Abstract
Background
Combined Lung Ultrasound (LUS) and Focused UltraSound for Intensive Care heart (FUSIC Heart - formerly Focused Intensive Care Echocardiography, FICE) can aid diagnosis, risk stratification and management in COVID-19. However, data on its application and results are limited to small studies in varying countries and hospitals. This United Kingdom (UK) national service evaluation study assessed how combined LUS and FUSIC Heart were used in COVID-19 Intensive Care Unit (ICU) patients during the first wave of the pandemic.
Method
Twelve trusts across the UK registered for this prospective study. LUS and FUSIC Heart data were obtained, using a standardised data set including scoring of abnormalities, between 1st February 2020 to 30th July 2020. The scans were performed by intensivists with FUSIC Lung and Heart competency as a minimum standard. Data was anonymised locally prior to transfer to a central database.
Results
372 studies were performed on 265 patients. There was a small but significant relationship between LUS score >8 and 30-day mortality (OR 1.8). Progression of score was associated with an increase in 30-day mortality (OR 1.2). 30-day mortality was increased in patients with right ventricular (RV) dysfunction (49.4% vs 29.2%). Severity of LUS score correlated with RV dysfunction (p < 0.05). Change in management occurred in 65% of patients following a combined scan.
Conclusions
In COVID-19 patients, there is an association between lung ultrasound score severity, RV dysfunction and mortality identifiable by combined LUS and FUSIC Heart. The use of 12-point LUS scanning resulted in similar risk score to 6-point imaging in the majority of cases. Our findings suggest that serial combined LUS and FUSIC Heart on COVID-19 ICU patients may aid in clinical decision making and prognostication.
Introduction
Coronavirus disease 2019 (COVID-19) is a respiratory tract infection caused by a novel coronavirus: SARS-CoV-2. It is a heterogeneous disease with a wide spectrum of severity and organ dysfunction. In the UK, during the first wave of the pandemic, approximately 20% of hospitalised patients required admission to High Dependency Units (HDU) or ICUs. 1
Point of Care UltraSound (PoCUS) typically combines lung and cardiac imaging to obtain goal-oriented information and answer-specific clinical questions. It is a non-invasive, non-radioactive, bedside modality of imaging. 2 In patients with COVID-19, PoCUS can aid diagnosis, risk-stratification and management decisions. 3 However, uncertainty remains about how to utilise PoCUS optimally.
LUS uses pleural surface changes and structural patterns of the lung to identify disease. LUS has a sensitivity between 90–100% for diagnosing infection, effusion, pneumothorax or embolic phenomena.4,5 Algorithms such as the BLUE protocol 6 provide accessible methods for interpreting findings and differentiating causes of respiratory failure.
Critical care echocardiography (CCE) can be a focused or advanced structural assessment, with the option of dynamic cardiac output monitoring. CCE has become a well-established adjunct to care for initial and serial assessments of the shocked patient. 7 Whilst CCE is operator-dependent, focused echocardiography can provide very good correlation with departmental echocardiography – achieving a sensitivity of 0.91 and specificity of 0.88 for determining any degree of left ventricular (LV) systolic impairment. 8
In patients with acute respiratory distress syndrome (ARDS), CCE features of acute cor pulmonale (ACP) are found in 20–25%.9,10 These include RV dilation, RV impairment, septal dyskinesia and pulmonary hypertension. Lung inflammation and hypoxic vasoconstriction contribute to the disease pathology, and high positive end-expiratory pressure (PEEP) strategies can be used in the management of ARDS. Once established, ACP is an independent risk factor for 28-day mortality in those with moderate-to-severe ARDS (60% vs 36%, ACP vs no ACP). 11
As COVID-19 spread towards the UK, observational data from China 12 and Italy 13 supported LUS similarities between ARDS and COVID-19 pneumonitis (thickened pleura, multiple or confluent B-Lines and consolidations) in spite of two clearly different phenotypes (compliant and non-compliant 14 ). Scoring mechanisms were proposed that could help enable COVID-19 diagnosis and scale illness severity, 15 potentially reducing the burden of pandemic on hospital resources. 16
Few studies have reported focused CCE alongside LUS in COVID-19, 17 but echocardiographic findings correlate with changes seen in ARDS, as well as case reports of acute pulmonary embolism, dilated cardiomyopathy and acute myocardial injury. 18 Across 74 patients with COVID-19, one study showed RV dilatation in 41% and RV dysfunction in 27%, whilst LV function was normal in 89%. 19
This paper is a nationwide service evaluation looking at the application and results of LUS and FUSIC Heart in combination during the first wave of COVID-19. This is a novel approach to the assessment of an ad hoc service managing a large number of patients with similar physiological needs. Data from ICUs within 12 registered trusts was analysed looking for observable trends that can help establish better national PoCUS practice and guidelines. 20
This paper describes findings from the intensive care arm of the CORONA study, a study jointly led by the Intensive Care Society and the Society of Acute Medicine.
