Abstract
Background:
Peripherally administered vasopressor infusions are used to support critically ill patients. The Intensive Care Society recently published guidelines supporting their use. Preliminary evidence suggests variability in peripheral vasopressor infusion use. We aimed to characterise current practice in the use of peripheral vasopressor infusions in the UK National Health Service and to capture the views of healthcare professionals caring for critically ill adult patients.
Method:
We conducted an online survey of healthcare professionals from December 2022 to March 2023. Our survey used Google Forms and was shared via email, X (formerly twitter), and mobile phone messaging within the UK. Health Research Authority approval was sought, and ethical approval was not required.
Results:
We received 227 responses and our survey showed variation in the use of peripherally administered vasopressor infusions in critically ill patients across UK National Health Service hospitals. About 87.7% of healthcare professionals initiated peripheral vasopressor infusions in their clinical role. Peripheral vasopressor use was limited to low dose, short durations of infusions, however, there was variability. Metaraminol was the most used peripheral vasopressor (90.3%). Only 22.5% of healthcare professionals used peripherally administered noradrenaline in practice. Our respondents agreed that equipoise exists.
Conclusion:
Our survey found variability in the use of peripheral vasopressors and in care delivered to critically ill patients in the UK. Our survey shows there are ongoing concerns regarding safety, dosing ranges and durations of use. Future research is required to explore the optimal role that peripheral vasopressor infusions can play in the care of critically ill patients.
Keywords
Introduction
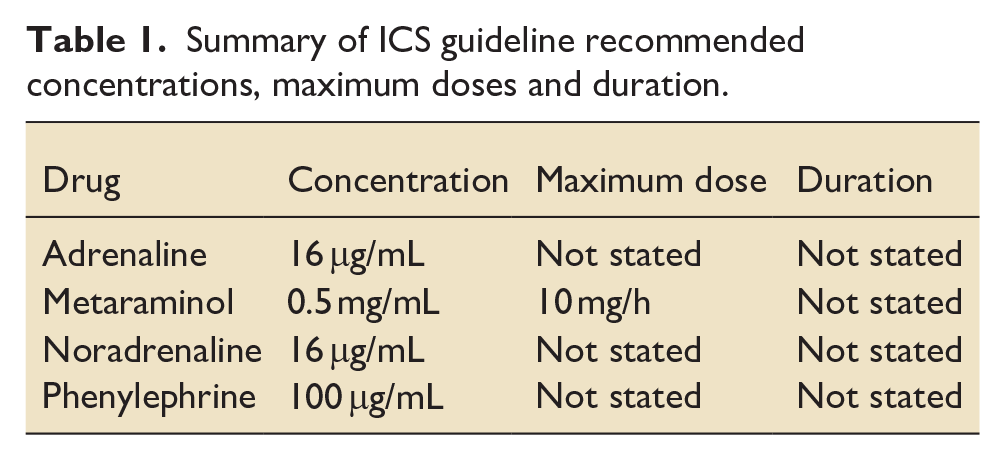
Peripherally administered vasopressor infusions (PaVi) such as metaraminol are used to stabilise and support adult critically ill patients in the United Kingdom.1,2 A recent systematic review concluded the risk of extravasation from PaVi is uncommon and unlikely to lead to major complications, challenging traditional beliefs and concerns regarding their use. 3 The current international Surviving Sepsis Campaign consensus recommendations 4 include a low-quality evidence recommendation to start PaVi in adults with septic shock rather than delaying initiation until central venous access is secured. The UK National Institute for Health and Care Excellence sepsis guideline offers no guidance on PaVi use. 5 Recently, the Intensive Care Society (ICS) has published guidance on the use of PaVi, 6 recognising there is limited data to support this practice. The guidance covers the use of adrenaline, metaraminol, noradrenaline, phenylephrine (Table 1) and suggests that the maximal safe dose and duration of peripheral infusions should be determined locally. Currently there are no published data that has sought to evaluate the use of these PaVi in the National Health Service (NHS).
Summary of ICS guideline recommended concentrations, maximum doses and duration.
Objectives
We aimed to conduct a survey of United Kingdom NHS healthcare professional (HCP) practices and attitudes towards the use of PaVi. We also aimed to capture HCP experiences when using peripheral and centrally administered vasopressor infusions (CaVi) for critically ill patients and gather information to ascertain whether there is community equipoise for further research.
Methods
The survey was aimed at doctors, nurses and critical care practitioners working within the NHS. An electronic survey was created which could be shared via email, twitter and mobile phone messaging. The survey was designed to be completed within 5–7 min. The survey proposal and survey questions were submitted to the Health Research Authority (HRA) and deemed not to require research regulatory approvals and ethical approval was not deemed necessary. A copy of the survey can be found in Appendix 1.
The survey was distributed by the UK Critical Care Research Group (UKCCRG) and Peri-Operative Medicines Clinical Trails Network (POMCTN) to its membership via email in December 2022. The survey was disseminated across all four constituent nations in the UK. We allowed approximately 3 months to receive responses after which the survey link was closed for analysis of our results.
Survey design
Survey questions were developed in line with our objectives and prepared in Google Forms (Google Forms, Mountain View, California, USA). The survey was piloted locally at our trust amongst doctors and nurses and refined using an iterative process. Participants were required to self-declare that they were a doctor/advanced critical care practitioner/nurse working in adult critical care currently or had worked in adult critical care within the last 12 months and agree to take part. This was mandatory and only those that were eligible and gave agreement were able to proceed with the survey.
