2 = 81%) and TAC (OR 1.79; 95% CI 0.58–5.56; P = 0.35; I2 = 35%). The DAPT group was associated with increased bleeding with an OR of 2.05 (95% CI 1.18–3.56; P = 0.01; I2 = 0%).
Conclusion:
Our study supports temporarily withholding DAPT or TAC (if clinically feasible) to minimize bleeding risks associated with percutaneous dilatational tracheostomy.
Introduction
The insertion of a tracheostomy is a common procedure in the critically ill, predominantly to facilitate gradual liberation from invasive ventilatory support (weaning). Other indications include actual or threatened upper airway obstruction or an inability to protect the airway from aspiration. A tracheostomy can be inserted in two ways: an open “surgical” technique (usually in the operating theatre) or a percutaneous dilatational approach (usually at the patient’s bedside in the intensive care unit (ICU)). Percutaneous tracheostomy (PT) is the dominant technique in the critically ill as it does not usually require a surgeon, operating theatre team and facilities, or an anaesthetic team. This facilitates timely insertion, reducing delays, recovery time, and costs. 1 Furthermore, meta-analyses have shown that PT is comparable to surgical insertion methods with comparable complication rates and bleeding risk.2,3
Between 5 and 7 per cent of tracheostomy insertions are complicated by bleeding. 4 While major haemorrhage is rare, even minor bleeding can lead to airway obstruction and pose a life-threatening risk to the patient. Bleeding can be managed via localized measures such as applied pressure, tamponade, packing, local application of epinephrine, haemostatic agents, surgical intervention such as electrical or chemical cautery, stoma revision, or removal of the tracheostomy tube. 5 More systemic interventions include the correction of coagulopathies, antifibrinolytics, transfusion, cardiovascular support, permissive hypotension and vascular intervention including interventional radiology.
Increasingly complex patients are admitted to ICUs and are often treated with anticoagulation (AC) or antiplatelet therapy (AP). This may be due to pre-existing or concurrent comorbidity or new interventions or diagnoses such as thromboembolism, extracorporeal support or coronary/vascular endovascular interventions and stents. Balancing the therapeutic benefits of these agents against the risk of bleeding risk is left to clinical decision-makers. Between 10 and 13 per cent of all ventilated ICU admissions will require tracheostomy, 6 meaning the clinician is confronted with the decision between the ST and PT without clear consensus as to which may be beneficial. It has been suggested that PT may be a relative contraindication in those with coagulopathy7,8 whilst others propose PT can be performed with caution.9,10 A relevant but historical survey of intensive care practice in the Netherlands found that less than half of units had a policy on anticoagulation management for PT insertion. 11 Furthermore, clinicians were uncertain if correction of anticoagulation or coagulopathy was necessary before tracheostomy insertion. 11
This systematic review aims to summarise current evidence for the risk of bleeding associated with anticoagulation and antiplatelet therapies in patients who undergo percutaneous tracheostomy insertion.
Methodology
This systematic review was reported as per PRISMA 2020 guidelines 12 with a predetermined protocol registered with PROSPERO (CRD 42022357685).
The outcome of interest
The primary outcome of this review was the occurrence of intra-operative bleeding from PT insertion, defined as any bleeding occurring up to 24 h postprocedure. The severity of bleeding was classified as: minor (terminated after pressure/tamponade, packing, cautery or additional suturing) or major (clinically defined, requiring blood transfusion, or surgical revision). 3 The outcome was further stratified based on the anticoagulant or antiplatelet therapy that the patient was receiving at the time of tracheostomy: single antiplatelet therapy (SAPT), dual antiplatelet therapy (DAPT), and/or if the patient was anticoagulated with a systemic agent with therapeutic intent (therapeutic anticoagulation; TAC). Exposure groups were compared with those anticoagulated with prophylactic intent, as this is routine clinical practice in the ICU patient cohort requiring a tracheostomy and associated with similar bleeding risk to those without any anticoagulation in percutaneous procedures. 13
Search and selection of studies
Medline, Embase, Cochrane and Scopus databases were searched using MeSH, Embase and actual terms. The literature search was detailed within the supplementary material in Tables 1 and 2. Keywords included “tracheostomy,” “tracheotomy,” “antiplatelet,” “dual antiplatelet therapy,” “anticoagulants,” “heparin,” “bleeding,” and “haemorrhage.” No time limits were applied to the search with the last search performed on 1st December 2023.
Manuscripts and abstracts identified were amalgamated, and duplicates were removed. The first round of screening was conducted using abstracts and titles before selecting studies, which were screened using full text. Screening was performed independently by two authors (MB and FE), with disagreement resolved by consensus with a third author, BM. Studies containing original data reporting bleeding outcomes with PT in the adult population and describing antiplatelet therapy or anticoagulation exposure were selected. Studies were pragmatically limited to the English Language. Case studies, studies without a control group and those including ST were excluded from this review.