Methods
A prospective service evaluation of PoCUS was completed between 1st February and 30th July 2020. Service evaluation methodology was employed to define and describe variations in approach and evaluate relationships with outcomes during routine clinical practice. All adult patients (aged ≥16 years) presenting to participating hospitals with confirmed or suspected COVID-19 were eligible for inclusion. All point-of-care LUS and FUSIC Heart images obtained for these patients (with confirmed or suspected COVID-19) during their acute care admission were eligible for inclusion. The analysis presented is a subset of the national evaluation for patients admitted to the ICU of each registered trust.
Demographic and clinical data were recorded using a standard proforma. All point-of-care imaging was undertaken by trained practitioners: FAMUS (Focused Acute Medicine Ultrasound) or FUSIC Lung and Heart or a higher qualification for transthoracic echocardiography. The decision to undertake LUS and the approach to imaging was determined by the treating clinician. The ultrasound device used was determined by local availability. Serial images on the same patient were identified using a unique study ID number.
A data capture tool based on standardised point-of-care imaging protocols was provided.
LUS findings could be recorded in a maximum of 14 predefined anatomical zones. Clinicians recorded whether a 6-point, 12-point or 14-point approach had been undertaken. A semi-quantitative severity score was recorded at each anatomical location. ● Score 0: A-lines or <2 B lines (normal aeration). ● Score 1: ≥2 well-spaced B-lines (moderate loss of aeration). ● Score 2: Coalescent B lines +/− small consolidations < 1 cm (severe loss of aeration). ● Score 3: Tissue-like pattern +/− frank consolidation +/− small consolidations > 1 cm (complete loss of aeration).
Summated LUS severity score was approach dependent – in the case of 6-point imaging it was between 0–18. Presence of pleural effusion was recorded, and a subjective assessment of the pleura for each lung recorded as normal or abnormal.
Bedside transthoracic echocardiography data collection tool was derived from the FUSIC Heart protocol. This involved visual assessment for LV and RV dilatation and function. The presence of ventricular dilatation was recorded as a binary variable. Ventricular dysfunction was defined as either a dilated or visually impaired left or right ventricle. Advanced, quantitative measurements of dilatation and functional impairment were recorded in some studies.
The indication for imaging and any immediate change of management was recorded. Patient outcome was recorded at 30-days after the imaging study.
Statistics
Analysis was performed using the R statistical software. Normally distributed continuous variables are described using mean and standard deviation, and non-normally distributed continuous variables are described using median value and interquartile range. Count data is described as proportions with 95% confidence intervals.
Logistic regression was used to explore the relationship between LUS, the PCR result and 30-day outcomes. Some patients had repeated imaging obtained. The relationship between severity score and mortality was undertaken on the initial scan. Primary analysis was restricted to scans performed within 7 days of admission to the ICU. The multivariate models included age, sex, interval between ICU admission and study detection of SARS-CoV-2 by PCR as fixed effects. Model coefficients are expressed using odds ratios where appropriate. The discriminatory performance of the ultrasound severity score to predict 30-day mortality was assessed using receiver operating characteristic curves and reported by calculating the area under the curve (AUC).
Ethical approval and consent to participation
All participating sites registered the evaluation locally and obtained permission to share anonymised data by the relevant Caldicott Guardian. Data were anonymised locally prior to transfer to the central database (hosted at the University of Birmingham using Research Electronic Data Capture (REDCap) software 21 ) and identified using a unique study identification number. The study had R&I approval locally (East Kent Hospitals University NHS Foundation Trust); R&I reference number: 2021/GAP/13.
Results
ICUs from 12 UK hospitals recorded 372 imaging studies in the database between 1st February 2020 and 30th July 2020. The median number of studies contributed by individual hospitals was 14 (IQR 3–40). A total of 372 imaging studies were recorded from 265 ICU patients, 72 (27.2%) patients had two studies and 35 (13.5%) patients had more than two studies recorded. Paired thoracic imaging and echocardiography were recorded in 333 (89.5%) studies. Isolated echocardiography in 32 (8.6%) and isolated thoracic imaging was undertaken in 7 (1.9%) studies. The median interval between ICU admission and the first imaging study was 2 days (IQR 0–8).
The mean patient age was 54 years (12.4), and 187 (70.6%) of patients were male. The SARS-CoV-2 nasopharyngeal PCR was positive in 219 (82.6%, 95%CI 77.6–86.7) patients at the point of the initial imaging study. The SARS-CoV-2 PCR status of 15 (5.7%) patients was unknown.
Invasive mechanical ventilation (IMV) was being provided to 320 (86.0%) patients at the time of imaging. Non-invasive respiratory support was being provided to 24 (6.5%) patients at the time of imaging (Continuous positive airway pressure = 14, high flow nasal support = 5 and bi-level positive airway pressure support = 5). Death within 30 days of imaging occurred in 37 (32.8% 95%CI 27.5–38.7) patients. The outcome at 30 days was not recorded for 15 (5.7%) patients.
Thoracic imaging studies were conducted using a 6-point protocol in 305 (89.7%) patients and a 12-point protocol in 35 (10.3%) patients. Missing values in relation to one or more or the six anatomical thoracic imaging locations occurred in 17 (5.0%) studies. A single value was missing in 15 (4.4%) studies, two values in 1 (0.3%) study and three values in one study (0.3%).
