Abstract
Importance
The role of anticoagulant therapy as an adjuvant treatment for sepsis remains controversial. Although observational studies have often suggested benefits, large-scale randomized controlled trials have yielded conflicting results, and the risk-to-benefit profile across different anticoagulant agents remains unclear.
Objective
To evaluate the efficacy and safety of anticoagulant therapy and its effect on all-cause mortality and major bleeding events in adult patients with sepsis as well as explore differences between study designs and anticoagulant types.
Introduction
Sepsis, defined as a life-threatening organ dysfunction caused by dysregulated host response to infection, 1 remains a leading cause of mortality in intensive care units (ICUs) and a major global health challenge. 2 The pathophysiology of sepsis is characterized by a complex and overwhelming interplay between the inflammatory and coagulation systems, a process widely recognized as thromboinflammation.3,4 This systemic activation of coagulation can lead to the development of sepsis-induced coagulopathy (SIC) 5 and, in its most severe manifestation, disseminated intravascular coagulation (DIC). 6 DIC is an independent predictor of mortality in sepsis, driven by widespread microvascular thrombosis, consumption of coagulation factors and platelets, and subsequent ischemic organ injury, which culminates into multiorgan failure.3,4 Given the central role of coagulation activation in the pathogenesis of sepsis, anticoagulant therapies have long been investigated as a potential adjuvant treatment to interrupt this vicious cycle.
Over the past decades, a variety of agents have been evaluated. Traditional anticoagulants, such as heparin, have been studied alongside novel therapies targeting specific pathways of the coagulation cascade, including recombinant human activated protein C (rhAPC), antithrombin III (ATIII), and recombinant human soluble thrombomodulin (rhTM). Despite a strong biological rationale, the clinical evidence supporting the use of these agents remains highly contentious. Although numerous observational studies have reported associations between anticoagulant use and improved survival, particularly in patients with overt DIC,7,8 these findings have often not been replicated in large-scale, high-quality randomized controlled trials (RCTs). Landmark trials such as PROWESS-SHOCK (for rhAPC) 9 and SCARLET (for rhTM) 10 have failed to demonstrate a mortality benefit, leading to the withdrawal of some agents from the market 9 and a general decline in enthusiasm for this therapeutic strategy. Furthermore, the inherent risk of bleeding associated with anticoagulation in critically ill patients, who often have multiple risk factors for hemorrhage, complicates the clinical decision-making process. This landscape of conflicting evidence leaves clinicians with significant uncertainty. Key unresolved questions include the following: (a) Does any form of anticoagulant therapy offer net benefit in sepsis? (b) Are certain patient subgroups, such as those with confirmed DIC, more likely to benefit? (c) Are there meaningful differences in the risk–benefit profiles of different anticoagulant agents? Therefore, we conducted this systematic review and meta-analysis to synthesize the evidence available from RCTs and observational studies. Our primary objectives were to determine the overall effect of anticoagulant therapy on all-cause mortality and major bleeding in adult patients with sepsis and to explore potential sources of heterogeneity by examining the influence of study design and anticoagulant type.
Methods
This systematic review and meta-analysis was designed, conducted, and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement. 11 The study protocol was registered a priori with the International Prospective Register of Systematic Reviews (PROSPERO, registration number: CRD420251123750).
Eligibility criteria
Studies were included if they met the following Population, Intervention, Comparison, Outcomes, Study (PICOS) design criteria:
Population. Adult patients (≥18 years) diagnosed with sepsis, severe sepsis, or septic shock in an ICU setting. Intervention. Any systemic anticoagulant therapy administered at therapeutic or prophylactic doses. This included traditional anticoagulants (e.g. unfractionated heparin and low-molecular-weight heparin) and novel anticoagulants (e.g. rhAPC, ATIII, rhTM, and protein C concentrate). Comparison. Placebo or standard of care without administration of the specific anticoagulant therapy being investigated. Outcomes. The primary outcomes of interest were all-cause mortality (at the longest reported follow-up) and the incidence of major bleeding events. The specific definitions of major bleeding as reported in each included study are detailed in Table S1. Study design. RCTs and retrospective or prospective observational studies (cohort or registry studies).
