Abstract
Background:
Pneumonia is a well-known complication in patients with severe alcohol withdrawal syndrome (SAWS). Antibiotic prophylaxis in ICU treated SAWS patients may be beneficial but data is lacking. The aims of this study were to investigate the effect of introduction of trimethoprim-sulfamethoxazole (TMP-SMX) prophylaxis, on use of broad-spectrum antibiotics and ICU length of stay (LOS) in SAWS patients with refractory delirium tremens (rDT).
Methods:
Retrospective observational cohort study comparing before (control group) and after the introduction of TMP-SMX prophylaxis in patients admitted to a single center ICU because of alcohol withdrawal induced rDT.
Results:
A total of 108 patients were included, 53 patients in the control group and 55 patients in the TMP-SMX group. Baseline characteristics did not differ between the groups (89% male, median age 52 years). The use of broad-spectrum antibiotics was significantly lower in the TMP-SMX group than in the control group (11 vs 70%, p < 0.001). The only variable associated with decreased use of broad-spectrum antibiotics in multiple logistic regression analysis was receiving TMP-SMX (0.06, 95% CI 0.02, 0.19, p-value: <0.001). Median LOS was shorter in the TMP-SMX group than in the control group (61 vs 72 h, p = 0.004). In multiple linear regression analysis receiving TMP-SMX SMX was associated with shorter LOS (−22.7, 95% CI: −41.3, −4.1, p-value: 0.02) and having one or more comorbidities with longer LOS (37.4, 95% CI: 10.4, 64.5, p-value: <0.01).
Conclusion:
Prophylaxis with TMP-SMX in ICU-treated rDT patients was associated with reduced use of broad-spectrum antibiotics and reduced ICU LOS.
Introduction
Alcohol use disorder (AUD) is common and estimated to be prevalent in 21–42% of all hospital-treated patients and 10–33% of intensive care unit (ICU) patients. 1 Patients with AUD are at risk of developing withdrawal symptoms, also known as alcohol withdrawal syndrome (AWS). 2 Even if most people with AWS experience mild symptoms, up to 5% develop severe alcohol withdrawal syndrome (SAWS), including seizures and/or delirium tremens (DT). 2 DT is a life-threatening condition characterized by an unregulated excess of excitatory signals in the brain and a substantial systemic autonomic response, with a mortality rate of 5–15%.1,2 Management of DT is focused on reducing these effects through the administration of sedative drugs targeting the GABA-receptor. Patients who do not respond to mild sedatives, usually benzodiazepines, are defined as having refractory DT (rDT) and are treated with more potent drugs that require care at an ICU due to the higher level of sedation.2,3 Pneumonia is a well-known complication of AUD and AWS, and patients with AUD have both an increased risk of pneumonia and an increased risk of worse outcomes of pneumonia.2,4 –6
S:t Göran’s Hospital (StGH) is a publicly funded, private 350-bed hospital in Stockholm, Sweden. Due to its location close to the largest clinic dedicated to addiction care in the Stockholm area, Beroendecentrum Stockholm (BCS; Stockholm Centre for Dependency Disorders), StGH admits 50–60 rDT patients needing ICU care annually. In November 2018, observations that more than 75% of rDT patients received intravenous broad-spectrum antibiotics during their ICU stay, most often because of pneumonia, prompted the introduction of antibiotic prophylaxis. Based on published etiology data in community-acquired pneumonia in Sweden and in patients with alcohol use disorders, prophylaxis targeting Streptococcus pneumoniae, Haemophilus influenzae, and Staphylococcus aureus was deemed desirable.7,8 Trimethoprim/sulfamethoxazole (TMP-SMX) was chosen based on the combination of being a relatively narrow-spectrum antibiotic, having acceptable activity against the chosen pathogens according to Swedish national resistance data, and being easy to switch from intravenous to oral administration. 9
This retrospective cohort study aimed to investigate the effect of the introduction of TMP-SMX prophylaxis on the use of broad-spectrum antibiotics and length of stay (LOS) for ICU-treated rDT patients.
Material and methods
This is a retrospective single-center cohort study comparing outcomes in patients with rDT in the StGH ICU before and after the introduction of TMP-SMX as antibiotic prophylaxis. The study was approved by the Swedish Ethical Review Authority (No. 2020-00091, May 25, 2020, and amendment No. 2020-05066, October 15, 2020) with a waiver of informed consent, and was conducted in accordance with the Helsinki Declaration of 1975.
Study design
The study period was originally planned to be July 2017 to June 2020 but was prolonged to June 2021 for the two groups to be balanced numerically, due to a sharp decrease in the number of patients with rDT treated at the StGH ICU during the COVID-19 pandemic.
