Abstract
Background:
Frailty is common and is present in up to 30% of patients admitted to the ICU. We do not yet understand the true impact of ICU admission on this population, certainly in terms of longer-term outcomes.
Method:
Random selection of patients admitted to each general ICU within the SWARM network between October 1st 2012 and January 31st 2013. To be eligible, patients had to be over the age of 18 and admitted to the ICU with either a medical diagnosis or following emergency gastrointestinal/vascular surgery.
Results:
1888 patients were eligible for random selection. 560 patients were selected for the final analysis. Three hundred and eighteen patients had an estimated frailty score between 1 and 3 (classified as not frail, 56.8%), whilst 112 had a score of 4 (classified as vulnerable, 20%) and 130 patients had a score of between 5 and 7 (classified as frail, 23.2%). The median survival for patients with an estimated frailty score of four (vulnerable) was lower than those with an estimated score of 5 or greater (331.5 vs 594 days, p < 0.0001). One-year survival was 79% (75, 84), whilst 5-year survival was 69% (65, 75). Hazard ratios for ‘vulnerable’ and ‘frail’ sub-groups were estimated as 2.6 and 2.4 respectively.
Conclusion:
There is significant evidence to suggest a strong causal association between frailty and survival after ICU admission. ‘Vulnerable’ status may well be associated with similar effect on long-term survival to that of ‘frailty’.
Introduction
Recent unprecedented pressure on intensive care medicine services has emphasised the shortfall in Intensive Care Unit (ICU) bed provision within the United Kingdom (UK). 1 This increase in ICU service pressure is partly driven by an ageing population with a mounting burden of co-morbidities, factors that compound our low bed capacity per head of population. 2 An inevitable consequence of this situation is the complexity of decisions that need to be made with regards to ICU admission and the instigation of invasive therapies. The process of complex decision-making requires careful clinical judgement, ethical consideration, and the principles of shared decision-making. Central to all three of these facets is an accurate understanding of the individualised risks and benefits of all treatment options, including the most invasive therapies offered in the ICU. Recent expert guidance has rightly emphasised this point, the importance of individualised care based upon those therapies most likely to benefit them. 3
Patients discharged from ICU may face significant health concerns, including physical, cognitive and mental health impairments that together comprise the ‘post-ICU syndrome’. 4 The impact of these impairments, alongside new co-morbidities, on longer-term mortality (or life expectancy) is not well understood. This makes the individualised risk benefit analysis or ‘best-interest’ decisions difficult; how can we weigh the benefits versus the risks when we have a limited understanding of what survival means for an individual in the longer-term? A better appreciation of population life expectancy and the impact that critical illness has on survival is important.
Frailty is common and is present in up to 30% of patients admitted to the ICU 5 ; it is a state of reduced physiological reserve which can encompass low muscle mass, reduced mobility, malnutrition and cognitive dysfunction.6,7 Frail patients are more susceptible to acute stressors and frailty is associated with increased short-term mortality, prolonged ICU admission, increased rates of discharge to residential care and greater health economic burden.8–18 Although frailty is more common in both older and more co-morbid adults, ageing and frailty are not synonymous.17,19 Younger individuals with significant chronic illness, multimorbidity or disability may present as part of a wider medically vulnerable group. 20 The ICU admission of a frail patient may not be within that individual’s best interests; however, we do not yet understand the true impact of ICU admission on this population, certainly in terms of longer-term outcome.
The South West Anaesthesia Research Matrix (SWARM, our regional trainee research network) conducted a retrospective, multi-centre observational study with two predominant aims; (1) to estimate life expectancy after ICU admission compared with that of the general UK population and (2) to estimate the impact of increasing frailty on life expectancy.
Methods
Selection of data
Each participating site collected data on up to 100, randomly selected patients admitted to each general ICU within the SWARM network (Royal Cornwall hospital; Royal Devon and Exeter hospital; Torbay hospital; Derriford hospital; North Devon district hospital; Musgrove Park hospital; Yeovil district hospital) between October 1st 2012 and 31st January 2013. The patients were selected using a random number generator which sorted and randomly picked the first 100 patients at each site. If a patient did not meet inclusion criteria the next randomly identified patient was selected.
To be eligible, patients had to be over the age of 18 and admitted to the ICU for greater than 48 h with either a medical diagnosis or following emergency gastrointestinal/vascular surgery. Emergency gastrointestinal and vascular surgical procedures were selected for inclusion in our cohort as a patient group with significant burden of critical illness common between each recruiting site. Exclusion criteria were age less than 18, admission to ICU following elective surgery or following emergency surgery other than specified above.
Data was collected from three separate sources: date of death from NHS digital, variables associated with the ICU stay from the ICNARC database (Intensive Care National Audit and Research Centre) and physical function/co-morbidity/laboratory result data from patient records. Researchers were core trainees in anaesthesia and intensive care medicine, specific to each site. Individual members of local data collection teams estimated patient frailty using the Rockwood et al. Clinical Frailty Score (CFS) 19 . Data for this estimation was taken from hospital and ICU admission notes found within the medical record. This was completed by a thorough review of each hospital admission record to collate all information pertaining to frailty status, including multidisciplinary team assessments, hospital and ICU admission documentation. Standardised data collection forms and guides on CFS estimation were distributed to each local site team, 19 in an effort to ensure consistency of clinical frailty scoring across all sites. Body mass index and estimated glomerular filtration rate (eGFR) were calculated.
Outcome and variable selection
The primary outcome was time to death, right censored on 1st February 2020. The data variables chosen for collection were based upon previous analyses, prognostic models, our own pilot analysis and clinical acumen.21–25
Missing data
The final dataset was assessed for missing data. Data was assumed to be missing at random. Multiple imputation by chain equations (MICE) was used for imputation of missing values; five separate, imputed, complete datasets were constructed and one chosen at random for final modelling.
Statistical analysis
Our primary survival analysis involved two separate steps:
(1) Frequency tables and Kaplan-Meier estimation of survival function for whole cohort and specific pre-identified sub-cohorts. These were Rockwood et al. Clinical Frailty Score (‘frailty’) group 19 (not-frail defined as a CFS of one to three, vulnerable defined as a CFS of four and frail defined as a CFS of five or more; vulnerable patients were categorised separately to not-frail or frail patients due to a hypothesised difference in survival), age group (ages 18–64, 65–79 and over 80)26,27 and invasive organ support groups (received advanced respiratory, cardiovascular or renal support).