Data extraction and synthesis
Two reviewers (MB and FE) independently extracted data. Study-level characteristics were extracted, including study design, protocol to withhold AP or AC therapy and the demographic of the study population. The definition of the control group, the definition of the exposure group (number with SAPT, DAPT, TAC) and outcome data (bleeding definition(s), counts, proportion, or odds ratio of bleeding events) were also extracted. Missing data were requested from authors if it was not available in published reports or supplementary material.
Extracted data were combined for synthesis in the final analysis. Odds ratios (OR) were produced from pooling studies using random effects models with the inverse variance method. Heterogeneity was estimated using the I2 statistic through RevMan (version 5.4.1, Cochrane Collaboration). Further investigation of effect modifiers and publication bias was explored using RStudio (version 4.2.2, RStudio, Boston) with the metafor package. Evidence of publication bias was explored using a funnel plot and Peter’s test.
Risk of bias
Assessment of bias was conducted by MB using the Risk Of Bias Tool for Non-Randomized Studies of Interventions (ROBINS–I), a recognized bias assessment tool that compares studies to a well-conducted randomized control trial. 14 The tool considers bias due to confounding, selection, classification of intervention, deviation from intended intervention, missingness of data, measurement of outcomes, and selection of reported results, which contributes to an overall score grade of the study as low, moderate, serious, and critical risk of bias.
Quality of evidence
A post-protocol addition was made to appraise the certainty of evidence using GRADE principles (Grades of Recommendation, Assessment, Development and Evaluation), which includes several domains including risk of bias, inconsistency, indirectness, imprecision and publication bias. 15 The rating of evidence was then sumarised as either: high, moderate, low or very low.
Results
Search selection
Using our search strategies, we identified 1686 records and screened 1187 after removing 499 duplicate records (Figure 1; PRISMA diagram). We excluded 1104 records by abstract and title. Of the records 83 were eligible for full-text screening, of which 61 were excluded from assessment for full text (19 included non-PDT populations, 17 with ineligible interventions, 16 with ineligible control groups, six with ineligible or composite outcome and three with duplicate populations).
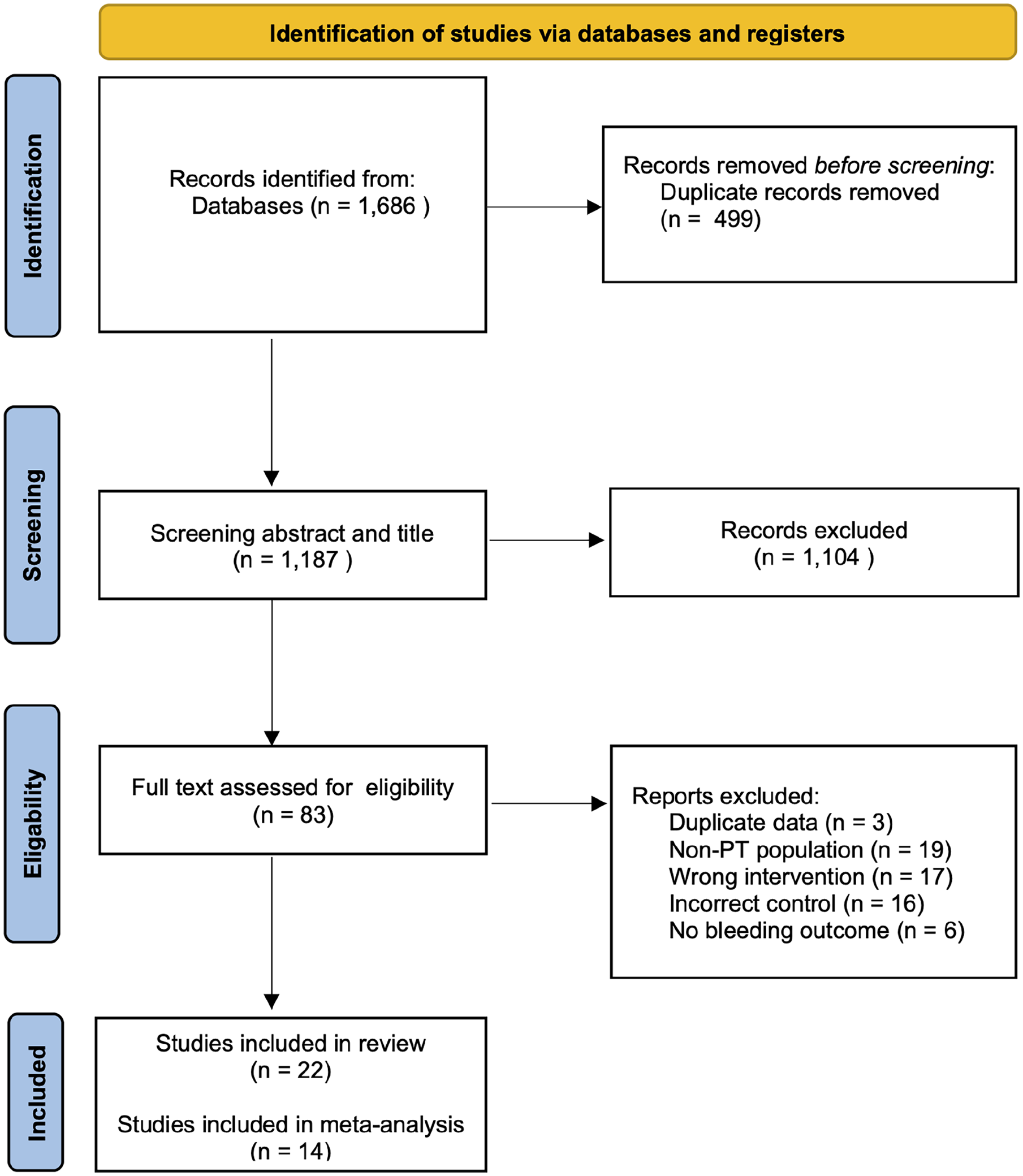
PRISMA reporting items for systematic reviews and meta-analysis.
