Abstract
Background
Acute onset atrial fibrillation is a common dysrhythmia experienced by patients following cardiac surgery which can often cause morbidity and extended hospital length of stay. The primary aim of the study was to explore adherence to National Institute for Health and Care Excellence (NICE) guidance which suggests the need for prophylaxis for postoperative atrial fibrillation (POAF). Secondary aims were to explore factors contributing to the development POAF and the impact of POAF on patient-centred outcomes.
Methods
An analysis consisting of descriptive statistics and regression models was conducted using 138 patient’s records who underwent cardiac surgery between January and March 2017.
Results
We identified 83 (62%) patients on prophylactic rate control medications prior to surgery. During the study period, a total of 50 patients (36%) developed POAF, of which 28 were on prophylactic medication prior to surgery. Patients who developed POAF had significantly prolonged hospital length of stay compared to those who did not develop POAF.
Conclusion
Our study identified a significant proportion of patients not being offered prophylactic rate control prior to cardiac surgery. It is clear that poor patient outcomes are associated with the development of POAF and therefore there is an important need to ensure preventative measures are implemented in guidance relating to the management of these patients. Our results also suggest that tight management of clinical and physiological risk factors prior and during cardiac surgery may improve outcomes in this group of patients and could be considered in future enhanced recovery after cardiac surgery protocols.
Keywords
Introduction
Development of atrial fibrillation (AF) following cardiac surgery such as coronary artery bypass graft (CABG) surgery or valve surgery is common, and can cause significant morbidity (including stroke, heart failure and renal failure) as well increase length of stay (LOS) in both hospital and intensive care unit (ICU).1–8
The reported incidence of AF after CABG is approximately 26%.6,7 A higher incidence has been noted in patients who have had valvular surgery (33–49%) and also those who have had combined valvular and CABG surgery (36–63%).1,8
As postoperative AF (POAF) is such a significant burden, the National Institute for Health and Care Excellence (NICE) guidelines in the United Kingdom (UK) state that any patient undergoing cardiothoracic surgery should be offered AF prophylaxis. 9 A recent descriptive survey undertaken by the Society of Cardiovascular Anesthesiologists (SCA) through collaboration with the European Association of Cardiothoracic Anaesthetists (EACTA) showed a spectrum of practice patterns for prevention and treatment of POAF which do not translate into best available evidence and practice. 10 The aim of our retrospective chart review was to assess whether AF prophylaxis was being administered routinely to patients undergoing elective cardiac surgery. We also set out to evaluate the relationship between perioperative physiological factors that could potentially increase the risk of POAF and assess the relationship between POAF and hospital LOS.
Methods
Study design and participants
We conducted a retrospective analysis of patients who underwent cardiac surgery between January 2017 and March 2017, identifying patients through electronic medical records. This retrospective analysis was conducted on routinely collected anonymised data on individuals within the critical care department at the Queen Elizabeth Hospital Birmingham following institutional approval (CARMS-13386). Adult patients undergoing CABG, valve surgery or a combination were included. Patients with AF prior to study start date, those requiring mechanical circulatory support, aged younger than 18 years of age, or those who have undergone aortic surgery; heart or lung transplants were excluded. One hundred and thirty-eight patients met the inclusion criteria.
Data collection
Parameters and variables collected included: age; gender; type of surgery; acidosis/acidaemia; hypokalaemia; hypomagnesaemia; hypocalcaemia; partial pressure of the arterial oxygen to fractional inspired oxygen (PaO2:FiO2) ratio; hospital LOS; AF prophylaxis; vasoactive drug support; renal replacement therapy (RRT), development of POAF and LOS from operation date. Postoperative AF was defined as new onset AF of at least 30 min duration detected by continuous telemetry electrocardiographic monitoring and 12-lead electrocardiogram. Electrolyte disturbances were defined using our institute’s cut off values: potassium (<4.0 mmol/L), magnesium (<0.7 mmol/L) and calcium (<2.1 mmol/L).
The PaO2:FiO2 ratio per patient was calculated in the first 48 h postoperatively. Hypoxaemia from arterial blood gas measurements was classified into categories defined by the Berlin Criteria 11 ; mild: 201–300 mmHg (≤39.9 kPa); moderate: 101–200 mmHg (≤26.6 kPa); severe: ≤100 mmHg (≤13.3 kPa). The impact of each of these categories on the development of POAF was assessed.