Responses were collected from 16th December 2022 to 10th March 2023. Data from Google Forms were downloaded from Excel (Microsoft Offices, Redmond, USA) for analysis.
Data collection
We collected the following data:
How PaVi are used in the UK: Which vasopressors are used Settings and contexts of use Reasons why HCP choose to, or choose not to, use PaVi HCP who initiate PaVi Whether PaVi such as metaraminol and phenylephrine are given using a Central Venous Catheter (CVC) Infusion rates and durations that HCP are comfortable to use in practice. These were standardised in line with the ICS guidance for peripheral vasopressor use (metaraminol (mg/h); phenylephrine (mg/h); noradrenaline (mcg/kg/min). When HCP decide to change from using a PaVi to a CaVi
Positive and negative experiences associated with the use of PaVi and CaVi
HCP equipoise for a future trial comparing PaVi to CaVi
Results
We received 227 responses to the survey from a variety of HCP (Table 2). The majority of responses were from England (86.8%). Consultants formed the majority of the respondents followed by junior doctors.
Respondents by professional group.
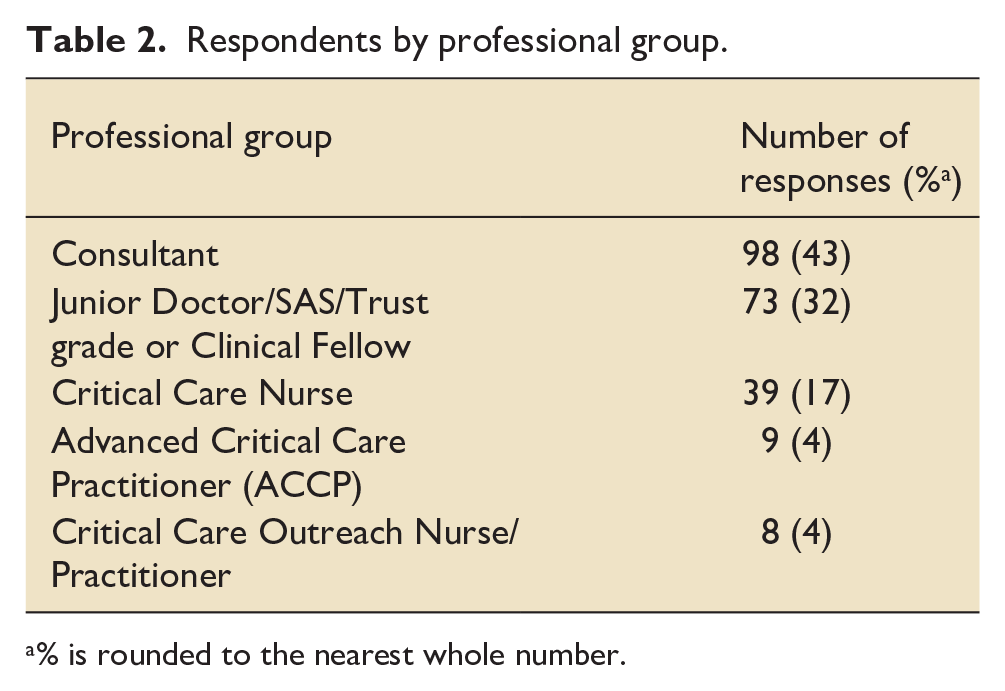
% is rounded to the nearest whole number.
HCP practices and attitudes
HCP surveyed used a variety of PaVi when supporting the critically ill patient; with metaraminol cited as the most commonly used agent (90.3%) followed by phenylephrine (41.4%), adrenaline (39.2%) and noradrenaline (22.5%). PaVi were utilised s in a variety of settings (Figure 1) particularly in critical care; the emergency department (ED) and theatres. Over 64% of respondents stated they would use PaVi in any area that required the stabilisation of a critically ill patient. The majority (87.7%) of respondents confirmed they initiated PaVi as part of their clinical role when treating critically ill patients.