Information sources and search strategy
A comprehensive systematic search was conducted in the following electronic databases from their inception to 7 August 2025: PubMed, Embase, Cochrane, and Web of Science. The search strategy combined Medical Subject Headings (MeSH), Emtree terms, and free-text keywords related to “sepsis” (e.g. “sepsis,” “septic shock,” and “septicemia”) and “anticoagulation” (e.g. “anticoagulants,” “heparin,” “thrombomodulin,” and “activated protein C”). To specifically identify RCTs, validated filters were applied where appropriate. The full search strategies for each database are provided in the Supplementary Appendix. Additionally, the reference lists of the included studies and relevant review articles were manually screened to identify any potentially eligible studies.
Study selection
All records identified from the database searches were imported into EndNote, where duplicate records were removed. Two reviewers (Jun Lu and Lina Wang) independently screened the titles and abstracts of the remaining records for eligibility. The full texts of potentially relevant articles were then retrieved and assessed independently by the same two reviewers against the predefined inclusion criteria. Any disagreements during the screening or eligibility assessment phases were resolved through discussion or, if necessary, consultation with a third reviewer (Zhirong Zhang).
Data extraction process
A standardized data extraction form was developed and pre-piloted. Two reviewers (Jun Lu and Lina Wang) independently extracted the following information from each included study: study characteristics, first author’s last name, year of publication, study design, country or region, and number of participating centers.
Patient characteristics included total sample size, baseline demographics (age and sex), severity of illness scores (e.g. Acute Physiology and Chronic Health Evaluation (APACHE II) and Sequential Organ Failure Assessment (SOFA)), and proportion of patients with DIC.
Intervention and control details included the type of anticoagulant, dose, route of administration, duration of treatment, and description of the control group.
Regarding outcome data, for dichotomous outcomes (mortality and major bleeding), the number of events and total number of patients in both intervention and control groups were extracted.
Risk of bias assessment
The methodological quality and risk of bias of each included study were independently assessed by two reviewers. The Cochrane Risk of Bias 2.0 (RoB 2.0) tool was used for RCTs, covering five domains: (a) bias arising from the randomization process; (b) bias due to deviations from intended interventions; (c) bias due to missing outcome data; (d) bias in measurement of the outcome; and (e) bias in selection of the reported result. The Risk of Bias in Non-randomized Studies–of Interventions (ROBINS-I) tool was used for observational studies, covering seven domains, including bias due to confounding and selection of participants. Each domain was judged as “low risk,” “some concerns,” or “high risk” for RCTs and “low risk,” “moderate risk,” “serious risk,” or “critical risk” for observational studies. Disagreements were resolved by consensus.
Data synthesis and statistical analyses
The primary effect measure was the relative risk (RR) with its corresponding 95% confidence interval (CI). Data were pooled using a random-effects model (DerSimonian–Laird method) to account for anticipated clinical and methodological heterogeneity across studies. Hartung–Knapp adjustment was applied to calculate the CIs for the summary effect as this method is more robust when the number of studies is small.
Statistical heterogeneity was quantified using the I2 statistic, with values of <25%, 25%–50%, and >50% considered to represent low, moderate, and substantial heterogeneity, respectively. The chi-square test for heterogeneity was also performed, with a p-value <0.10 indicating significant heterogeneity.
To explore potential sources of heterogeneity and the robustness of our findings, we conducted the following analyses:
Subgroup analysis. We performed a subgroup analysis based on study design (RCTs vs. observational studies). Sensitivity analysis. A leave-one-out sensitivity analysis was performed by sequentially removing one study at a time and recalculating the pooled effect estimate. We also performed a sensitivity analysis by restricting the analysis to only RCTs.
Publication bias was assessed by visual inspection of funnel plots and formally tested using Egger’s linear regression test, with a p-value <0.05 indicating significant asymmetry. All statistical analyses were performed using R software (version 4.5.1; R Foundation for Statistical Computing, Vienna, Austria), primarily utilizing the meta package.
