Abstract
Background
Despite oxygen being the commonest drug administered to critically ill patients we do not know which oxygen saturation (SpO2) target results in optimal survival outcomes in those receiving mechanical ventilation. We therefore conducted a feasibility randomised controlled trial in the United Kingdom (UK) to assess whether it would be possible to host a larger national multi-centre trial to evaluate oxygenation targets in mechanically ventilated patients.
Methods
We set out to recruit 60 participants across two sites into a trial in which they were randomised to receive conservative oxygenation (SpO2 88–92%) or usual care (control – SpO2 ≥96%). The primary outcome was feasibility; factors related to safety and clinical outcomes were also assessed.
Results
A total of 34 patients were recruited into the study until it was stopped due to time constraints. A number of key barriers to success were identified during the course of the study. The conservative oxygenation intervention was feasible and appeared to be safe in this small patient cohort and it achieved wide separation of the median time-weighted average (IQR) SpO2 at 91% (90–92%) in conservative oxygenation group versus 97% (96–97%) in control group.
Conclusion
Whilst conservative oxygenation was a feasible and safe intervention which achieved clear group separation in oxygenation levels, the model used in this trial will require alterations to improve future participant recruitment rates in the UK.
Introduction
Oxygen is one of the commonest drugs used in the management of acutely unwell patients with respiratory failure requiring mechanical ventilation on an intensive care unit (ICU). Despite this, there is little available evidence or guidance on what level of arterial oxygenation that favours improved survival. It has been proposed that reducing arterial oxygenation targets (termed ‘permissive hypoxaemia’) may not only be safe but might also improve clinical outcomes. 1 A number of studies have attempted to explore the relationship between oxygenation and survival in critically ill patients by analysing retrospective datasets.2–4 The results have been varied and the conclusions must be interpreted with care as it is difficult to avoid confounding by treatment intention (i.e. the more unwell a patient is the greater likelihood of over-oxygenation) using this methodological approach. A recent study of hyperoxaemia in patients admitted to ICU in England used novel methods to reduce confounding and concluded that there was an association between hyperoxaemia and mortality, but did not explore the relationship between hypoxaemia and survival. 5 Three recently published, moderate sized randomised controlled trials evaluating conservative oxygen therapy protocols have shown conflicting results in the critically ill. The first, a single-centre study (n = 434) comparing an oxygen saturation (SpO2) target range of 94-98% to usual therapy was stopped early, reporting a substantial reduction in morality in the intervention group. 6 The second, a multi-centre study (n = 1000) comparing an SpO2 target range of 91-96% to usual care, showed no difference in ventilator-days or survival between oxygenation groups. 7 The third, a multi-centre study of patients with acute respiratory distress syndrome (ARDS) (n = 205, comparing an SpO2 target range of 88–92% to ≥96%) was also stopped early and showed no difference in 28 day survival. 8 The situation is further complicated by the fact that each study recruited different critically ill populations (in terms of severity of illness and diagnosis) and implemented different intervention and control group oxygenation targets. It is therefore impossible to draw any clear conclusion about oxygenation and survival in mechanically ventilated patients based on the current published evidence except to say that there is strong historical evidence of increased mortality for critical care patients with very low oxygen saturation levels (SpO2 <85%) and there is moderate evidence of harm from marked hyperoxaemia with an arterial partial pressure of oxygen (PaO2) >200 mmHg or 26 kPa, equivalent to an SpO2 of 99%-100%.
We therefore set out to conduct a randomised controlled trial to assess the feasibility of recruiting mechanically ventilated patients admitted to ICUs in England into a study evaluating a conservative oxygenation intervention. The purpose of the study was to determine criteria that could be used in a subsequent large trial to evaluate the efficacy and cost effectiveness of the intervention.