Of the remaining 22 eligible studies, 14 contributed data for quantitative synthesis, representing 3,485 participants undergoing PT insertion. Of these, intraoperative bleeding outcomes during PT insertion were reported in six studies with SAPT (953 participants),16 –21 six with DAPT (224 participants),19 –24 and six with TAC (221 participants).25 –29 Eight of the studies were excluded from the quantitative meta-analysis.30 –37 Among these, four focused on extracorporeal membrane oxygenation (ECMO), while the remaining were omitted due to insufficient detail, hindering their inclusion in the quantitative data analysis.
The majority of studies reporting intra-procedural bleeding risk reported both minor and major intraoperative bleeding. Postoperative bleeding was less frequently reported and in insufficient numbers for meta-analysis. One study reported postoperative outcomes for all SAPT, DAPT and TAC groups 20 and another for DAPT. 24
Study characteristics
All studies included in the meta-analysis were cohort studies (Table 1); 11 were retrospective, and three were conducted prospectively.25,27,29 There were differences in practice among studies for withholding both AP and AC therapy. Of the nine studies reporting SAPT or DAPT, four continued therapy,16,22,23,38 one study withheld antiplatelets 24 h prior to procedure 19 and four did not specify a protocol.17,18,21,24 Anticoagulation was continued during the procedure in one study. 25 Other studies ceased anticoagulation 24 h, 23 five hours 26 and four hours 38 before PT insertion. Two studies reporting TAC did not specify an anticoagulation cessation protocol.28,29
Description of study level characteristics.
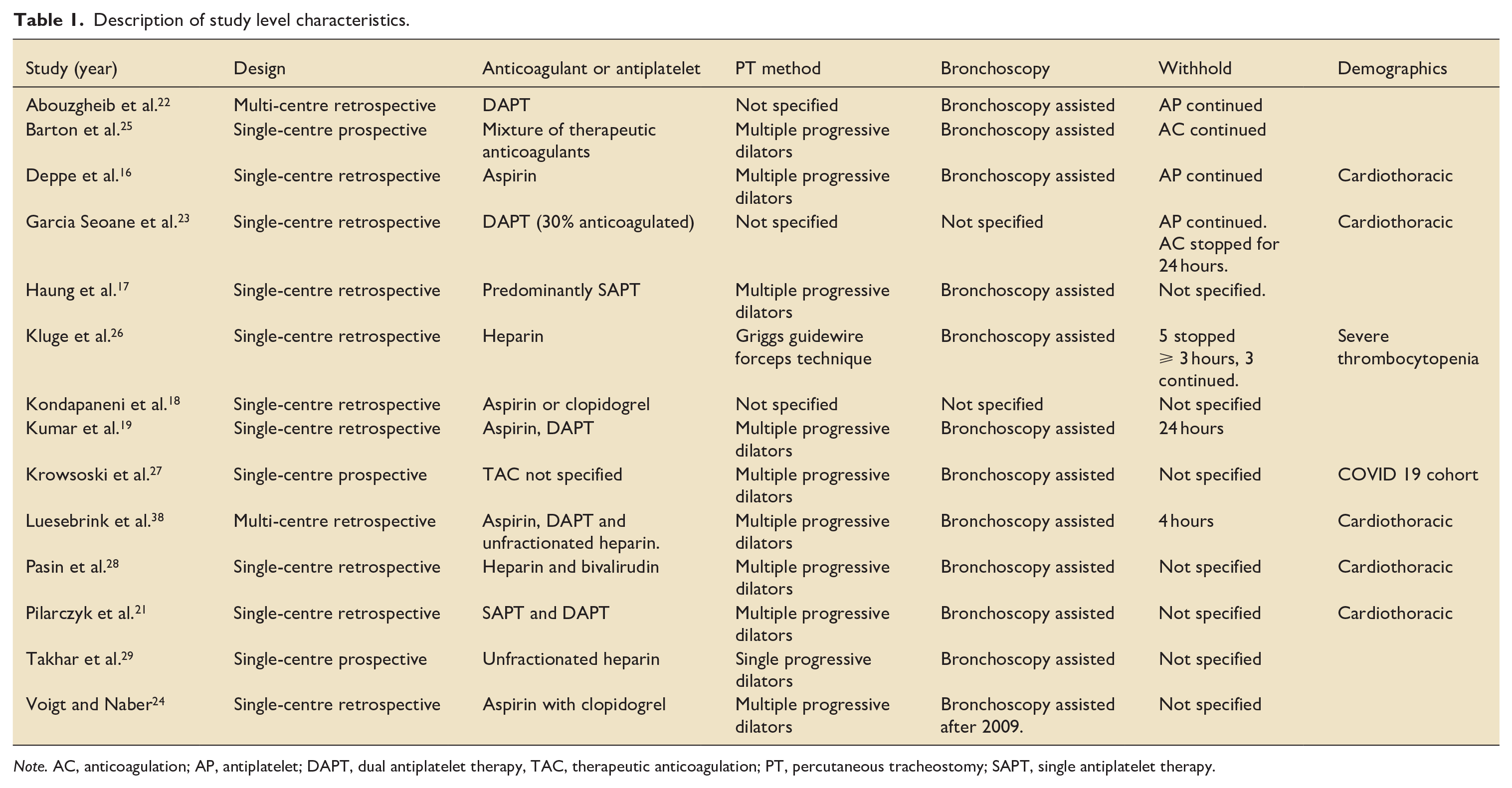
Note. AC, anticoagulation; AP, antiplatelet; DAPT, dual antiplatelet therapy, TAC, therapeutic anticoagulation; PT, percutaneous tracheostomy; SAPT, single antiplatelet therapy.