(2) A Relative Survival Analysis graphically comparing survival of the whole cohort and certain sub-groups to an age and sex matched United Kingdom population using data from the Office of National Statistics. 28 As a secondary analysis we went about building a multiple-variable accelerated failure time (AFT) model. This model investigated the association between our primary exposure variable (CFS) and our primary outcome (time to death) by adjusting for multiple, confounding variables.
A Direct Acyclic Graph (DAG, see Figure 1) was constructed to identify confounding pathways and therefore the candidate variables that the model would need to adjust for to estimate any (causal) association between CFS and outcome. 29 This DAG was constructed using local expert opinion across multiple ICUs, including clinicians both within and outside of Intensive Care. We hoped to limit the number of variables (if possible) to maintain an event per parameter (EPP) ratio of at least 20:1. We also did not utilise any formalised process for candidate variable pre-selection for example, backwards stepwise selection, beyond our DAG. All DAG identified variables were introduced into our model.
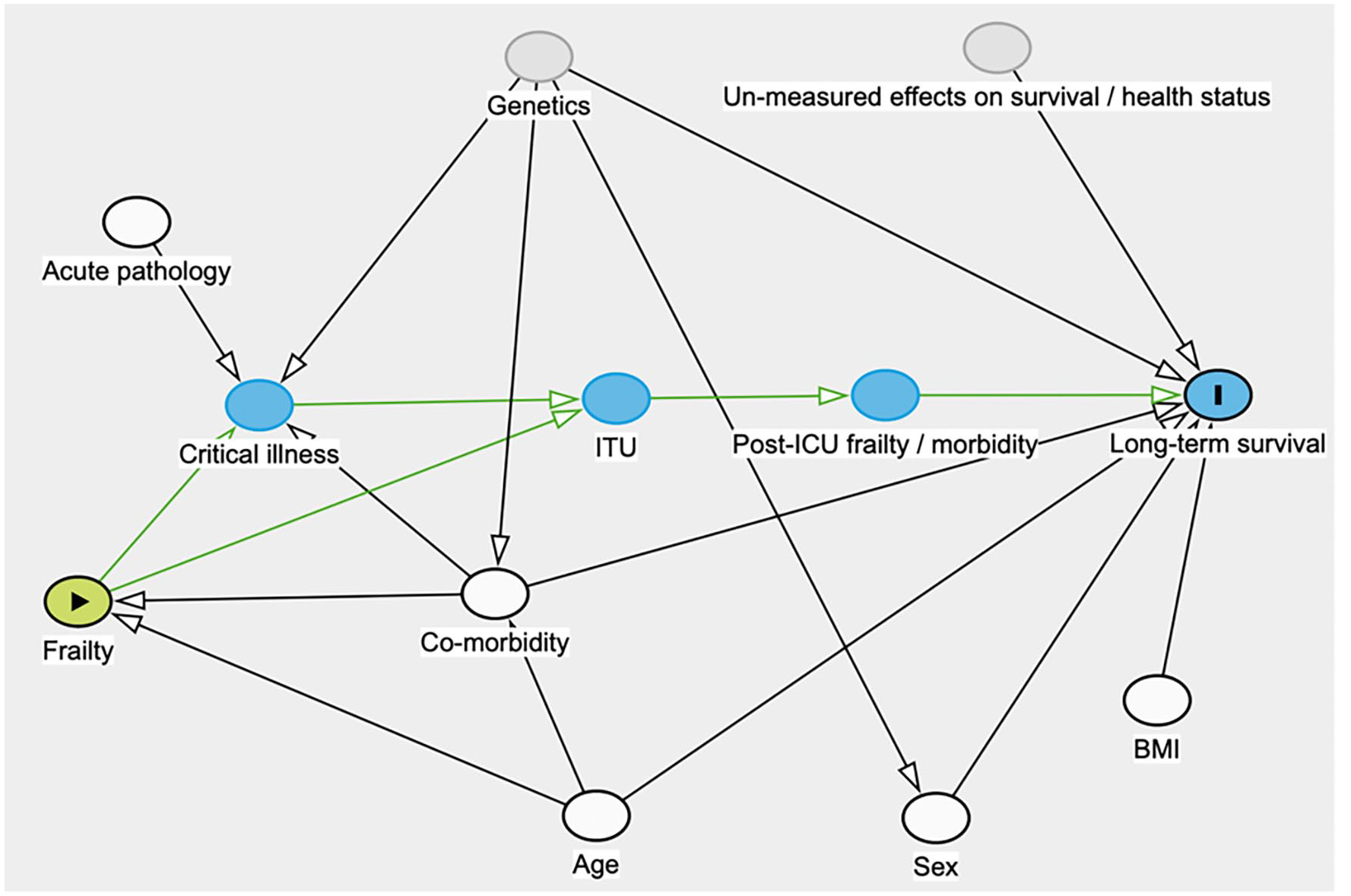
Direct acyclic graph (DAG) depicting the association between frailty and time to event (time to death). White nodules are co-variates included in the model. Adjustment for these enables estimation of the total effect of frailty on time to event. Blue nodules represent co-variates that, if adjusted for, will block the causal pathway. This then produces a spurious effect size estimate. Grey nodules represent un-measured co-variates. Arrows are directional and describe a ‘cause and effect’ relationship between co-variates. Within the blue ‘ITU’ nodule it is assumed that there are three confounding pathways relating to advanced respiratory, cardiovascular and renal support. We used the ICNARC score as a measure of acute illness/pathology.
AFT model distribution was selected according to best fit determined by Akaike information criterion (AIC) and simplicity. Continuous variables were assessed for non-linearity and if significant, penalised splines with three knots were used to adjust. Time-variation of co-variates was assessed using Schoenfeld Residual plots. Data was clustered on site of admission. Model assumptions were tested using visual inspection of log-log plots.
