Abstract
Introduction:
Mechanical ventilation (MV) is a common and often live-saving intervention on the Intensive Care Unit (ICU). The optimisation of sedation to mechanical ventilation is fundamental, and inappropriate sedation has been associated with worse outcomes. This scoping review has been designed to answer the question ‘
Methods:
The protocol was designed using the PRISMA-ScR checklist and the JBI manual for evidence synthesis. Data were extracted and reviewed by a minimum of two reviewers.
Results:
Searches of electronic databases (PubMed, OVID, Scopus, Web of Science) produced 726 results; 45 citations were identified for further eligibility assessment, an additional five studies were identified through keyword searches, and 12 through searching reference lists. Of these 62 studies, 27 studies were included in the final review: 6 case reports/case series, 11 retrospective cohort/observational studies, 1 prospective cohort study, 9 prospective randomised studies.
Conclusion:
We found a lack of high-quality well-designed studies investigating the use of continuous ketamine sedation on ICU. The available data suggests this intervention is safe and well tolerated, however this is of very low certainty given the poor quality of evidence.
Background
Mechanical ventilation (MV) is a common intensive care intervention (approximately 20 million patients per annum 1 worldwide) that accounts for much intensive care unit (ICU) resource utilisation. Despite efforts, patients requiring MV unfortunately still experience high mortality and morbidity. 2
Most critically ill patients requiring MV are medically sedated with optimisation of sedation and analgesia being fundamental to management. International guidance has been aimed at improving outcomes 3 owing to reported associations between deep sedation and negative prognostic markers, 4 however these results have not been consistently demonstrated 5 and agent selection is a balance of risks and benefits. Providing adequate sedation, maintaining comfort, reducing pain, and minimising agitation and delirium have been identified as top priorities for ICU research by the intensive care community through the James Lind Alliance. 6
In a survey of ICUs in the UK, propofol combined with either alfentanil or fentanyl was the most common sedation-analgesia regime; 92.2% of units reported propofol as first line agent. 7 A third of units reported using other non-ketamine sedative agents either ‘frequently’ or ‘very frequently’, including benzodiapines (29.4%), clonidine (35.3%), and dexmedetomidine (11.8%).
Most traditional sedatives (including propofol, benzodiazepines, and alpha-2-agonists) are associated with multiple significant and potentially problematic adverse effects, (commonly hypotension, bradycardia, and prolonged mechanical ventilation ). The predominant mechanism is attenuation of external stimulation, reduced sympathetic tone, and vasodilation. 8
Sedatives, in particular benzodiazepines significantly increase the risk of delirium in ICU.3,9 Delirium during critical illness is associated with higher mortality, longer ICU stay, and poorer long-term outcomes. 10
Ketamine is an N-methyl D-aspartic acid (NMDA) receptor antagonist that has been used since the 1970s to provide cataleptic, amnesic, analgesic, and dose dependant anaesthetic effects. 11
Ketamine has been particularly successful in military 12 and pre-hospital 13 settings owing to its ability to stimulate the sympathetic nervous system, preserving heart rate and blood pressure, whist avoiding respiratory suppression, 14 as a result ketamine has become increasingly popular as an anaesthetic agent for emergency surgical procedures in hypotensive patients. 15
Although ketamine has been in clinical practice for approximately 50 years; ketamine by continuous infusion remains an unlicensed and rarely used sedative to facilitate MV. 7 Ketamine infusions have however been used successfully in mechanically ventilated patients on ICU in the context of severe asthma, 16 utilising ketamine’s bronchodilatory properties.
