Abstract
Background:
Dysphagia places a substantial burden on the critically ill, affecting 12%–84% of this cohort, and is independently associated with worse outcomes. Pharyngeal electrical stimulation (PES-treatment) is a novel dysphagia therapy with an emerging evidence base. This retrospective observational study describes our dysphagia service and reports the use of PES-treatment as a standard of care in recovering critically ill patients at a single-site tertiary UK hospital.
Methods:
Patients admitted to Acute or Cardio-Thoracic adult intensive care units between 1st July 2017 and 30th June 2022 were routinely referred to Speech and Language Therapy (SLT) following tracheostomy, or suspected dysphonia/dysphagia. Clinical assessments and direct laryngeal visualisation using Fibreoptic Evaluation of Swallowing (FEES) were performed. Severe dysphagia was defined as Penetration-Aspiration Score of ⩾6 and patients were offered PES-treatment when staffing allowed.
Results:
Of 289 patients with severe dysphagia, 19 underwent a course of PES-treatment with the remaining patients receiving standard care. PES-treatment patients were significantly less likely to remain nil-by-mouth (11.1% vs 62.5%, Chi2 p < 0.001) or to have an enteral feeding tube in situ at discharge from critical care (27.8% vs 62.5%, p = 0.006) than those receiving standard dysphagia care. Both groups demonstrated an improvement in Penetration-Aspiration Score at repeat FEES: PES-treatment mean difference −2.0 (p = 0.003); non-PES-treatment −1.68 (p < 0.001); (61% PES-treatment improved vs 40% non-PES-treatment, p = 0.09).
Conclusion:
Our observations suggest that PES may be effective in the general critical care population. PES may offer new treatment options for patients and healthcare staff managing severe dysphagia and its significant consequences.
Introduction
Oropharyngeal dysphagia places a substantial burden on critical care patients, with the reported incidence of swallowing difficulty ranging from 12% to 84% in the general Intensive Care Unit (ICU) population 1 and from 11% to 93% in patients with a tracheostomy within an acute care facility. 2 Whilst the cause is often multifactorial, commonly proposed mechanisms include direct laryngeal trauma, central or peripheral neurological impairment, gastroesophageal reflux, dyssynchronous breathing and ICU-acquired myopathy.3,4 The presence of dysphagia complicates care and worsens outcomes. Prolonged inpatient stays, increased re-intubation frequency, aspiration pneumonia, dehydration, malnutrition, increased placement of feeding tubes, chronic psychological problems and increased morbidity and mortality have all been reported.1,5 In patients with tracheostomies, dysphagia is independently associated with failed decannulation. 6 Prolonged dysphagia can significantly impact recovery and rehabilitation in up to one-quarter of patients, 6 months after ICU discharge. 7
Dysphagia must be identified early to mitigate adverse outcomes. One of the most effective diagnostic methods is direct visualisation of the larynx using Fibreoptic Endoscopic Evaluation of Swallowing (FEES).8,9 Following initial clinical assessment, bedside FEES assists in detecting and managing dysphagia, dysphonia, secretion management and laryngeal pathology, helping to guide clinical decisions around rehabilitation and weaning.
Dysphagia treatment should be initiated promptly following diagnosis. 4 However, there is a limited evidence base for acute and critical care dysphagia interventions, with most studies focusing on stroke. 10 Established treatments include compensatory strategies such as dietary modifications (volume, viscosity or texture of food) and postural changes (chin tuck or head tilt). Rehabilitation strategies include therapeutic swallowing exercises and therapeutic oral trials, with many patients requiring supportive nutrition and/or enteral tube feeding until dysphagia symptoms improve. 11 These interventions may not benefit the critically ill or those with tracheostomies.4,10,11 Novel treatments for neurogenic dysphagia include Pharyngeal Electrical Stimulation (PES). PES stimulates the pharyngeal wall using an intraluminal electrode catheter combined with a nasogastric feeding tube. The electrode delivers a low-level electrical current to the pharyngeal muscles which stimulates the peripheral and cortical neural control of swallowing. 12
Usual PES treatment courses consist of three to six consecutive daily periods of stimulation for 10 min. The majority of PES literature to date has focused on neurogenic dysphagia due to stroke. Improvements following a treatment course are thought to be associated with cerebral plasticity driven by stimulation of afferent sensory swallowing pathways.4,13 –16
Three studies have examined PES in tracheostomised stroke patients whose only barrier to decannulation was severe dysphagia.13,17,18 PES treatment significantly improved dysphagia and facilitated protocolised decannulation in 75% of the PES-group compared with 20% non-PES-group (study participants n = 30) 17 ; 49% versus 9% (n = 69) 18 ; and 61% of PES-treated patients (n = 23). 13 Dysphagia of non-neurological origin has also been studied, with one Austrian case series describing 15 out of 19 tracheostomised critically ill patients being successfully decannulated after PES treatment. 19
Whilst dysphagia in the critically ill is often multifactorial, these data suggest that PES has a role in dysphagia treatment of any aetiology and may improve swallow function (by peripheral sensory stimulation, exciting motor pathways to restore neurological swallow integrity) and pharyngo-laryngeal function, in patients who have required tracheostomy as part of their critical care journey. This paper aims to describe our baseline services for dysphagia identification and management in our recovering critically ill patients. Our objectives were to describe the severity and progression of dysphagia in our patients, measured by repeated FEES and clinical assessment, and the impact that we observed following the introduction of PES as a rehabilitation intervention for severe dysphagia.
