Abstract
In recent years, point-of-care ultrasound has become a useful technique for assisting in rapid diagnosis or exclusion of pneumothorax. It has been shown to have excellent sensitivity and specificity. Despite this, we present a case of a large pneumothorax that was not detected by bedside ultrasound. We consider the reasons for failure and discuss pitfalls and limitations described in the literature.
Introduction
Ultrasound (US) is a valuable bedside tool in the intensive care setting for aiding diagnosis and facilitating procedures. Lung ultrasound can be particularly helpful in conditions commonly seen in critically ill patients, including pleural effusion, pulmonary oedema, consolidation and pneumothorax. It offers several advantages over computed tomography (CT), which is considered the gold standard. 1 These include lack of radiation, lack of need for patient transportation, reduced cost and ready availability. These factors lead to more timely diagnosis with safer procedures, such as real-time drain insertion. Several studies have confirmed its high sensitivity and specificity for the diagnosis of pneumothorax.1–5 However, there are occasions where US may fail to identify a pneumothorax, resulting in false reassurance and diagnostic bias.
Case study
A 67-year-old female patient underwent an elective left lower lobectomy for lung carcinoma. Post-operatively, she was admitted to the intensive care unit (ICU) with progressive hypoxaemia due to presumed right-sided pneumonia and pulmonary oedema. She was initially managed with non-invasive ventilatory support, diuresis and antibiotics. A bedside lung ultrasound on the day of ICU admission, performed by an experienced operator, demonstrated the phenomenon of B-lines suggesting oedema but no evidence of effusion, consolidation or pneumothorax.
She further deteriorated and was subsequently intubated. She underwent a CT chest and pulmonary angiography. This excluded pulmonary embolism but revealed widespread ground glass opacification within the right lung and a small left pneumothorax. Two left-sided intercostal drains were already in situ following her surgery. A bronchoscopy revealed only watery secretions. She was successfully extubated the following day and managed with high flow oxygen via nasal cannulae and non-invasive ventilation. However, due to worsening hypoxaemia, she failed extubation and was re-intubated.