Results
Study selection and characteristics
Our systematic search yielded a total of 4792 records. After removing duplicates and screening, 20 full-text reports were assessed for eligibility. Finally, 10 studies (8 RCTs and 2 retrospective observational studies) met our inclusion criteria and were included in the final review (Figure 1). The 10 included studies involved a total of 7480 patients. The characteristics of the included studies are summarized in Table 1. The 8 RCTs, comprising 4817 patients, were published between 1998 and 2016. The 2 observational studies, comprising 2663 patients, were published in 2016 and 2019, respectively. The anticoagulants investigated included heparin, ATIII, rhAPC, rhTM, and protein C.

PRISMA 2020 flow diagram illustrating the study selection process. The diagram details the number of records identified, screened, assessed for eligibility, and ultimately included in the systematic review and meta-analysis. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Characteristics of the included studies.

ICU: intensive care unit; RCTs: randomized controlled trials; rhAPC: recombinant human activated protein C; rhTM: recombinant human soluble thrombomodulin; DIC: disseminated intravascular coagulation; DrotAA: drotrecogin alfa (activated); DAA: drotrecogin alfa (activated).
Risk of bias assessment
The risk of bias assessment for the eight included RCTs is presented in Figure 2. Overall, two RCTs were judged to be at low overall risk of bias, five had some concerns, and one was at high risk of bias. The primary concerns were related to the selection of the reported results for several early studies and a lack of blinding in the trial by Hagiwara et al. 18 The detailed risk of bias assessment for the two observational studies using the ROBINS-I tool is available in the Supplementary Material (Figure S1). Both observational studies were judged to be at serious overall risk of bias, mainly due to confounding.

Risk of bias summary for the included randomized controlled trials (RCTs). Assessment was performed using the Cochrane Risk of Bias 2.0 (RoB 2.0) tool across five domains: D1: Bias arising from the randomization process; D2: Bias due to deviations from intended interventions; D3: Bias due to missing outcome data; D4: Bias in measurement of the outcome; D5: Bias in selection of the reported result. Green, yellow, and red circles indicate low risk, some concerns, and high risk of bias, respectively.