All studies included patients located in ICU with five studies based in cardiothoracic units.16,20,21,23,28 One study 27 was specific to the COVID-19 population and another from patients with severe thrombocytopenia in a medical ICU. 26 Studies primarily reported multiple progressive dilator techniques for PT insertion, and all but two studies reported the routine use of bronchoscopy to guide initial needle insertion. Further descriptions of study-level characteristics with the potential for confounding were summarised in Supplemental Table 1.
All studies reported bleeding of varying severity. Definitions of the severity of bleeding were similar overall, with major bleeding frequently defined as a reduction in haemoglobin, requiring transfusion or surgical intervention (Supplemental Table 1). Minor bleeding included the application of pressure or packing to the wound site.
Bias assessment
Ten studies had a serious risk of bias, with two having a critical risk of bias (Figure 2). The patient domains with the highest proportion of at least moderate risk included bias due to confounding and selection of reported results. Many studies did report confounders by SAPT, DAPT, and TAC groups. All studies had a low risk of missing data. A critical risk of bias was found in the selection of participants in one study 26 and the classification of the intervention. 17 A detailed assessment of the risk of bias and its domains across all studies can be found in the supplementary material in Figure 1.
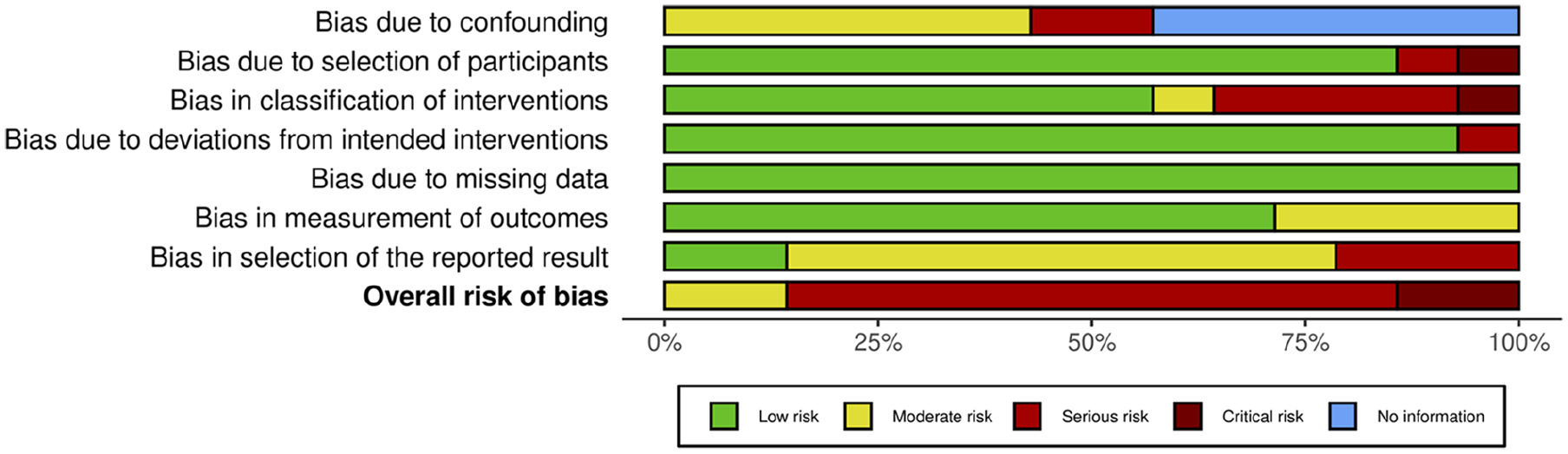
Summary of outcomes from the risk of bias assessment.