We reported number, percentage and median values with interquartile ranges for descriptive data, median time to event in days with 95% confidence intervals for Kaplan-Meier estimates of survival functions, Pearson’s chi-squared or Fisher’s exact test for comparison of categorical data, Kruskal-Wallis rank sum test for non-parametric continuous data, log-rank test for univariate analysis of Kaplan-Meier survival estimations (include pair-wise comparisons with no correction for multiple comparisons made) and Hazard Ratios (HR) with 95% confidence intervals for multivariate model co-variate effect size estimates. A p-value of <0.05 was taken as significant to refute the null hypothesis that frailty did not impact long term survival. All statistical analyses were undertaken using Rstudio version 1.4.1106.
Ethical approval
Formal Research Ethical Committee approval was waived for this study following Healthcare Research Authority submission via the Integrated Research Application System. It involved only secondary use of routinely collected data. All researchers involved in the statistical analysis remained independent from the local clinical research teams.
Results
A total of 1888 patients were eligible for random selection. Five hundred and sixty patients were selected for the final analysis (see Figure 2; consort diagram). There were 310 events in total during the follow-up period. Baseline demographic data is summarised in Table 1.
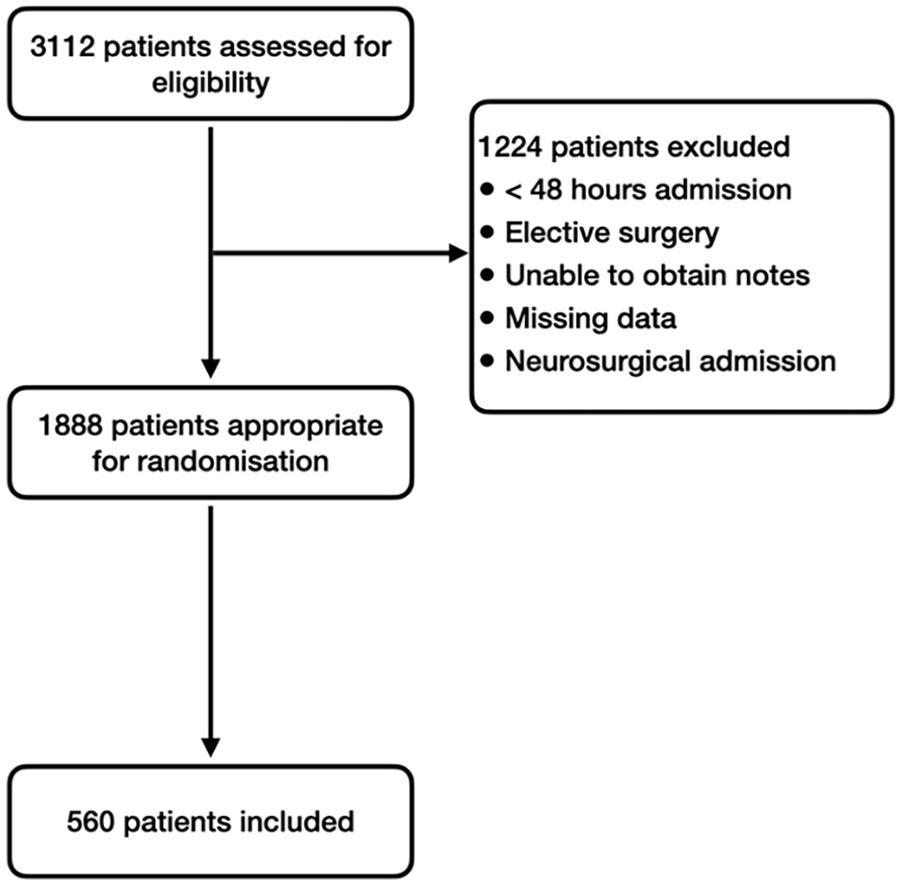
Consort diagram.
Basic demographics of whole cohort and frailty sub-groups.
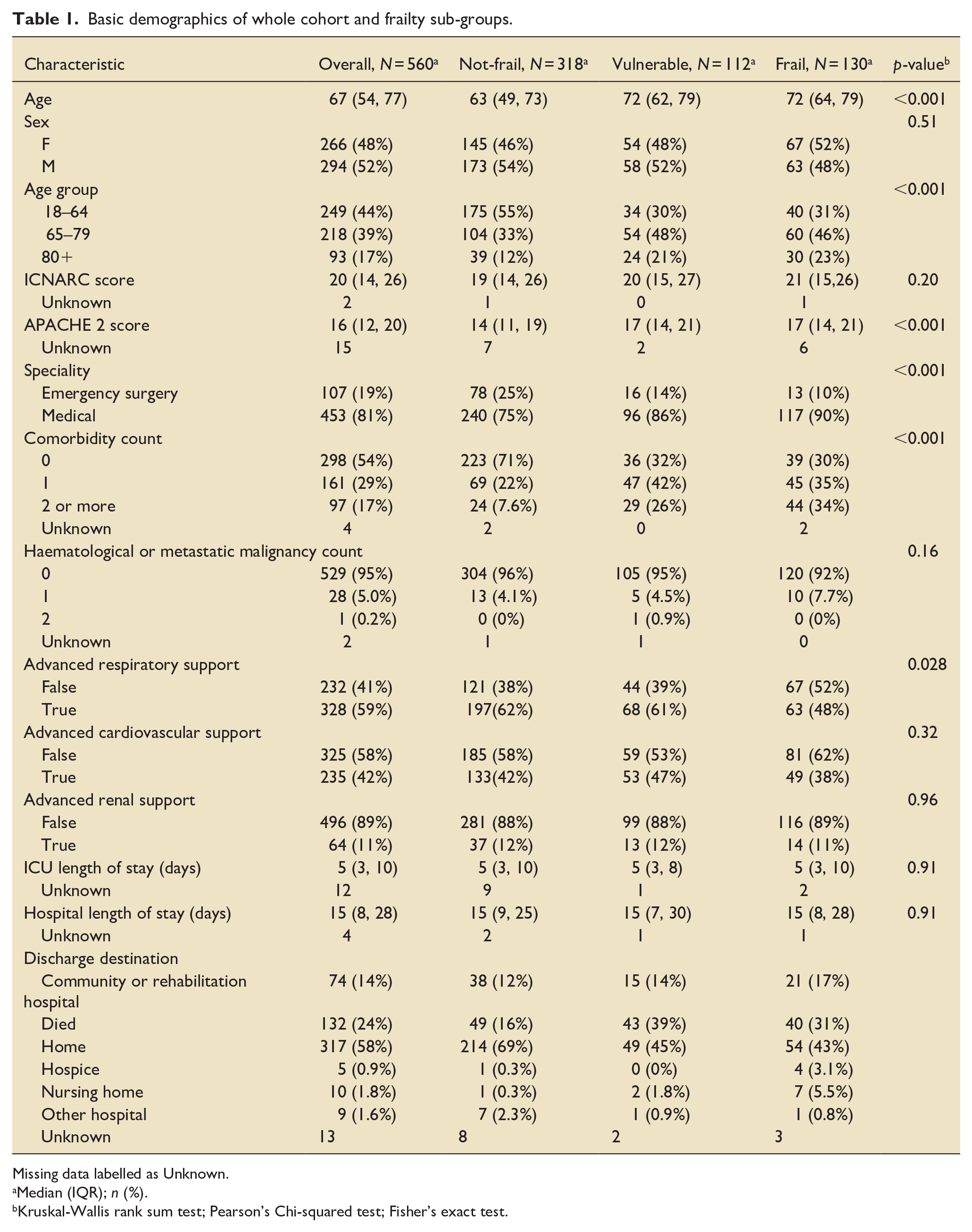
Missing data labelled as Unknown.