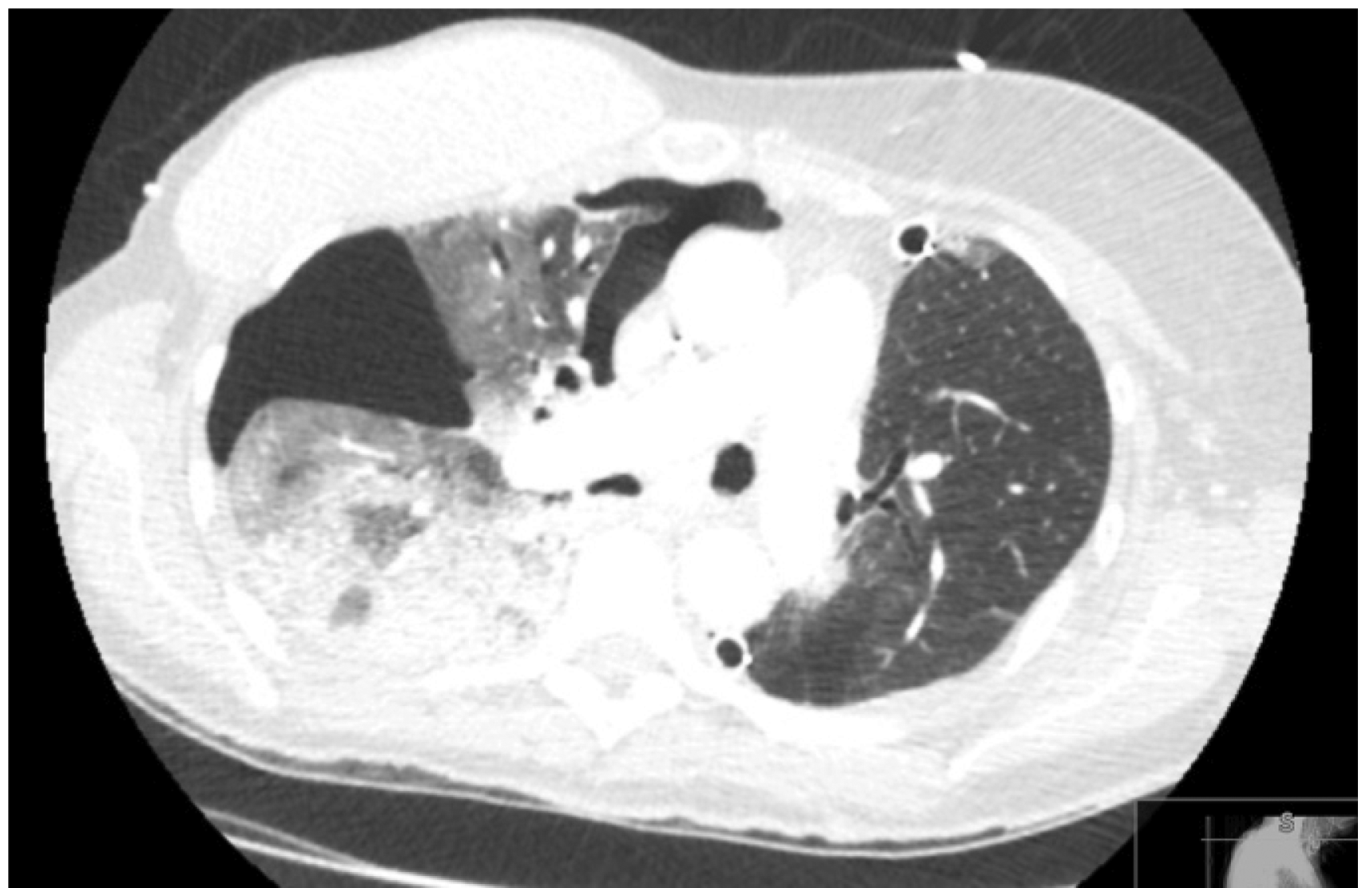
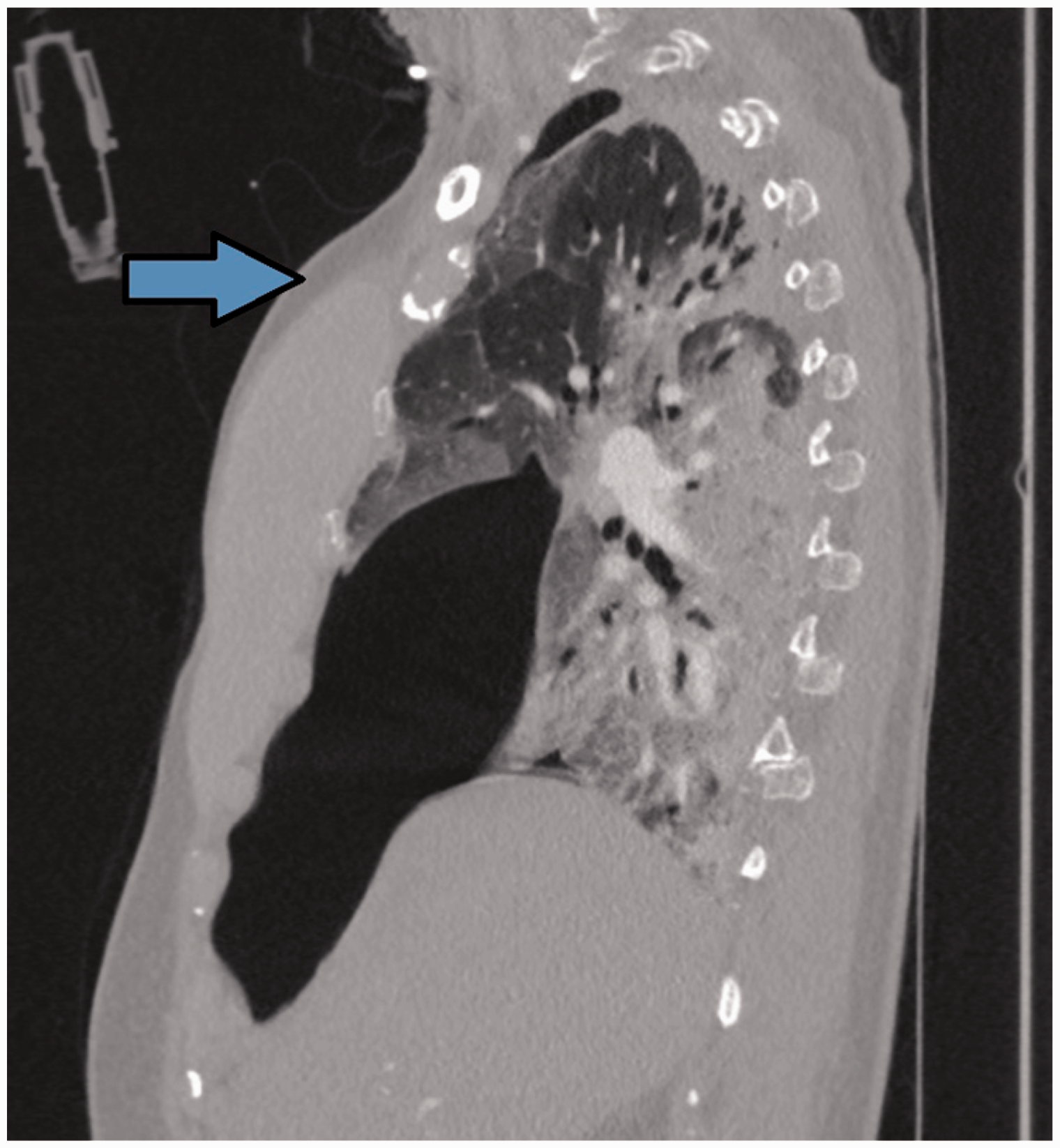
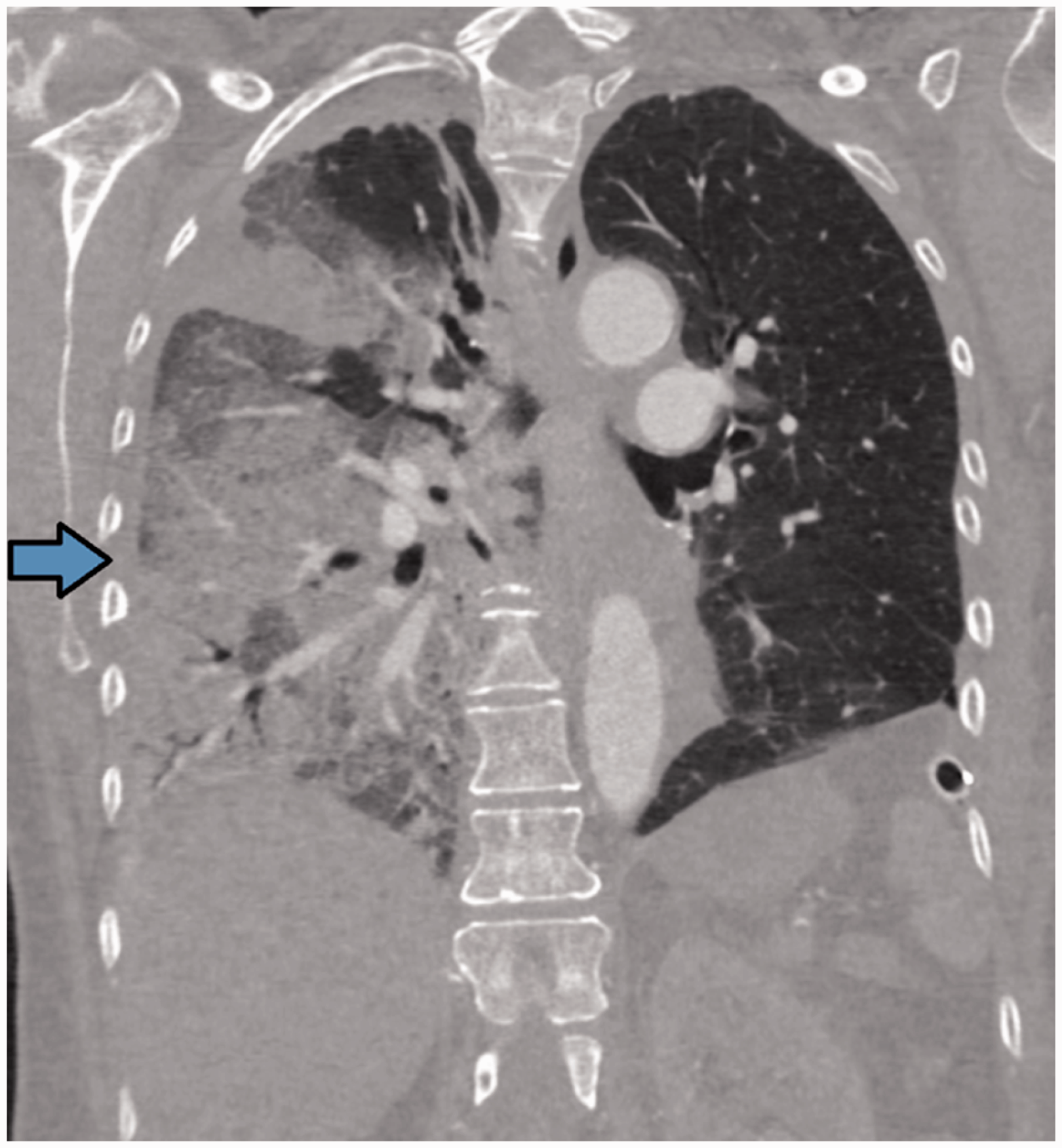
A bedside ultrasound was performed at this time to exclude worsening of the known small pneumothorax in the context of positive pressure ventilation. This did not reveal any evidence of pneumothorax on either side. It was performed by a consultant experienced in bedside thoracic ultrasound in both the standard anterior and lateral chest positions. A repeat CT was undertaken to investigate causes of the respiratory deterioration. Selected images are shown below and demonstrate a large right-sided anterior pneumothorax (Figures 1 and 2). However, as can be seen, portions of the lung remain tethered to the chest wall, particularly in the superior anterior and posterior lateral areas, where one may well place the US probe (Figures 3 and 4). In these areas, US did not demonstrate the sonographic signs characteristic of pneumothorax. A right mastectomy and subpectoral breast prosthesis are also noted. A right-sided intercostal drain was inserted uneventfully, and the patient’s oxygenation improved.