Bleeding risk
SAPT
Single antiplatelet therapy may have little to no effect on risk of any bleeding during percutaneous tracheostomy insertion (OR 1.58; 95%CI 0.72–4.41; I2 = 81%; Table 2; figure 3) when compared to those without therapeutic anticoagulation or antiplatelet therapy. However, the certainty of the evidence was very low due to very serious inconsistency between studies and extremely serious imprecision of the pooled effect, leading to a downgrading of evidence. Subgroups of bleeding severity confirmed a lack of a significant association for those receiving SAPT (Table 3) with risk of minor (OR 1.96; 0.76–5.05; I2 = 80%), major (OR 5.38; 0.97–29.64; I2 = 0%) or any postoperative bleeding (OR 1.25; 0.39–3.95).
Summary of findings. The effect of antiplatelet and anticoagulation therapy on bleeding risk during percutaneous tracheostomy insertion with assessment of evidence certainty using the GRADE principles (Grades of Recommendation, Assessment, Development and Evaluation).

Note. CI, confidence interval; DAPT, dual antiplatelet therapy; SAPT, single antiplatelet therapy; TAC, therapeutic anticoagulation; OR, odds ratio.
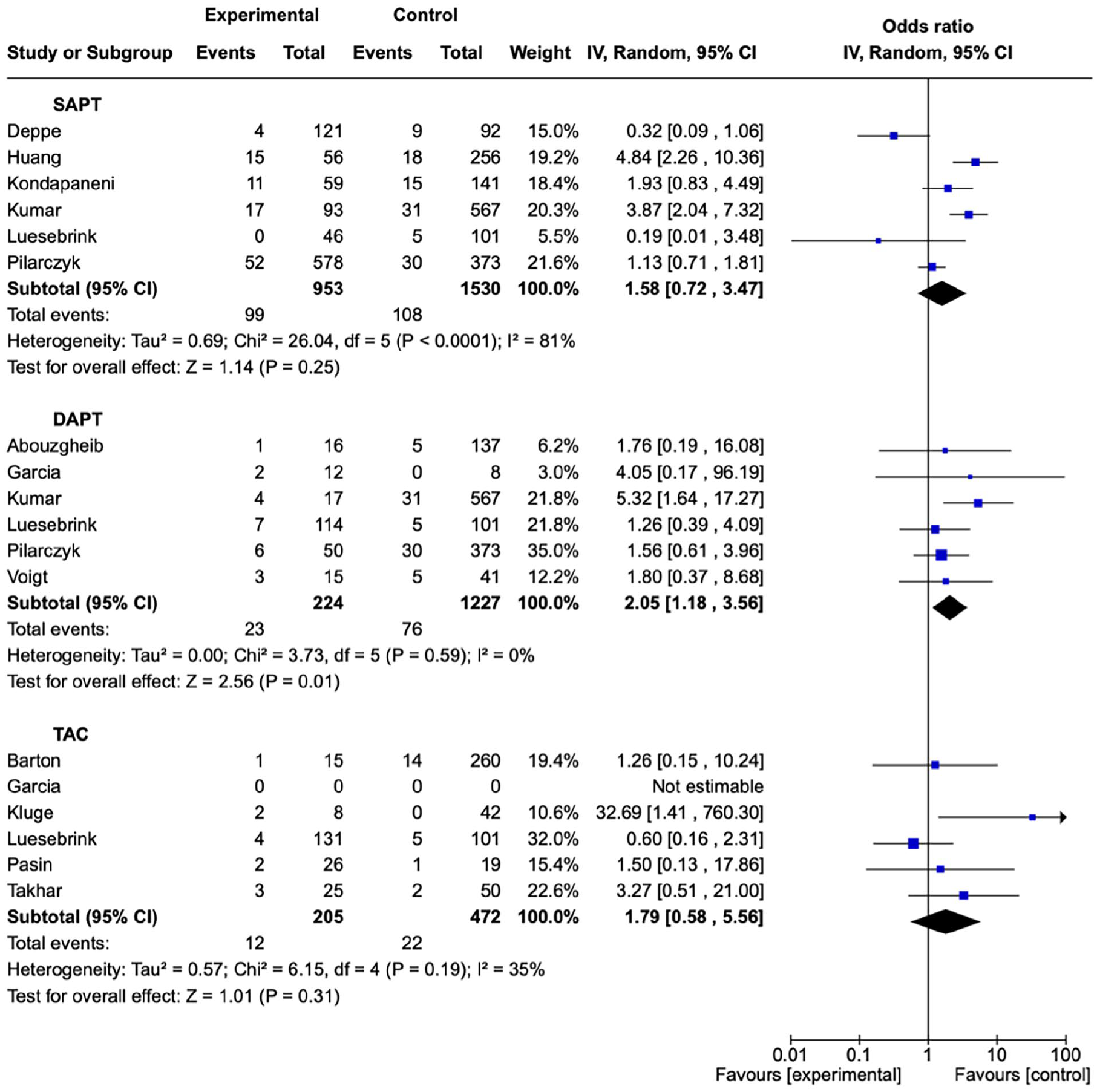
Forest plot for intraoperative bleeding risk during percutaneous tracheostomy insertion.