Patients were identified from the electronic medical record system using the International Classification of Disease 10th Revision (ICD-10) code F10.4 (Mental and behavioral disorders due to use of alcohol, withdrawal state with delirium). All charts were reviewed manually, and data collected retrospectively. Inclusion criteria were age ⩾18 years and having received the diagnosis code F10.4 during their ICU stay. Exclusion criteria were being admitted to the ICU for a reason other than rDT, having received ICU care and/or antibiotics within 4 weeks prior to admission, being transferred to the ICU from a hospital other than BCS, having an ongoing infection at ICU admission defined as ongoing antibiotic treatment or fever and C-reactive protein (CRP) >50 mg/L, and receiving a diagnosis of COVID-19 during their ICU stay. For the TMP-SMX group, an additional exclusion criterion was not starting prophylaxis during the first 24 h. For patients with multiple admissions, each admission was included as a separate episode provided that more than 4 weeks had passed since the previous admission.
Treatment of rDT
All patients were initially treated at BCS using a standardized AWS treatment protocol (S2 Table). In short, AWS symptoms were assessed using the Clinical Institute Withdrawal Assessment for Alcohol (CIWA-Ar) scoring system. 10 If the CIWA-Ar score was ⩾8, benzodiazepines were given using a symptom-triggered, front-loading strategy to reduce withdrawal symptoms. In the presence of DT, including continuous withdrawal symptoms despite high doses of oral oxazepam, diazepam was administered intravenously to induce sleep. If sleep was not achieved using a maximum of 600 mg diazepam in 24 h, patients were considered to have rDT and were transferred to the StGH ICU.
The ICU at StGH uses a local rDT protocol (S3 Table). Briefly, the patients are sedated with continuous infusion of propofol, in combination with clonidine if needed. The sedation target is set to Richmond Agitation Sedation Scale (RASS) −3/−4, corresponding to moderate or deep sedation, and is maintained until first daytime after 48 h. Sedation level RASS −3/−4 without intubation was chosen since studies targeting RASS −4/−5 have reported significant side effects, including hypotension and prolonged ICU stay, possibly caused by the high propofol dose and routine intubation and mechanical intubation. 11 The dose of propofol typically used is 25–45 μg/kg/min and approximately half of the patients receives clonidine as adjunctive therapy. Patients receive crystalloids to achieve normovolemia as judged by the attending physician. Respiratory support consists of heated and humidified oxygen delivered by high-flow nasal cannula (HFNC) or non-invasive ventilation with continuous positive airway pressure, with intubation only performed in patients with progressive severe respiratory distress. Routine ICU monitoring is conducted in the form of ECG, invasive blood pressure, respiratory rate, pulse oximetry, and urinary catheter. Arterial blood gas tests are analyzed both regularly and on clinical indication. One qualified nurse and one assisting nurse take care of one or, maximum, two patients, and always have visual surveillance of the patient. There is always at least one qualified anesthesiologist present at the ICU. Decisions regarding the need for intubation and the administration of drugs outside the protocol (e.g., antibiotics) are made by the anesthesiologist in charge. The key features of rDT treatment with sedation without intubation includes bedside presence of a nurse at all times, access to an attending ICU specialists/anesthesiologists at all times, close monitoring and adjustment of sedation levels, and use of HFNO with frequent airway management. Airway management includes regularly nasopharyngeal/nasotracheal suctioning, regularly changes of patient position, and airway recruitment by use of PEP masks. The attending nurse, or assisting nurse, are responsible for this airway management.
Starting in November 2018, the local rDT protocol included antibiotic prophylaxis with 160 mg trimethoprim and 800 mg sulfamethoxazole administered intravenously every 12 h during the ICU stay.
ICU antibiotic policies during the study period
No changes in the ICU antibiotic policies were made during study period. A senior Infectious Diseases (ID) consultant performs daily rounds and decides on all antibiotic treatments. De-escalation of antibiotic treatment is considered daily and performed as early as possible. Pharmacists are not included in decision making unless the ID consultant thinks it is needed. After the round, the ID consultant are available for on-site consultation until 4.30 pm and by telephone until 10 pm during weekdays, and both on-site and telephone consultations until 6 pm during weekends. During the night there is a centralized Infectious Diseases service in Stockholm greater area that can be reached by telephone. If the intensivist in charge during night time is a senior physician, decisions on antibiotic treatment are sometimes made without an ID consultation, while more inexperienced physicians usually use the ID service. Since an ID consultant most often is involved in the decision to start antibiotic treatment in the ICU, it is unusual to stop antibiotics before a clinically evaluation can be made, usually after 48–72 h.
Data collection
Clinical data, including present and previous medical history, ongoing medication, radiology findings, and laboratory findings, were obtained from electronic medical records. Data regarding physiological parameters, intubation, and drug administration in the ICU were extracted from both scanned and electronic medical charts.