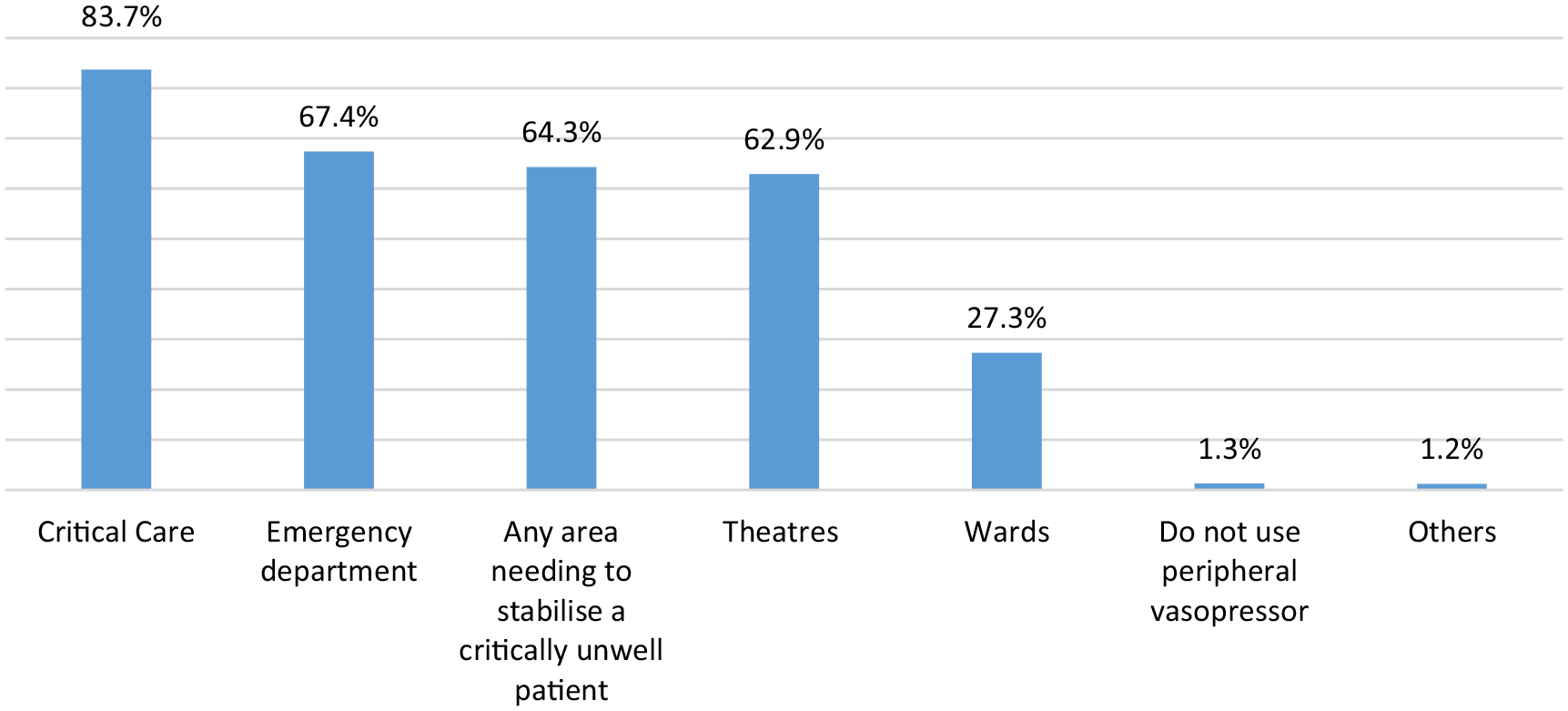
Clinical settings of PaVi use.
Respondents gave a variety of reasons for using PaVi to support critically ill patients (Figure 2). Anticipated short duration of use; low dose use; use when a CVC was not available or until a CVC could be inserted were the commonest reasons for the use of PaVi.
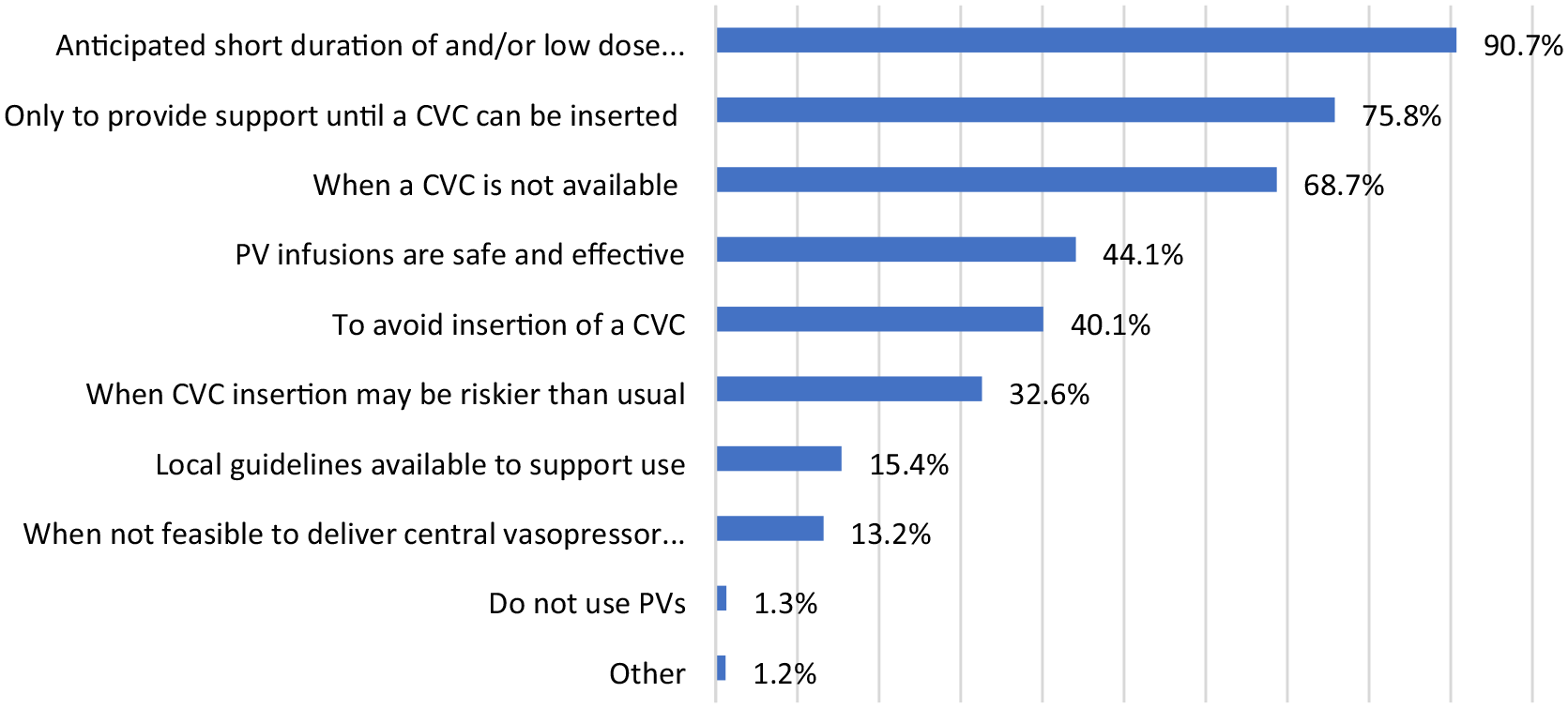
Reason for selecting PaVi.