The patient proceeded to have a tracheostomy inserted and was successfully weaned from ventilation several weeks later.
Transverse CT image demonstrating the heterogeneous nature of the pneumothorax within the right hemithorax. Transverse CT image at a lower level demonstrating the heterogeneous nature of the pneumothorax within the right hemithorax. Sagittal plane CT image demonstrating the anterior nature of the pneumothorax and with lung remaining in contact with the chest wall at the superior anterior aspect. The arrow indicates a likely anterior probe position. Coronal plane CT image showing the posterior aspect of the right lung extending to the lateral chest wall with associated consolidation. The arrow indicates a possible lateral probe position.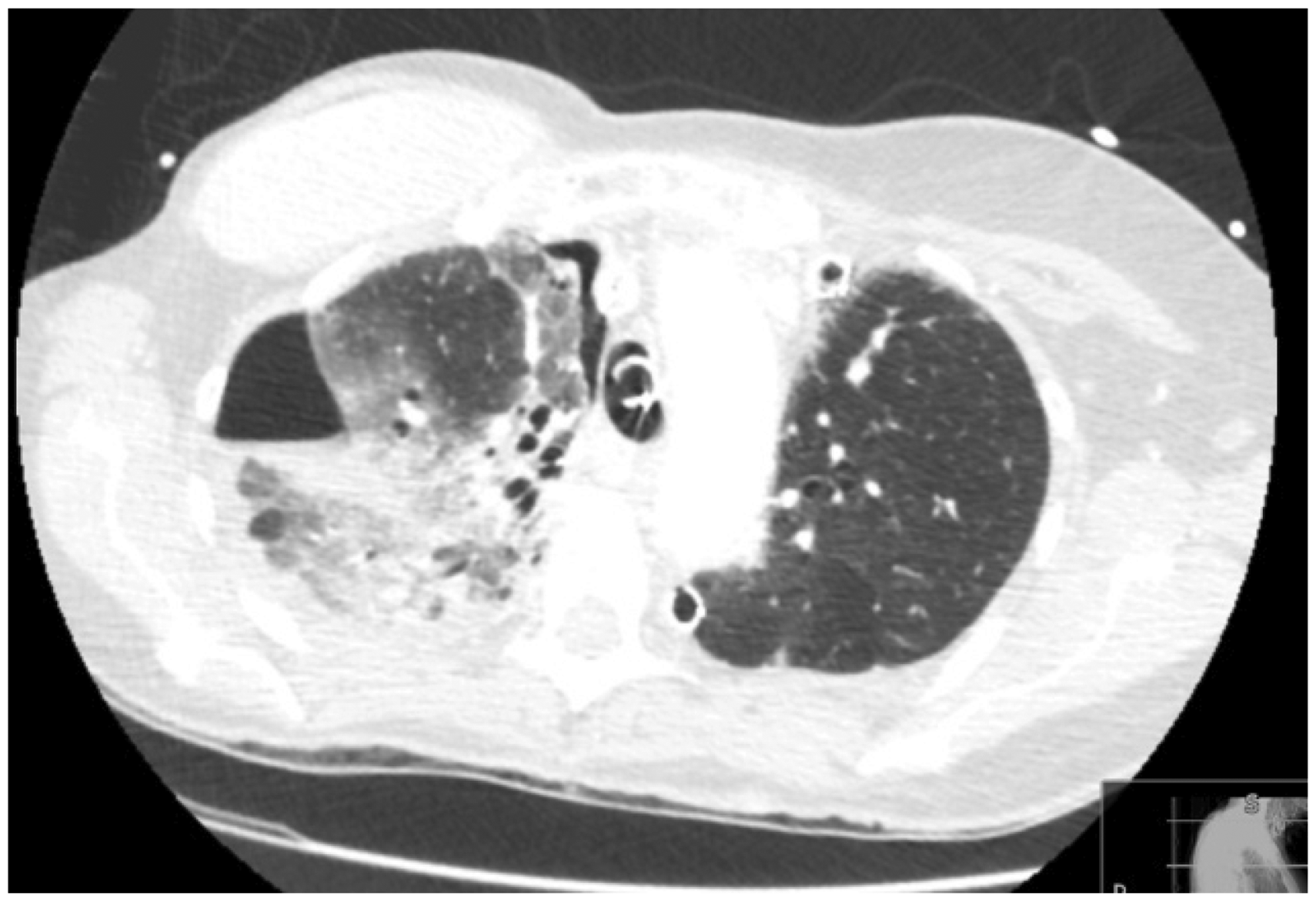
Discussion
The use of US to assist in the diagnosis of pneumothorax was first described in a horse in a veterinary journal in 1986. 6 Wernecke et al. 7 reported this use of US in humans. Since then, a multitude of studies have confirmed its value in clinical practice. Several have investigated the sensitivity of ultrasound for the detection of pneumothorax using CT as the gold standard and found US to be superior to chest radiography.1–5,8 A meta-analysis in 2011 reported a pooled sensitivity and specificity of 88% and 99%, respectively, for ultrasonography, and 52% and 100%, respectively, for chest radiography. 4 As a result, it has steadily gained popularity, especially amongst intensivists and emergency physicians.
Sonographic features of pneumothorax include the absence of lung sliding, presence of A-lines and the ‘lung point’, with the absence of B-lines. 9 Lichtenstein et al. 1 demonstrated that for the diagnosis of occult pneumothorax, the abolition of lung sliding alone had a sensitivity of 100% and a specificity of 78%. Absent lung sliding plus the ‘A-line sign’ had a sensitivity of 95% and a specificity of 94%. The ‘lung point’ had a sensitivity of 79% and a specificity of 100%. 1
Following our experience, we explored instances where ultrasound may fail to detect a pneumothorax or incorrectly diagnose pneumothorax. In our case, despite scanning by an experienced operator, a significant pneumothorax was missed. Upon retrospective review of the CT images, it can be seen that the pneumothorax is not spread uniformly throughout the hemithorax, but rather that portions of the lung remain in contact with the chest wall. In this case, the standard probe positions are where these parts of lung remain inflated and tethered to the chest wall, leading to false reassurance and diagnostic bias that pneumothorax was not present. This is of interest when considering ‘standard’ probe positioning to exclude this pathology.