Definitions
The date of birth was used to calculate the patient’s age at the time of admission. When there was only a year registered, June 30 was attributed as the day of birth (n = 15 patients). Broad-spectrum antibiotics were defined as any dose of a third-generation cephalosporin (fourth-generation cephalosporins were not available during the study period), piperacillin/tazobactam, and a carbapenem, while narrow-spectrum antibiotics used during the study period was defined as first and second generation penicillins (Penicillin G, amoxicillin, cloxacillin), TMP-SMX and doxycycline; fever was defined as a temperature ⩾38.5°C at admission or >38.0°C after sedation, and immunosuppression was defined as an immunosuppressing disease or immunosuppressive treatment, such as corticosteroids or other immunomodulators. A diagnosis of pneumonia required positive chest X-ray and/or positive respiratory culture from the lower respiratory tract, plus fever and/or significant increase in CRP. Significant microbial findings were defined as growth of Streptococcus pneumonia, Haemophilus influenza, Staphylococcus aureus, or Klebsiella pneumonia in a specimen derived from a deep respiratory location (i.e., sputum, trachea, or bronchoalveolar lavage). A significant increase in CRP was defined as a maximum value ⩾50 mg/L, together with a minimum 100% increase in the CRP value compared to admission. A positive X-ray was one in which the radiologist found a definitive infiltrate and all other findings, including suspected infiltrates, were defined as negative. Comorbidity was defined as chronic obstructive pulmonary disease (COPD), diabetes mellitus, malignancy, and immunosuppression.
Outcomes
Primary outcome was use of broad-spectrum antibiotics and secondary outcome was ICU LOS.
Statistical analysis
Statistical analyses were performed using STATA version 16.1 (StataCorp, TX, USA). A power calculation for the primary outcome, “use of broad-spectrum antibiotics,” was conducted. Based on previous clinical observations, we estimated a 75% frequency of broad-spectrum antibiotic use in the control group and a 40% frequency in the prophylaxis group. To achieve 90% power with a significance level of 95%, a total of 80 evaluable patients were required. Allowing for at least a 10% failure rate, the target number of patients was set to 100.
Differences in baseline characteristics between the treatment groups were assessed using the chi-squared test for categorical variables and the unpaired t-test for continuous variables. To identify factors associated with the primary endpoint, the use of broad-spectrum antibiotics, we performed logistic regression for continuous variables and Fisher’s exact test for binary variables. A multiple logistic regression analysis for the primary outcome included age, Simplified Acute Physiology Score 3 (SAPS3), sex, homelessness, comorbidity, fever, and TMP-SMX prophylaxis based on literature and clinical importance. The results were presented as odds ratios with 95% confidence intervals and p-values.
The secondary outcome, length of ICU stay, was analyzed using linear regression for continuous variables, with results presented as coefficients with 95% confidence intervals. For binary variables, the unpaired t-test was used. A multiple linear regression analysis for the secondary outcome included the same variables as in the analysis of the primary endpoint. A p-value of less than 0.05 was considered statistically significant. The model’s fit was evaluated using McFadden’s Pseudo R-squared.
Results
Patients
During the study period, 175 patients received an ICD-code of F10.4 during their ICU stay: 78 patients before the introduction of TMP-SMX prophylaxis and 97 after. Of these, a total of 108 patients (62%) could be included, 53 in the control group and 55 in the TMP-SMX group (Figure 1). There was a large predominance of males (89%), and the median age was 52 years. We found no significant differences in any of the baseline characteristics between the two groups, including SAPS3, number of days in hospital before transfer to the ICU, fever, and CRP level at admission to the ICU (Table 1).
