Abstract
Lung ultrasonic B-lines have high accuracy in diagnosing extravascular lung water (ELW) but have not been systematically subcategorized to differentiate the varied etiologies of ELW. This brief communication describes subcategories of B-lines into “inflammatory” and “transudative” patterns, based on their location, pleural morphology and associated subpleural pathologies. This subcategorization was derived using information from trainees undergoing lung ultrasound training in the Learning Ultrasound in Critical Care program, pathophysiological principles and their corresponding ultrasound correlates. This subcategorization helped trainees differentiate inflammatory pathologies of ELW (e.g. pneumonia, acute respiratory distress syndrome) from transudative (congestive) pathologies (e.g. fluid overload, cardiac failure).
Introduction
In lung ultrasound in critically ill (LUCI),1,2 “B-lines” are dynamic, vertical ring-down artifacts from the pleural line to identify extravascular lung water (ELW).2 –7 ELW from increased hydrostatic pressure have uniform B-line distribution, while acute respiratory distress syndrome (ARDS) shows pleural line abnormalities and uneven tissue patterns. 8 However, these subcategorizations are not incorporated in diagnostic protocols.3,9
Since 2014, through the muti-center, longitudinal educational research program Learning Ultrasound in Critical Care (LUCC), using data from trainees’ LUCI interpretation and experiential learning to iteratively improve training processes, 10 we incorporated pathophysiological principles to classify B-lines into “inflammatory” and “transudative” patterns. This brief communication presents B-line subcategorization and a pilot study to evaluate the ultrasound patterns (Figure 1).
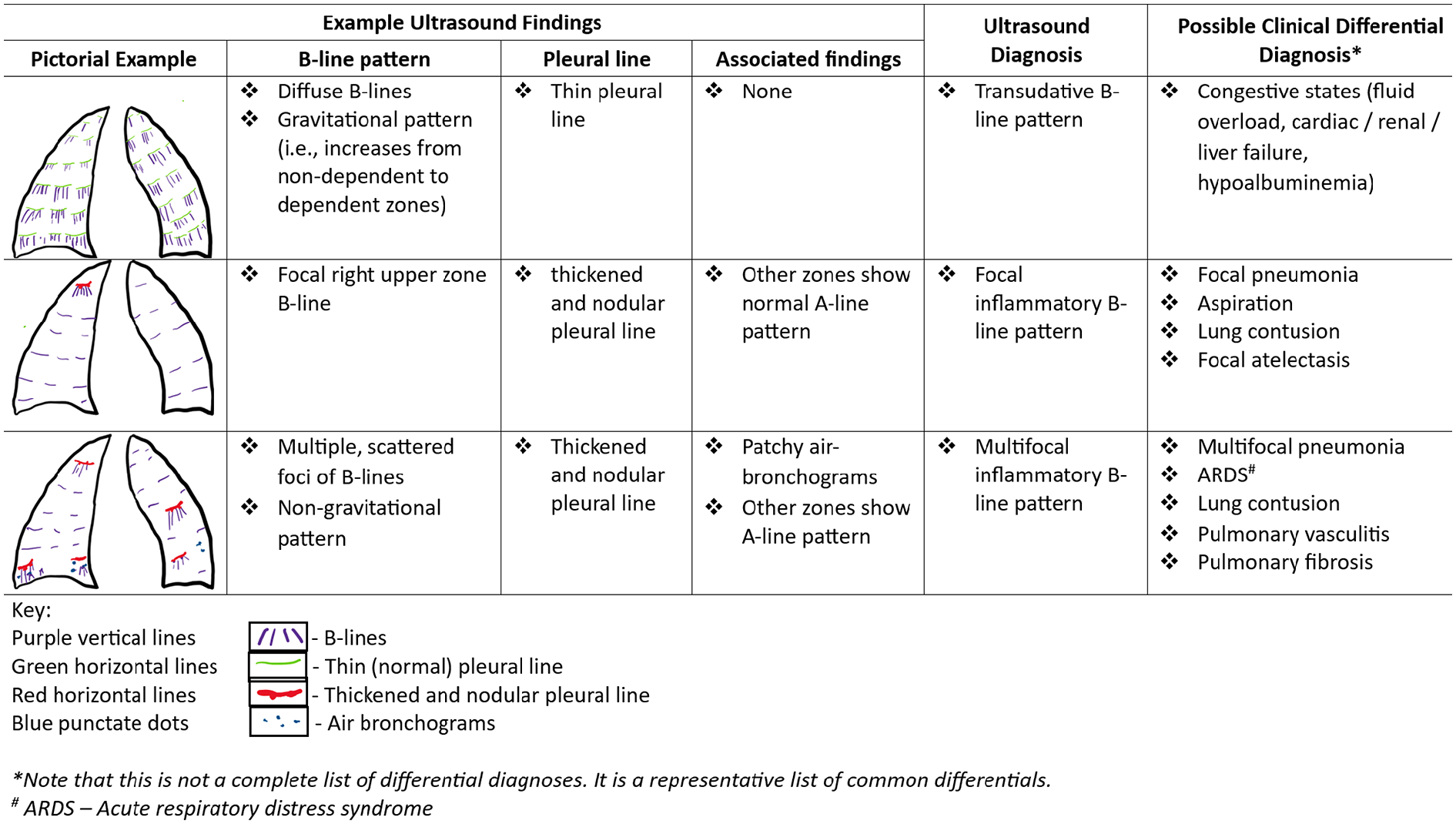
Pictorial representation of B-line patterns for the whole lung.