The post-operative surgical population and the acutely hypotensive patient groups were the most common contexts in which PaVi were used (Figure 3).
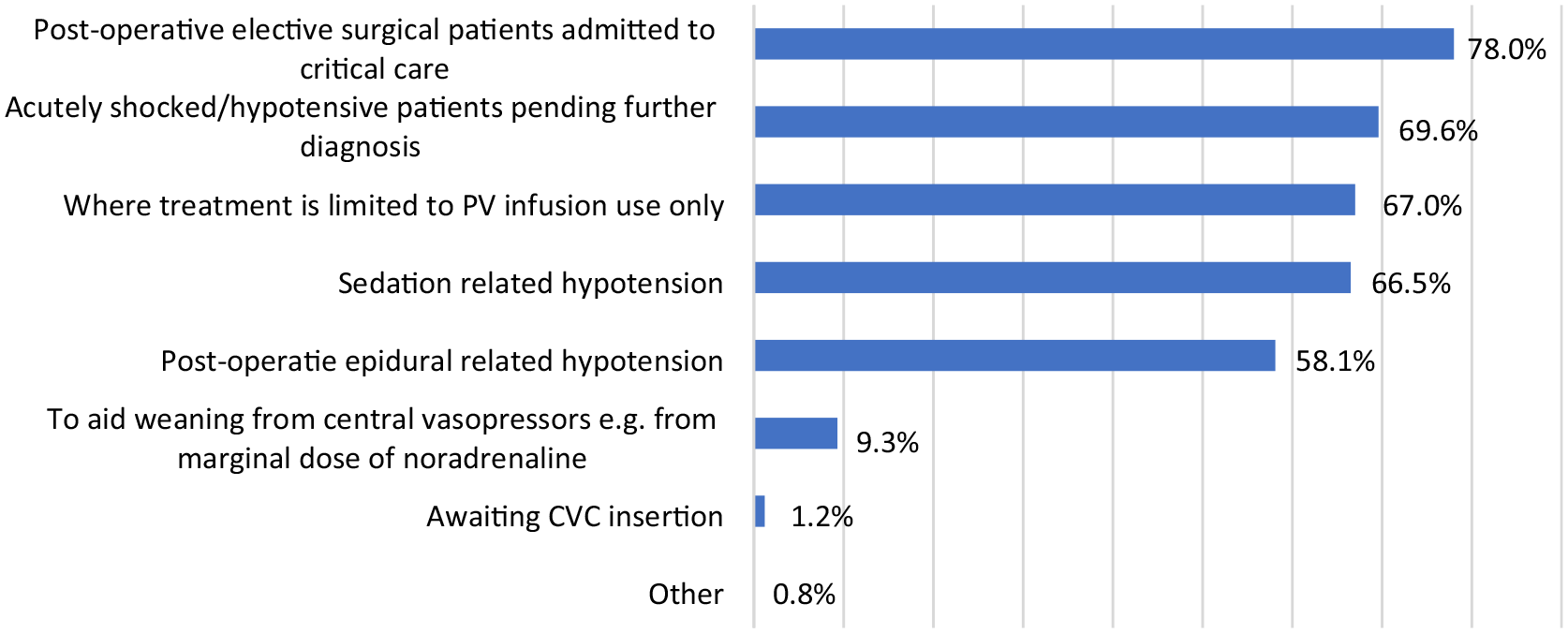
Context for use of PaVi.
We also explored reasons why potentially some HCP may not use PaVi in their practice. A preference for CaVi (11.9%); local departmental practice (6.6%) and safety and effectiveness concerns were amongst other reasons cited (Figure 4).