Methodology
This service evaluation describes the observed impact of introducing PES treatment into routine ICU clinical practice as part of a service development. The report follows the STROBE checklist for observational reports (See STROBE Checklist, Supplemental Figure 1). 20 The NHS Health Research Authority decision tool (www.hra-decisiontools.org.uk) confirmed that this service development did not require ethical approval and the study was conducted under the governance structure of the University of Manchester Academic Critical Care Research Group. The study was carried out at Wythenshawe Hospital, part of Manchester University NHS Foundation Trust (MFT). Our hospital is a tertiary referral centre with 64 critical care beds, including general ICU, cardiothoracic surgery and Extra-Corporeal Membrane Oxygenation (ECMO) services.
Dysphagia management pathway
Our ICU clinical dysphagia management pathway commenced in 2004 with Speech and Language Therapist (SLT) input and introduction of FEES into assessments. By July 2017, the pathway included pre- and post-dysphagia-treatment FEES if clinically indicated and comprehensive data collection using the established SLT database as part of the clinical record. PES was introduced in our ICU in 2017, as a commercially available validated treatment for dysphagia, but PES was not routinely available in UK ICU’s at the time.
An experienced SLT undertook FEES using a XION Medical EndoFLEX video nasopharyngoscope (model EV-NC with DiVAS software; DP Medical Systems, Chessington, UK) whilst the patient was managed in the critical care areas. Images were captured, reported and archived as part of the electronic patient record. Bedside interpretation utilised validated scoring systems to evaluate laryngeal physiology and function and included a narrative report. Scoring systems included Penetration-Aspiration Scale (Pen-Asp) 21 and Secretion Rating Scale (SRS) 22 (see Supplemental Table 1 for details).
Patients were considered to have severe dysphagia when significant sensory impairment was detected during fibreoptic laryngoscopy. This was defined as one or more of: silent aspiration, lack of response to secretions in the larynx, lack of response to aspiration and residue or lack of response to the FEES scope or laryngeal airflow. In the eight-point Penetration-Aspiration clinical severity scale, severe dysphagia translated into scores of 6 to 8 inclusive (the most severe categories), considered unsafe for oral intake. 23 The Penetration-Aspiration scale is often referred to as ‘PAS’ but to clearly distinguish the score from PES, we have referred to it as ‘Pen-Asp’ henceforth. The Pen-Asp rating assigned is for the most severe type of aspiration observed over multiple attempts at oral intake. For example, a patient might score an 8 on thin fluids, but a 5 on thickened fluids, and therefore receive a Pen-Asp score of 8 (the worst score) and yet still be considered safe for trials of oral thickened fluid (on a clinical risk basis). Briefly, the Secretion Severity Rating Scale is a four-point scale ranging from: 0 (normal, no visible secretions in the hypopharynx), 1 (secretions visible bilaterally surrounding the laryngeal vestibule), 2 (secretions that change from scale point 1 to 3 during the observation period), to 3 (most severe, secretions in the laryngeal vestibule). Feeding outcomes were evaluated at the time of the final FEES in the ICU using the International Dysphagia Diet Standardisation Initiative (IDDSI) scale (see Figure 1 for details). A management plan was made by the multidisciplinary team and discussed with the patient. Patients who remained dysphagic after the final ICU FEES were followed up by SLT on the ward or as an out-patient.
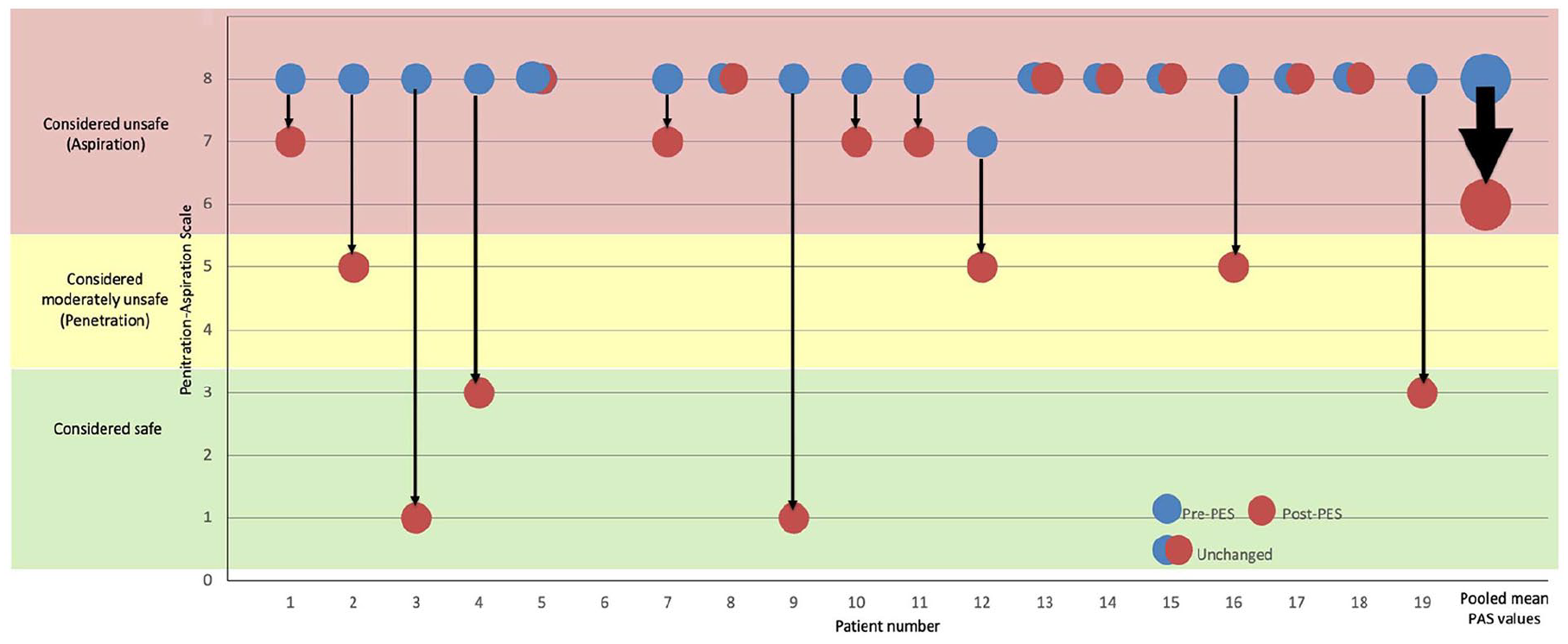
Pre- and post-PES Penetration-Aspiration (pen-Asp) Scores for each of the 19 patients receiving PES therapy. Patient 6 did not undergo a final FEES examination due to delirium.