Pathophysiology (Figure 1)
Most inflammatory lung pathologies cause pleural morphological changes—pleural-line broadening with patchy/focal irregular/nodular ultrasound deformities. Subpleural/parenchymal inflammatory pathologies cause ultrasonic B-lines, C-lines, lung hepatization, and/or air-bronchograms. Inflammatory B-lines have heterogenous, multifocal, and non-gravitational distribution. Transudative pathologies have thin/smooth pleura, no subpleural inflammatory findings, and bilaterally homogenous/symmetrical distribution with gravitational preponderance.
Video_1 (https://photos.onedrive.com/share/2B874E3E657B5810!214420?cid=2B874E3E657B5810&resId=2B874E3E657B5810!214420&authkey=!APTsaYhAy_bxpBM&ithint=video&e=pjG6KG) and Video_2 (https://photos.onedrive.com/share/2B874E3E657B5810!214421?cid=2B874E3E657B5810&resId=2B874E3E657B5810!214421&authkey=!ANphCOg-ar-gNh0&ithint=video&e=GKuhKb) are LUCI images representing inflammatory and transudative B-lines respectively.
Methods
In phase_1, ten novice trainees underwent standardized LUCI training on BLUE protocol and international recommendations. 11 In phase_2, they underwent 1-h training on B-line subcategorization. After each phase, they answered 10 different multiple-choice questions of LUCI B-line video-clips, randomly chosen from a bank of 100 video-clips of varying etiologies (phase_1 = cases 1–10 and phase_2 = cases 11–20). Throughout, no feedback was given. Trainees rated the two approaches on a 3-point Likert scale (agree/neutral/ disagree). Their diagnoses were compared with expert (AR’s) diagnosis for accuracy and level of Fleiss’ Kappa agreement using R software.
Results
All 10 trainees agreed that B-line subcategorization improved their mechanistic understanding of B-lines. For the BLUE protocol, six were neutral and four disagreed.
Figure 2(a) shows a heatmap of trainees’ incorrect and correct interpretation in the pilot study. In phase_2, there were more correct diagnoses, indicating an improvement in diagnostic accuracy following B-line subcategorization training, with reduced variability compared to phase_1. Figure 2(b) shows that the Fleiss’ Kappa agreement between the trainees’ and expert’s diagnoses improved from 0.18 [0.09, 0.28] in phase_1 to 0.88 [0.79, 0.96] in phase_2.