Outcomes from meta-analysis for any minor and major intraoperative and postoperative bleeding. All comparisons are with those who did not receive antiplatelet or therapeutic anticoagulation therapy.
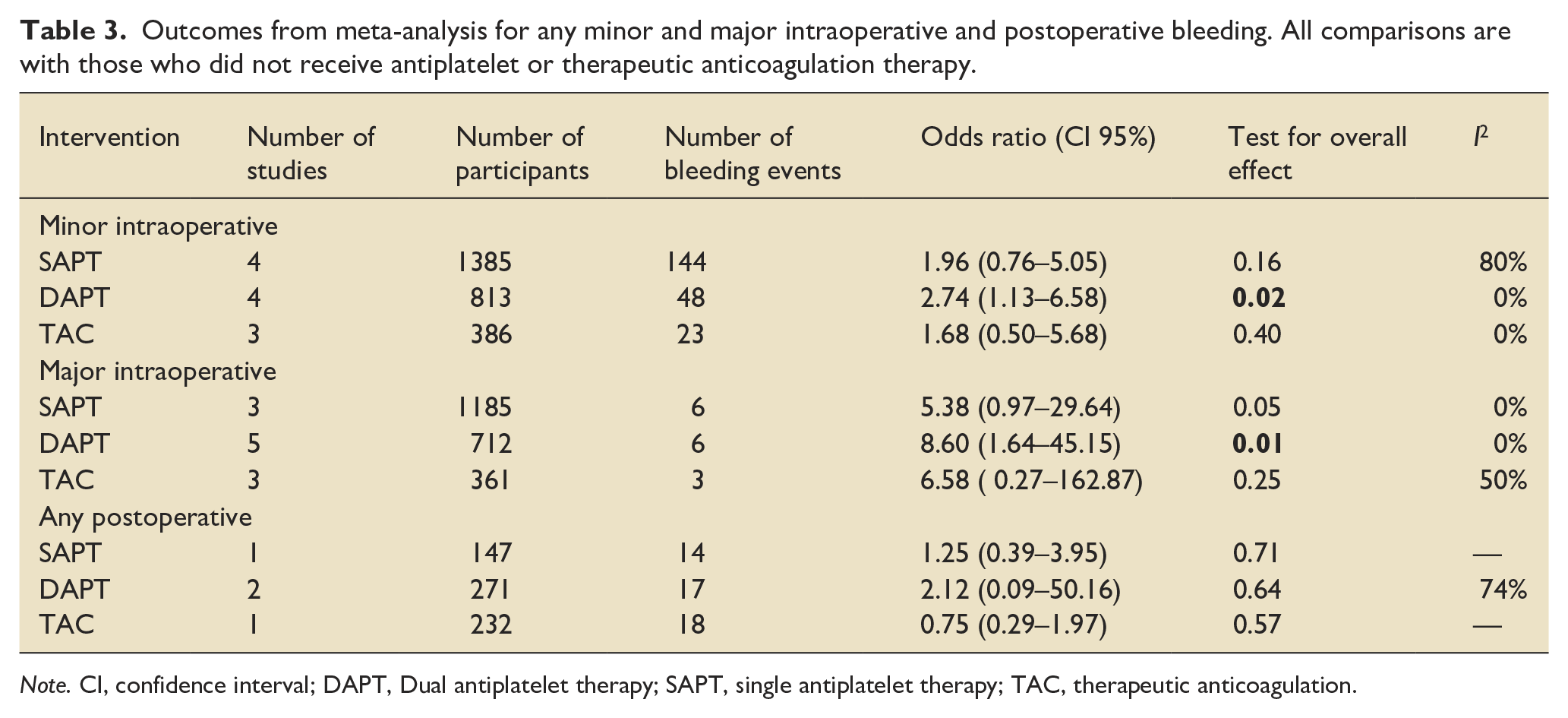
Note. CI, confidence interval; DAPT, Dual antiplatelet therapy; SAPT, single antiplatelet therapy; TAC, therapeutic anticoagulation.
DAPT
Dual antiplatelet therapy would likely increase intraoperative bleeding risk (OR 2.05; 1.18–3.56; I2 = 0%) compared to patients who did not receive antiplatelet or therapeutic anticoagulant therapies (Table 2). The certainty of the evidence was low due to nature of observator studies and risk of bias assesement. However, a strong association was observed due to the magnitude of the effect. Further subgroups of bleeding severity suffered from further imprecision for minor (OR 2.12; 0.09–50.16; I2 = 80%) major (OR 8.60; 1.64–45.15; I2 = 0%) and any postoperative bleeding risk (2.12; 0.09–50.16; I2 = 74%).
TAC
The quality of evidence for therapeutic anticoagulation was very low. Uncertainty remained due to the cessation of anticoagulation prior to insertion of a percutaneous tracheostomy. This was assessed to diminish the true effect observed; therefore, the association of TAC and bleeding risk was not directly observed. Meta-analysis of TAC found no association with intraoperative bleeding (OR 1.79; 0.58–5.56; I2 = 35%) with similar effects observed for minor (OR 1.68; 95%CI 0.50–5.68; I2 = 0%), major (OR 6.58; 95% CI 0.27–5.68; I2 = 50%) and postoperative bleeding (OR 0.75; 0.29–1.97).