Standard therapeutic dysphagia options included: secretion management (physical suctioning and/or pharmacological interventions with anticholinergics and/or salivary gland Botox), postural training, swallowing exercises, modified tracheostomy care (sub-glottic suction clearance, cuff deflation trials with positive pressure ventilation and one-way valve, Above Cuff Vocalisation) and oral dietary modification or risk feeding.24,25
Pharyngeal electrical stimulation as a therapeutic option
Phagenyx™ (Phagenesis Ltd, Manchester, UK) is a commercially available PES treatment purchased by the SLT and multidisciplinary tracheostomy team in 2017. Patient selection criteria for patients in a general ICU were developed by our team. As part of our service development, patients with severe dysphagia identified at FEES (Pen-Asp ⩾6) were offered a PES treatment course. Patients had to be managed in the critical care areas of the hospital, and be calm, cooperative and able to obey commands. The SLT team trained in the use of PES had to be available to complete the proposed course of treatment, which included baseline (pre-PES-treatment) and follow-up (post-PES-treatment) FEES, and the provision of daily PES therapy. Exclusions were based on the product literature and included: patient refusal, patients with planned Magnetic Resonance Imaging scans during proposed treatment window, implantable electrical devices such as pacemakers, defibrillators or ventricular assist devices, clinical instability or delirium (treatment requires a degree of cooperation). SLT staff were trained and supported by Phagenesis to deliver PES, and therapy was delivered using the following procedure:
Swap the standard nasogastric feeding tube for a stimulating catheter/feeding tube. Nutrition Specialist Nurse assisted with catheter placement following training by Phagenesis.
Check the position of the stimulating catheter prior to each treatment (distance markers visible externally at the nose and visualisation of coloured catheter markers in the posterior pharynx by looking in the mouth).
PES Threshold: the current is slowly increased until the patient reports a tingling sensation, in the throat.
PES Tolerance: the current is increased until the patient finds the sensation very uncomfortable.
PES Treatment: an algorithm determines the optimal treatment current based on the threshold and tolerance values (a value around 2/3 of the difference between the two values, above the threshold).
Typically, three measurements are taken for each determination of threshold and tolerance, and average values are used to calculate the treatment current. Treatment currents are applied for up to 10 min and treatment occurs on 3 consecutive days. Post-PES-treatment FEES was undertaken within 2–4 days of completion of the PES treatment course. Additional PES treatments (more than the recommended three treatments) were offered if the post-PES-treatment FEES only detected a minimal functional improvement on the post-PES-treatment FEES. These further rounds of treatment were not protocolised but are described in the PES stroke literature. Patients may have had multiple FEES dysphagia assessments to diagnose and guide treatment. We report FEES data from the immediate pre-PES-treatment FEES and from the FEES that followed the last session of PES therapy (the post-PES-treatment FEES).
Patient identification and statistical considerations
The SLT ICU database was searched for patients who had an initial Pen-Asp score of ⩾6 and more than one FEES (baseline/diagnostic and follow-up/post-treatment). Within this severe dysphagia cohort, patients were grouped into those who had undergone PES therapy and those who did not (non-PES). Data were extracted into Microsoft Excel spreadsheets and statistical analysis was performed using SPSS 27 (IBM Corp). Changes in scores were calculated between the paired FEES assessments for each patient. Data are presented as mean (standard deviation (SD), range) or median (interquartile range (IQR), range) depending on distribution. Paired t tests were used to assess differences between paired FEES data. Contingency Chi-Squared tests were used to assess categorical group differences and Linear Regression with Analysis of Covariance (ANCOVA) tests were used to evaluate differences between continuous variables. Statistical significance was accepted at p < 0.05 (two-sided).
Results
Between 1st July 2017 and 30th June 2022, 484 patients underwent 764 FEES examinations in our ICUs. Tables 1–3 summarise the FEES procedures and findings. Briefly: 289 patients were identified with severe dysphagia (Pen-Asp ⩾6); 160 patients had more than one FEES; 138 patients had both severe dysphagia and more than one FEES to evaluate progress. Nineteen patients (19/289, 6.6%) were offered a course of PES therapy, with 18/19 patients undergoing a second post-PES-treatment FEES (one patient deteriorated and died before the follow-up FEES could be undertaken). Seventeen of the PES-treatment patients had a tracheostomy at the time of therapy (see Supplemental Tables 2 and 3 and Supplemental Figure 2). The COVID-19 pandemic and its aftermath had an impact on SLT staffing and our ability to deliver PES-treatment.
Demographical data for all patients with severe dysphagia.