Outcome measures
Adherence to the NICE guidance exploring the use of AF prophylaxis was the primary outcome. Secondary outcomes assessed included exploring (1) the development of POAF and its association with LOS and (2) the association between: hypokalaemia, hypomagnesaemia, hypocalcaemia, vasoactive drug support, postoperative renal replacement therapy, PaO2:FiO2 ratio, and their association with development of POAF.
Statistical methods
Baseline characteristics were descriptively compared. Continuous variables were compared using t tests or Wilcoxon rank sum depending on normality of the data and categorical and nominal variables were assessed using chi-squared tests. Where a regression model was applied, the odds ratio (OR) was calculated using a multiple logistic regression conducted on STATA v14.2, adjusting for age, type of surgery (CABG and valve surgeries) and sex, which have a relationship with the development of AF. 9 The threshold of statistical significance was set at an (two-sided) α level of .05. Box-plot graphs were constructed to illustrate the relationship between POAF development with LOS and PaO2:FiO2 ratio.
Results
One hundred and eight patients (79%) were male. In the total cohort, 50 (36%) patients developed POAF. Mean age was 65 years and the most common type of surgery in our cohort was CABG (n = 90/138, 65.2%), followed by aortic valve replacement (n = 20/138, 14.5%).
We identified that 83 (62%) patients were on prophylactic rate control medications prior to surgery. Twenty-eight of these patients went on to develop POAF. Within the cohort, we did not observe a statistically significant association between receiving a rate control agent prior to surgery and a reduction in the incidence of POAF (OR 0.71; 95% CI: 0.3–1.5, p = 0.380).
Baseline characteristics of patients who developed postoperative atrial fibrillation vs. those who did not.
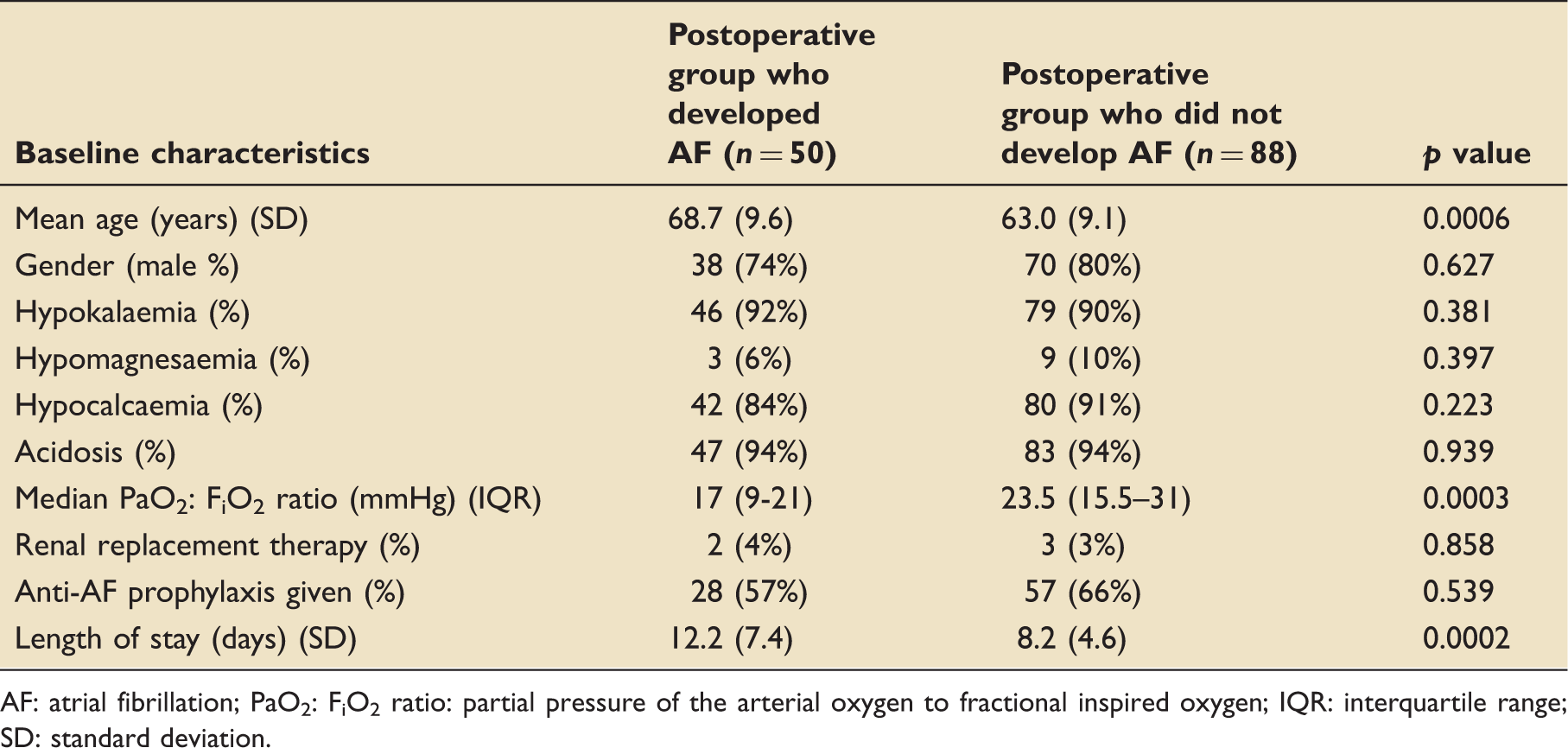
AF: atrial fibrillation; PaO2: FiO2 ratio: partial pressure of the arterial oxygen to fractional inspired oxygen; IQR: interquartile range; SD: standard deviation.