Methods
Trial design
The Targeted OXYgen therapY in Critical illness (TOXYC) trial was designed according to the standard protocol items: recommendations for interventional trial (SPIRIT) statements. 9 Its purpose was to determine the feasibility of conducting a randomised controlled trial of a lower than normal (conservative) SpO2 target in adult critically ill patients requiring mechanical ventilation in a National Health Service (NHS) setting. The study was approved by the London Harrow Research Ethics Committee (reference 17/LO/1334) along with approval from the Health Research Authority. The trial protocol and statistical analysis plan were published prior to completion of participant recruitment. 10 The aim was to recruit a total of 60 patients across two sites in England (the Royal Free Hospital, London and Southampton General Hospital) in 15 months; recruitment commenced in January 2018.
Patients
Eligible patients were mechanically ventilated adults (18 years of age or older) within 24 hours of an unplanned admission to ICU, who had a diagnosis of respiratory failure and where it was thought mechanical ventilation was expected to be required for 72 hours or more. Exclusions to enrolment included: admission following surgery (elective or unplanned), patients expected to die within 24 hours of admission to ICU, pregnant women, admission post-cardiac arrest, patients with chronic lung disease known (or highly suspected) to have baseline SpO2 in the range of the intervention arm (88-92%), admission post-trauma (including traumatic brain injury), known sickle cell trait or disease, ongoing significant haemorrhage or profound anaemia, severe peripheral vascular disease, severe pulmonary hypertension, other medical conditions where mild hypoxaemia would be contraindicated, and participation in another interventional clinical trial. Agreement to participate in the study was sought from the patient (if they were deemed to have capacity), a personal consultee, or professional consultee as appropriate. 10 All patient consent and consultee agreement procedures adhered to the Mental Capacity Act (2005).
Randomisation and treatment
Patients were randomly assigned on a 1:1 basis (www.sealedenvelope.com) into either the conservative or control group, stratified by study site, using random permuted blocks of different sizes. In the conservative oxygen therapy group, the fractional inspired oxygen concentration (FIO2) was titrated to achieve an SpO2 of 88%–92%; guidance was provided to bedside staff but the process was not protocolised. In the control group, FIO2 was adjusted to maintain an SpO2 at or above 96%. The study intervention was continued until extubation, formation of tracheostomy, transfer to another ICU or death. Due to the nature of the intervention, neither the research nor the clinical teams were blinded to participant group allocation.
Participants were reviewed by the research team on a daily basis in order to assess compliance with the SpO2 criteria they were allocated to. Where a participant was briefly transferred out of ICU (e.g. investigation, or imaging) the trial was paused until they returned to ICU. Aside from the designated SpO2 targets, all other aspects of care remained the same between the intervention and control cohorts. Regular arterial blood gases were taken during the trial, according to local clinical guidelines. Treating clinicians were able to withdraw participants from the study at any point if were there any medical concerns.
Protocol major amendments
During the course of the trial, it was necessary to amend the study protocol; the major amendments are summarised below:
January 2018 (NOSA001): An addition to the inclusion criteria to allow patients intubated whilst on ICU to be considered for enrolment within 24 hours of intubation. In the inclusion criteria, the expected duration of a potential participant remaining intubated was reduced from >72 hours to >24 hours.
September 2018 (NOSA002): An additional level of agreement to participate in the study was introduced for patients who were deemed to lack the capacity to consent prior their current acute illness.
February 2019 (NOSA003): The removal of the requirement to maintain an SpO2 of ≥96% for the participants allocated to the control group; i.e. patients allocated to the control group had their oxygenation managed purely by the clinical team, without any restrictions. This was necessary as clinicians claimed that the ≥96% target did not reflect usual practice.
Outcome measures
The primary outcome of the study was feasibility; this was defined as the ability to recruit patients and the rate of participant withdrawal from the study. Support for the trial from clinicians and the reasons for withdrawal from the study were also assessed. Feasibility of recruitment was evaluated by monitoring patient screening and their subsequent agreement to participate, along with any withdrawal of consent during or after the study. As part of the feasibility assessment, adherence to the oxygenation targeting component of the study protocol was assessed by monitoring hourly SpO2 and any logged protocol deviations.