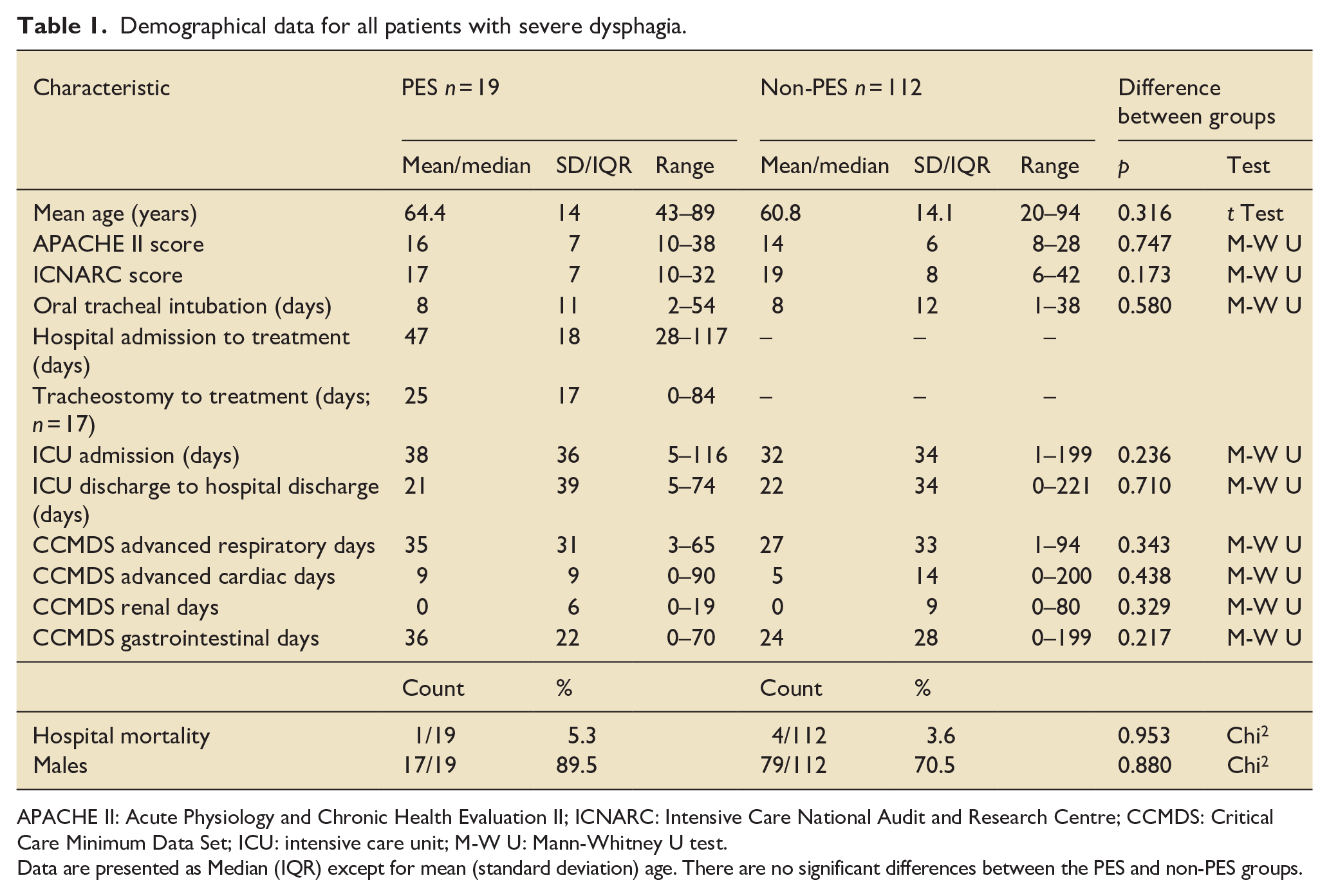
APACHE II: Acute Physiology and Chronic Health Evaluation II; ICNARC: Intensive Care National Audit and Research Centre; CCMDS: Critical Care Minimum Data Set; ICU: intensive care unit; M-W U: Mann-Whitney U test.
Data are presented as Median (IQR) except for mean (standard deviation) age. There are no significant differences between the PES and non-PES groups.
Swallowing and functional laryngeal scores derived from FEES examination at baseline and after final FEES.
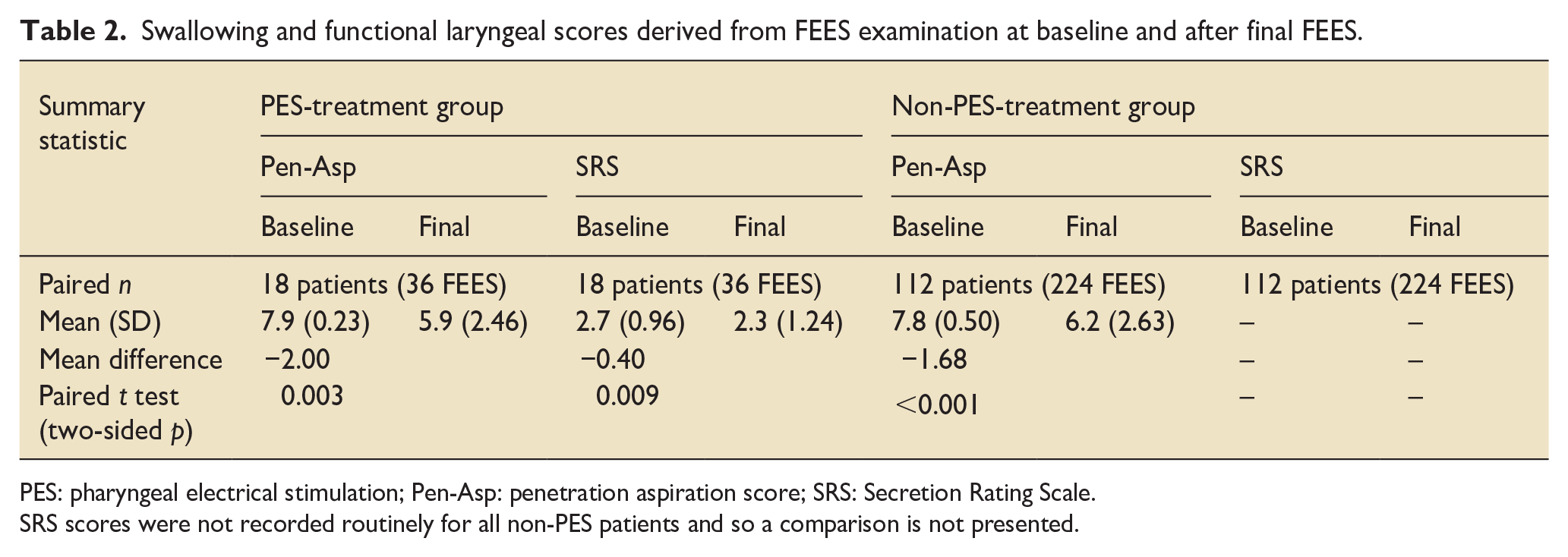
PES: pharyngeal electrical stimulation; Pen-Asp: penetration aspiration score; SRS: Secretion Rating Scale.