The development of POAF was associated with a significantly increased hospital LOS in comparison to those who did not develop AF (Table 1). The mean LOS (days) in patients with POAF was 12 (SD 7.4) compared to 8 (SD 4.6) days in those who did not develop AF (p < 0.001). Figure 1 demonstrates the difference in LOS between these two groups.
Box plot showing hospital length of stay for patients who developed AF and those who did not. The horizontal line is the median value, the box is the interquartile range and the whiskers extend out to the adjacent values. Outliers are plotted individually as filled circles. AF: atrial fibrillation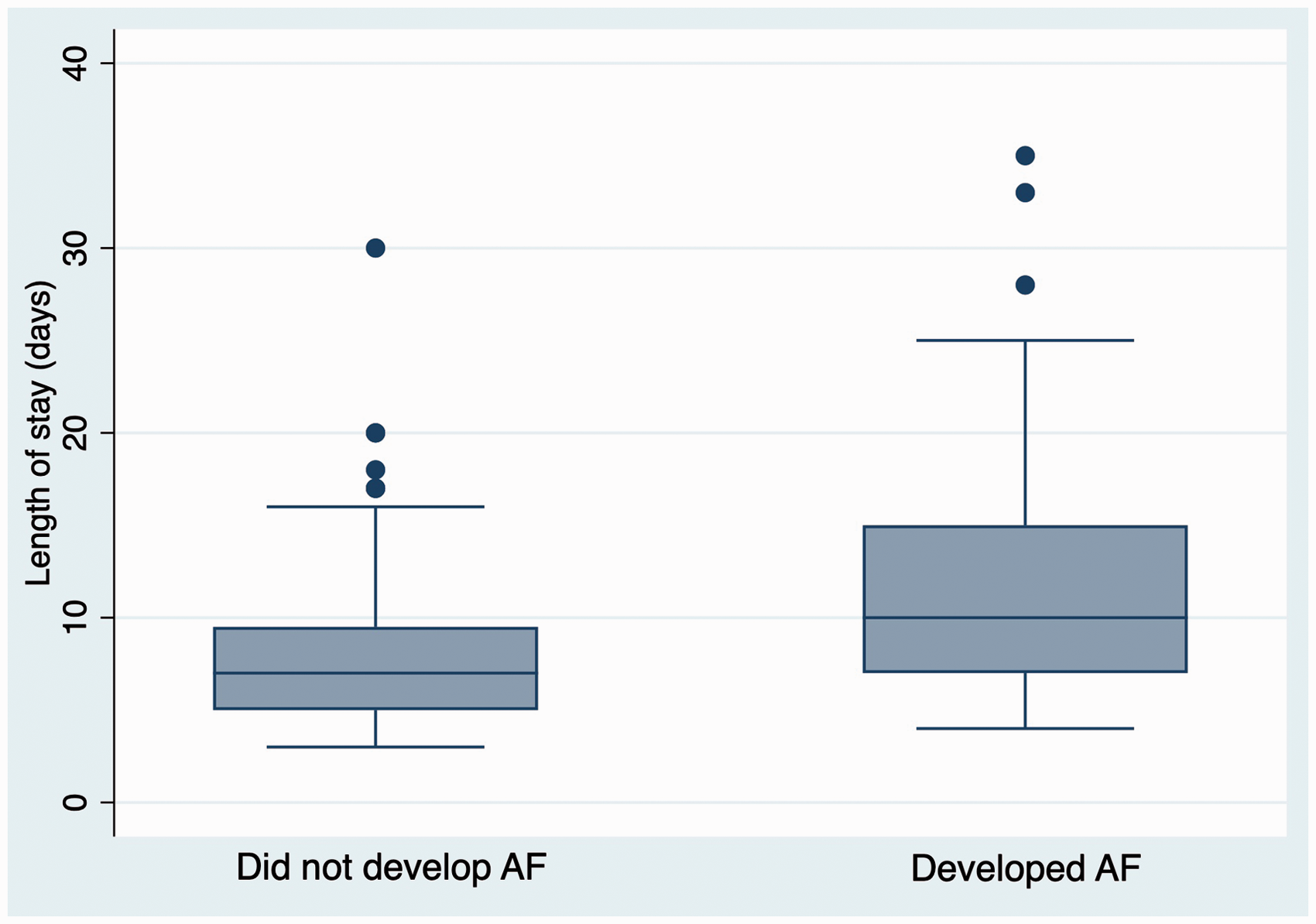
Hypokalaemia (OR 1.56; 95% CI: 0.4–5.9, p = 0.511), perioperative acidaemia (OR 1.03; 95% CI: 0.2–4.9, p = 0.967), vasoactive drug use (OR 5.0; 95% CI: 0.5–47.45, p = 0.157), renal replacement therapy (OR 1.1; 95% CI: 0.2–8.1, p = 0.915), hypomagnesaemia (OR 0.55; 95% CI: 0.1–2.3, p = 0.413) and hypocalcaemia (OR 0.37; 95% CI: 0.1–1.2, p = 0.087) were not significantly associated with the development of POAF.
Out of 60 patients with Berlin criteria defined mild/moderate hypoxaemia, 31 developed POAF. None of the patients within this cohort developed severe Berlin hypoxaemia. On multiple regression, we identified that mild/moderate hypoxaemia when compared to normoxia was associated with the development of POAF (OR 3.81; 95% CI: 1.7–8.4, p = 0.001). 11
Discussion
The primary aim of this study was to explore adherence to the NICE guidance regarding adherence to pre-operative prophylaxis for AF. In this cohort, we identified 83 (62%) patients were on prophylactic rate control medications prior to surgery, which suggest poor adherence to guidance. A meta-analyses investigating the efficacy of beta-blockade in POAF prevention have mostly concluded that beta-blockers significantly reduce the risk of developing AF after cardiac surgery.2,3,12–14 Interestingly, we did not find a significant association between receiving pharmacological prophylaxis and incidence of POAF. This could be due to either our study being underpowered to identify this association, and in addition, we had excluded patients with pre-existing AF, which is a major risk factor for developing uncontrolled POAF. 7
When assessing the whole cohort of 138 patients, 50 (36%) developed POAF. This is in agreement with published observational literature reporting an incidence of 17–47%.1,6,7 Our secondary aim was to explore possible risk factors relevant to the development of POAF. We found an association between mild/moderate postoperative hypoxaemia (as defined by Berlin criteria for severity of hypoxaemia) 11 in the first 48 h and new onset POAF. There is currently a lack of data on oxygenation targets for post-cardiac surgery patients. Of note, Yogaratnam et al. showed that patients who received hyperbaric oxygen prior to commencement of cardiopulmonary bypass for CABG had significantly fewer postoperative complications including POAF. 15
However, the ‘ideal’ oxygenation target in cardiac surgical patients is yet to be determined and least injurious ventilation strategies in the context of cardiac surgery as well as optimal oxygenation targets in the ICU are currently being investigated.16–18