A number of secondary outcome measures were evaluated in order to determine relevant endpoints for future trials and to explore potential biological mechanisms. Clinical secondary outcomes included ICU and hospital length of stay, survival at ICU discharge, 30 and 90 days, and pre-defined adverse events, and change in the sequential organ failure assessment (SOFA) score over time. Key physiological measures were also recorded during the intervention period. Blood samples were taken at baseline and on days 2, 3, 5 and 10 after recruitment to measure an array of selected of biomarkers of oxidative stress (not reported here).
Statistical analysis
As the primary outcome measure of this trial was feasibility, no sample size calculation was performed. 11 In view of a predicted mortality of approximately 30% in the study cohort we chose to set our sample size on the higher end of what is usually considered to be acceptable for a feasibility study. Data were collected from bedside charts and entered into an electronic clinical record form (eCRF). Data analysis was conducted blinded to specific group allocation. Primary and secondary outcome measures were presented using summary statistics. Missing data, non-compliers and withdrawals were analysed to determine if was bias seemed likely. Daily time-weighted mean values of FIO2, SpO2, PaO2 and arterial partial pressure of carbon dioxide (PaCO2) were calculated as an area under the curve using the area of trapezoids by multiplying the mean of the measured individual values by the duration of the interval, divided by the complete time of observation, then finding the sum of all values per participant in a 24 hour period. Similarly, for treatment time-weighted means the sum of all values was calculated for the total time on treatment (between 1 and 21 days). For each patient, the proportion of time spent within the randomisation determined SpO2 limits were calculated and summarised by treatment arm. Adverse events were tabulated and grouped according to seriousness, severity and causality.
Results
The two sites recruited a total of 34 participants between February 2018 and October 2019. Recruitment of patients was terminated after the 34th patient as the trial had reached the end of its extended recruitment window.
Baseline and randomisation
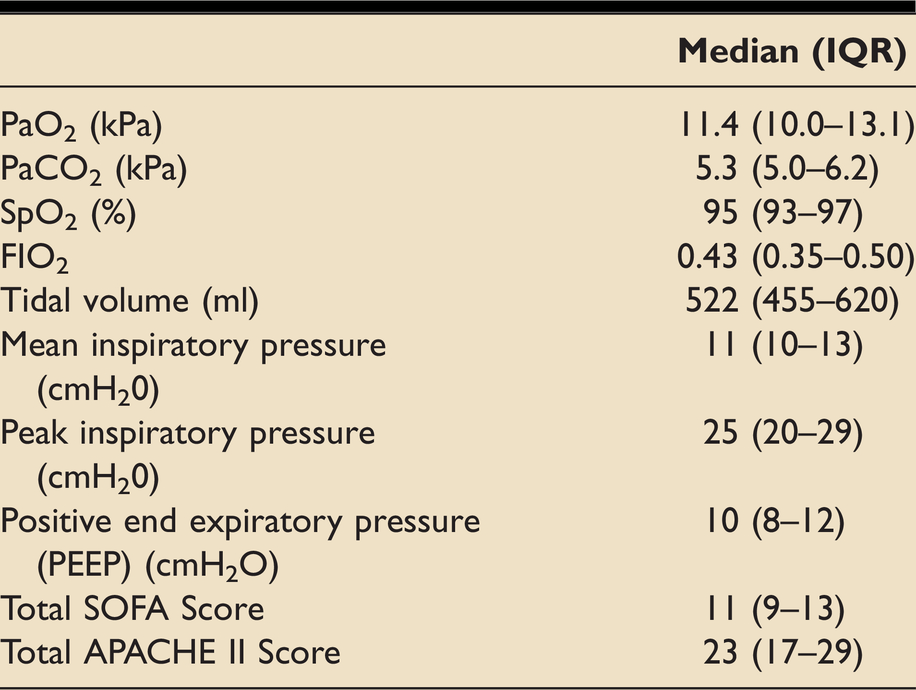
Primary respiratory diagnosis and underlying comorbidities of participants are shown in Tables 1 and 2 respectively; 22 (64.7%) participants were male and the median age was 66 years (IQR 58–74). Key baseline respiratory measures are displayed in Table 3. 27 (79.4%) participants were on a mandatory mode of ventilation at the start of the intervention, 4 (11.8%) on a spontaneous mode and 4 (11.8%) on a mixed mode. Randomisation was balanced between the two treatment groups (17:17) and was also balanced at each site (10:10 at site one and 7:7 at site two). A summary of missing data is shown in the online supplementary information; no evidence of bias was detected.