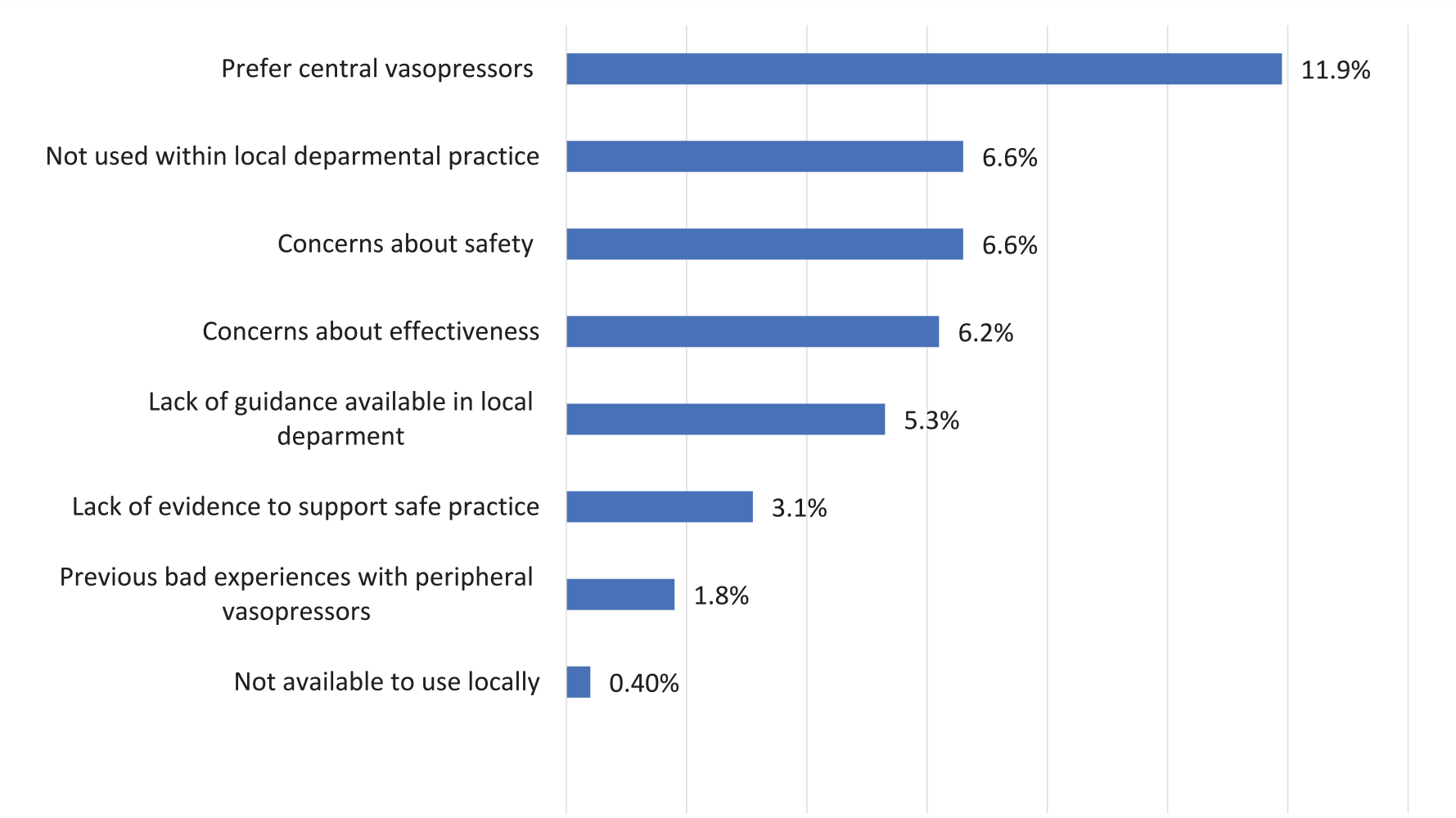
Reasons for not using PaVi to support critically ill patients.
We asked respondents when using PaVi what doses they were comfortable with (Figure 5). 21/227 (9.3%); 111/227 (48.9%) and 143/227 (63%) of respondents said this was N/A to them for metaraminol, phenylephrine and noradrenaline, respectively. This may reflect where HCP were not comfortable with or did not use the vasopressor peripherally.
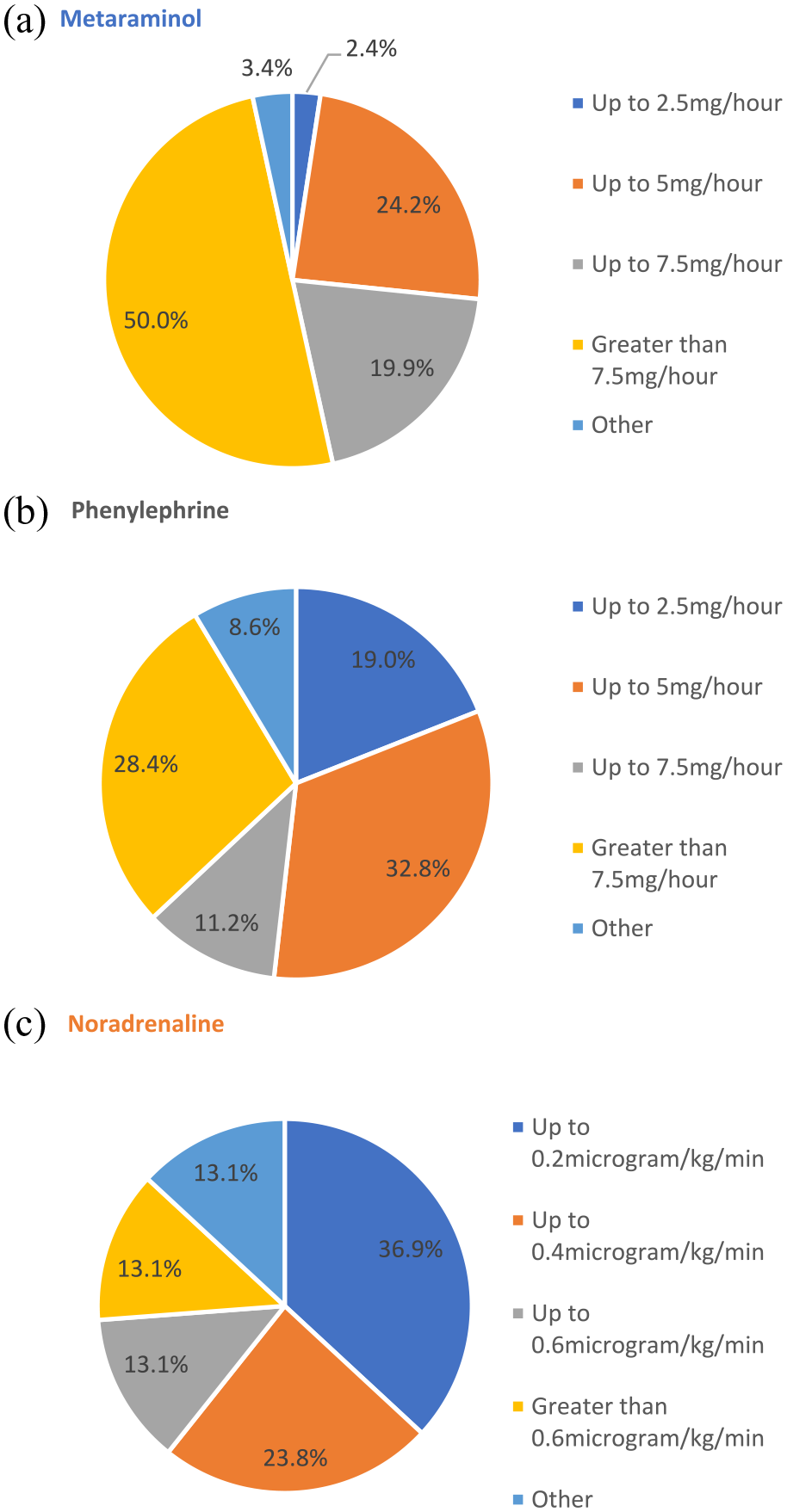
PaVi doses HCP are comfortable with: (a) metaraminol, (b) phenylephrine, and (c) noradrenaline.