Efficacy outcome: all-cause mortality
The overall pooled analysis of all 10 studies showed no significant difference in all-cause mortality between patients receiving anticoagulant therapy and those in the control group (RR, 0.92; 95% CI, 0.77–1.11). There was substantial heterogeneity among the studies (I2 = 56.5%; p for heterogeneity = 0.014) (Figure 3).
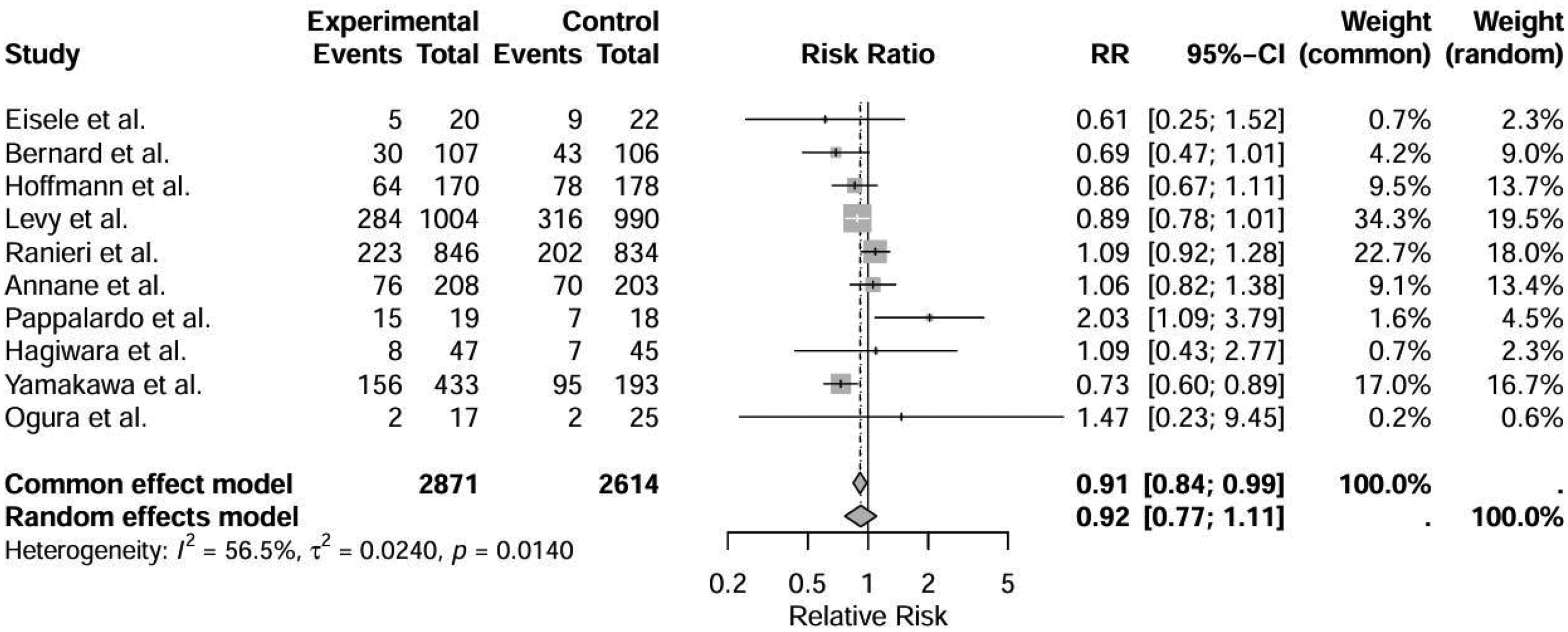
Forest plot of the effect of anticoagulant therapy on all-cause mortality across all included studies. Results are reported as relative risk (RR) with 95% confidence intervals (CIs). The analysis was performed using a random-effects model. The diamond represents the pooled effect estimate.
In a pre-specified subgroup analysis stratified by study design, there was a significant difference between the subgroups (p for subgroup difference = 0.018). The pooled estimate from the eight RCTs showed no significant effect on mortality (RR, 0.96; 95% CI, 0.78–1.18), whereas the pooled estimate from the two observational studies suggested a significant reduction in mortality (RR, 0.74; 95% CI, 0.30–1.81), although with a very wide CI (Figure 4). Further subgroup analysis restricted to RCTs based on anticoagulant type is presented in Figure 5. No specific agent demonstrated a statistically significant survival benefit, and there was a trend toward a difference in the treatment effect across the different anticoagulant subgroups (p for subgroup difference = 0.10).

Subgroup analysis of all-cause mortality based on the study design. The analysis compares the pooled effect estimate from randomized controlled trials (RCTs) with that from observational studies.

Forest plot of the effect of anticoagulant therapy on major bleeding events. Results are reported as relative risk (RR) with 95% confidence intervals (CIs) using a random-effects model.
Safety outcome: major bleeding events
The meta-analysis of nine studies that reported major bleeding events (eight RCTs and one observational study) showed that patients receiving anticoagulant therapy had a nonsignificant, 29% higher risk of bleeding (RR, 1.29; 95% CI, 0.90–1.87). Heterogeneity for this outcome was moderate (I2 = 31.2%; p for heterogeneity = 0.17) (Figure 6).