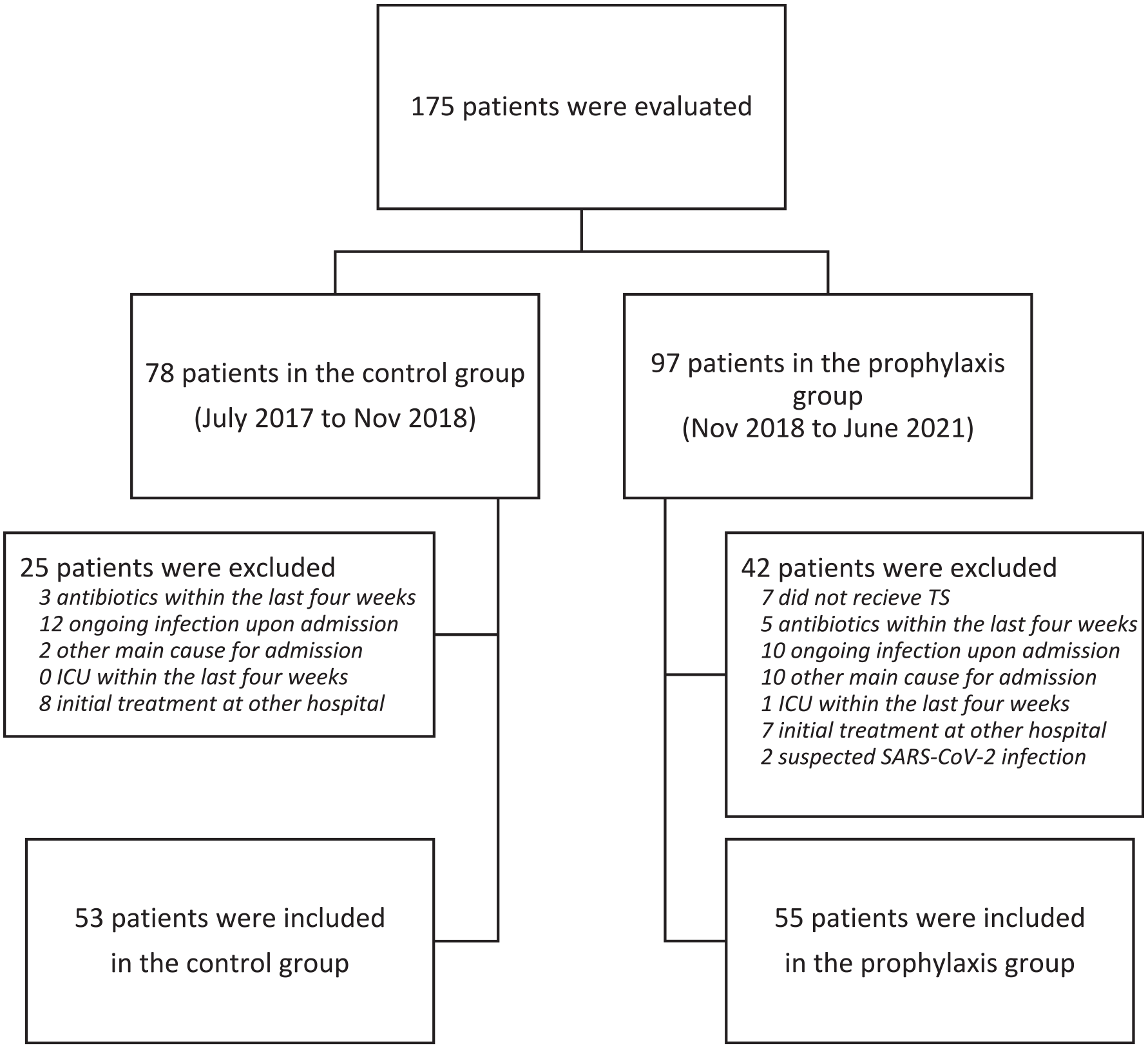
Flow chart showing inclusion and exclusion.
Patient characteristics.
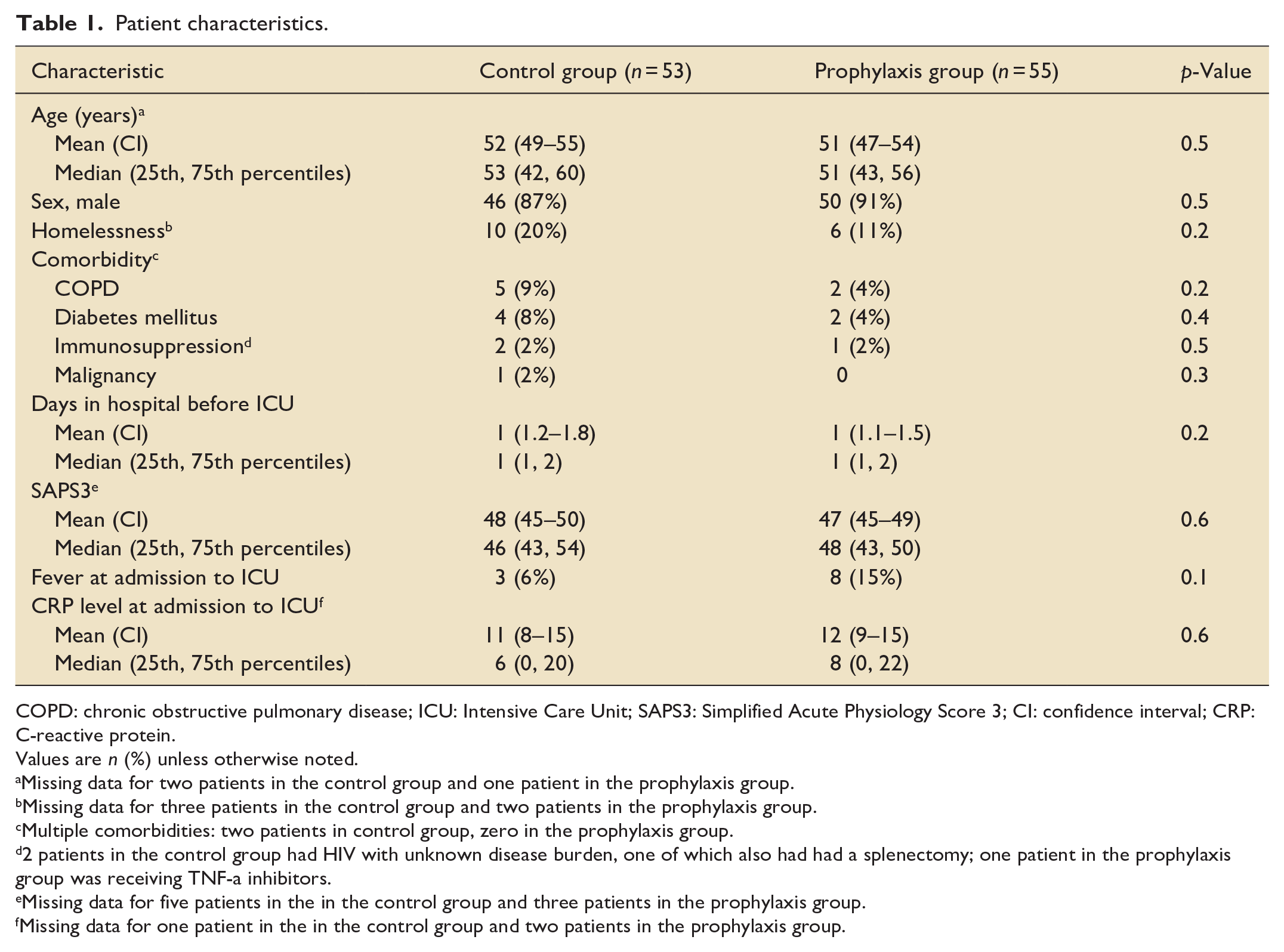
COPD: chronic obstructive pulmonary disease; ICU: Intensive Care Unit; SAPS3: Simplified Acute Physiology Score 3; CI: confidence interval; CRP: C-reactive protein.
Values are n (%) unless otherwise noted.
Missing data for two patients in the control group and one patient in the prophylaxis group.
Missing data for three patients in the control group and two patients in the prophylaxis group.
Multiple comorbidities: two patients in control group, zero in the prophylaxis group.
2 patients in the control group had HIV with unknown disease burden, one of which also had had a splenectomy; one patient in the prophylaxis group was receiving TNF-a inhibitors.