Unpublished literature
Two observational studies were found to be unpublished and applicable to our review. One comparing SAPT as clopidogrel monotherapy was withdrawn; we were unable to contact the author of this study for unpublished results (ClinicalTrials.gov ID: NCT02074254). Another study investigating DAPT is undertaking recruitment until the end of 2024 (ClinicalTrials.gov ID: NCT05058469).
Sensitivity analysis and heterogeneity
Overall heterogeneity was moderate or low (Table 2), except for intraoperative bleeding risk for SAPT demonstrating substantial heterogeneity (I2 = 81%). A linear mixed effects model investigation of moderators found that studies based at cardiothoracic ICUs were associated with reduced bleeding risk (Qm = 10.4, P = 0.001). Exploration of other explanators of hetrogeneity was limited by the completion of data provided by studies. Exploration of publication bias was precluded due to the insufficient quantity of studies.
The sensitivity analysis of groups was repeated with the exclusion of studies with a critical risk of bias, which did not affect estimates produced for DAPT bleeding risk. Furthermore, excluding studies with critical risk also did not significantly influence SAPT and TAC estimates (Supplemental Table 2). The substitution of inverse variance for the Mantel-Haenszel method did not change the primary analysis results (Supplemental Table 3). Supplemental Figure 4 shows the Forest plots for any grade of bleeding but restricted to the post-operative period. SAPT, DAPT or TAC were not associated with any differences in post-operative bleeding.
Discussion
We examined the risk of perioperative bleeding in patients undergoing PT insertion while on antiplatelet or therapeutic anticoagulant therapy, comparing it to those not on such therapy or those receiving prophylactic anticoagulation. We found an increased risk of bleeding for patients who received DAPT and no association was found for SAPT and TAC. However, anticoagulation was discontinued for varying lengths of time in the included studies. Previously, there was a lack of substantial evidence as to the risk of these common treatments on bleeding during PT insertion. Our results can support clinical decision-makers in continuing or discontinuing anti-platelet or TAC therapy before the insertion of PT.
The overall methodological quality of the studies we evaluated was poor compared to a well-conducted randomized control trial. However, the nature of the clinical question and diversity of approaches to PT in the critically ill means that observational studies dominate the reported literature. It was clear from our review that there are a range of practices surrounding withholding therapy prior to PT insertion, in keeping with established literature. 11 A previous systematic review of bedside procedures could not conclude if anti-platelet or AC were associated with differences in bleeding risk in PT insertion. 39 Hermann et al. 39 included a greater diversity of studies than our meta-analysis and concluded that there was inconsistent or inconclusive evidence for bleeding risk from SAPT, DAPT, or TAC. Since their publication, several more rigorous studies have been published and, despite their remaining incertitude, these studies met the criteria to included in this meta-analysis.
Further questions remain regarding the recommencement of anti-platelet or AC following tracheostomy and its effect on postoperative bleeding risk. A minority of studies from our analysis reported postoperative bleeding after PT insertion. Heuristic reasoning would suggest that postoperative bleeding risk would reflect the associations observed in the intraoperative setting. On the other hand, prolonged cessation of anti-platelet or AC is unlikely to be feasible given the risks of thromboembolism and cardiovascular complications in the critically ill.
In our study, the bleeding risk was not associated with SAPT therapy and substantial heterogeneity was displayed between studies. The SAPT studies included patients taking Asprin and other pharmacological classes of antiplatelet drugs in common use such as adenosine diphosphate (ADP) receptor inhibitors. Aspirin may not confer an increased risk of bleeding in minor surgical procedures 40 and regional approaches in anaesthesia. 41 Aspirin has been used in clinical practice during the perioperative period as bridge therapy by replacing other SAPT to mitigate the risk of cardiovascular events. However, numerous observational studies 42 have suggested the continuation of aspirin did not increase bleeding risk while contradictory randomised control trials have provided opposing evidence for non-cardiac surgery. 43 Without the availability of high-quality evidence, the decision to continue or withhold SAPT should involve the balancing the risks and benefits on a case-by-case basis.
The relationship between the continuation of TAC during the PT insertion remains unclear, with very low certainty of evidence and substantial heterogeneity. Wide variations in practice surrounding the cessation of TAC prior to PT insertion preclude an accurate and precise interpretation of bleeding risk if TAC were to be continued. Therefore, our analysis reflects current cessation practices rather than the continuation of TAC. An alternative strategy to time-based TAC cessation would be the use of point-of-care tests as an endpoint to allow safe PT insertion by the adequate reversal of coagulation. Consensus strongly supports the practice of performing anticoagulation tests before PT insertion. 44 Several point-of-care tests used in current clinical practice which may aid clinical decision-making are the international normalised ratio (INR) for evaluating the impact of warfarin therapy or coagulopathies. 45 activated clotting time (ACT) for heparin, or thromboelastography (TEG) techniques for platelet function. A reassuring interpretation is the current practice appears to ameliorate the assumed increase in bleeding risk of TAC that was not present in our analysis.