The distribution of 6-point scores stratified by the interval between admission and imaging is provided in Figure 1(A). The distribution of scores was asymmetric with skew towards higher scores. The distribution of scores was broadly similar when stratified by the interval between ICU admission and scan. The median score using the 6-point protocol was 11 (8–13 IQR). Ultrasound severity scores obtained utilising a 12-point protocol were compared to the 6-point score derived from the same study. The 12-point scan did not result in a reclassification in the risk quartile in 28 (80.0%) studies. The 12-point scan resulted in re-classification into a higher risk quartile in 5 (14.3%) studies and into a lower risk quartile in 2 (5.7%). Lung severity scoring. (A): Distribution of 6-point score stratified by the interval between admission and imaging. (B): Distribution of scores stratified by anatomical location.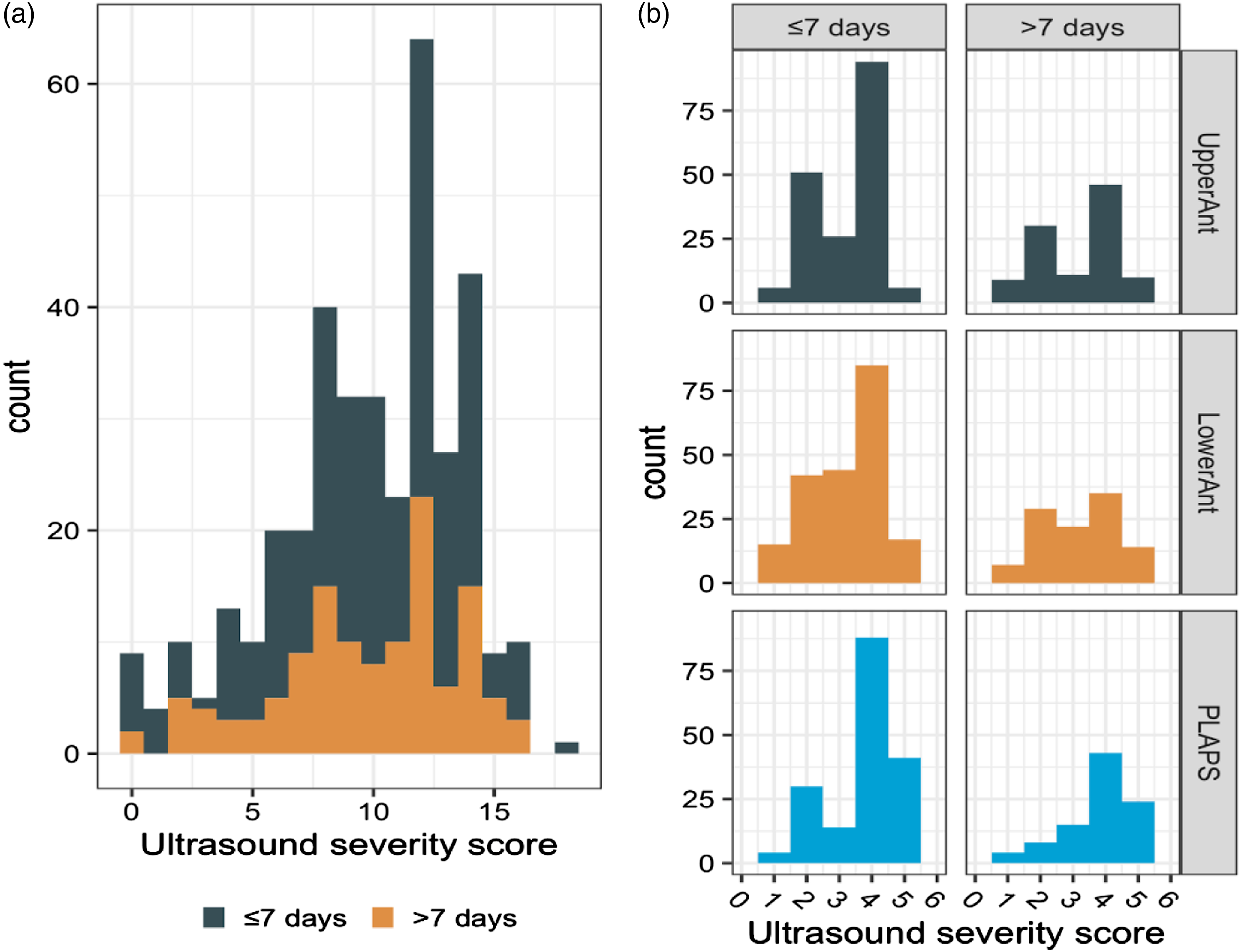
Pleural abnormalities were detected in 306 (90.0%) studies, abnormalities were unilateral in 19 (5.6%) studies and bilateral in 287 (84.4%) studies. Unilateral pleural effusions were detected in 29 (8.5%) and bilateral pleural effusions were detected in 14 (4.1%) studies. Of these patients, 5 (35.7%, 95% CI 16.3–61.2) had simultaneous evidence of LV impairment. Preserved A- lines in all six anatomical zones were observed in 9 (2.6%) studies of which 7 (77.8%) were negative for SARS-CoV-2 by PCR.