Missing data for five patients in the in the control group and three patients in the prophylaxis group.
Missing data for one patient in the in the control group and two patients in the prophylaxis group.
Use of broad-spectrum antibiotics
Use of TMP-SMX prophylaxis was associated with lower use of broad-spectrum antibiotics as compared to no prophylaxis (11 vs 70%, p < 0.001; Table 2). The only variable associated with decreased use of broad-spectrum antibiotics in multiple regression analysis was receiving TMP-SMX (0.06, 95% CI 0.02, 0.19, p-value: <0.001) (Table 3).
Primary and secondary outcomes.
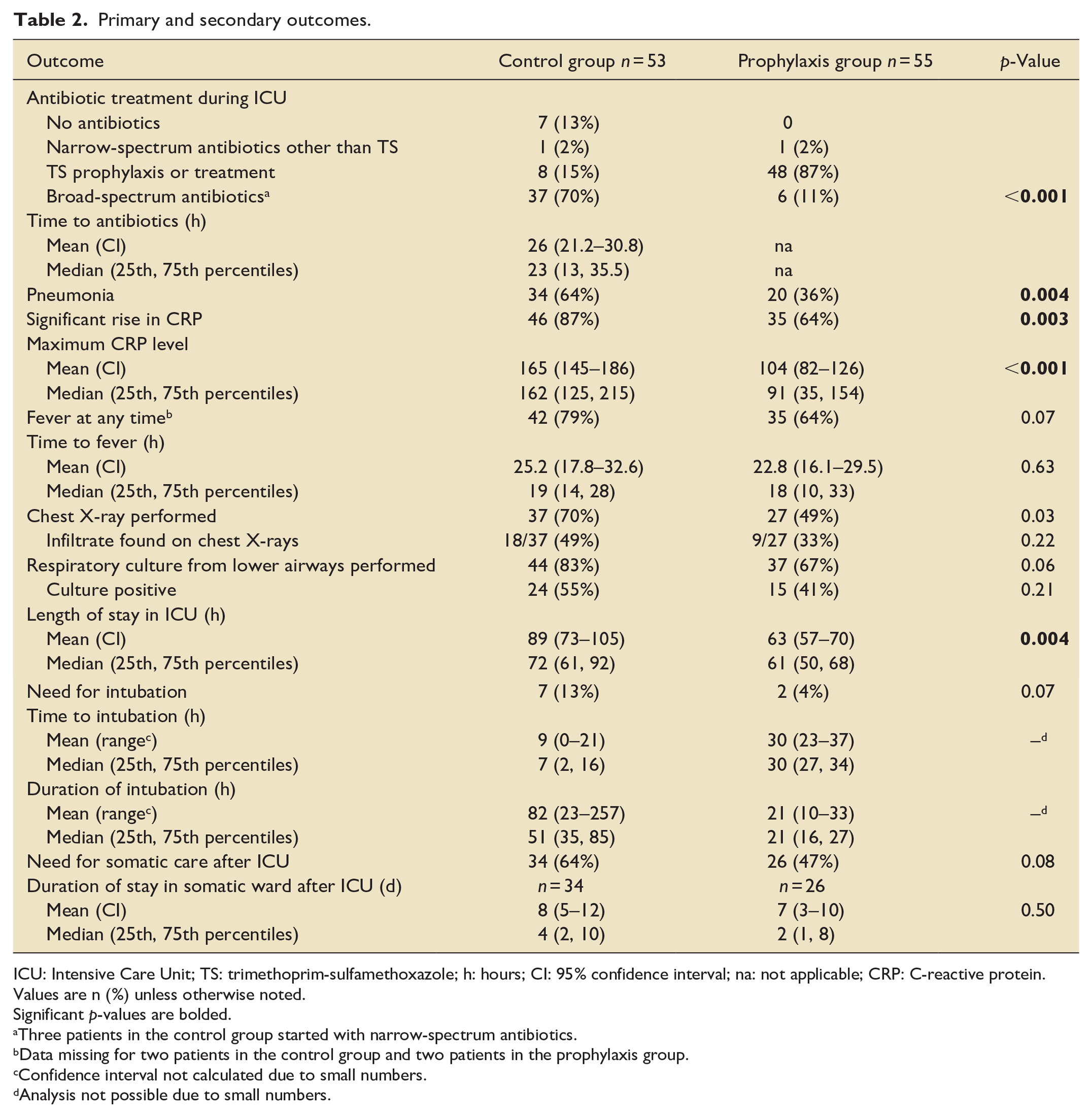
ICU: Intensive Care Unit; TS: trimethoprim-sulfamethoxazole; h: hours; CI: 95% confidence interval; na: not applicable; CRP: C-reactive protein. Values are n (%) unless otherwise noted.