Primary respiratory diagnosis of study participants at enrolment.

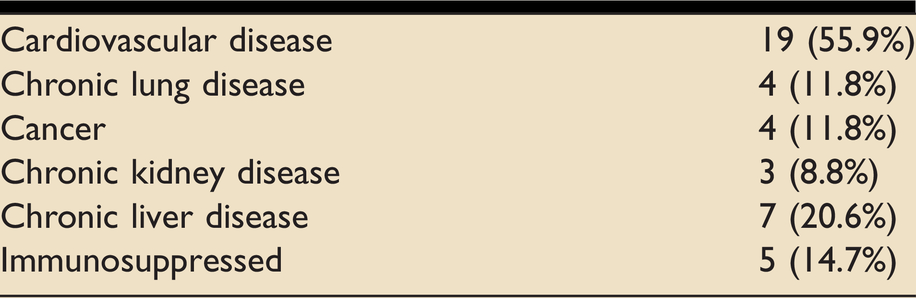
Underling comorbidities of participants at enrolment.
Baseline respiratory characteristics of all participants.
Feasibility
The 34 participants were recruited over 622 days, an average rate of 20 participants per year or 1.7 per month. There were two withdrawals of consent from the study by Personal Consultees. Four participants were withdrawn from the study by the clinical team; reasons for this were i) development of a new stroke (intervention group); ii) concerns over excessively high FIO2 in a patient with bronchiectasis on a new CT scan (control group); iii) development of ischaemic colitis (intervention group); and iv) concerns over high FIO2 to maintain the SpO2 target (control group). A number of factors were identified that may have contributed to the low recruitment rate observed in this study: Method of obtaining consent from participants/agreement from consultees Control group SpO2 parameters. Low number of recruitment centres Narrow inclusion criteria.
Intervention
The overall median (IQR) duration of intervention was 6 (3-10) days. The reason for the intervention being terminated in each participant (either within protocol or for other reasons) are summarised in Table 4.
Reasons for terminating the intervention.

aPredetermined stop points for the intervention.
bSpecified in the protocol as no ethics permission in receiving hospital.
Protocol adherence
Figure e2 in the online supplementary information shows the daily time-weighted mean values for SpO2 according to randomisation group. Of the 17 participants randomised to the conservative oxygenation group, 73.1% of daily time-weighted mean values were between 88-92% SpO2. Of the 17 participants randomised to usual care, 75.2% of daily time-weighted mean values were between 96 to 100% SpO2.
Oxygenation measures
The median (IQR) time-weighted mean SpO2 for participants in the conservative oxygen therapy group was 91 (90-92)% and for those in the control group it was 97 (96-97)% (Table 5). The daily time-weighted mean values for FIO2, SpO2, PaO2 and PaCO2 are shown in the online supplementary data (figures e1-e4 respectively).
Treatment time-weighted averages [median (IQR)].

Adverse events
There was a total of 75 adverse events reported in 24 participants; the details are summarised in the online supplementary information (Table e2). 37 of these adverse events were in the conservative oxygenation group and 38 in the control group. The adverse events appeared well balanced between the two groups in terms of severity, causality and expectedness. There was a total of 23 serious adverse events; 10 in the conservative oxygenation group and 13 in the control group. There were 5 deaths in the conservative oxygenation group and 4 in the control group. There appeared to be marginally more respiratory and cardiovascular adverse events in the conservative oxygenation than the control group (65% vs. 53%, and 71% vs. 53% respectively).