Indications for scan
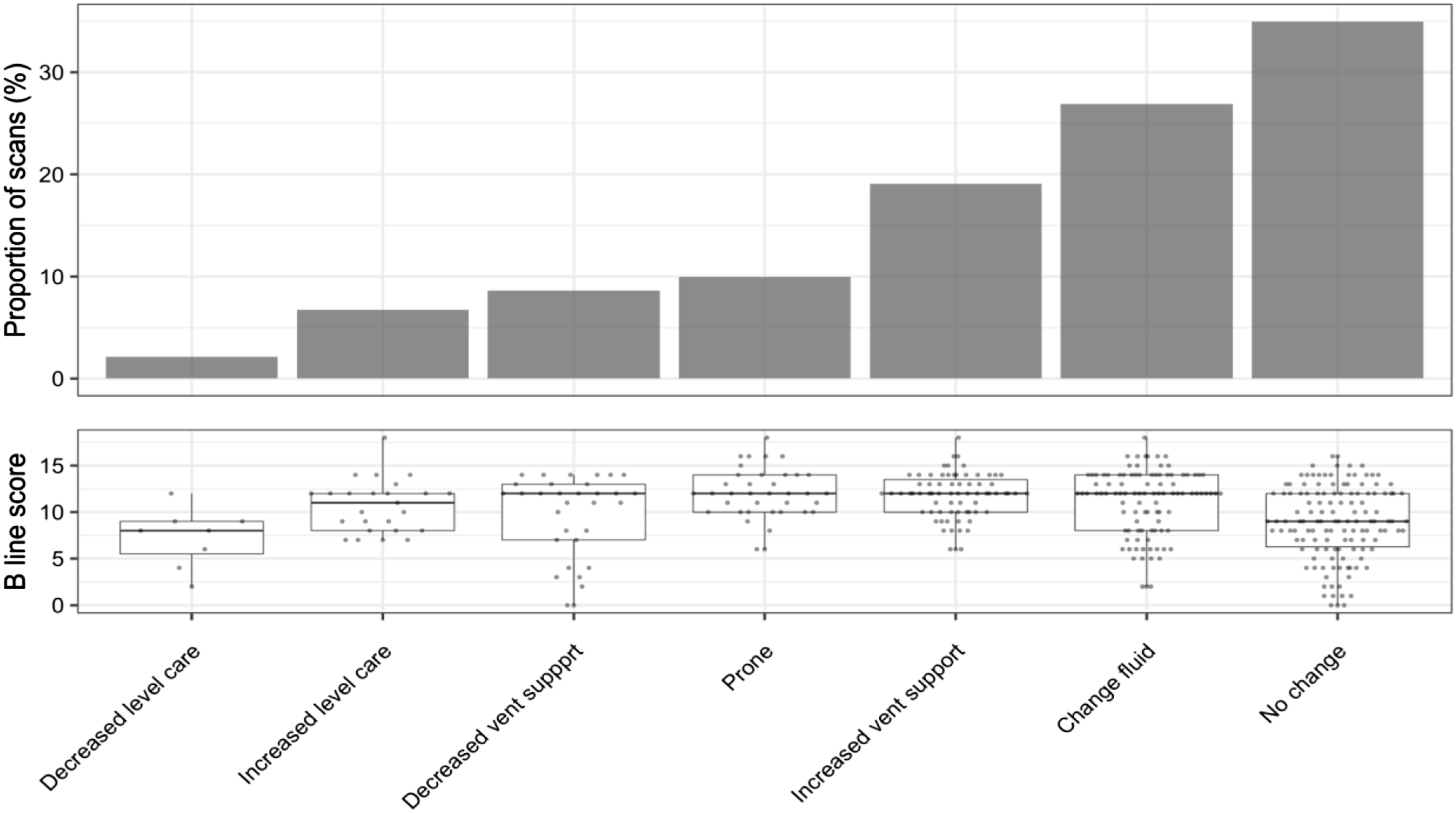
The indication and outcome of studies were recorded in the database. The most common reason for undertaking imaging was for routine surveillance accounting for 164 studies (44.3% 95% CI 39.3–49.4). The indication for imaging and associated distribution of 6-point lung ultrasound scores associated with each group is provided in Figure 2, and the outcome of scans is provided in Figure 3. Assessment of extravascular lung water (EVLW) was quoted as the indication for imaging in 116 (31.4%, 95%CI 26.8–36.2) studies. Imaging was employed to aid decision making in relation to ventilation strategy in 146 (39.5%, 95%CI 34.6–44.5) studies. Indication for lung assessment. Outcome following PoCUS scan.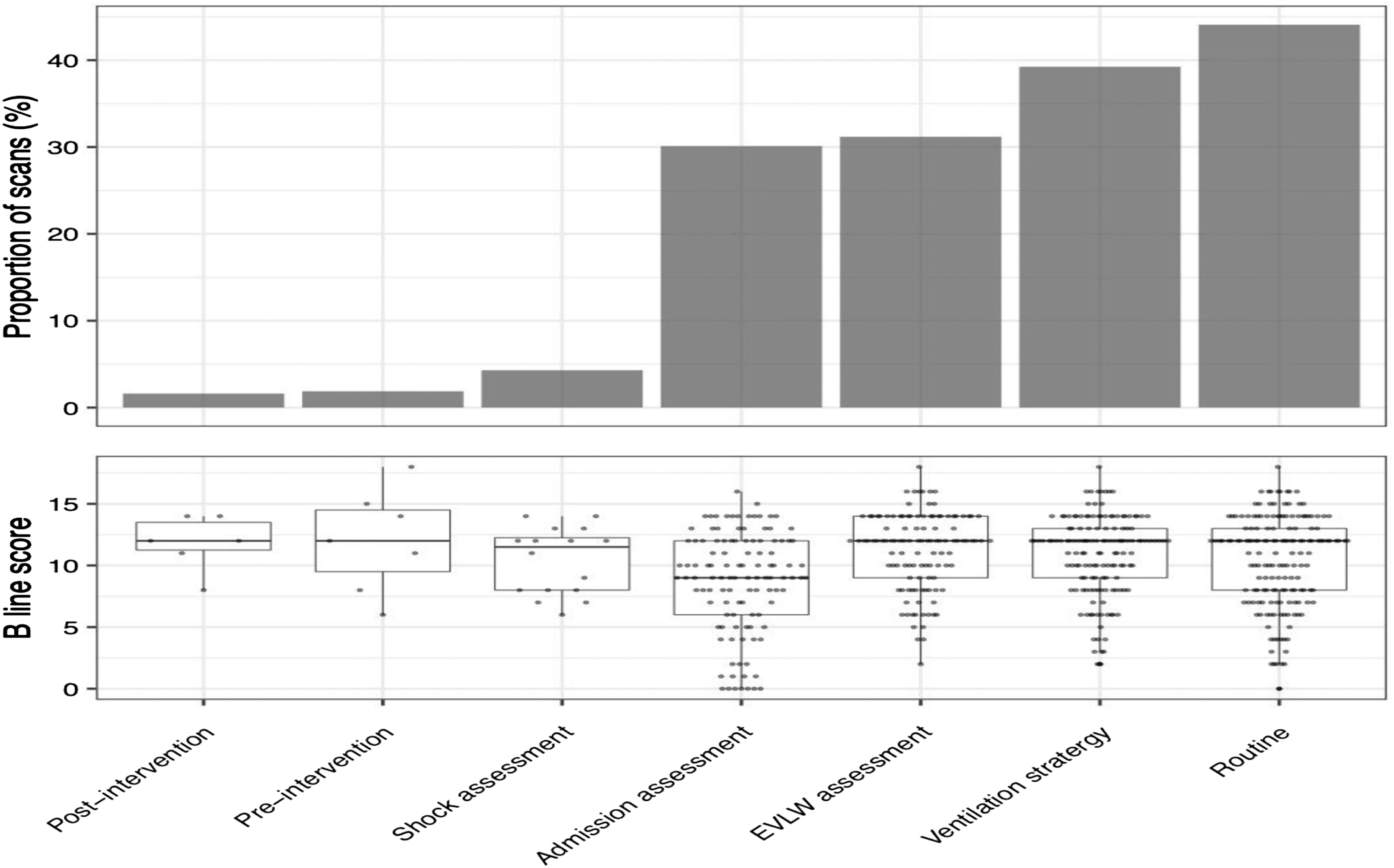
Lung involvement and mortality