Reluctance around more routine use of ketamine is likely multifactorial and may relate to lack of clinical familiarity (as demonstrated in two surveys of UK sedation practices7,17), perceived contraindications such as raised intracranial pressure (ICP), and possible side effects such as ‘
Emergence reactions are psychomotor symptoms experienced by some patients when waking from ketamine anaesthesia. The reported incidence of these reactions varies from 5% to 30%. 18
Milder manifestations such as floating sensations, disorientation, excitement, dysphoria, vivid dreams, and amnesia, through to more marked reactions including frank delirium, hallucinations, and agitation18,19 are well documented in the context of general anaesthesia. To our knowledge these have not been examined in the context of the critically ill, where neuropsychiatric dysfunction and risk-benefit ratio differ markedly from those in general anaesthesia.
Ketamine was recently licenced for major depressive disorder (MDD) treatment following reviews that highlighted transient psychotomimetic effects with single-dose administration.20,21
MDD and post-traumatic stress disorder (PTSD) are common amongst the survivors of ICU, with the prevalence of MDD ranging in the literature from 17% to 43%22,23 and PTSD ranging from 21% to 35%.22,24 Associations between perceived risk factors and PTSD have been extensively examined with varied results. 23 Duration of ICU delirium, early post-ICU depressive symptoms, the use of benzodiazepines, and physical and cognitive impairment during recovery and rehabilitation have all been identified as significant risks for developing MDD.24 –26
To our knowledge there is currently no evidence suggesting administration of ketamine for the purpose of sedation during mechanical ventilation can influence longer-term psychological outcomes. This is of interest in this review given the prevalence of MDD and PTSD and could represent an area for potential future research including clinical trials specifically investigating ketamine and critical care recovery and rehabilitation.
As part of this scoping review we aim to establish what literature exists regarding the use of continuous ketamine sedation on ICU, particularly in relation to cardiovascular effects, delirium and the psychological aspects of ICU.
Methods
Aim
The aim of this scoping review is to review current literature in order to answer the following research question: ‘
Objectives
To search relevant databases to identify peer-reviewed literature investigating ketamine’s efficacy, safety, and clinical effects when used as a continuous infusion in MV patients on ICU.
Protocol
This study was designed using the Preferred Reporting Items for Systematic Reviews and Meta-analysis extension for Scoping Reviews (PRISMA-ScR) checklist 27 and the JBI Manual for Evidence Synthesis. 28
The protocol is available from Research Square (https://doi.org/10.21203/rs.3.rs-910472/v1).
Search strategy and terms
Full details of the search strategy employed can be found in the appendix (see Appendix B)
A three-step literature search was used in accordance with JBI methodology for scoping reviews 28 :
A preliminary search of PubMed was conducted using the terms: ‘(Ketamine) AND (Intensive Care) AND ((Sedation) OR (Analgesia))’.
Subsequent searches were conducted across all included databases (PubMed, Embase, OVID, Scopus, Web of Science) using words and phrases contained in the title, abstract, and index terms of papers retrieved during the preliminary search.
Reference list of identified reports and articles were analysed for additional sources and reports.
Eligibility criteria
Only full-text reports published in English in peer-reviewed literature were included.
Inclusion criteria:
- Adult patients (⩾18 years old)
- Use of continuous ketamine infusions for the purpose of primary or adjunctive analgesia or sedation during mechanical ventilation
Accepted methods included experimental and quasi-experimental designs (e.g. randomised controlled trials, non-randomised controlled trials, and interrupted time-series studies), analytical observational studies including prospective and retrospective cohort studies, case-control, case series, individual case reports and descriptive cross-sectional studies.
Systematic reviews and meta-analyses were not included, but their references were screened for appropriate studies that met the inclusion criteria. Conference abstracts, qualitative studies and opinion papers were not considered for inclusion in this scoping review.
Information sources
Searches were made across PubMed, Embase, OVID, Scopus, and Web of Science using the inclusion criteria above.
Screening of sources
Two reviewers (NDR and WW) independently screened titles and abstracts for inclusion. Full texts were obtained for relevant identified articles and citations were collated in EndNote X6.
Disagreements arising between reviewers at each stage of the selection process were resolved through discussion, without the need for an additional independent reviewer.