Median (IQR); n (%).
Kruskal-Wallis rank sum test; Pearson’s Chi-squared test; Fisher’s exact test.
The median age was 67 (interquartile range 54–77). Two hundred and sixty-six patients were female (48%). The median BMI was 27 (23, 32). Four hundred and fifty-three patients were admitted with a medical diagnosis (81%) whilst 107 patients were admitted following emergency gastro-intestinal or vascular surgery (19%). The median APACHE 2 and ICNARC scores were 15 (11, 20) and 20 (14, 26). Three hundred and eighteen patients were not-frail (56.8%, estimated CFS between 1 and 3), whilst 112 were vulnerable (20%, estimated CFS of 4) and 130 patients were frail (23.2%, estimated CFS between 5 and 7). The median ages (IQR) of these three groups were 63 (49, 73), 72 (62, 79) and 72 years old (64, 79), whilst the proportions of patient over the age of 80 in these three frailty sub-groups were 12%, 21% and 23% respectively.
The ICNARC scores for the three sub-groups were 19, 20 and 21 whilst the proportion of patients receiving advanced respiratory support were 62%, 61% and 48% and the proportion of patients receiving advanced cardiovascular support were 42%, 47% and 38%.
There were 249 patients in the ‘18 to 64’ age sub-group (44.4%), 218 patients in the ‘65 to 79’ age sub-group (38.9%) and 93 patients in the ‘80 and over’ age-subgroup (16.6%). Three hundred and twenty-eight (59%) patients received advanced respiratory support; 235 (42%) patients received advanced cardiovascular support and 64 (11%) patients received renal support. Four hundred and twenty-eight patients were discharged from hospital alive (76%). The median length of hospital stay was 15 days (8, 28).
Table 2 summarises median, one-year and five-year for the whole cohort as well as both the frailty and age sub-groups. The median survival for the whole cohort was 2074 days (5.7 years, Figure 3). Median survival estimates for the three pre-defined frailty groups were not reached, 332 and 594 days (Figure 4). Although the median survival for vulnerable patients was shorter than for frail patients, this was not statistically significant (p = 0.67). Median survival estimates for the three pre-defined age groups were not reached, 1664 and 702 days (Figure 5). Log-rank p values for all other frailty and age sub-group category comparisons were < 0.01 (not corrected for multiple comparisons). Figure S1 shows Kaplan-Meier estimates for each CFS separately (rather than grouped).
One-year, 5-year and median survival (95% confidence intervals) for whole cohort and frailty / age sub-groups (% and days).
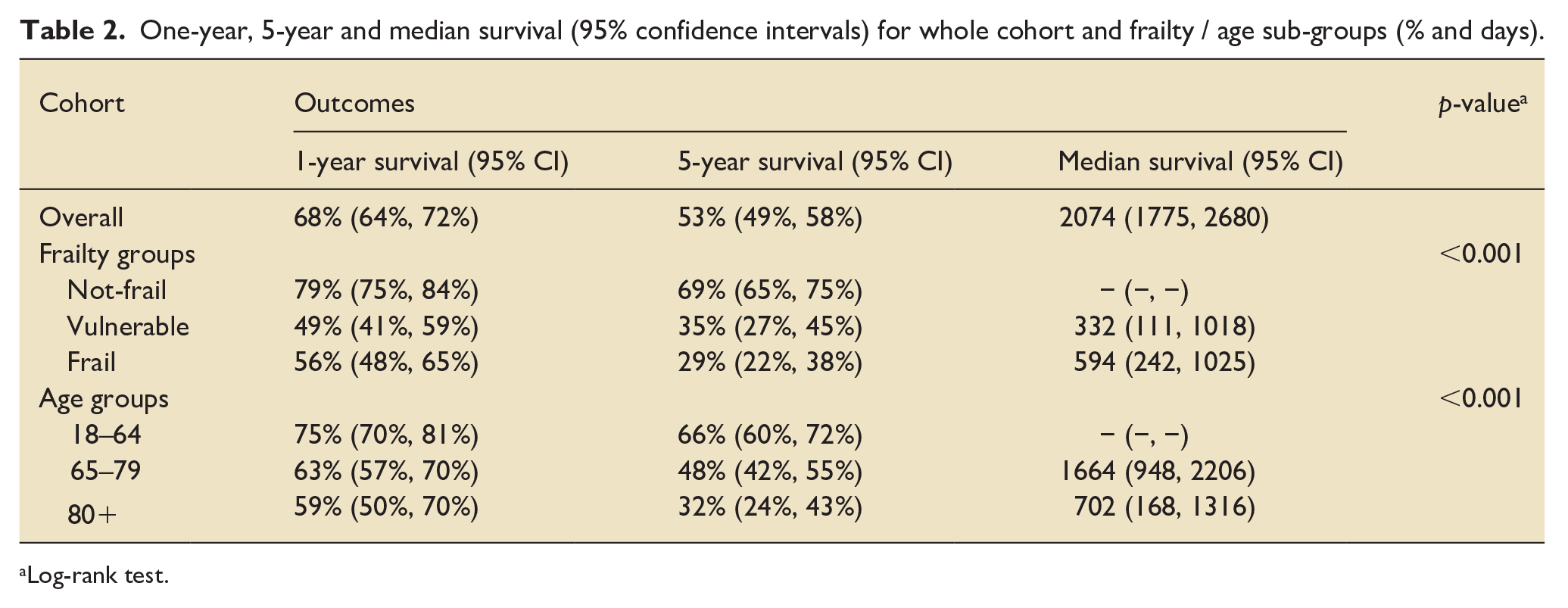
Log-rank test.