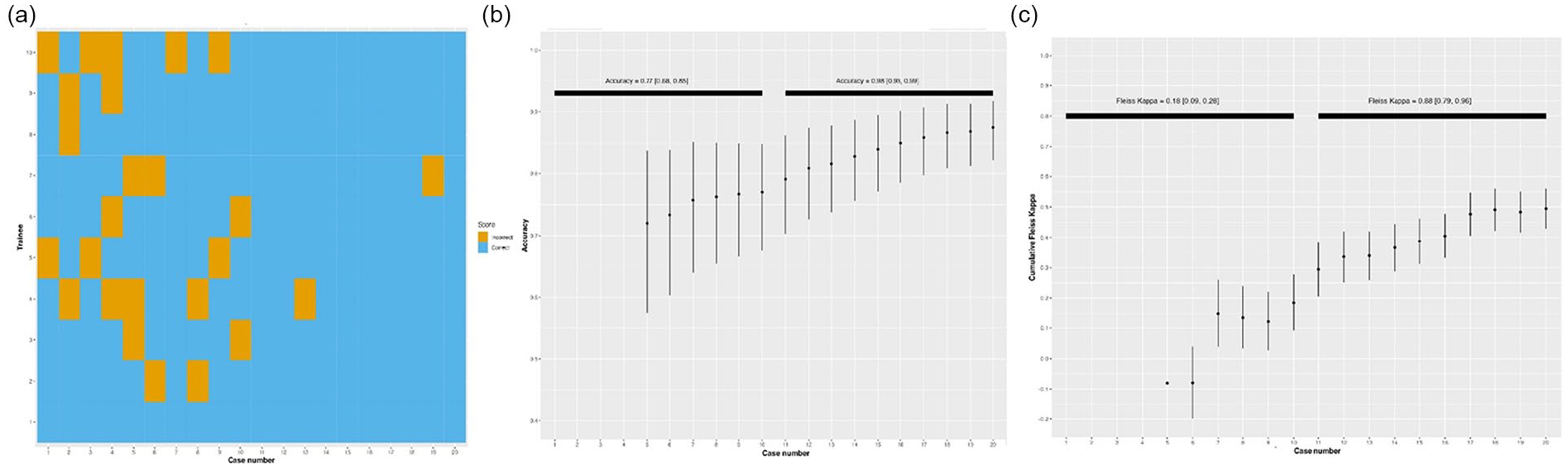
(a) Heatmap of correct and incorrect diagnoses made by trainees. (b) Accuracy graph depicting level of accuracy in diagnoses made by trainees. (c) Level of agreement between diagnoses made by trainees and expert using Fleiss’ Kappa.
Discussion
In this brief communication, using pathophysiological principles and experiential data from trainees undergoing longitudinal LUCI training, we present a modified approach to B-line analysis, subcategorizing them into inflammatory and transudative patterns.
Our approach is based on four facts: one, the reliable association between interstitial edema and ultrasonic B-lines 2 ; two, the common involvement of the pleura with almost all acute respiratory disorders 12 ; three, the cause of edema being either inflammatory or transudative; four, pleural inflammation causes morphological and ultrasonic abnormalities of the pleural line, differentiating inflammatory from cardiogenic/transudative pathologies. 13 During the LUCC program, trainees found our approach easy to understand and implement. We propose that that this approach may help trainees and practitioners interpret LUCI images to make relevant clinical diagnoses with more clarity.
The pilot study showed that all trainees reported that the training was effective, which was confirmed in the improvement in the number of correct diagnoses in the post-training phase. The combination of good accuracy and good agreement implies that this the subcategorization is effective and accurate.
Our report has the strengths of being based on sound pathophysiological principles and iterative development based on the experience gained from teaching trainees.
There are several limitations. First, sonographic differences between ARDS and cardiogenic edema have been described previously.5,8,13 However, ours is the first systematic incorporation of pathophysiology and B-line subcategories. Second, the algorithm for our training curriculum was based on the BLUE protocol which was described in 2005. 1 However, our training curriculum incorporated more contemporary evidenced-based recommendations. 11 Third, focused echocardiography/LUCI as part of whole-body ultrasound may differentiate inflammatory from transudative lung pathologies. However, cardiac dysfunction does not necessarily mean fluid overload as a cause of B-lines. For example, patients with cardiomyopathy may be hypovolemic from vomiting, yet have inflammatory B-lines from aspiration pneumonitis. Fourth, the exposure of the novice trainees to the BLUE protocol in Phase 1 may have contributed to their improved interpretation in Phase 2. Finally, our pilot study lacked statistical power. Hence, future studies with adequate power are required to validate this approach and help develop a new diagnostic algorithm for LUCI.
Conclusion
This small pilot study categorizes lung ultrasonic B-lines into inflammatory and transudative patterns to help trainees in their analysis of LUCI. The clinical and educational utility of this approach requires further exploration.
Footnotes
Author Contribution
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