HCP were comfortable to use high doses, 7.5 mg/h or greater, of metaraminol provided the patient was achieving a target mean arterial pressure (MAP) (Figure 5(a)). About 63% were unfamiliar with using noradrenaline peripherally. Those familiar with peripheral noradrenaline infusions were comfortable with doses up to 0.2 mcg/kg/min. The majority of respondents were unfamiliar with phenylephrine use and those that did use this agent, would be comfortable to administer up to 5 mg/h. The survey also showed 43.6% of HCP administer metaraminol centrally via a central line, followed by 21.1% who administer phenylephrine centrally.
We asked respondents when using PaVi when they would consider changing to a CaVi. The majority who used metaraminol said they would consider this if the metaraminol requirement was on an increasing trajectory, or the infusion rates was greater than 5 mg/h. For those who used peripheral noradrenaline and phenylephrine some respondents would think of switching to a CaVi if vasopressor requirements were on an increasing trajectory. We also explored how long HCP were comfortable to use PaVi. There was significant variation in practice (Figure 6). Shorter durations of PaVi were preferred, although others remained comfortable to use PaVi as long as the patient was achieving their target MAP.
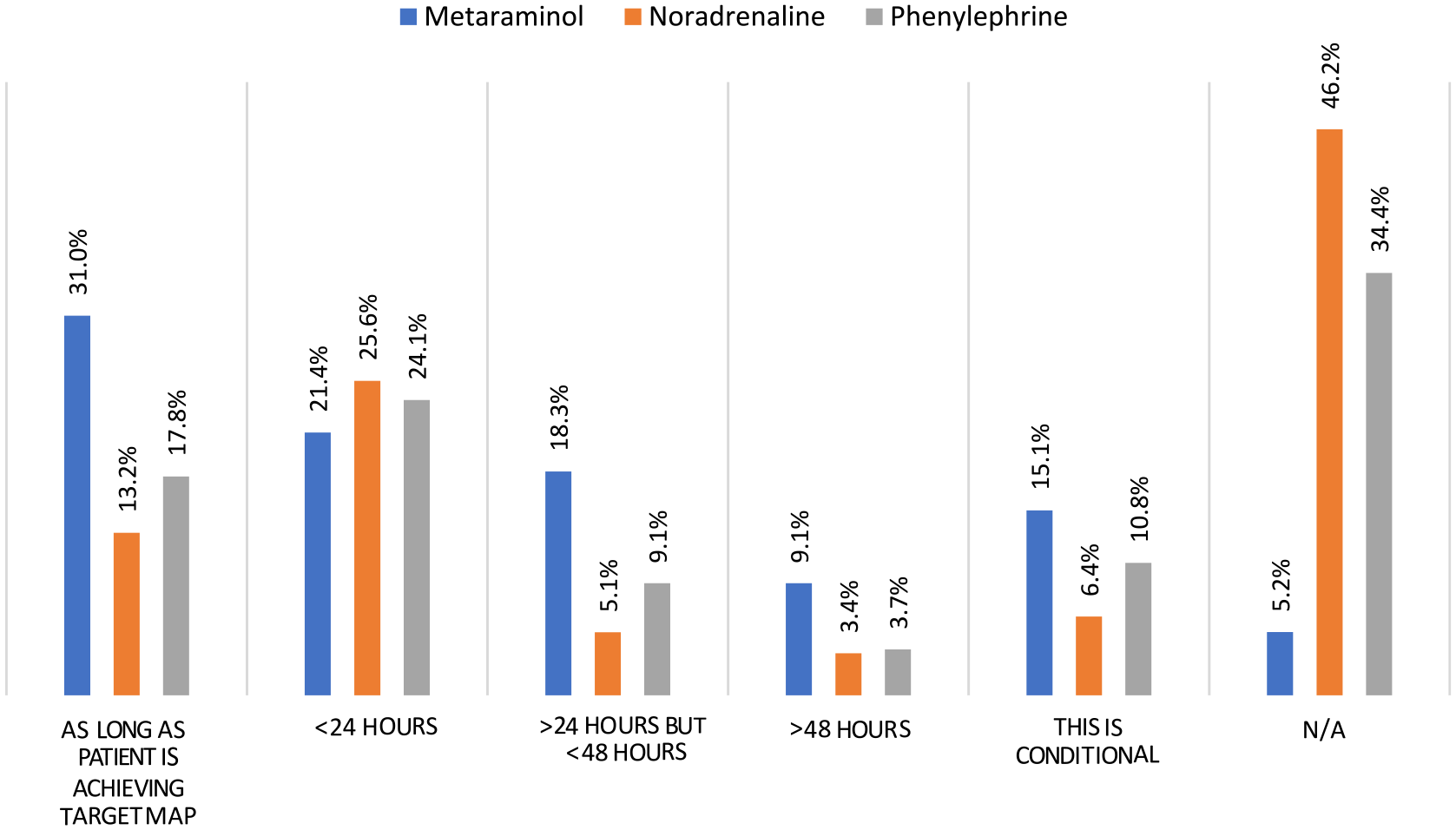
How long are you comfortable to continue a PaVi?