The observed prolonged LOS in the POAF subgroup of patients is in agreement with published observational studies showing that patients who develop AF have an increased postoperative hospital and ICU LOS.1–8 One study demonstrated that the association with increased LOS had an adjusted ratio of mean to be 1.27 (95% CI: 1.20–1.34: p < 0.0001). 5 This could also be explained by data showing that AF patients are more likely to develop complications such as infection, renal failure and stroke also leading to poor outcomes when compared with patients without AF.1–5 However, due to improving perioperative cardiac care and enhanced recovery programmes, LOS is getting shorter. 4
Age has been previously demonstrated as an important factor relating to the development of POAF, with one study suggesting that individuals over the age of 72 were more than five times as likely to develop POAF than those under 55, indicating the clear importance of adjusting for this factor in our statistical modelling. 19
We found no significant association relating to electrolyte imbalances (potassium, magnesium or calcium) and acid–base status to the development of POAF. This is contrary to previous studies which have shown that electrolyte disturbances have been shown to increase the risk of developing POAF.20–23 A pilot randomised controlled trial of prevention of postoperative atrial fibrillation (SPPAF) showed that patients with a potassium level of 3.9 mmol/L or less were more likely to develop POAF in comparison to those whose potassium levels were greater than 4.4 mmol/L (50.7% vs. 32.9% respectively). 20 Potassium is well known to play an important role in the cardiac action potential cycle, in the repolarisation stage. Although the role of magnesium in AF prophylaxis is well documented, the actual mechanism remains controversial.15,20,21 It is thought that magnesium interacts with the calcium channels in the myocytes membrane thus playing an important role in cardiovascular tone. 2 A meta-analysis of 17 randomised controlled trials (n = 2069) examining the effect of prophylactic magnesium on the incidence of supraventricular arrhythmias in cardiac surgery showed that magnesium reduced the incidence of POAF by 29%. 21 However, a more recent meta-analysis focusing on well-conducted studies (adequately powered, double-blind, intention to treat studies where AF was the primary end-point) did not support prophylactic use of magnesium to prevent POAF in cardiac surgery. 22
Our pilot study has several limitations: first, the small sample size mainly due to limited electronic data during the study period (rendering the study underpowered) and the large confidence intervals led to insignificant results in contrast to current literature reports, for instance variables such as hypokalaemia, hypomagnesaemia and not receiving AF prophylaxis are reported to be associated with POAF; however, we did not find these to have a significant association in our analysis;1–3,9,12,20,21 secondly, the retrospective study design has an effect on the validity of the results, for example relying on accurate record keeping and not being able to control other variables that the patients may be exposed to, which may have affected the outcome; third, we did not perform a propensity score matched analysis on the effect of POAF on patient-centred outcomes thus increasing the possibility of bias. In addition, due to limitations in data available, we were unable to collect data on the breakdown of anti-arrhythmic agents used postoperatively, in