Clinical secondary outcomes
Table 6 displays the key clinical secondary outcome measures by randomisation group. There was a trend towards higher survival and shorter ICU and hospital length of stay in the control group. No statistical analyses were performed as the trial was not powered to detect a difference in these secondary measures.
A summary of key clinical outcomes by randomisation group.
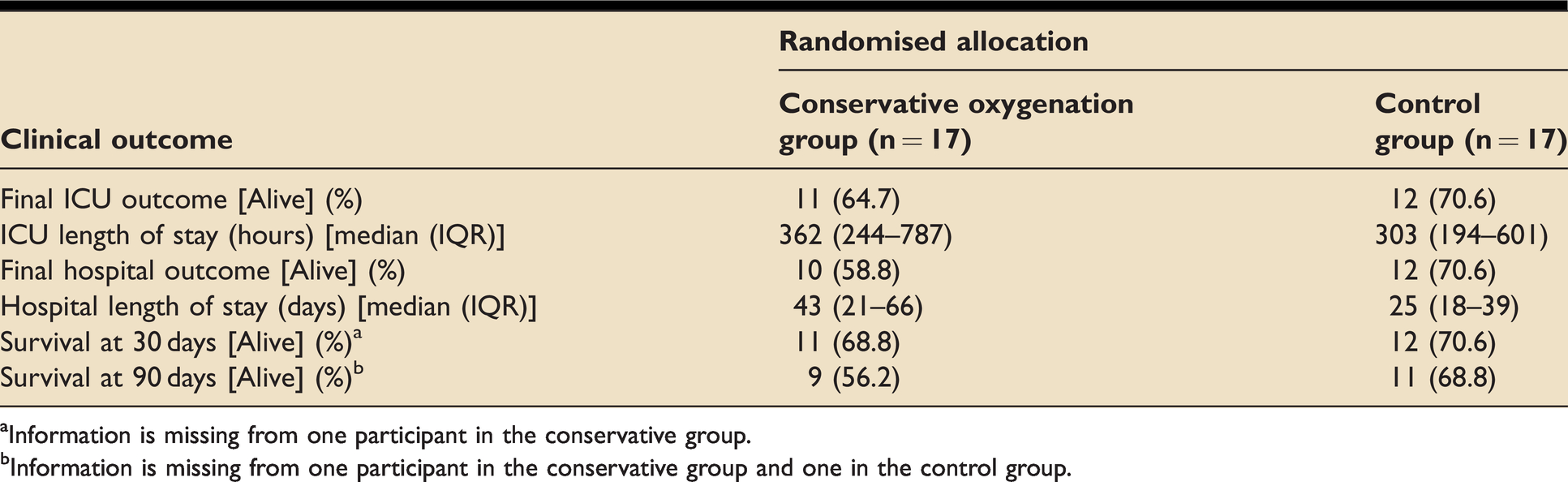
aInformation is missing from one participant in the conservative group.
bInformation is missing from one participant in the conservative group and one in the control group.
Discussion
Whilst a number of moderate sized RCTs have set out to address the issue of determining the optimal oxygenation targets in critically ill patients, none of them have been conducted in the setting of the UK NHS, and to date, no clear answer has emerged. For these reasons, we undertook a study to assess the feasibility of conducting a trial in which mechanically ventilated NHS patients were enrolled and randomised to assess a conservative approach to oxygenation (SpO2 88–92%). The primary outcome of this trial was an assessment of feasibility which included the ability to recruit participants, deliver the intervention, retain patients in the study and collect meaningful data.