Significant p-values are bolded.
Three patients in the control group started with narrow-spectrum antibiotics.
Data missing for two patients in the control group and two patients in the prophylaxis group.
Confidence interval not calculated due to small numbers.
Analysis not possible due to small numbers.
Primary outcome analysis of factors associated with use of broad-spectrum antibiotics.
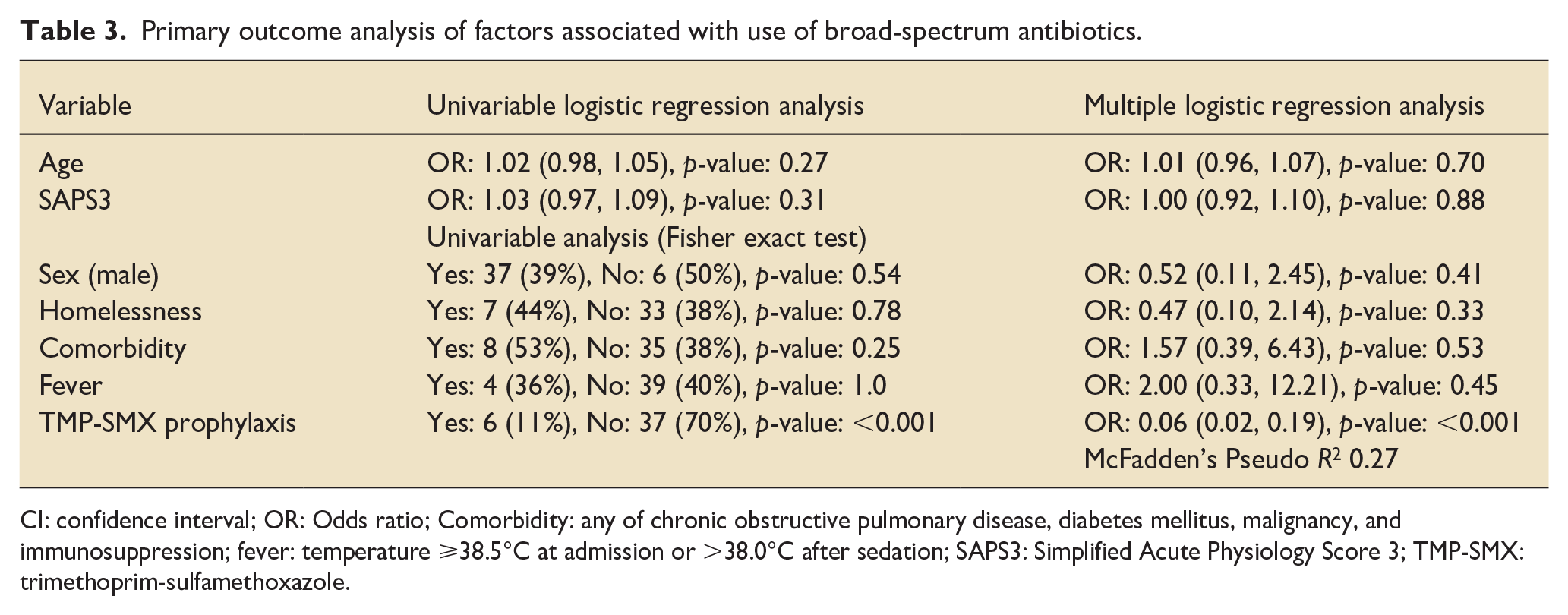
CI: confidence interval; OR: Odds ratio; Comorbidity: any of chronic obstructive pulmonary disease, diabetes mellitus, malignancy, and immunosuppression; fever: temperature ⩾38.5°C at admission or >38.0°C after sedation; SAPS3: Simplified Acute Physiology Score 3; TMP-SMX: trimethoprim-sulfamethoxazole.