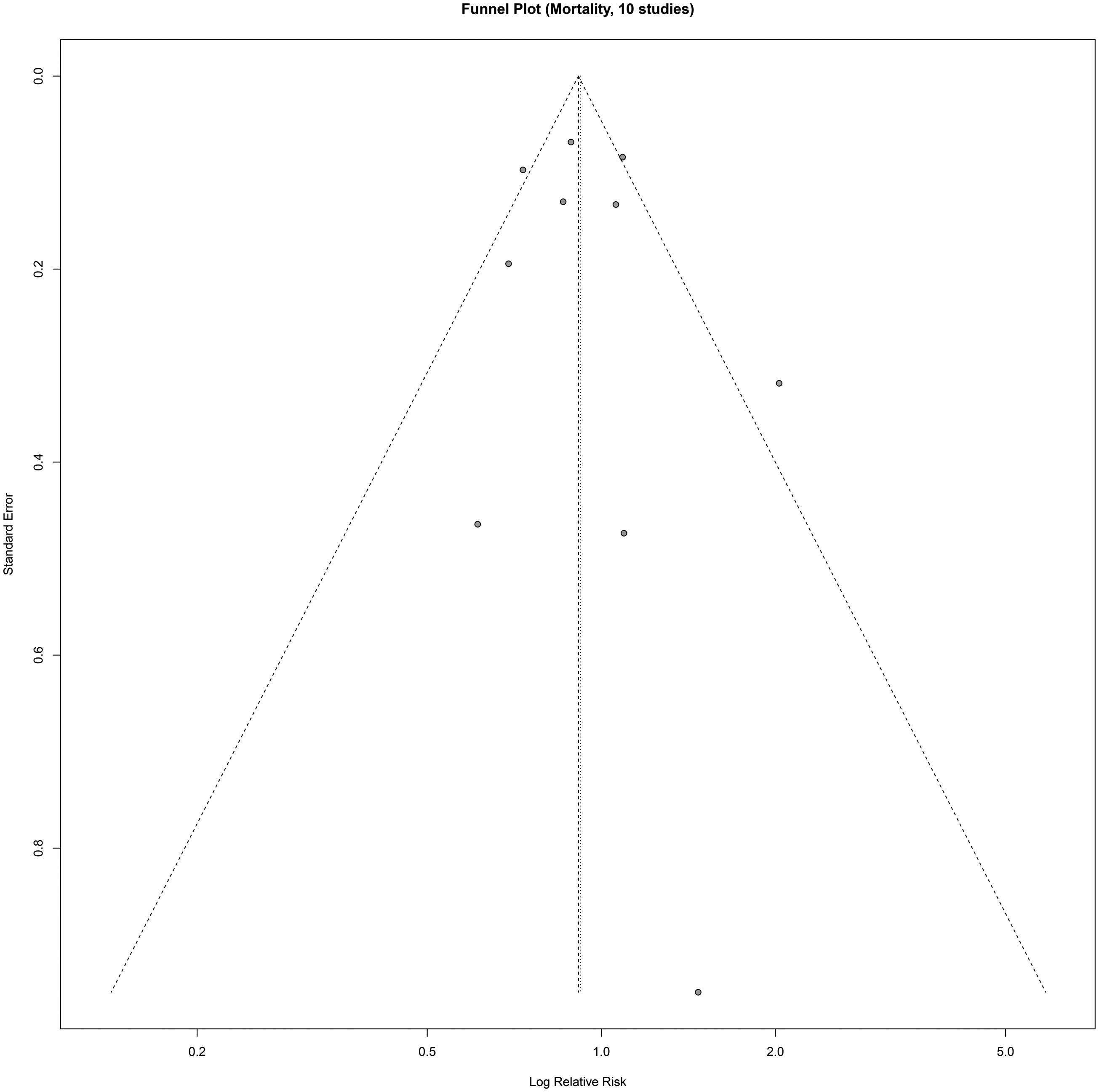
Funnel plot for the assessment of publication bias for the outcome of all-cause mortality. The y-axis represents the standard error of the log RR, and the x-axis represents the log RR. Each point represents an individual study. RR: relative risk.
Sensitivity and publication bias
The robustness of mortality-related findings was confirmed in a leave-one-out sensitivity analysis of the eight RCTs, where the exclusion of any single study did not materially alter the overall null effect (Figure S2). Visual inspection of the funnel plots for mortality (10 studies) did not reveal significant asymmetry (Figure 7). This was supported by Egger’s test, which showed no evidence of small-study effects (p = 0.03). The funnel plot and sensitivity analysis for major bleeding are provided in the Supplementary Material (Figures S3 and S4).
Funnel plot for the assessment of publication bias for the outcome of major bleeding events.
Discussion
Summary of main findings
In this comprehensive systematic review and meta-analysis, our primary finding was that anticoagulant therapy, when applied to a broad population of adult patients with sepsis, does not significantly reduce all-cause mortality (RR, 0.92; 95% CI, 0.77–1.11). This conclusion is robust, primarily driven by the pooled evidence from eight high-quality RCTs. Furthermore, our analysis indicated a clinically relevant, but statistically insignificant, trend toward an increased risk of major bleeding events (RR, 1.29; 95% CI, 0.90–1.87).
