Abstract
There are instances where hospitals may experience a relative shortage of oxygen, for example, future pandemics, natural disasters or wartime. We developed a modification to a domiciliary ventilator that captures oxygen normally vented during expiration into the atmosphere, delivering it to the patient. The modification significantly increases PaO2 in mechanically ventilated patients at their baseline FiO2 and baseline FiO2 +1, +2 and +3 L/min compared to no modification. The modification reduces walled oxygen consumption by median (IQR) −1.0 (−1.25 to −1.00) L/min whilst maintaining PaO2. This modification may be a valuable in circumstances where there is a relative shortage of oxygen.
Introduction
Healthcare institutions can experience a relative shortage of oxygen if there is high demand or a shortage of supply. During the COVID pandemic there were numerous instances of hospitals running out of oxygen due to high demand.1 –4 Similarly, in poorly resourced healthcare systems or during natural disasters or wartime, oxygen may be in short supply.
Domiciliary ventilators were frequently used during the pandemic due to a shortage of ICU ventilators. In 2020 we showed, in vitro, that a simple modification to a domiciliary ventilator could capture oxygen normally vented into the atmosphere during expiration to increase FiO2 delivered by the ventilator. 5 Here we tested this modification in vivo in critically ill, mechanically ventilated patients.
Methods
The study was approved by the Wales research ethics committee 6 (Reference 212/WA/0264) on 11 October 2022. To capture oxygen vented into the atmosphere during the expiratory phase we machined an adaptor from medical grade acetal plastic (POM C, Ensinger, Germany). This was attached to the air inlet (which, as with many other ventilators also acts as an oxygen exhaust) on a Breas Nippy 4+ (Breas Medical, Sweden) domiciliary ventilator. A T-piece connector, bacterial filter and reservoir bag were fitted to form the modification (Figure 1), which in the expiratory phase, captures oxygen usually vented into the atmosphere into the reservoir bag. During inhalation, the oxygen rich gas captured in the reservoir bag mixes with the constant oxygen supply, providing a higher oxygen concentration gas mixture to the patient.

Breas Nippy 4+ ventilator with modification fitted.
Participants were switched to the Breas Nippy 4+ for the study. Entrained oxygen (L/min) was adjusted to match the FiO2 delivered on their ICU ventilator. After 10 min FiO2, PaO2 and PaCO2 were measured. The modification was then fitted to the ventilator; after 10 min FiO2, PaO2 and PaCO2 were measured. This process was repeated after increasing oxygen delivered to the ventilator by 1, 2 and 3 L/min compared to baseline with the modification fitted (or not) in random order (Sealed Envelope, Oxford, UK). The modification was then fitted to the ventilator to conserve oxygen; entrained oxygen delivered to the ventilator was reduced to maintain baseline FiO2; after 10 min FiO2, PaO2 and PaCO2 were measured.
Comparison of variables with and without the modification were analysed using the Wilcoxon signed rank test using SPSS v29.0.1.0 (IBM, Chicago, USA).
Results
Ten patients consented to the study; all were receiving spontaneous ventilation (continuous positive airway pressure ± pressure support) via tracheostomy. Baseline characteristics: median (IQR) age 66.5 (60.2–78.2) years; BMI 28.5 (25.1–30.6); Length of mechanical ventilation 13.5 (7.3–28.3) days; FiO2 baseline 0.30 (0.29–0.34); Sequential Organ Failure Assessment score 7.0 (5.75–8.25).
The modification fitted to the ventilator significantly increased PaO2 at baseline FiO2, and with 1, 2 and 3 L/min additional oxygen entrained by a median (IQR) of 1.63 (1.35–2.17) kPa, 1.60 (0.87–2.80) kPa, 2.15 (3.02–8.42) kPa and 3.65 (2.47–4.57) kPa respectively compared to no modification, without significantly affecting PaCO2 (Table 1).
PaO2 FiO2 and PaCO2 with no modification and with modification attached.
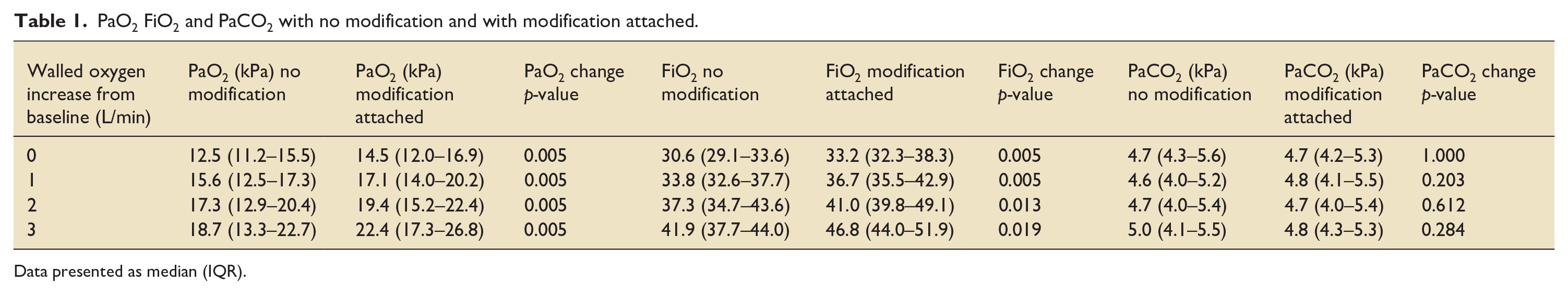
Data presented as median (IQR).
When the modification was fitted to conserve oxygen it significantly reduced wall oxygen consumption by median (IQR) −1.0 (−1.25 to −1.00) L/min (p = 0.003), without affecting PaO2 (p = 0.760), PaCO2 (p = 0.508) or FiO2 (p = 0.759).
Discussion
Our modification offers the potential either to deliver a higher FiO2 to an individual patient using a domiciliary ventilator or, if applied to many patients, to reduce the oxygen consumption of an institution without reducing the FiO2 delivered to patients. By machining an adaptor with the addition of components that are globally and cheapy produced, it could provide a local solution in situations where oxygen demand is increased,1 –4,6 or in natural disasters or conflict where supply may be interrupted or reduced.6,7 The modification has other possible benefits; it may prevent the occasional requirement for a second oxygenator in patients requiring high levels of FiO2 on home ventilation, and has potential environmental and economic benefits by reducing oxygen production requirements.8,9
Limitations
Our ethical committee forbade us enrolling patients by gaining consent from their next of kin and stipulated participants be able to give their own consent. Thus, participants were at the recovery stage of critical illness with modest FiO2. This could limit the generalisability of this data where patients require higher levels of FiO2, 10 but interestingly the modification reduced walled oxygen consumption more in participants with higher FiO2 requirements (0.35 and 0.40) than with lower FiO2 requirements (0.28–0.33) by 2 L/min versus 1 L/min respectively.
Concluding, a simple modification can increase in FiO2 and PaO2 without requiring more oxygen from the wall supply, which may be a valuable modification in circumstances where there is a relative shortage of oxygen.
Footnotes
Acknowledgements
The authors gratefully acknowledge Dr Donna Hall, Robin Hardie, and the medical and nursing staff at Harefield Hospital for facilitating this study, and the patients for agreeing to participate in the study.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MIP is a paid consultant for Philips Respironics; however, Philips had no role in this study. TOJ, PG, TMS, YM declare no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Royal Brompton and Harefield Hospitals Charity Fellowship, who had no role in the design, collection, analysis, interpretation of the data or writing the manuscript.