There was an association between the severity of lung involvement (as assessed by 6-point severity score within 7 days of admission) and 30-day mortality (adjusted OR 1.1 95%CI 1.0–1.2).
The discriminatory performance of the ultrasound severity score to predict 30-day mortality was poor (AUC 0.58). The observed 30-day mortality associated within quartiles of lung severity score stratified by the time interval between ICU admission and the initial thoracic imaging study is shown in Figure 4(A). 30-day mortality and prevalence of RV dysfunction: (A): 30-day mortality stratified by ultrasound severity score and interval between admission and imaging. (B): Prevalence of RV dysfunction stratified by ultrasound severity score and interval between admission and imaging.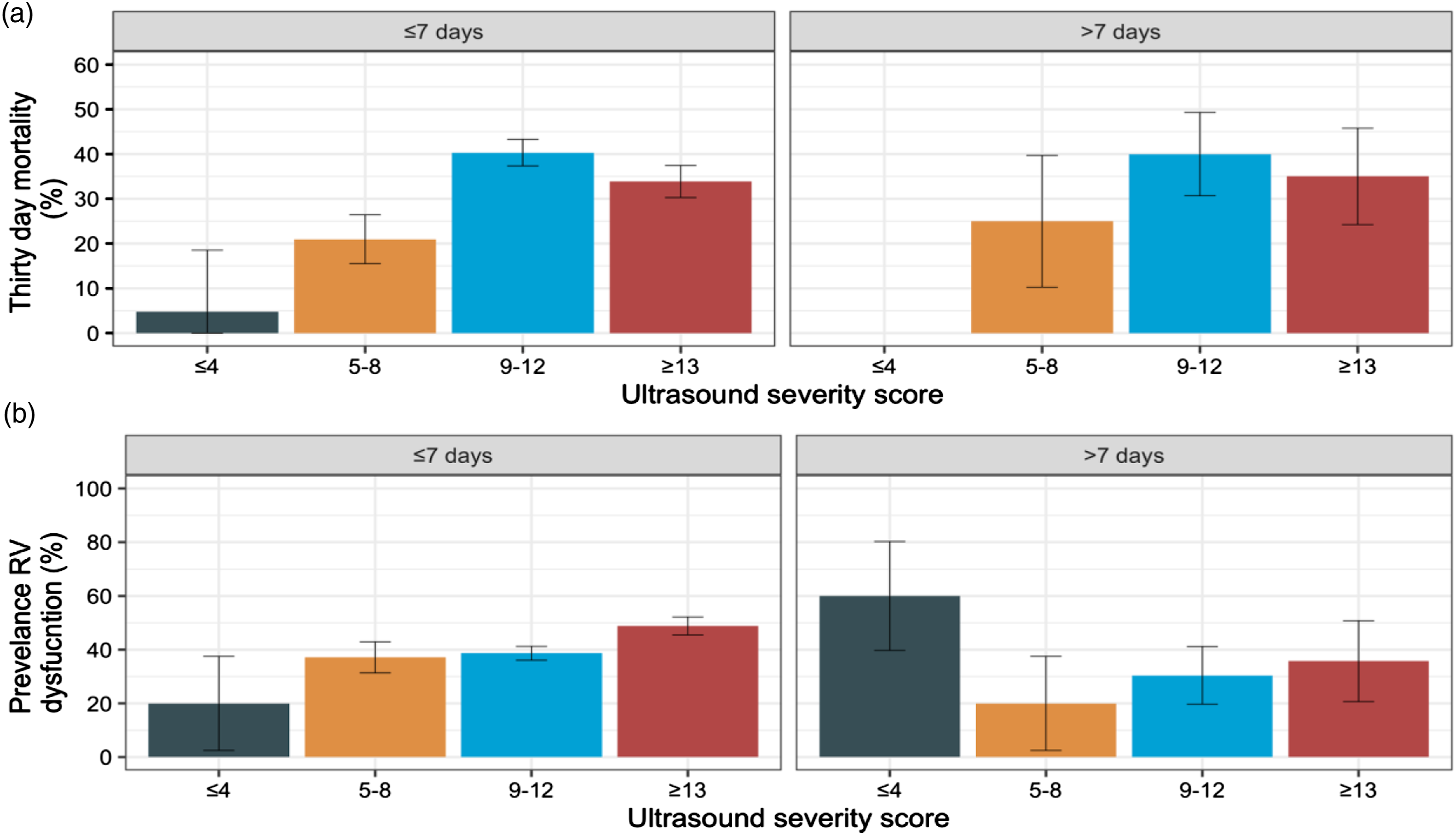
Echocardiography
Echocardiographic studies were recorded from 241 individual patients, of which 174 (72.8%) were focused studies and 67 (27.2%) were advanced bedside studies. RV dysfunction was identified in 83 (37.7%, 95%CI 31.5–44.3) of the initial studies. RV function was not recorded in six studies (2.5%). Observed 30-day mortality was significantly higher in patients with RV dysfunction compared with patients with normal RV function (49.4% 95%CI 38.9–59.9 vs, 29.2% 95%CI 22.2–37.3) (Figure 4(B)). There was no significant difference in the prevalence of RV dysfunction comparing focused echocardiographic studies with advanced bedside studies (35.1%, 95%CI 29.4–41.3 vs 36.7%, 95%CI 27.4–47.0). LV dysfunction was identified in 23 (9.5%, 95%CI 6.4–13.9) of initial studies of which 13 patients died within 30 days (56.5%, 95%CI 36.8–74.4).