Much of the literature on this subject focuses on positive aspects of US in this setting. There is a relative paucity of information regarding the instances where US may fail to correctly rule a pneumothorax in or out. Clearly, it is vital that the operator has a firm understanding of sonographic features. It follows that operator experience may influence accuracy.4,5 If it is assumed the operator can correctly identify the presence or absence of all features, are there occasions where we may be misled due to other pathology? False-positive diagnosis of pneumothorax using US has been described in bullous lesions and severe chronic obstructive pulmonary disease.1,10 It is also thought that sensitivity and specificity decreases after the first 24 h and morbid obesity may present difficulty due to loss of resolution. 1 A review by Volpicelli 11 notes that subcutaneous emphysema may mimic pleural motion and other artefacts similar to B-lines. Similarly, parietal emphysema or pleural calcifications may hinder the visualisation of the pleural interface. 1 Volpicelli also highlights that air may be trapped in different areas if the visceral and parietal pleura for some reason adhere. In such cases, the ‘lung point’ may be inaccurate in indicating the extent of the pneumothorax.
The use of medical ultrasound has evolved to include a plethora of uses. It is prudent to be mindful that novel applications of this technology would not have undergone a rigorous testing as say a new drug therapy would. Thorough training and robust evaluation are essential to ensure appropriate use of the technology, including delineating clinical conditions and patient groups where it may not be possible to apply general principles. Technology is not a substitute for clinical assessment; it is important to avoid fixation error or diagnostic bias, but to utilize USS as a powerful complementary adjunct for prompt bedside management in conjunction with all clinical history and findings.
Conclusion
Lung ultrasound is a useful aid to the diagnosis of a variety of conditions, including pneumothorax. We present a case for shared learning that highlights limitations of this technique and mindfulness to avoid bias and potential clinical misdiagnosis. Despite increasing popularity and clinical use, CT remains the gold standard.
Footnotes
Acknowledgements
We would like to thank Dr. Vikram Raju, consultant radiologist, for his expertise.
Consent
Published with written consent from the patient.
Declaration of conflicting interests
The author(s) declared no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