Luesebrink et al.38,46 conducted a multicentre observation study, further delineating anti-platelet and AC groups from those represented in our analysis. Notably, a cohort of patients who have a cardiac plaque rupture or thrombosis will receive triple therapy by incorporating both TAC and DAPT. In their study, there was no association with intraoperative bleeding risk (OR 0.93; 95%CI 0.49–1.66; P = 0.82). 38 As in our planned analysis, further stratifying anti-platelet by TAC was not performed due to a lack of available data. Given that we found DAPT was associated with increased bleeding risk, the addition of TAC to produce a triple therapy would be expected to produce an increased risk of bleeding. This is relevant to cardiothoracic ICUs where triple therapy is used more often.
Several studies were found as a result of the search strategy but were ineligible for quantitative analysis, including those undergoing extra-corporeal membrane oxygenation (ECMO).32 –35 With ECMO, the balancing of bleeding risk is more challenging, given the added risk of circuit thrombosis. For patients receiving ECMO, no standard protocol for the cessation of anticoagulation for procedures exists, due to differences between centres, anticoagulation practices, and individual clotting response to the ECMO circuit itself. Instead, the dangers of bleeding from tracheostomy insertion and circuit thrombosis due to anticoagulation are left to the clinic decision maker, based on the individual patient's needs and guided by clotting tests.
There are several limitations applicable to this meta-analysis. Firstly, there was a dearth of high-quality evidence free from the effects of bias, as found in previous systematic reviews. 39 Unreported confounders such as anticoagulant cessation prior to the procedure or the experience of operators likely distorted the true causal effect from our analysis. Efforts were made to contact authors for operator modifiers of bleeding risk such as pre-operative sonography and experience of interventionalists, however, minimal response from authors precluded inclusion in this analysis. Lastly, there was a lack of discrimination regarding single-agent effects, which would further aid clinical decisions on monotherapy, such as clopidogrel or aspirin.
Conclusion
We aimed to support clinical decision-makers in balancing the risks and benefits of continuing anti-platelet or AC therapy on perioperative bleeding associated with PT. Our analysis suggests that SAPT does not significantly increase the risk of major bleeding associated with PT. However, careful consideration should be given to balance the increased risk of bleeding for patients receiving DAPT. What remains unknown, and a target for future research, is whether an open surgical approach to tracheostomy influences the risk of bleeding for patients receiving DAPT, or whether all DAPT should be converted to SAPT prior to any tracheostomy. Stopping antiplatelet drugs before tracheostomy requires considerable planning due to typically long drug half-lives. The decision to suspend or continue non-antiplatelet anticoagulation based on the lack of significant effect seen in the TAC group could be due to the routine cessation of anticoagulant therapy before PT insertion reported in the candidate studies we identified. A future trial to elucidate best practice would help to clarify risks associated with bleeding, especially whether an open surgical or percutaneous tracheostomy presents different risk profiles for patients taking DAPT. However, the timing and method of tracheostomy in recovering critically ill patients is always a balance of risks and a trial would be difficult if there isn’t a clear sense of clinical equipoise.
In summary, the evidence we present supports temporarily withholding DAPT or TAC (if clinically feasible) to minimize bleeding risk associated with percutaneous dilatational tracheostomy.
Supplemental Material
sj-docx-1-inc-10.1177_17511437251314298 – Supplemental material for Antiplatelet and anticoagulation use and risk of bleeding from percutaneous dilatational tracheostomy insertion: Systematic review and meta-analysis
Supplemental material, sj-docx-1-inc-10.1177_17511437251314298 for Antiplatelet and anticoagulation use and risk of bleeding from percutaneous dilatational tracheostomy insertion: Systematic review and meta-analysis by Mark Brown, Fayez Elsawy, Benjamin Allison and Brendan McGrath in Journal of the Intensive Care Society
Supplemental Material
sj-docx-2-inc-10.1177_17511437251314298 – Supplemental material for Antiplatelet and anticoagulation use and risk of bleeding from percutaneous dilatational tracheostomy insertion: Systematic review and meta-analysis
Supplemental material, sj-docx-2-inc-10.1177_17511437251314298 for Antiplatelet and anticoagulation use and risk of bleeding from percutaneous dilatational tracheostomy insertion: Systematic review and meta-analysis by Mark Brown, Fayez Elsawy, Benjamin Allison and Brendan McGrath in Journal of the Intensive Care Society
Footnotes
Acknowledgements
None.
Abbreviations
AC – anticoagulation
AP – Antiplatelet
DAPT – Dual antiplatelet therapy
ECMO – Extra-corporeal membrane oxygenation
ICU – Intensive care unit
PT – Percutaneous tracheostomy
SAPT – single antiplatelet therapy
ST – Surgical tracheostomy
TAC – therapeutic anticoagulation
Authors’ Note
Presented in part at the Anaesthesia Research Society Meeting, London, May 2024.
Details of authors’ contributions
Literature search: MB
Screening of search: MB, FE
Study design, data analysis: MB, BM
Writing: MB, FE, BA, BM
Final approval of the version to be published: all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