Paired studies
Comparisons were made between the initial studies and a second study taken at a later time point in the 71 cases where serial studies were recorded. The median interval between the initial and second study was 4 days (IQR 3–5). There was no statistical difference in mean score comparing the initial and later study (mean 9.0 SD 3.6 vs 9.3 SD 3.8, p value = 0.6). The trajectory of lung severity scores between scans is provided in Figure 5(A). A unit increase in lung severity score was associated with a small increase in 30-day mortality in a model adjusted for the time interval between imaging studies (adjusted OR 1.2 95%CI 1.0–1.5, p value = <0.05). Paired imaging studies: (A): Paired ultrasound severity scores. (B): Initial B line severity score plotted against change in score from the second study.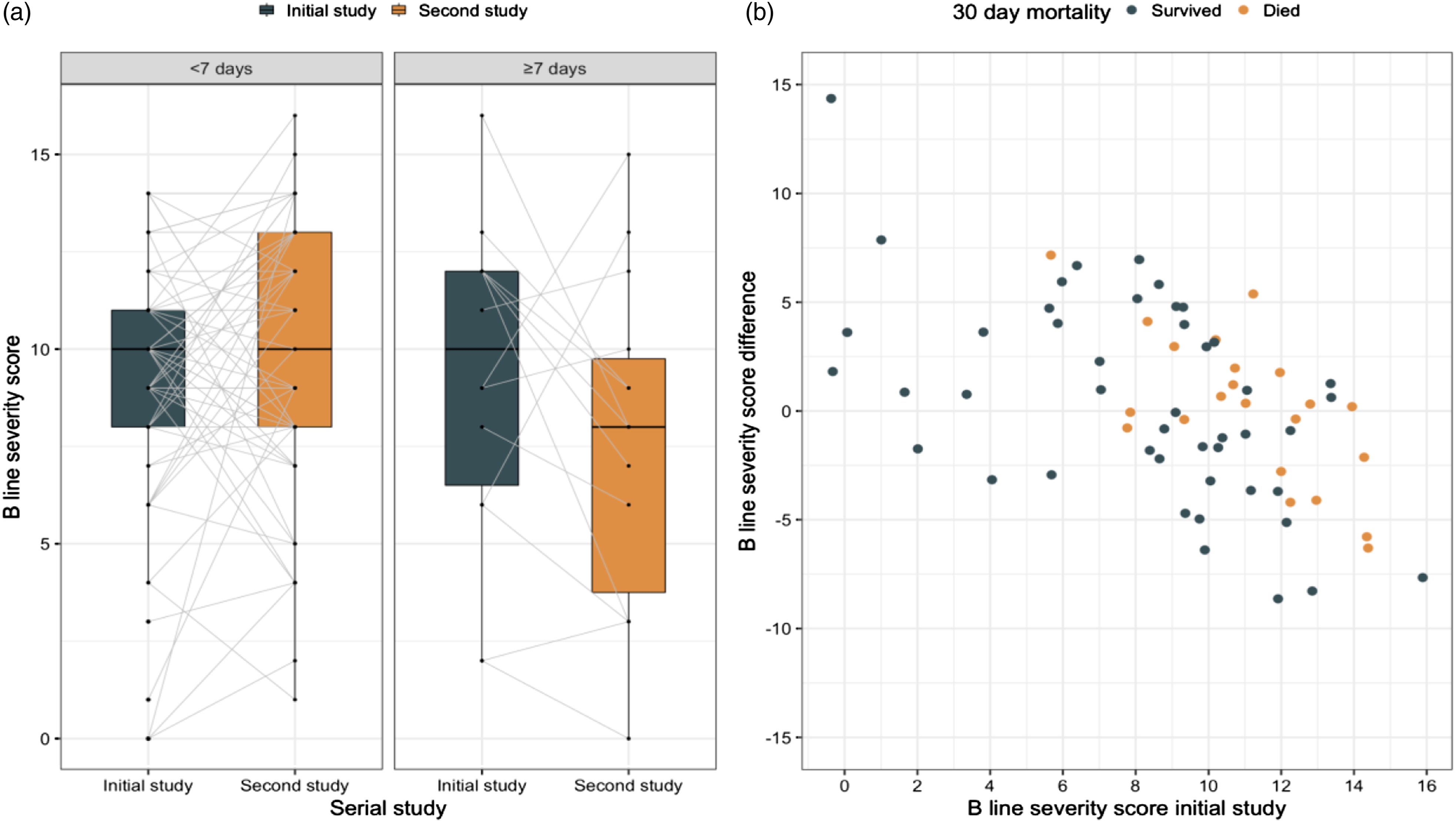
Discussion
This service evaluation is the first national study of its kind at the time of publication. It included patients admitted with respiratory failure during the COVID-19 pandemic from 12 different UK trusts. Patient demographics from this cohort closely matched ICNARC data 22 , indicating that we studied a generalisable sample.
There was a mean time to scan of 2 days from ICU admission, and only 39.5% of patients received two or more scans. Serial scan data in our study and other papers have shown increased mortality with worsening lung scores. 23 This suggests that there is currently suboptimal provision for serial ICU PoCUS services across the trusts involved and potentially nationwide.
However, in our evaluation, combined LUS and FUSIC Heart were performed in 89.5% of total patients enrolled. This indicates that, even in severely critically ill COVID-19 patients, combined LUS and FUSIC Heart were deliverable alongside the high intensity ICU clinical workload across the trusts. Maximum benefit of an ICU POCUS service would more likely be achieved with serial scanning of patients with subsequent changes in management rather than a focused scan done on ICU admission alone.
The correlation between 12-point and 6-point scores supports the role of the 6-point LUS protocol in ICU patients.24,25 The PosteroLateral Alveolar and/or Pleural Syndrome (PLAPS) is easily achievable and provides good discrimination from the anterior views.26-29 Clearly, this is beneficial as movement of COVID-19 patients for posterior views requires additional manpower, can be time-consuming and can cause instability.
Typical LUS findings have a good correlation with COVID-19 PCR positivity. This has been well studied and published previously.30-35 Preserved A-lines in all six zones made the diagnosis of COVID-19-related respiratory failure extremely unlikely. 36 Alveolar-interstitial syndrome associated with a normal LV function indicates a non-cardiogenic cause for respiratory failure.