Adequate recruitment of participants is key to the success of any trial and is determined by factors such the suitability of inclusion criteria for the population to be screened, willingness of patients (or their next of kin) to enrol, the intervention (including an understanding of its potential benefits and harms) and the method and duration of data collection/sample collection. Recruitment to this trial was considerably slower than was predicted, which led to the study being stopped before reaching the target number of participants. The original plan was to recruit 60 patients at two centres in 15 months; this equates to 4 patients per month. A total of 34 patients were recruited to the study at a mean rate of 1.7 per month. Recruitment rate was reviewed regularly during the study, both at Trial Management Group (TMG) and Trial Steering Committee (TSC) meetings. The factors highlighted to have contributed to the low recruitment rate would need to be fully addressed to improve recruitment in a future trial:
In summary, a future trial of conservative oxygenation in an NHS setting should consider broad inclusion and minimal exclusion criteria, an unrestricted usual care comparator group, a deferred consent model for enrolment and a large number of simultaneously recruiting sites.
The intervention was halted prematurely (outside of a protocolised reason to end the intervention period) in a total of 10 (29.4%) patients. The commonest reason (40%) was a clinician withdrawing the patient from the study. In these cases, clinicians expressed concerns regarding oxygenation in both the conservative oxygenation (20% of clinician withdrawals) and control (20%) groups. This was an unexpected finding and efforts to rectify concerns in the control arm of the study led to the protocol change outlined above. Adherence to the designated SpO2 targets was comparable between the two groups (Figure e2) and achieved for the majority of the time. Separation of oxygenation measures between the randomisation groups was good, as highlighted in Table 5 and Figures e1-4. There was a difference of 3.2 kPa between median PaO2 values in the conservative and usual practice groups (Table 7), which compares to 1.6 kPa in the recently published ICU-ROX trial. 7 Of note, this was achieved despite only a small difference between median FIO2 in the two groups of our study (0.35 versus 0.37 in conservative and usual care groups respectively) (Table 5).
Separation of intervention groups in terms of arterial partial pressure of oxygen for recent trials of conservative oxygen therapy.
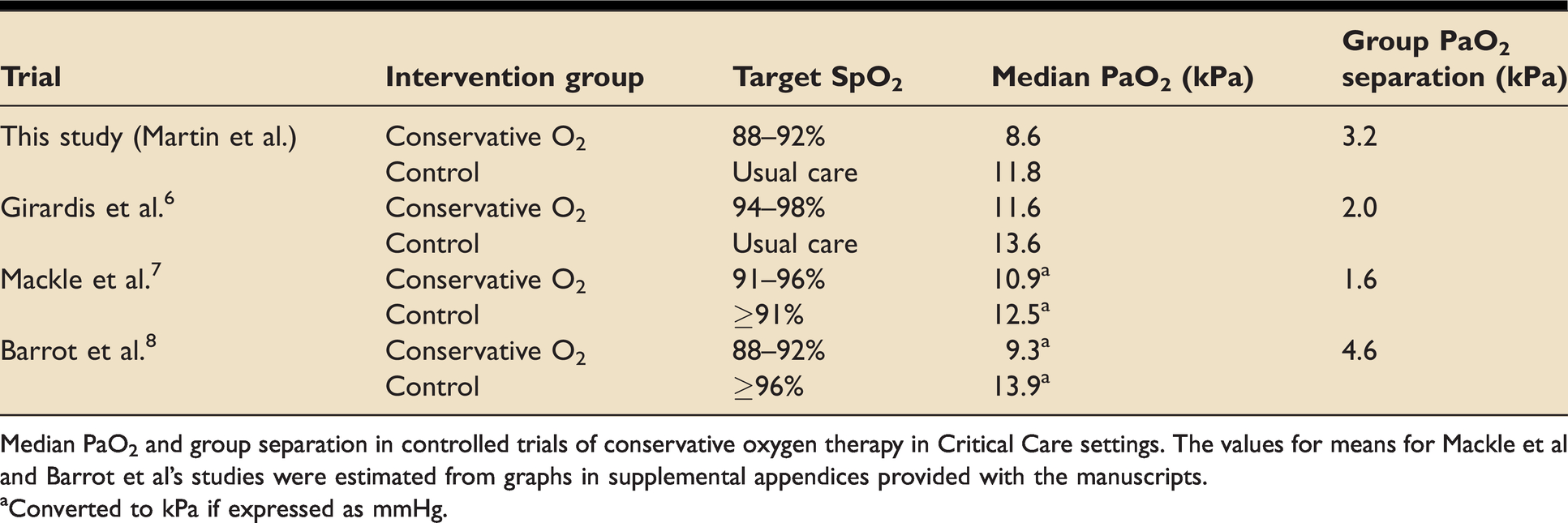
Median PaO2 and group separation in controlled trials of conservative oxygen therapy in Critical Care settings. The values for means for Mackle et al and Barrot et al’s studies were estimated from graphs in supplemental appendices provided with the manuscripts.