Interpretation of key findings and subgroup analyses
Our overall null finding on mortality reinforces the conclusions of several landmark RCTs, such as the PROWESS-SHOCK 15 and SCARLET trials, 21 as well as challenges the routine use of anticoagulants in unselected septic patients. However, the most insightful findings of our meta-analysis emerged from the subgroup analyses, which revealed critical nuances in the evidence.
A significant discrepancy was observed between the results of RCTs and observational studies (p for subgroup difference = 0.018). The pooled RCTs showed no benefit; however, the observational studies suggested a survival advantage. This highlights the substantial risk of confounding by indication in nonrandomized research on this topic. Our findings are broadly consistent with previous large-scale meta-analyses. For instance, Zarychanski et al. 7 and Umemura et al. 22 also concluded that anticoagulants did not provide an overall survival benefit in a general sepsis population. However, the borderline trend toward benefit for heparin in our analysis (RR, 0.89; 95% CI, 0.78–1.01) echoes the findings of Wang et al. 8 who suggested a potential benefit specifically in patients with severe sepsis. Similarly, retrospective studies by Liu et al. 23 have indicated benefits in specific phenotypes. This underscores that although routine use is unsupported, the question of efficacy in precisely selected high-risk subgroups remains pertinent. It is plausible that clinicians preferentially administer anticoagulants to patients with more severe coagulopathy (such as DIC), as suggested by Wada et al. 24 and Levi et al., 25 creating an apparent association that is not borne out in randomized settings.
Our most compelling finding could be the signal of heterogeneity among different classes of anticoagulants (p for subgroup difference = 0.10). This suggests that viewing “anticoagulant therapy” as a monolithic intervention is an oversimplification. Our analysis revealed a spectrum of effects, as described below.
Protein C is an agent associated with a significant increase in mortality (RR, 2.03; 95% CI, 1.09–3.79). This aligns with the eventual withdrawal of rhAPC from the market following the PROWESS-SHOCK results. 15 Previous systematic reviews by Martí-Carvajal et al. 26 and Fourrer 27 have highlighted the lack of benefit and increased bleeding risks, contradicting the previous promising results of the PROWESS trial. 9 Trials by Annane et al. 16 and Levy et al. 14 further confirmed the lack of efficacy in broad populations.
Heparin, a traditional anticoagulant, showed a borderline nonsignificant trend toward mortality reduction. This potential benefit might have been missed due to insufficient statistical power in single large-scale trials such as the HETRASE study by Jaimes et al. 28 Future research needs to focus on high-risk subgroups, a concept supported by current guidelines 2 and pathophysiology reviews.3,4
Other novel agents such as ATIII and rhTM showed neutral effects. The high-dose ATIII trial by Warren et al. 29 and subsequent studies by Hoffmann et al. 13 and Eisele et al. 12 failed to show mortality reduction, potentially due to the interference of concomitant heparin use, as noted by Kienast et al. 30 Similarly, despite strong biological rationale described by Iba et al., 5 trials on rhTM (ART-123) by Vincent et al.10,21 and Hagiwara et al. 18 did not demonstrate significant survival benefits, although post-hoc analyses in coagulopathy subgroups20,31 suggest potential utility. Trials on other agents such as tifacogin also yielded negative results. 32
Strengths and limitations
This meta-analysis has several strengths, including a comprehensive search strategy and adherence to the PRISMA 2020 guidelines. 11 We employed rigorous statistical methods, including the DerSimonian–Laird model 33 and Hartung–Knapp adjustment 34 to ensure the robustness of our pooled estimates. Publication bias was assessed using Egger’s test. 35
However, our study has certain limitations. First, significant statistical heterogeneity was present, likely reflecting the diversity of the included studies in terms of patient populations and evolving diagnostic criteria for sepsis (from Sepsis-1 to Sepsis-3) 1 and DIC6,36 over two decades. Second, our subgroup analysis was limited by the small number of studies for some agents. Third, the definition of “major bleeding” varied across studies. Finally, the observational studies included had a serious risk of bias as assessed using the ROBINS-I tool 37 ; RCTs were assessed using RoB 2. 38 The inherent complexity of sepsis pathophysiology, involving thromboinflammation 39 and varying host responses, 40 further complicates the interpretation of the aggregate data.