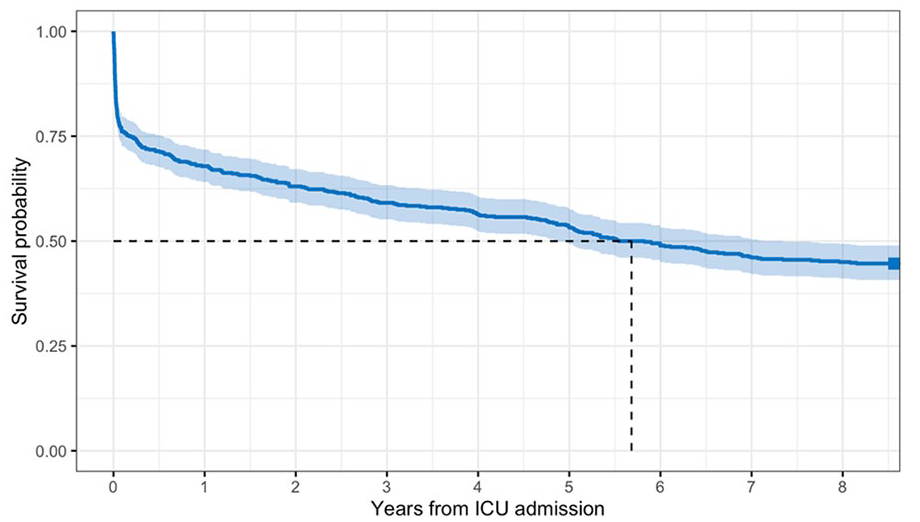
Kaplan-Meier curve for whole cohort.
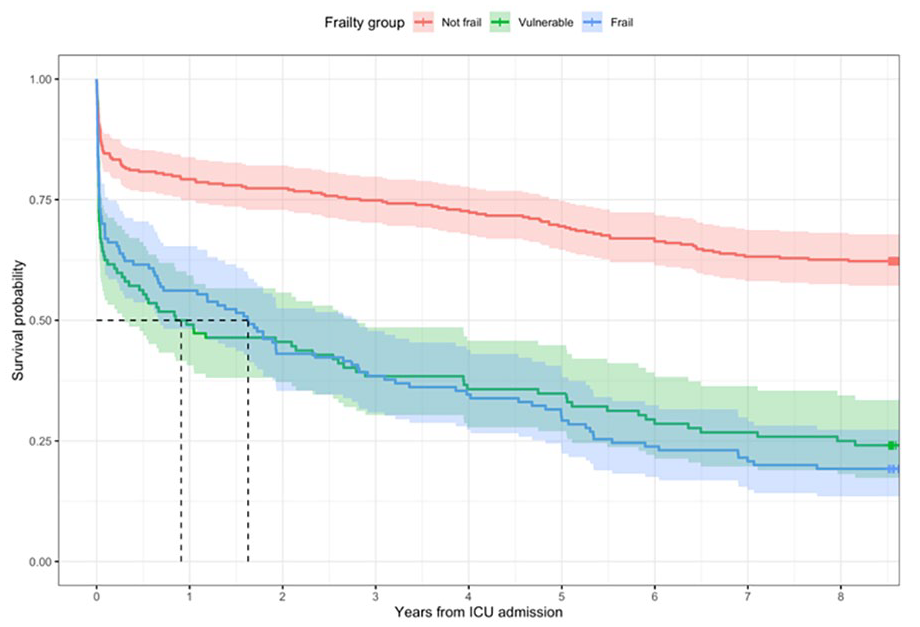
Kaplan-Meier curve for three frailty sub-groups.
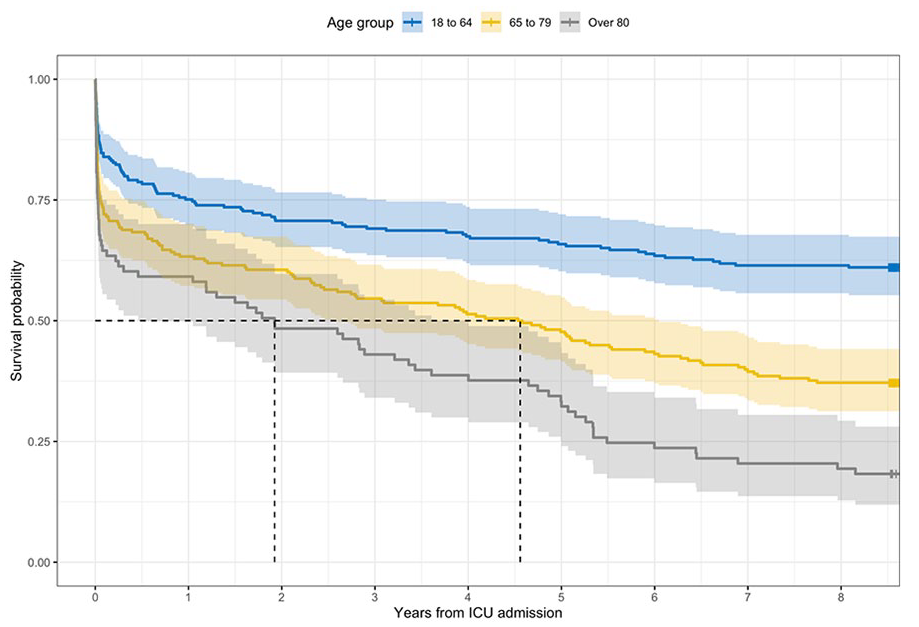
Kaplan-Meier curve for age group: 18 to 64; 65 to 69 and over 80 years.
There was no statistically significant difference in median survival for those patients who received either advanced respiratory, advanced cardiovascular or advanced renal support versus those who did not (see Figure S2).