aConverted to kPa if expressed as mmHg.
This study was not powered to formally assess the safety and efficacy of the intervention as compared to practice in the control group. The values in Table 6 show a trend toward better survival in the control group, accompanied by a shorter ICU length of stay; along with a similar pattern for hospital survival and length of stay. Whilst it is impossible to place any meaning to these findings due to the design of this study, it is noteworthy that a recent trial of conservative oxygen therapy (target SpO2 88–92% in patients with ARDS reported a 28 day mortality of 34.3% in the conservative oxygen group and 26.5% in the ‘liberal’ oxygen group (SpO2 ≥96%), a difference of 7.8% (95% confidence interval −4.8 to 20.6). 8 The trial was stopped early by the data and safety monitoring board because of safety concerns and a low likelihood of a significant difference between the two groups for the primary outcome.
Many of the limitations of this study design have been outlined above. The purpose of the study was to determine whether patients could be enrolled into a trial that delivered an intervention to generate separation in terms of delivered oxygen concentration and arterial oxygenation. An additional limitation was what appeared to be a high degree of missing data in this study (Table e1 in the online supplementary information). Many of the missing figures were due to these data not being part of routine ICU data capture at the frequency stated in the CRF. For example, the high degree of missing values for arterial blood gases is because they are not routinely taken hourly; they tend to be taken 4-6 hourly, at the discretion of the clinical team. Finally, the administration of oxygen therapy cannot be delivered in a blinded manner, therefore it is possible that knowledge of group allocation could have led to bias in the study results.
Conclusion
Whilst this trial was feasible in an NHS setting, it would require a number of fundamental alterations to the design of the study in order for it to be successful on a larger scale. There remains no answer to the question of whether a conservative oxygen intervention is beneficial to mechanically ventilated ICU patients, nor the precise SpO2 target for the intervention. The methodology of this feasibility study delivered a clear separation of SpO2 and PaO2 between the intervention group and the control groups.
Supplemental Material
sj-pdf-1-inc-10.1177_17511437211010031 - Supplemental material for A feasibility randomised controlled trial of targeted oxygen therapy in mechanically ventilated critically ill patients
Supplemental material, sj-pdf-1-inc-10.1177_17511437211010031 for A feasibility randomised controlled trial of targeted oxygen therapy in mechanically ventilated critically ill patients by Daniel S Martin, Margaret McNeil, Chris Brew-Graves, Helder Filipe, Ronan O’Driscoll, Jia Liu Stevens, Rachel Burnish, Andrew F Cumpstey, Norman R Williams, Michael G Mythen and Michael PW Grocott in Journal of the Intensive Care Society
Footnotes
Acknowledgements
We would like to thank the Trial Steering Committee (Anthony Gordon, Magdalena Wojtowicz, David Brealey, Ulrike Naumann and Mark Peters) and members of the public who contributed to this study.
Disclaimer
This publication presents independent research funded by the National Institute for Health Research (NIHR). The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DSM and MGM are in part funded by the UCLH/UCL NIHR Biomedical Research Centre. MPWG is in part funded by the Southampton NIHR Biomedical Research Centre.
Funding
This project was funded by a National Institute for Health Research (NIHR) award (PB-PG-0815-20006).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