Implications for clinical practice and future research
The findings of this meta-analysis have clear implications. For clinicians, our results strongly oppose the routine use of anticoagulant therapy in unselected adult patients with sepsis. It is critical to distinguish this from the standard-of-care use of prophylactic-dose heparin for venous thromboembolism (VTE) prevention. 2 For researchers, the era of “one-size-fits-all” trials is over. Future research should leverage precision medicine, using specific criteria for SIC 41 or DIC 36 to identify responders. New trials should focus on heparin use in high-risk subgroups by re-evaluating the effect of heparin in patients with confirmed DIC or high SOFA scores, using biomarker-guided strategies that incorporate biomarkers, as suggested by Yamakawa et al., 19 to stratify patients and by abandoning harmful agents. Further research on agents such as protein C for broad populations is not warranted.9,26
Conclusions
Anticoagulant therapy does not significantly reduce mortality in the general sepsis population and may increase the bleeding risk. Although its routine use is not recommended, the potential benefit of heparin in specific high-risk subgroups warrants investigation in future high-quality trials.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251409939 - Supplemental material for Efficacy and safety of anticoagulant therapy in sepsis: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605251409939 for Efficacy and safety of anticoagulant therapy in sepsis: A systematic review and meta-analysis by Jun Lu, Zhirong Zhang and Lina Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251409939 - Supplemental material for Efficacy and safety of anticoagulant therapy in sepsis: A systematic review and meta-analysis
Supplemental material, sj-pdf-2-imr-10.1177_03000605251409939 for Efficacy and safety of anticoagulant therapy in sepsis: A systematic review and meta-analysis by Jun Lu, Zhirong Zhang and Lina Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605251409939 - Supplemental material for Efficacy and safety of anticoagulant therapy in sepsis: A systematic review and meta-analysis
Supplemental material, sj-pdf-3-imr-10.1177_03000605251409939 for Efficacy and safety of anticoagulant therapy in sepsis: A systematic review and meta-analysis by Jun Lu, Zhirong Zhang and Lina Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-4-imr-10.1177_03000605251409939 - Supplemental material for Efficacy and safety of anticoagulant therapy in sepsis: A systematic review and meta-analysis
Supplemental material, sj-pdf-4-imr-10.1177_03000605251409939 for Efficacy and safety of anticoagulant therapy in sepsis: A systematic review and meta-analysis by Jun Lu, Zhirong Zhang and Lina Wang in Journal of International Medical Research
Supplemental Material
sj-pdf-5-imr-10.1177_03000605251409939 - Supplemental material for Efficacy and safety of anticoagulant therapy in sepsis: A systematic review and meta-analysis
Supplemental material, sj-pdf-5-imr-10.1177_03000605251409939 for Efficacy and safety of anticoagulant therapy in sepsis: A systematic review and meta-analysis by Jun Lu, Zhirong Zhang and Lina Wang in Journal of International Medical Research
Footnotes
Acknowledgments
The authors wish to thank Dr Wei Zhang, a senior statistician at our institution, for the insightful guidance regarding the meta-analysis methodology. We are also grateful to the peer reviewers and editorial team of the journal for their constructive comments, which have significantly improved the quality of this manuscript.
Author contributions
J.L. and L.W. conceived and designed the study. J.L. and Z.Z. were responsible for the literature search, data extraction, and quality assessment. J.L. performed the statistical analyses. J.L. and L.W. wrote the initial draft of the manuscript. All authors contributed to the critical revision of the manuscript for important intellectual content and approved the final version to be submitted. No artificial intelligence (AI) tools were used in the writing of this manuscript.
Data availability statement
All data generated or analyzed during this study are included in this published article and its supplementary information files. The data are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that they have no conflicts of interest.
Funding
This research did not receive any specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