Length of stay in the ICU
Prophylaxis with TMP-SMX was associated with shorter median ICU LOS as compared to no prophylaxis (61 vs 72 h, p = 0.004; Table 2). Variables associated with a significant effect on LOS in multiple regression analysis were receiving TMP-SMX (reduced LOS, −22.7, 95% CI: −41.3, −4.1, p-value: 0.02) and having one or more comorbidities (prolonged LOS, 37.4, 95% CI: 10.4, 64.5, p-value: <0.01) (Table S4).
Other outcomes
There were non-significant trends toward the need for less intubation in the TMP-SMX group (4 vs 13%, p = 0.07) and less need for continuation of hospital care for reason other than continued AWS treatment at discharge from the ICU (47 vs 64%, p = 0.08). No patients died during the ICU stay. One patient, in the control group, died during hospitalization, nine days after discharge from the ICU, due to complications of cholecystitis. In the TMP-SMX group, fewer patients had fever (62 vs 80%, p = 0.04) and significant increase in CRP (64 vs 87%, p = 0.03), the maximum CRP level was lower (91 vs 162 mg/L, p < 0.0001) and criteria for pneumonia were less often met (36 vs 66%, p = 0.007). Even if the protocol stipulated that airway cultures should be performed before start of TMP-SMX prophylaxis, cultures were performed after start of prophylaxis in 12 (22%) patients and not at all in 18 (33%) patients. Staphylococcus aureus was the most common finding in both groups (Table 4).
Significant bacterial findings in respiratory cultures.

Discussion
The main findings in this study are that the introduction of TMP-SMX prophylaxis was associated with a reduced use of broad-spectrum antibiotics and a reduction in ICU LOS in ICU-treated patients with alcohol withdrawal induced rDT. Pneumonia is a well-known complication of AWS and is associated with both worse outcomes and increased mortality.12,13 Although many studies have found an association between AUD/AWS and pneumonia, few have looked at pneumonia prevalence according to the severity of AUD/AWS.4 –6,14 A recent North American study by Carlson et al., that looked specifically at pneumonia in patients with SAWS, found a total ICU pneumonia incidence of 55%, with 73% of cases already present at ICU admission. 4 The 66% pneumonia incidence found in the control group in the present study appears to be well in line with these results. The finding by Carlson et al. that most cases of pneumonia were present at admittance to ICU is interesting. SAWS patients are usually admitted to ICU only after failing therapy with mild to moderate sedatives outside the ICU setting. Sedation of delirious patients without continuous airway surveillance reduces the cough reflex and increases the risk for aspiration which may facilitate development of pneumonia. 15 Like in the Carlson study, pneumonia was probably already present in many patients at the time of ICU admission also in the present study, as indicated by the fact that even if administration of TMP-SMX reduced the diagnosis of pneumonia, most patients still developed fever and a significant rise in CRP. Furthermore, in the non-prophylaxis group empiric broad-spectrum antibiotics were started as early as a median of 23 h after admittance to ICU. Thus, TMP-SMX appears have been more of an early treatment than prophylaxis in many patients, which could explain that administration of TMP-SMX led to a significant reduction in ICU LOS as early as within the first 3 days in the ICU.