Figure 6 shows the survival of patients discharged alive from hospital versus the expected life-expectancy of an age and sex-matched UK general population (n = 428), as well as the same for each of the age sub-groups (n = 208, 159 and 61 respectively). The whole cohort had a significantly reduced survival versus the age and sex-matched UK population with a relative one- and five-year survival of 0.97 (0.96, 0.98) and 0.86 (0.81, 0.89) respectively (see Figure S3). This held true for the ‘18 to 64’ and ‘65 to 79’ age sub-groups, with their one- and 5-year relative survival estimations being 0.97 versus 0.95 and 0.85 versus 0.79. However, the ‘80 and over’ sub-group had a survival that matched the UK population (one- and five-year relative survival of 1.0).
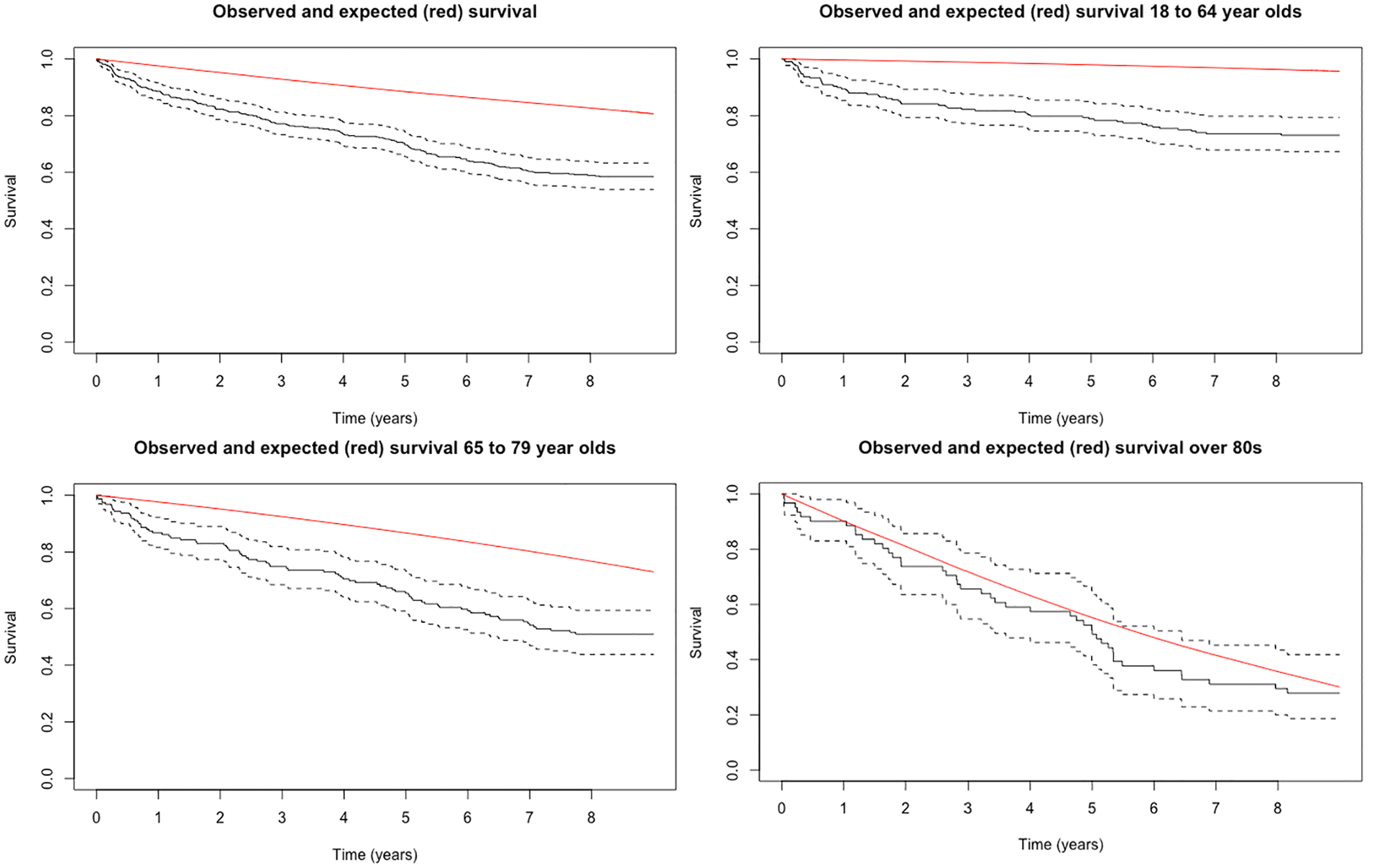
Observed versus expected survival for cohort of patients discharged alive from hospital (a), as well as for the 3 age sub-groups (b-d).
The results of our primary, multivariate, accelerated failure time (AFT) parametric model can be seen in Table 3. Ninety-eight pieces of data were found to be missing overall and were therefore manufactured using multiple imputation by chained equations prior to building of our model (missing data was predominantly baseline creatinine, height and weight). The final model included the co-variates pre-admission CFS group, age (as a categorial variable), sex, BMI, ICNARC score (as measure of acute pathology), baseline eGFR, major comorbidity count (one point for each of ischaemic heart disease, cerebral-vascular disease, significant respiratory disease, heart failure and diabetes mellitus), diagnosis of either metastatic or haematological malignancy (one point for each), advanced respiratory, cardiovascular and renal support, whilst it clustered the data upon site of data collection. This analysis showed that there were similar, statistically significant effect size estimates for both ‘vulnerable’ and ‘frail’ patients, with adjusted HRs of 2.6 (1.7, 4.0) and 2.4 (1.8, 3.3) respectively. These effect size estimates were markedly different (lower) than the univariate estimates, these being estimated as 3.1 and 3.2 respectively.
Effect size estimates from univariate analyses and AFT multi-variable model.