HCP experiences of using PaVi and CaVi
The top 3 positive experiences cited when using PaVi were (1) PaVi can be established quickly (88.5%); (2) it is easier to site a peripheral cannula than a central line (67.8%) and (3) peripheral canulae can be sited or re-sited by the wider MDT (55.1%). The top 3 negative experience were (1) peripheral cannula can become displaced easily risking hypotension or extravasation (52.8%); (2) a peripheral cannula needs to be re-sited more often compared to CVC (47.1%) and (3) inserting peripheral cannula can be challenging in patients that are shocked (38.8%). A similar set of questions were asked related to CaVi. The top 3 positive experiences cited were (1) ease of adding in other vasopressors if a central line is already present (87.7%); (2) preference for one CVC over multiple cannulas in the critically ill patient (75.3%) and (3) central lines less likely to become displaced (61.2%). The top 3 negative experiences cited were (1) central lines take longer to insert and can delay vasopressor initiation (59.9%); (2) there tend to be fewer trained individuals that can insert CVCs (43.6%) and (3) inserting or re-siting a central line can be challenging (35.2%). We asked if HCP would be willing to enrol and randomise patients to a study comparing PaVi to CaVi. About 60% of respondents unanimously said yes and 24% said they would consider it.
Discussion
The principle finding of our survey was there is great variability in UK practice with use of PaVi, with a tendency of clinical staff to favour shorter durations and low dose infusion. PaVi are used to acutely stabilise critically ill patients and as a bridge to CVC insertion, as well as in circumstances where a CVC insertion is avoided and in post-operative surgical patients. Metaraminol is the most common agent in UK practice and our survey shows there has been low adoption of peripheral noradrenaline despite the presence of ICS guidelines. 6 HCP cited a variety of pros and cons for PaVi use in our survey however concerns about effectiveness, safety and efficacy, and a lack of evidence to support practice remain.
The use of peripheral vasopressor infusions to support critically ill patients has been an area of recent interest and research. Our survey showed metaraminol is the most commonly used PaVi in UK practice. This agrees with previous work by Grauslyte et al., 1 a small survey of 30 centres, which showed 88% of UK centres use metaraminol infusions. Furthermore, recent work by Australian colleagues 7 in an international survey of peripheral vasopressor infusions showed 82% of UK institutions use metaraminol as the first like PaVi in adult intensive care units. The ICS guidelines support PaVi doses for metaraminol up to 10 mg/h. Our survey suggests that only 50% of HCP were comfortable to infuse 7.5 mg/h or greater. Notably in UK practice there is also discrepancy in the NHS Injectable Medicines Guide also known as Medusa. 8 Medusa is available to every NHS Trust in the UK as the main reference source on injectable medicines and it recommends metaraminol be ‘preferably’ administered via a CVC and PaVi only be considered if a CVC is unavailable. This may explain why in our survey 43.6% of HCP administer metaraminol centrally.
Our survey showed relatively little use of (22.5%) peripheral noradrenaline infusion in clinical practice despite ICS guidelines supporting this practice. The guidance makes no recommendation for dosing range or maximal dose with this left to clinician discretion. The evidence to support this practice extends from studies examining risks of extravasation.9 –11 Peripheral infusion rates in these studies ranged from 0.01 to 0.7 mcg/kg/min and extravasation rates ranged from 0.035% to 4%. A single centre study 12 from the US reporting local implementation opted for a maximal dose of 0.2 mcg/kg/min for peripheral noradrenaline before transition to CVC in their in-house protocol. We also note the NHS Injectable Medicines Guide suggests noradrenaline be ‘preferably’ administered via the central route and PaVi considered in the absence of a central line which may further act as a barrier to the adoption of peripheral noradrenaline use in many critically ill patients requiring CVC access for other reasons. Furthermore, a recent prospective study 13 comparing peripheral metaraminol and noradrenaline concluded peripheral noradrenaline appeared to be safe and effective and significantly cheaper than metaraminol.
To date there have been several systematic reviews that have examined peripheral vasopressor safety.3,14 –17 Two recent systematic reviews and meta-analysis14,15 looking at adverse events associated with peripherally administered vasopressors reviewed 23 and 9 studies that included 16,055 and 1835 patients, respectively, determining the incidence of adverse events to be low. Findings by Tian et al. 3 showed that PaVi for limited duration were unlikely to cause major complications. Another recent systematic review 17 sought to compare safety of PaVi administration with CaVi. A total of 85 studies were included, of which the majority were case studies or series with only one randomised controlled trial. There were not trials within this review which directly compared PaVi to CaVi. The authors sought to examine reported incidence of adverse events. Withstanding significant limitations associated with the small sample size and reporting bias for adverse events within this review, there were more adverse events such as local tissue injury, skin and tissue necrosis, gangrene and vasopressor extravasation observed in the PaVi group than the CaVi. Thus, the relative safety of PaVi and CaVi remains to be determined.