SRS scores were not recorded routinely for all non-PES patients and so a comparison is not presented.
Summary of patients, FEES and resulting Pen-Asp scores, with the PES-therapy sub-group highlighted.
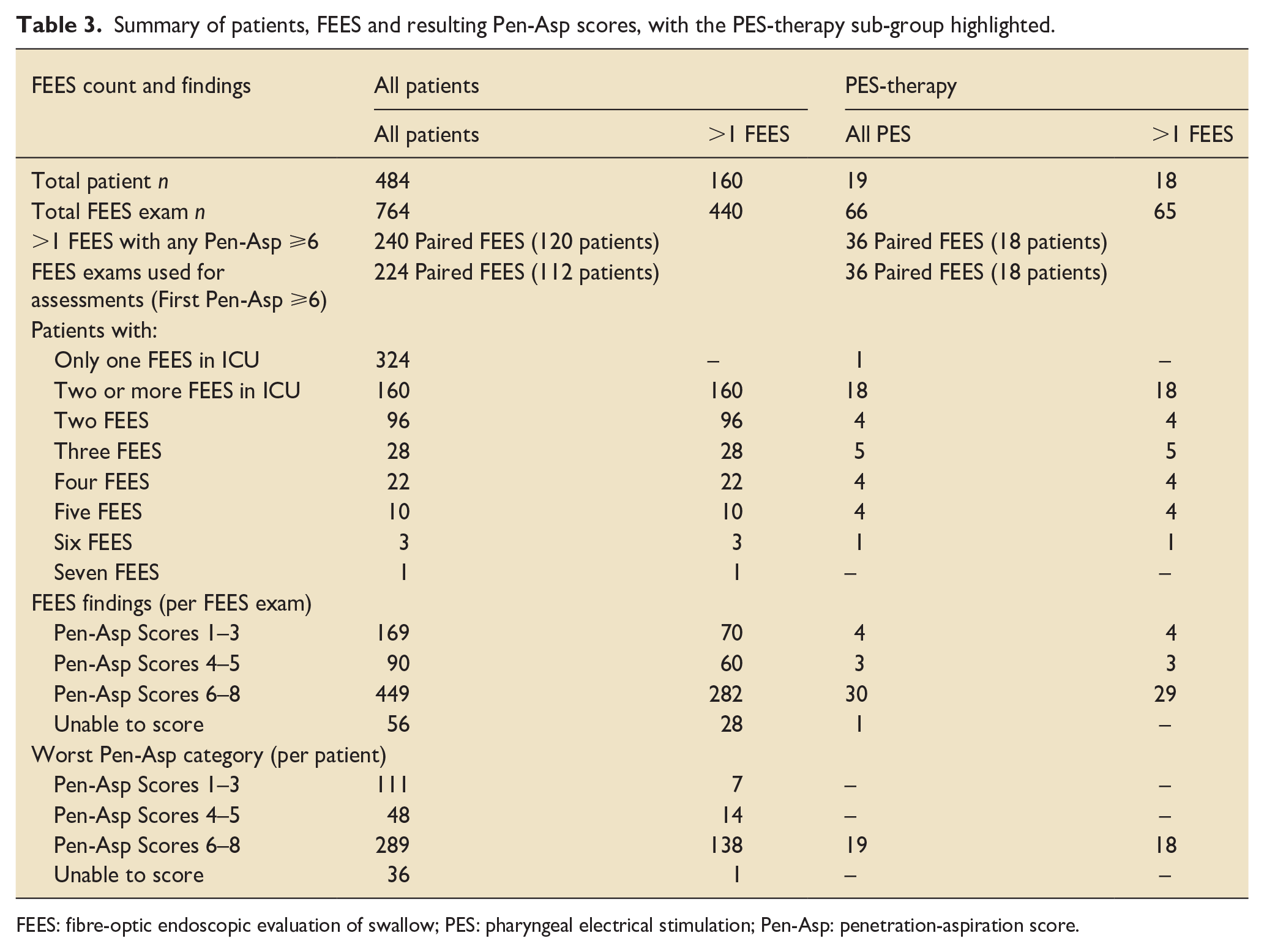
FEES: fibre-optic endoscopic evaluation of swallow; PES: pharyngeal electrical stimulation; Pen-Asp: penetration-aspiration score.
Demographic data for the PES-treatment and non-PES-treatment cohorts are shown in Table 1, with Supplemental Figure 3 summarising the range of patients’ primary medical specialties, reflective of our hospital population. There were no significant differences between the severity of illness and patient journey metrics between the groups (Table 1). For the PES-treatment group, the median APACHE II score at ICU admission was 16 (7; 10–38), patients were predominantly male (18/19) and had a mean age of 64.4 (14; 43–89) years. The median duration of initial tracheal intubation following ICU admission was 8 (11; 2–54) days. Patients were in hospital for a median 47 (18; 28–117) days before PES treatment, with a median of 25 (17; 0–84) days from tracheostomy to therapy for the 17 tracheostomised patients.