LUS was able to identify those who had a higher 30-day mortality, particularly if the total score was >8 or there was a high score in the PLAPS zones. 37 The incidence of RV dysfunction was more prevalent in our study in those with higher LUS scores (indicating more extensive COVID lung involvement).38,39 This study has demonstrated worse outcomes in patients with RV dysfunction. This association has been extensively shown in ICU patients with RV dysfunction in the context of ARDS and COVID-19.9,40-44 Lazzeri et al. 17 noted LUS score and right:left ventricle ratio (representing RV dilatation) were both independent predictors of ICU mortality in their observational study of 47 ICU patients.
Most clinical guidelines used fraction of inspired oxygen concentration (FiO2), oxygen saturations and respiratory rate as parameters to identify patients who would benefit from critical care referral. 45 This study suggests that combined LUS score and RV assessment may aid in identifying those patients in a worse prognostic group. Patients with high LUS scores and RV dysfunction may benefit from earlier ICU admission and closer monitoring. However, as this was a service evaluation study, formal research studies will need to be done to assess this further.
65% of patients who had POCUS scans had a subsequent change in ICU management (Figure 3). Management strategies were adapted from the literature 46 and utilised on a recommendation basis from clinical advisory groups. Many of these guidelines included the need for LUS and FUSIC Heart to aid decision making. 47 These included optimal PEEP strategies based on lung compliance and right heart function, decision to prone and haemodynamic management strategies aimed at reducing pulmonary hypertension or augmenting the systolic RV function.
A systematic review of PoCUS use in patients with undifferentiated shock 48 documented two ICU-based studies where focused echocardiography resulted in a change in patient management in 41–51% of patients.49,50 These studies focused primarily on shock management as an end point. COVID-19 pathophysiology is complex, multi-system, and the potential interventions are numerous and interdependent. This may explain why our percentage of intervention was higher. However, this study did not measure whether interventions led to changes in outcome. There is inherent bias due to this study being performed by enthusiastic PoCUS practitioners and so caution must be taken when interpreting this observation.
Given the association between LUS score >8, RV dysfunction and outcome, acquiring this information becomes important. The utility of this information is clear: triaging to a clinical area of higher monitoring, instigating pre-emptive management strategies or altering existing therapies are all possible.
The combination of these two imaging modalities, LUS and FUSIC Heart, in COVID-19 patients at ICU admission (or ideally before) provides dynamic clinical information and informs the complex interactions between the heart and lungs and the effects, if any, of therapeutic intervention. It is therefore crucial to the overall management of this condition.
Limitations
Service evaluation methodology provides insight into how LUS and FUSIC Heart are utilised in the delivery of routine care at the expense of a defined study protocol which ensures consistency in approach. Based on the observational methodology, the results should be interpreted within the constraints and limitations of a service evaluation study.
There was considerable variation in the time to first imaging study and intervals between studies. Including scans performed in the initial 7 days from ICU admission introduces a degree of survivor bias. The use of a semi-quantitative score may be an oversimplification of a fundamentally subjective assessment. The salient information which dictated changes may not have been captured within the ordinal scale used.
This is an evaluation of a service that for most was set up de novo to meet the needs of the pandemic. This may explain some of the discrepancies in the results. It seems less likely that a patient admitted to the ICU with severe respiratory failure due to COVID-19 pneumonitis had a lung score of zero, which may reflect issues with PoCUS sonographers' training or reporting.
In this study, 86% of patients were being mechanically ventilated. The ICNARC data from 1 September 2020 to 30 April 2021* reports 30.6% of patients were mechanically ventilated within the first 24 h. Therefore, caution needs to be taken with regard to interpreting the results in this study, as significantly more patients were mechanically ventilated compared to current practice. In this study, 6.5% of patients were receiving NIV; LUS and FUSIC Heart scanning in these patients are of equal importance and, indeed, may influence management changes such as escalation of care and decision to prone.
It is important to note that RV assessment is more challenging than first appreciated. If the left ventricle is hyperdynamic, one may expect the right to be in a similar state. The fact that the right merely appeared to be ‘normally’ functioning may actually represent dysfunction at this stage in context. The basic level of training has enabled a larger number of patients to have an assessment scan. However, the tradeoff is that subtle echo signs such as apical RV dilatation or early systolic dysfunction may have not been appreciated as this would have required an advanced skill set not reliably available on ICUs.
This raises questions around ‘quantity versus quality’ – is it better to provide a screening service where most patients have a scan or is it desirable to have a detailed analysis in a few, sicker patients? This question is beyond the scope of this study and requires further investigation to understand the optimum way to organise a PoCUS service.
Conclusions
Our service evaluation suggests that serial, combined LUS and FUSIC Heart can perform an important clinical role in identification of COVID-19, risk-stratification of patients, monitoring disease progress and guiding interventions on ICU. There was an association between lung ultrasound score, RV dysfunction and mortality in this study. A 12-point ultrasound scanning resulted in similar risk predictions to a 6-point scan in the majority of case. Further research studies need to be done to further identify the role of LUS and FUSIC Heart in COVID-19 ICU patients.
Footnotes
Acknowledgements
We would like to thank all of the members of participating hospital’s PoCUS teams without whose hard work the results of the study could not have been acquired.
Declaration of competing interests
The authors declare that they have no competing interests.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Availability of Supporting Data
The datasets used during the critical care study are available from the corresponding author at reasonable request.