Our survey demonstrated variability in doses and durations of PaVi used in current practice (Figures 5 and 6). We asked respondents which doses of PaVi they were comfortable to deliver (Figure 5). For phenylephrine the ICS states a dose range between 1.8 and 3.6 mg/h. Most respondents that were familiar with phenylephrine use, stated they would be comfortable to administer doses up to 5 mg/h. Within the third of respondents who were able to comment on their use of peripheral noradrenaline, there remained variability in doses that these individuals were comfortable to use. We saw similar variability in doses clinicians were comfortable to administer with metaraminol, however many more respondents were able to provide data here. This variability in practice in doses of peripheral vasopressors used may reflect the relatively poor evidence base to support practice and why ICS guidelines lend dosing and duration of use of peripheral vasopressor infusions to clinician discretion. This may also explain why within our study, clinicians tended to use shorter duration, lower dose infusions more commonly. Wider published studies18,19 to date have supported the idea of low doses of PaVi and this correlates with our survey findings where 90.7% of HCP were inclined to use PaVi where they anticipated low dose vasopressor requirements. However, what our survey adds at a granular level is practice is mixed and dependent on the vasopressor employed and context for use.
Our survey also captured significant practice variation with how long HCP were comfortable to continue PaVi (Figure 6). The majority of HCP using metaraminol appeared to be comfortable to deliver infusion for as long as the patient was achieving their target MAP. However, the majority of those using peripheral noradrenaline and phenylephrine, tended to limit infusion to less than 24 h. A large systematic review 3 reported a mean PaVi duration (range) of 22 (8–36) h and previous observational studies with less surveyed numbers have shown many UK intensive care units use time-limited vasopressor infusions for between 12 and 24 h.3,20,21 We also asked respondents when they would consider switching to CaVi, and for all three agents metaraminol, phenylephrine and noradrenaline, the majority stated they would escalate if vasopressor requirements were on an increasing trajectory.
Looking collectively at data, there appear to be potentially 4 factors influencing continued use of PaVi once initiated: (a) the trajectory of the dose required or a dose end-point, (b) a set duration of infusion end point, (c) a physiological end point and (d) a perceived optimal time to transition to a CVC. What is clear from our survey is the initiation of PaVi is not the issue however once a HCP starts PaVi, what happens thereafter is very variable and not evidence based.
Strengths and weaknesses
We believe the strength of our survey results are in that it is representative of the clinical settings in which these therapies are applied. We asked HCP to confirm they had recently worked in an adult critical care setting within the last 12 months so our results should be reflective of current UK practices. We have also tried to capture the views of the wider multi-disciplinary team and engaged with respondents in providing their opinions. Weaknesses ascribed can be this was a voluntary survey and not an extensive evaluation. Our survey has captured what individuals are able to disclose through this method, however, we acknowledge that qualitative work such as interviews or ethnography are methods that would allow a better understanding of factors influencing practice. Geographically we had minimal (<1%) response rates from Wales and as such, our finding may not reflect practice across the UK universally.
Conclusion
Despite systematic reviews demonstrating the safety of peripheral vasopressor infusions and ICS guidelines support their use in practice, our survey found great variability in their use and thus in care delivered to critically ill patients. Our survey suggests that there remain ongoing concerns regarding dosing ranges, safe durations of use and safety concerns and this may explain why lower doses, shorter durations on infusions are used and why peripheral vasopressors are used as holding measures until CVCs are inserted. Our respondents agreed that equipoise exists, there is uncertainty about best practice.
Future directions for research
Further research is required to develop the evidence base for optimal dose and duration of PaVi to support safe use. There is a need to explore whether there is a role for the use of peripheral vasopressors as sole interventions and which patients and/or settings would benefit the most from this. A better understanding is required of the barriers and concerns of HCP in their use of peripheral vasopressor infusions. In parallel, we recommend a review of the NHS Injectable Medicines Guide as it currently supports the use of peripheral vasopressors only if a central route is not available. This is not in line with current ICS guidance and may well be a barrier to prescription and use of peripheral vasopressors. Future research will need to explore the optimal role that peripheral vasopressor infusions can play in the care of critically ill patients.
Supplemental Material
sj-docx-1-inc-10.1177_17511437241259443 – Supplemental material for Peripherally administered vasopressors in critically ill adult patients: A survey of UK healthcare professional practices, attitudes and experiences
Supplemental material, sj-docx-1-inc-10.1177_17511437241259443 for Peripherally administered vasopressors in critically ill adult patients: A survey of UK healthcare professional practices, attitudes and experiences by Nauman Hussain, Tomas Jovaisa, Zudin Puthucheary, Rupert Pearse and Mandeep K Phull in Journal of the Intensive Care Society
Footnotes
Acknowledgements
The authors acknowledge the support of the UK Critical Care Research Group and Perioperative Medicine Clinical Trials Network for their support in disseminating the survey.
Author contribution
Nauman Hussain and Mandeep K Phull designed the study, performed the survey, analysed the data, prepared figures and/or tables, authored and reviewed drafts of the paper, and approved the final draft. Tomas Jovaisa, Rupert Pearse and Zudin Puthucheary designed the study, authored or reviewed drafts of the paper, and approved the final draft.
Data sharing
Further information is available from the corresponding authors upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