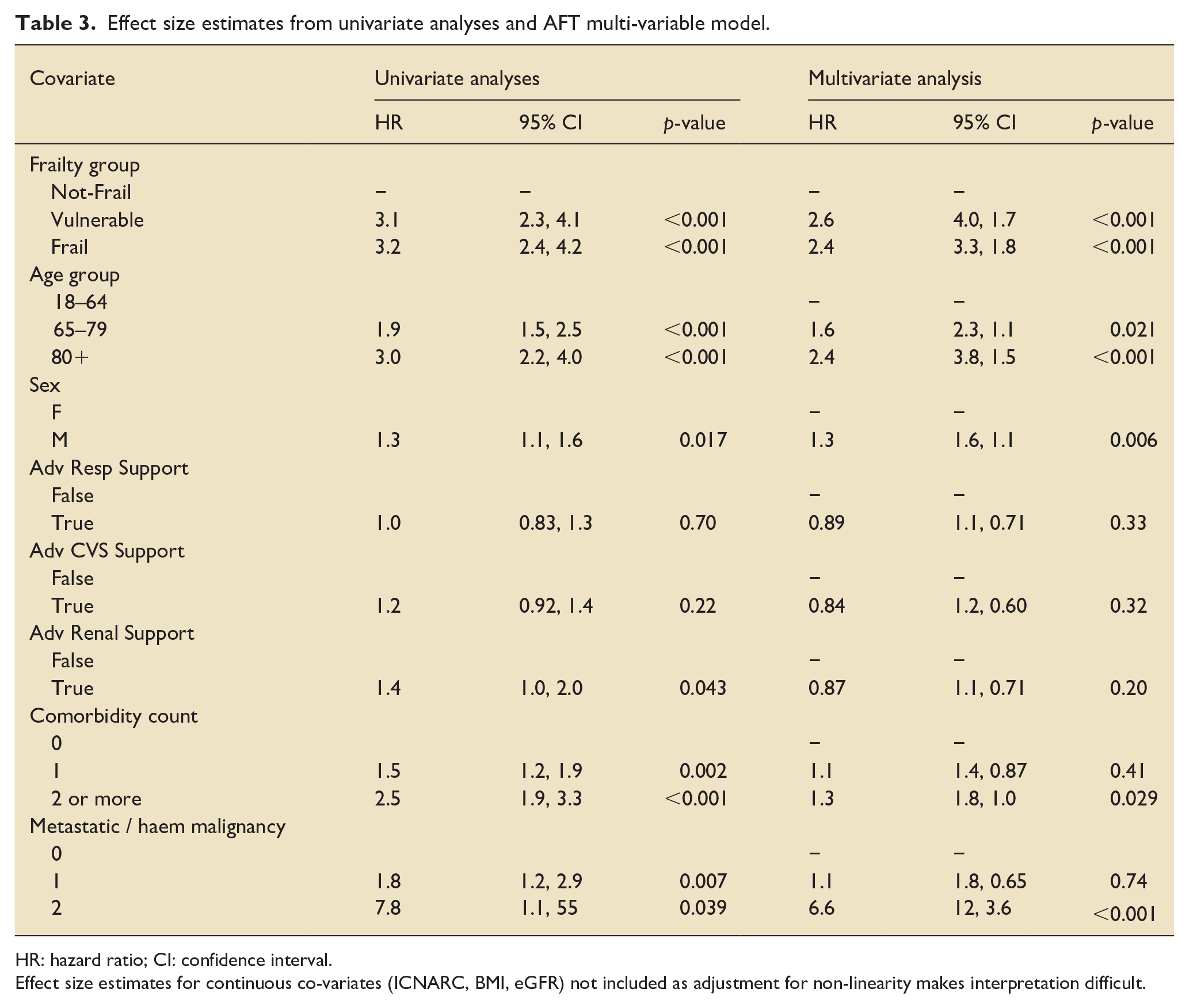
HR: hazard ratio; CI: confidence interval.
Effect size estimates for continuous co-variates (ICNARC, BMI, eGFR) not included as adjustment for non-linearity makes interpretation difficult.
Discussion
We have shown that there is a strong association between pre-admission CFS and life-expectancy for patients admitted to the ICUs across our region. This relationship was as strong for patients estimated to be ‘vulnerable’ (with a Rockwood CFS of four) as it was for those estimated to be ‘frail’ (CFS five to seven). The median unadjusted survival for these two sub-groups was short, estimated as 332 and 594 days respectively by the Kaplan-Meier method (no statistical difference), whilst 5-year survival was estimated as 35% and 29% (vs 69% for not-frail patients). Our multi-variable model provided further evidence of this association, whilst also adjusting for multiple confounders such as age, with the hazard ratios for the effect of vulnerable and frail status on outcome being estimated as 2.6 and 2.4. As a comparison, patients aged 80 years or older had a slightly longer unadjusted median survival time of 702 days (32% frail).
We have also showed that longer-term survival in patients discharged from ICU is significantly shorter when compared against the general UK population (matched for age and sex). This finding appears limited to patients aged less than 80 years old. This may suggest that patients who survive ICU admission do so with significant morbidity that alters life-expectancy.
This is the first study to our knowledge that has shown a relationship between ICU admission, frailty and long-term post-ICU survivorship in the UK. Our one-year survival estimates are perhaps more positive than those recently published for UK cohorts. 21 FRAIL-FIT estimated one-year survival to be 68.5% in non-frail patients and 46.8% in frail patients (in comparison to ours of 79% and 56%), although this study incorporated vulnerable patients into the non-frail cohort, perhaps negatively impacting their survival estimate, whilst the median ages were lower and the APACHE scores were higher. 21 An alternative recent non-UK estimate of one-year survival for patient admitted to ICU was 59%, in comparison to ours of 68%. 22 This study concluded that increasing age, as well as cumulative co-morbidity count, reduces 1 year survival in patients admitted to the ICU, with an estimated odds of death increasing to 2.91 (1.72, 4.9) for patients over the age of 80. Our analysis confirms that age is an important cofounder that needs to be adjusted for when inferring frailty’s effect on long-term outcome.