To our knowledge no study of antibiotic prophylaxis in sedated non-intubated ICU patients have been published. However, two large randomized studies of antibiotic prophylaxis in intubated patients have been published recently, the ANTHARTIC and PROPHY-VAC.16,17 The ANTHARTIC study included patients mechanically ventilated after out-of-hospital cardiac arrest and found that 2 days of prophylaxis with intravenous amoxicillin–clavulanate was associated with a reduced incidence of early ventilator-associated pneumonia, 32 versus 19%. 16 The PROPHY-VAC study included comatose adult patients who required mechanical ventilation for at least 48 h after acute brain injury and found that one dose of intravenous ceftriaxone was associated with a reduced incidence of early ventilator-associated pneumonia, 51 versus 14%, with no microbiological impact and no adverse effects attributable to ceftriaxone. 17 Even if there are substantial differences between these studies and the present one, it appears that antibiotic prophylaxis to ICU patients not being able to defend their airways decreases pneumonia.
An important weakness of the study is that sedation with continuous propofol without airway protection (i.e., no intubation) is not routinely used internationally and not allowed due to regulatory reasons in some countries. As sedation without airway protection may lead to an increased risk of developing pneumonia, generalization of the results may be questioned. 15 However, as discussed above, the fact that pneumonia was a very early event after ICU admission makes it unlikely that the sedation protocol was a significant factor for the high incidence in the control group. Taken together these results indicate that early administration of TMP-SMX, or another appropriate antibiotic depending on the local resistance pattern, may be beneficial in all SAWS patients admitted to ICU.
It could be debatable if the 11-h (15%) reduction in median ICU LOS with TMP-SMX, 72 h in the control group versus 61 h in the TMP-SMX group, is clinically meaningful. However, the mean total ICU LOS in the present study was only 3.2 days (76 h) compared to typically being between 4 and 9 days in studies using protocols with benzodiazepines only, or benzodiazepines with adjunctive phenobarbital.18 –22 A 15% reduction in the ICU LOS as seen in the present study, would in these studies correspond to a reduction in ICU LOS between 0.6 and 1.4 days. One explanation for the short total ICU LOS seen in the present study may be that a sedation target of RASS −3/−4 allows for the use of HFNC and or CPAP, reducing the risk for complications associated with deeper sedation, intubation, and invasive mechanical ventilation.23 –26 If the ICU LOS of 3 days in SAWS patients found in the present study can be confirmed in prospective controlled studies, it would mean a substantial gain to both the individual rDT patients and the healthcare system. 27
Other weaknesses of this study include the lack of an international standard for the definition of pneumonia, and the retrospective design with a risk of researcher bias. We chose to use a conservative definition of pneumonia, requiring a positive chest X-ray or positive respiratory culture from the lower respiratory tract plus fever or a significant increase in CRP. As administration of TMP-SMX prophylaxis turned out to be associated with a lower frequency of X-rays and respiratory cultures, there is a risk of a false low pneumonia incidence in the prophylaxis group. One reason for not performing diagnostic investigations in patients with fever and/or increased CRP, often stated in the medical records, was that the physicians in charge deemed that the prophylactic administration of TMP-SMX was a valid treatment for bacterial pneumonia. This may also have impacted the decision on whether to start broad-spectrum antibiotics or continue with TMP-SMX when patients developed fever and/or an increase in CRP. An interesting finding was that all six patients with growth of Staphylococcus aureus in respiratory cultures performed 2–24 h after start of prophylaxis successfully continued treatment with TMP-SMX only. This indicates that the time for respiratory culture to become negative after start of antibiotics might be longer for Staphylococcus aureus than other respiratory bacteria such as Streptococcus pneumonia.28,29 Finally, weaknesses in the present study also includes lack of safety data and data regarding potential development of antibiotic resistance.
The strengths of this study include the large homogenous patient cohort with two well-balanced groups, with prophylaxis with TMP-SMX being the only treatment difference introduced during the study period. As our hospital is the primary referral center for the majority of rDT patients in the Stockholm region, we think that the patient cohort is representative of this group of patients, allowing for a broad generalization of the results.
Conclusions
Administration of TMP-SMX prophylaxis reduced the use of broad-spectrum antibiotics and ICU LOS in ICU-treated rDT patients. The retrospective study design should be considered when interpreting the results, and prospective controlled studies are warranted to confirm these findings.
Supplemental Material
sj-docx-1-inc-10.1177_17511437241298518 – Supplemental material for Antibiotic prophylaxis reduced broad-spectrum antibiotics and length of stay in ICU patients with alcohol withdrawal induced refractory delirium tremens
Supplemental material, sj-docx-1-inc-10.1177_17511437241298518 for Antibiotic prophylaxis reduced broad-spectrum antibiotics and length of stay in ICU patients with alcohol withdrawal induced refractory delirium tremens by Dunia Al-Hashimi, Martin Kåberg, Anders Krifors, Michael Wanecek and Ola Blennow in Journal of the Intensive Care Society
Footnotes
Acknowledgements
The authors thank statistician Fredrik Johansson, Danderyd Hospital, for help with statistical calculations.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