Dysphagia treatments
In the PES-treatment group, patients underwent a median of four PES treatments (range 2–6). Overall, there was no significant difference in PES threshold, tolerance and treatment currents applied between first and last PES treatments (p = 0.420, p = 0.698 and p = 0.409 respectively; see Supplemental Table 2 and Supplemental Figure 2). Three patients experienced discomfort and requested treatment dose to be reduced but completed the planned treatment course. Three patients did not complete the proposed complete course of PES treatment: one patient had laryngopharyngeal oedema identified as a potential complication (completing 3 treatments); one patient had increased stridor with treatment (completing 4 treatments); one patient did not wish to undergo their final planned treatment (completing 2 treatments). There were no lasting consequences to any of these potential complications identified at the final FEES examination and they may have been explained by other medical factors. 9
Pre- and post-PES-treatment analysis
One patient could not undergo follow-up FEES due to delirium, and later died due to complications of their critical illness. Of the 18 remaining patients with paired FEES assessments, 11 (61.1%) patients’ Pen-Asp Scores improved following PES treatment and seven were unchanged (mean Pen-Asp scores fell from −7.9 to −5.9, mean difference −2.00, p = 0.003, Table 2). Clinically, this is significant, and translated into seven of the 18 patients who had silent aspiration demonstrated on initial FEES improving to conscious sensation of laryngeal secretions at the post-treatment FEES (Figure 1). There was also a significant improvement in Secretion Rating Scale (mean difference −0.40, p = 0.009). Lower SRS scores indicate improved secretion management, suggesting improvements in laryngopharyngeal sensory response and swallowing.
Non-PES-group description of cases
For patients undergoing standard dysphagia care (non-PES-treatment group), 120 had more than one FEES assessment; 112 with a baseline Pen-Asp scores of ⩾6. Forty-five (45/112, 40.2%) patients’ dysphagia improved based on their Pen-Asp scores over time; 63 (63/112, 56.3%) patients remained unchanged; and four (4/112, 3.6%) patients got worse. There was an overall mean difference in Pen-Asp score of −1.68 (p < 0.001, Table 2) from baseline to final FEES. Although a greater proportion of patients had improved Pen-Asp scores at follow-up in the PES-treatment group, this was not significant (11/18, 61.1% vs 45/112, 40.2%, p = 0.090). However, the mean (SD) time between first and final FEES was significantly shorter in the PES-treatment group (13.3 (7.5)) days when compared with the non-PES-treatment group (21.8 (21.1)) days (p = 0.008). Figure 2 demonstrates that the Pen-Asp scores in the PES-treatment group improved to a greater extent and more quickly than the non-PES-treatment group. The adjusted mean Pen-Asp scores were 7.2 (95%CI 6.6–7.9) in the PES-treatment group and 7.1 (6.8–7.3) in the non-treatment group with a non-significant difference between the slopes of the regression lines (ANCOVA p = 0.693).
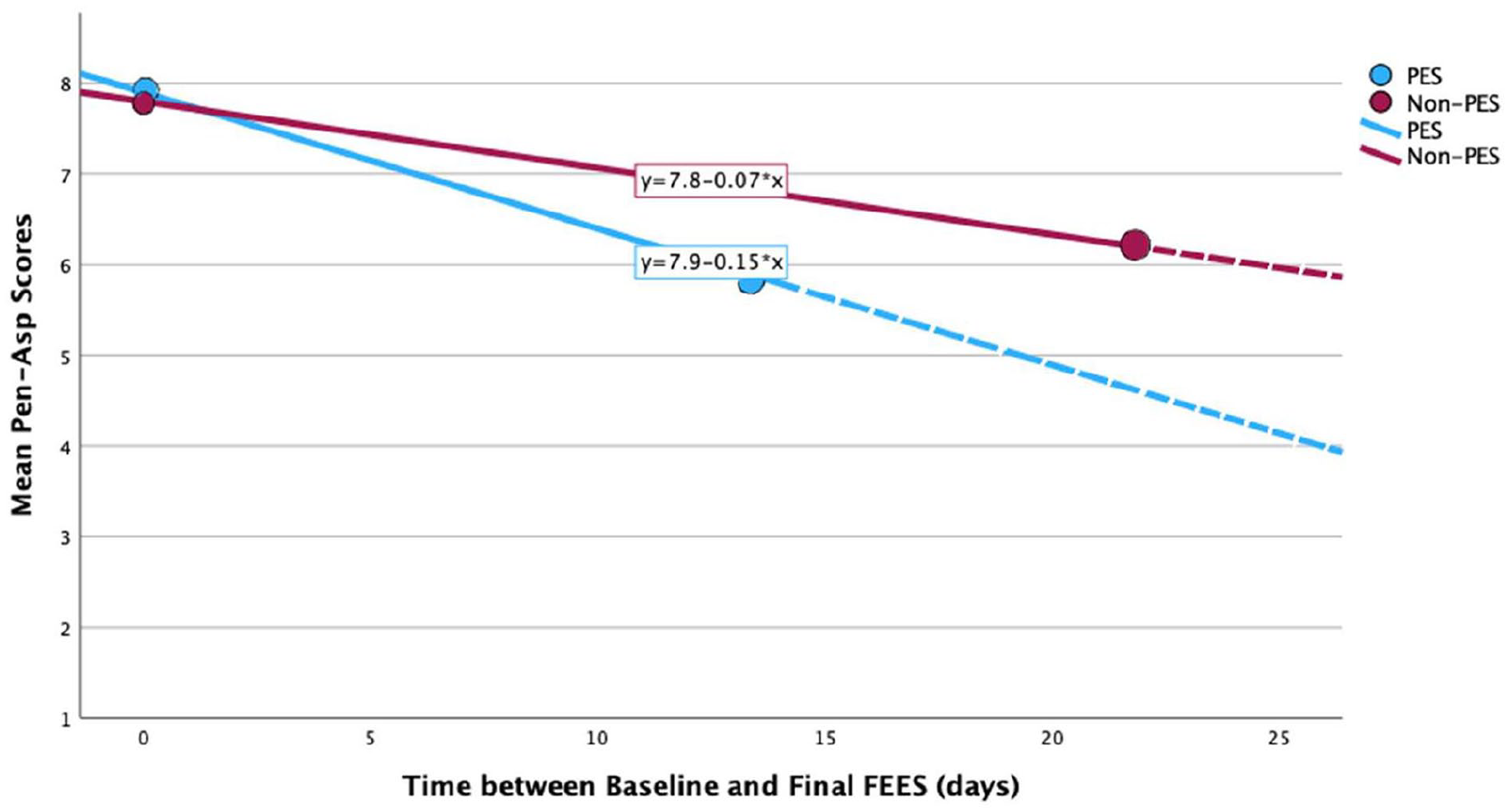
Simplified scatter plot and regression lines of mean Penetration-Aspiration (Pen-Asp) Scores versus time between first and final FEES grouped into PES-therapy and non-PES therapy cohorts.