We have reported a similar 5-year survival to that previously published for a UK population. 30 A large (7656 patients), 2005 cohort analysis on all adult patients admitted to Scottish ICU’s primarily looked at mortality and long-term expense to health care. They showed an improved 1-year mortality of 10.9% (95% CI, 10–11.7) and 5-year mortality of 32.3% in comparison to our estimates of 32% and 47%. The discrepancy in results between our studies may reflect our inclusion criteria of ICU length of stay greater than 48 hours and the exclusion of elective surgery, in contrast to all admissions. Lone et al. also showed that ICU admission increased 5-year mortality compared to an age and sex-matched general population (32.3%vs 13.4%, HR not given). This shortening of survival is consistent with our results and supports the concept of a ‘post-ICU syndrome’ that results in significant impact on long-term health. We found a five-year relative survival of 0.86 for our cohort in comparison to an age and sex-matched general population. This detrimental effect of ICU admission on long-term survival was more marked for patients between 65 and 79, with a relative survival of 0.79. However, we did find that the relative survival for patients aged 80 or over was not reduced, estimated as 1.0. These results might suggest that well defined ICU admission practices for elderly patients with rapidly reversible pathology do not result in overly burdensome impacts on long-term health. On the other hand, it is also clear that this age group does have a short life-expectancy (median of 702 days, one-year survival 59%, five-year survival 32%) and this is important to appreciate in the context of intervention decision-making.
Increasing numbers of recent studies have shown the significant impact of frailty on short-term outcome, with odds ratio estimates for in-hospital mortality as high as 7.83, 9 hazard ratio estimates for 30-day mortality of 2.12 8 and odds ratio for 30-day mortality increasing by 1.44 for each one-point increase in CFS. 10 We have attempted to show a causal association between frailty and longer-term survival by constructing a DAG to identify relevant confounding pathways that need to be adjusted for in a multi-variable model. Our results estimate an adjusted hazard ratio of 2.6 for ‘frail’ patients versus not-frail patients and 2.4 for ‘vulnerable’ patients versus not-frail. This may suggest that vulnerable patients should be more accurately dichotomised as frail rather than not-frail, counter to current common practice.
Limitations
Our study has limitations that need consideration. Firstly, the data has retrospectively been sourced from 2012. Although this has enabled beneficial long-term follow-up of patients, decisions regarding treatment escalation and admission criteria to the intensive care unit may have changed. Therefore, it may not be appropriate to generalise our results to current ICU admissions.
Our inclusion criteria selected medical and emergency gastro-intestinal / vascular admissions. We did not select neurosurgical, cardiac, trauma or orthopaedic patients (e.g.). Our results cannot therefore be generalised to different surgical populations or elective patients, who may have different burdens of critical illness or different characteristics relevant to longer-term survival.
The apparent equity of impact on long-term survival between vulnerable and frail sub-groups may be an artificial finding induced by selection error or allocation bias. Frail patients may have been admitted due to a confident, optimistic view of prognosis or a perception that the acute illness would have a short-lived impact on health and function (i.e. a low-acuity pathology i.e. readily reversible with a rapid recovery). Vulnerable patients may have been admitted to ICU despite a prognosis that was more pessimistic or a pathology with greater physiological impact, perhaps due to a perception of greater potential gain in life-expectancy. It is possible that this bias by allocation may have artificially improved the estimated survival for frail patients. Patient selection may also be a feature of local practice and population health. This group may therefore not represent other selected frail or vulnerable populations around the UK, making our findings less generalisable to other geographical areas.
A degree of measurement error may well have introduced inaccuracy into the CFS data captured and our effect estimates. All CFSs were estimated retrospectively from notes, with different teams of researchers working at different locations. Inter-observer variability may well be an issue, although there is evidence that the retrospective and prospective assessment of frailty produces similar estimations and doesn’t impact significantly on outcome effect sizes9,31
The results from our study are not evidence that frailty can be used as a tool for prediction that is, our frailty effect size estimates are not measures of how predictive frailty is of outcome. Our results merely describe a strong relationship between frailty and life-expectancy in those patients admitted to ICU. This should be seen as an association rather than of predictive significance.
Simpson’s paradox – we have conditioned on both hospital admission and ICU admission. We cannot quantify the effect size of pre-ICU frailty on long-term survival for either the general population or the ward-based population. The magnitude of the correlation we identified may well be significantly different in these larger populations.
Our Direct Acyclic Graph may be flawed or inaccurate. We have adjusted our model to remove confounding and therefore produce an un-biased estimate of the association between frailty and life expectancy. There may well be other biasing pathways that we have not depicted and adjusted for, meaning that our estimates remain biased or imprecise. It is also clear from our DAG that there are known co-variates that simply remain un-measured. As we are measuring survival over a long follow-up period, some of the most important determinants remain features of chronic health. We have not measured accumulation of new comorbidities and assessments of frailty or function after ICU admission, rather we have used pre-ICU frailty and co-morbidity as a surrogate of these, limiting any inference. Finally, our attempts at adjustment for known cofounders for example, acute pathology, may be incomplete. In our case, we have used ICNARC score as a surrogate for this co-variate, which may be inappropriate or provide only partial adjustment.
Conclusions
There is significant evidence to suggest a strong association between frailty and survival after ICU admission. Vulnerable status may well be associated with a similar effect on longer-term survival to that of frailty. It may be inappropriate to consider these patients as not-frail. Life-expectancy after ICU admission also appears shortened in comparison to that expected for the general population. Future work must focus on how frailty scores may be used for prognostication and thereby better inform complex decision-making regarding ICU admission. This is likely to require the validation of frailty scoring systems as a predictor variable. We must also investigate the effect of frailty on longer-term functional recovery outcomes, rather than just mortality.
Footnotes
Acknowledgements
We wish to acknowledge the contributions of the following individuals: Dr Ash Williams, Dr Emma Fisher, Dr Tom Woodward, Dr George Graham, Dr Ferenc Los, Dr Aaron Sutton, Dr Andrew McCardle, Dr Ben Dingle, Dr Briony Adams, Dr Carla O’Hagan, Dr Catherine Archer, Dr David Stewart, Dr Emma Bartlett, Dr Emma Endean, Dr Emma Fisher, Dr Emma-Tina Segall, Dr Jennifer Ridenton, Dr Kate Smurthwaite, Dr Katherine Fok, Dr Katie Adams, Dr Kerry Hunter, Dr Laura Markham, Dr Lorna Abraham, Dr Patrick Van Den Burg, Dr Ria Smith, Dr Jonathon Carter, Dr Toby Lewis, Dr Tor Tuckey, Dr Zahra Essackjee, James Gladwin, Robert Hutton, Dr Agnieszka Kubisz-pudelko, critical care research nurse Sarah Bean and Consultant Anaesthatist Rob Price.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