terms of both drug type, indication and efficacy. Future research could elaborate if particular agents are more efficacious than others. We were also unable to collect further baseline characteristics beyond those described in the results section above. Other factors such as baseline comorbidities (hypertension, chronic obstructive pulmonary disorder, diabetes, etc.), or type of AF prophylaxis used, would be beneficial in understanding the cause of POAF and future work should aim to collect this information. Another limitation which could be addressed in future research is that we were only able to confidently capture POAF during ICU stay when patients were on continuous monitoring. This would have allowed the possibility to capture asymptomatic subclinical occasions of POAF; however, we cannot be confident to have captured all cases of POAF following discharge to the ward or post-hospital destination.
Considering the limitations of the study, our results still suggest some important wider considerations.
There is a clear need for an integrated POAF care model as a component of the transdisciplinary (anaesthetists, cardiac surgeons, intensivists, cardiologists, electrophysiologists, allied health professionals), multimodal ‘enhanced recovery after cardiac surgery’ bundle. Enhanced recovery programmes in non-cardiac surgery have been shown to reduce perioperative complication rate and LOS. 24 Recently, the Enhanced Recovery After Surgery (ERAS) Society published recommendations/guidelines for perioperative care after cardiac surgery; however, POAF management strategies were not included in the consensus recommendations. 25 A dedicated perioperative recovery pathway and evidence-based care (risk stratification, prevention, rhythm/rate control, short/long term anticoagulation) incorporating the recently developed ‘Best Practice Advisory for the prevention and treatment of POAF after Cardiac Surgery’ by the SCA and EACTA 10 could potentially become an element of ERAS programme and accelerate validated measures of recovery.
Conclusions
It is clear that adherence to the NICE guidance on POAF prophylaxis is poor. Considering the morbidity associated with POAF it is paramount that clear, up to date and evidence-based guidelines and protocols are in place to effectively optimise cardiac surgical patients in order to prevent POAF. It would therefore stand to reason that POAF prevention and treatment (taking into account the recently developed SCA/EACTA best practice advisory tool) 10 is targeted in the existing enhanced recovery programmes as an individual intervention that could potentially affect clinically important outcomes in cardiac surgical setting.
Footnotes
Acknowledgements
An abstract of this paper has been presented in part as a poster at the UK Association of Cardiothoracic Anaesthesia and Critical Care (ACTACC) meeting (June 2017) at the Birmingham Repertory Theatre, UK and at the Critical Care Canada Forum (October 2017) at Sheraton Centre Toronto Hotel, Toronto, ON, Canada (CCCF 2017 Abstracts. Can J Anesth Can d’anesthésie 2018; 65: 1–131).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Zochios is supported by an Academic Clinical Fellowship from the National Institute for Health Research (ACF-2016-09-011). Dr Joht Singh Chandan is also supported by an Academic Clinical Fellowship from the National Institute for Health Research.