Feeding outcomes
With regards to feeding outcomes, patients improved a median of two International Dysphagia Diet Standardisation Initiative (IDDSI) food levels 26 and a median of three IDDSI fluid levels following PES treatment (both p = 0.001, Figure 3). Five of the 18 patients (27.8%) who completed PES therapy retained a naso-gastric feeding tube to deliver at least some enteral nutrition and medications after the final FEES, although only two patients (11.1%) remained entirely ‘nil-by-mouth’. Sixteen of the 17 patients with tracheostomies were decannulated (the patient who died had a tracheostomy) and 18 of the 19 patients went home.
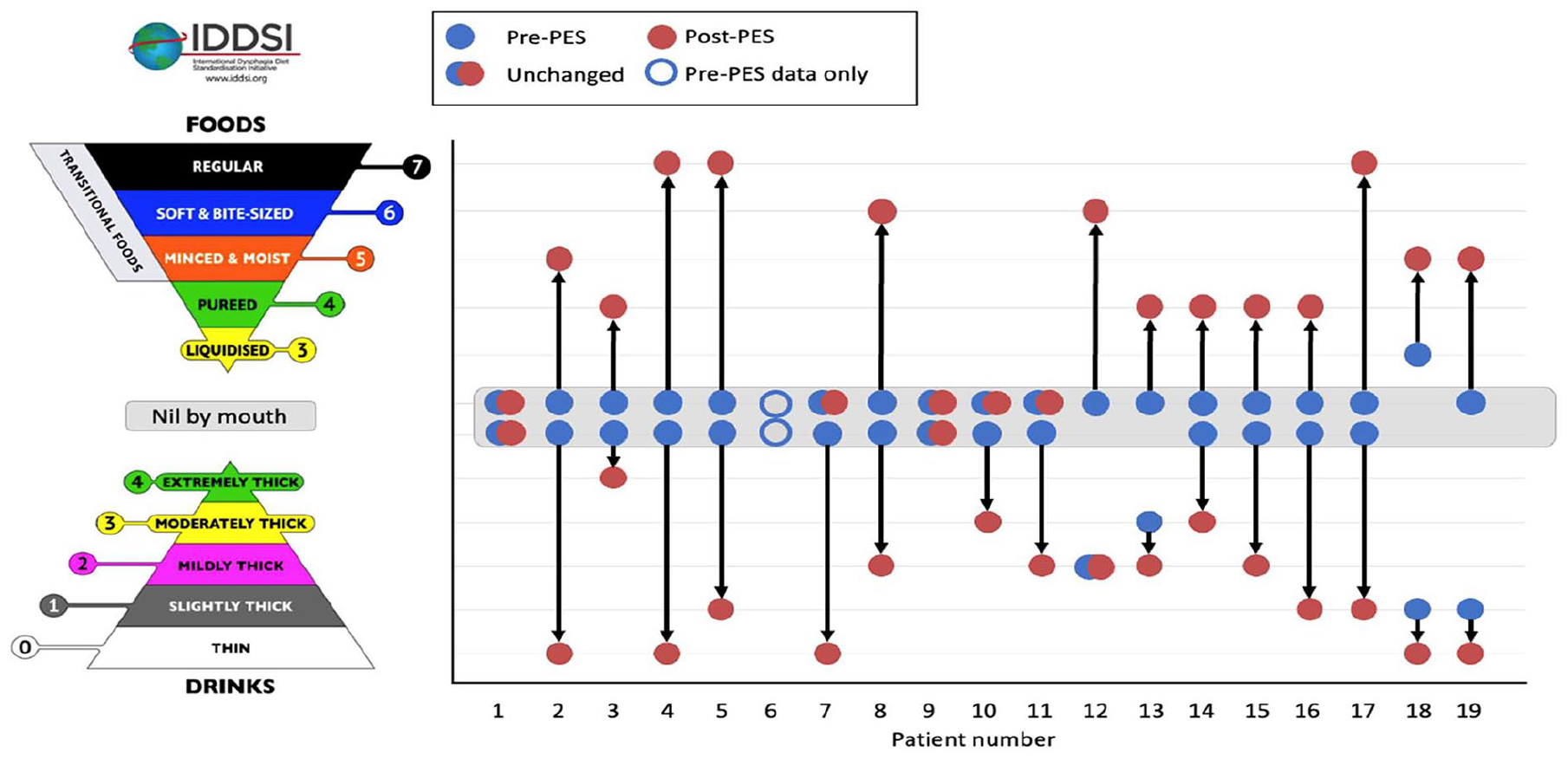
Clinical changes in food and fluid intake pre- and post-PES treatment, categorised into International Dysphagia Diet Standardisation Initiative (IDDSI) levels. An additional category of ‘nil by mouth’ has been added and food and fluid levels are reported separately for each patient. Overall, patients improved by a median of two IDDSI food levels and a median of three IDDSI fluid levels following PES-treatment (p < 0.001).
In contrast, 70 of the 112 patients (62.5%) who did not receive PES therapy remained nil-by-mouth following their final FEES on the basis of their assessment, prior to critical care discharge due to persistent dysphagia. Fifty-three patients retained a naso-gastric feeding tube and a further 17 patients were referred for percutaneous enteral gastrostomy (PEG) feeding. These patients were followed up on the ward and as outpatients. Significantly fewer patients who received PES-treatment retained an enteral feeding tube or remained nil-by-mouth following their final FEES assessment in ICU (p = 0.006 and p < 0.001 respectively).
Discussion
This service development evaluation suggests that PES treatment can significantly improve laryngeal function and feeding outcomes in a carefully selected patient population recovering from critical illness with confirmed dysphagia following FEES. Whilst our small case series cannot demonstrate how PES directly impacts outcome, improving pharyngo-laryngeal function and swallow through sensory stimulation and consequent motor recovery due to neuroplastic reorganisation would be expected to shorten time to recovery, promote an earlier return to patient-focused outcomes such as eating, drinking and vocalisation, and promote successful and safe tracheostomy decannulation. 27
PES was acceptable to a wide range of general ICU patients. Other studies have also demonstrated that PES is well tolerated in critical illness, but mainly populations with primary neurological problems. 28 PES is a relatively resource-intensive treatment, requiring specialists skilled in FEES and trained (and available) staff to deliver therapy. Our results highlight the importance of rigorous multidisciplinary dysphagia assessment in high-risk populations. Studies report that clinical examination alone has a low sensitivity for identifying laryngeal pathology (missed in up to 62% of patients when assessed clinically by SLT and in up to 97% when evaluated by medical staff). 9 The initial FEES in our patient group identified high rates of silent aspiration, which adds to the growing evidence base for the routine use of FEES in critical care patients. 29
Patient-focussed functional outcomes related to oral food and fluid intake were significantly improved following PES treatment in our patient group. Impaired laryngeal sensation is associated with the development of dysphagia following airway instrumentation and critical illness. 30 It is therefore not surprising that PES treatment, designed to promote laryngeal sensorium, resulted in better feeding outcomes. Facilitating safe oral nutrition in tracheostomised patients can positively impact a patient’s psychological well-being.31,32 Conversely, chronic dysphagia is a significant issue associated with prolonged hospital stays, higher morbidity and mortality and impaired overall rehabilitation.11,33 It is plausible to hypothesise that PES may improve these hospital outcomes, and our work may provide the stimulus for further research in this area.
This paper has several limitations, not just limited to the small sample size, non-protocolised selection criteria for PES therapy, and the retrospective observational nature of the evaluation. Whilst the ratings were performed by the same experienced SLTs, the results were not protocolised or reviewed independently, blindly or anonymously, which although consistent, may introduce bias. It is important to note that whilst there were no significant demographic differences between the PES-treatment and non-PES-treatment groups (Table 1) the groups were unmatched and any comparisons between the groups must be interpreted with caution. PES-treatment was offered to eligible patients based primarily on the availability of the senior SLT team for the duration of therapy. The presence of hyperactive delirium also limited access to PES-treatment for some eligible patients as it requires a degree of patient cooperation. Our case series likely does not reflect a ‘typical’ general ICU population, with our complex patients having notably long ICU stays. However, these may be precisely the patients who may benefit from more invasive treatments, having exhausted standard dysphagia therapies. In some, significant dysphagia may be the limiting factor in tracheostomy decannulation. 27 Even though PES treatment led to only a mean two-point improvement on the Pen-Asp scale, it is important to appreciate that for many patients, these score thresholds may represent the difference between silent aspiration and the patient being consciously aware and trying to expel aspirated laryngopharyngeal material. Small improvements may allow better secretion management, promote tracheostomy tube cuff deflation, subsequent progression with weaning, and even cautious (modified) oral intake. The Pen-Asp scale is just one measure of laryngeal function and risk, and functional outcomes, such as the IDDSI levels, are more important to the patient. Based on the improvements in laryngeal function identified by ourselves and others, PES may provide a more significant benefit if utilised earlier in ICU stay, and future prospective studies should explore optimum timings for therapy in addition to the identification of specific patient cohorts who could particularly benefit from early intervention. Delirium often delayed PES therapy in our cohort, although as the evidence base emerges, early therapy may be more easily justified.
Conclusion
PES is an established treatment for neurological conditions that cause dysphagia, with an emerging evidence base for the multifactorial pathologies that cause dysphagia in the critically ill. FEES is an ideal tool for dysphagia identification, characterisation of dysphagia and appropriate patient selection for PES, and evaluation of treatment efficacy. Our observations suggest that PES is effective in the general critical care population, particularly for patients with prolonged ICU stays and tracheostomies. By describing and evaluating our SLT-driven dysphagia service and the impact of introducing PES therapy for patients with severe dysphagia, we have demonstrated that effective service improvement is possible, with benefits to patients.
Supplemental Material
sj-docx-1-inc-10.1177_17511437241270244 – Supplemental material for Research report: Management of dysphagia using pharyngeal electrical stimulation in the general intensive care population – A service development
Supplemental material, sj-docx-1-inc-10.1177_17511437241270244 for Research report: Management of dysphagia using pharyngeal electrical stimulation in the general intensive care population – A service development by Thomas Williams, Elizabeth Walkden, Karishma Patel, Naomi E Cochrane, Brendan A McGrath and Sarah Wallace in Journal of the Intensive Care Society
Footnotes
Authors’ note
Presented in part at the Anaesthesia Research Society Meeting, Imperial College, London, May 2024. All material presented in this manuscript is our own original work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical approval
Data availability
Anonymised original data for this study is available from the authors on request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