Data charting process
NDR and WW extracted data from all included publications using the data extraction tool developed by the reviewers (see Appendix A).
The data extracted included publication details, study design, setting, intervention details, primary and secondary objectives, and key findings.
Critical appraisal of sources
Sources were critically appraised using the relevant aspects of the CASP checklists (https://casp-uk.net/casp-tools-checklists/) by two independent reviewers (NDR and WW).
Synthesis of results
Results are presented with frequency counts of concepts, populations, and study characteristics, accompanied by a descriptive summary. Studies are grouped based on methodological design, along with outcome measures, and relevance to this review.
Results
Selection of sources of evidence
Searches of electronic databases produced 726 results. After duplicate removal and initial screening of titles and abstracts, 45 potentially relevant citations were identified for full-text retrieval and further eligibility assessment. Common reasons for exclusion at this stage were review publications, incorrect patient population (paediatric or intra-operative), incorrect intervention (non-continuous ketamine), and non-human studies.
Five studies were identified from keyword searches and searching reference lists of the full-text and review articles identified 12 further studies for eligibility screening.
Of the 62 studies undergoing eligibility screening, 35 were excluded: 10 were judged not to be original quantitative research (e.g. review articles, commentaries), 10 were unavailable in English or full-text form, and 15 either did not include the study population of interest or outcomes of interest (paediatric populations, intra-operative populations, participants not intubated and ventilated) (Figure 1). The remaining 27 studies are included in this review and are reported in Table 1.
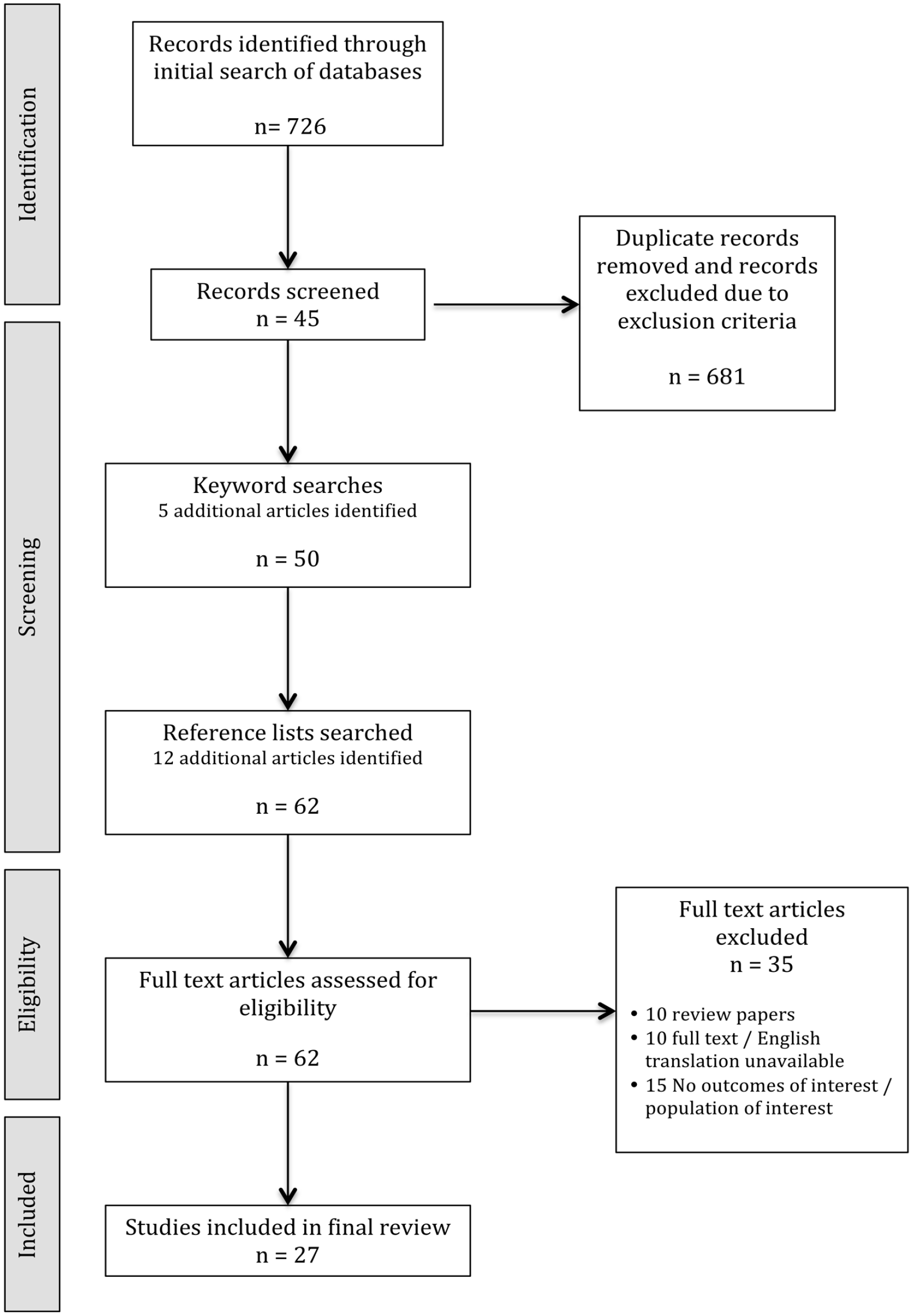
Searches, inclusion, and exclusion flow diagram.
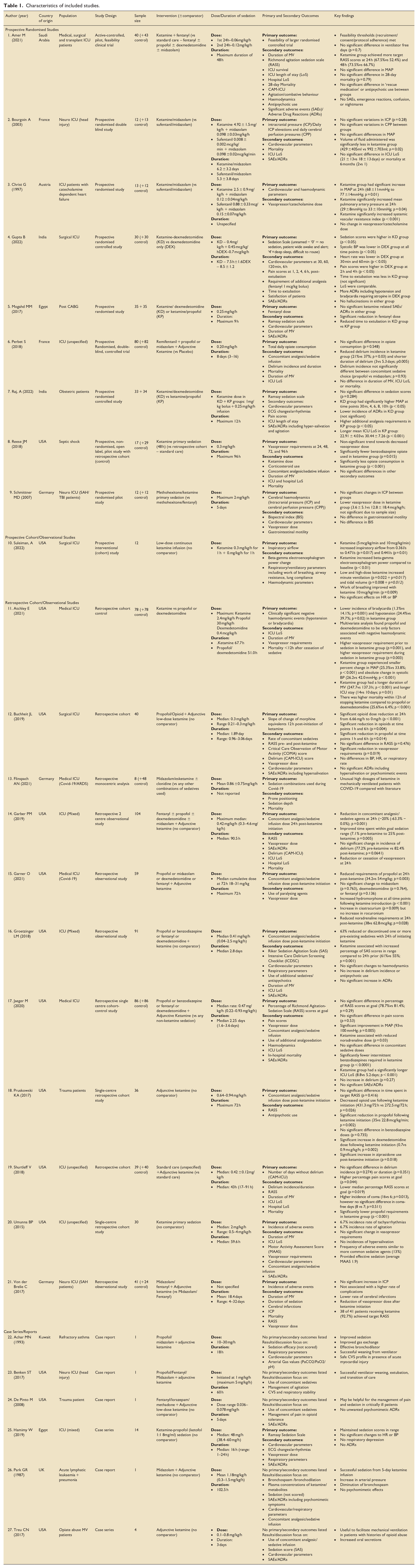
Characteristics of included studies.
Critical appraisal
Given the range of employed methodologies and apparent quality of evidence, sources were critically apprised using the relevant aspects of the CASP checklists (https://casp-uk.net/casp-tools-checklists/), which can be found in Table 2 in the appendix.
Critical appraisal of included studies.
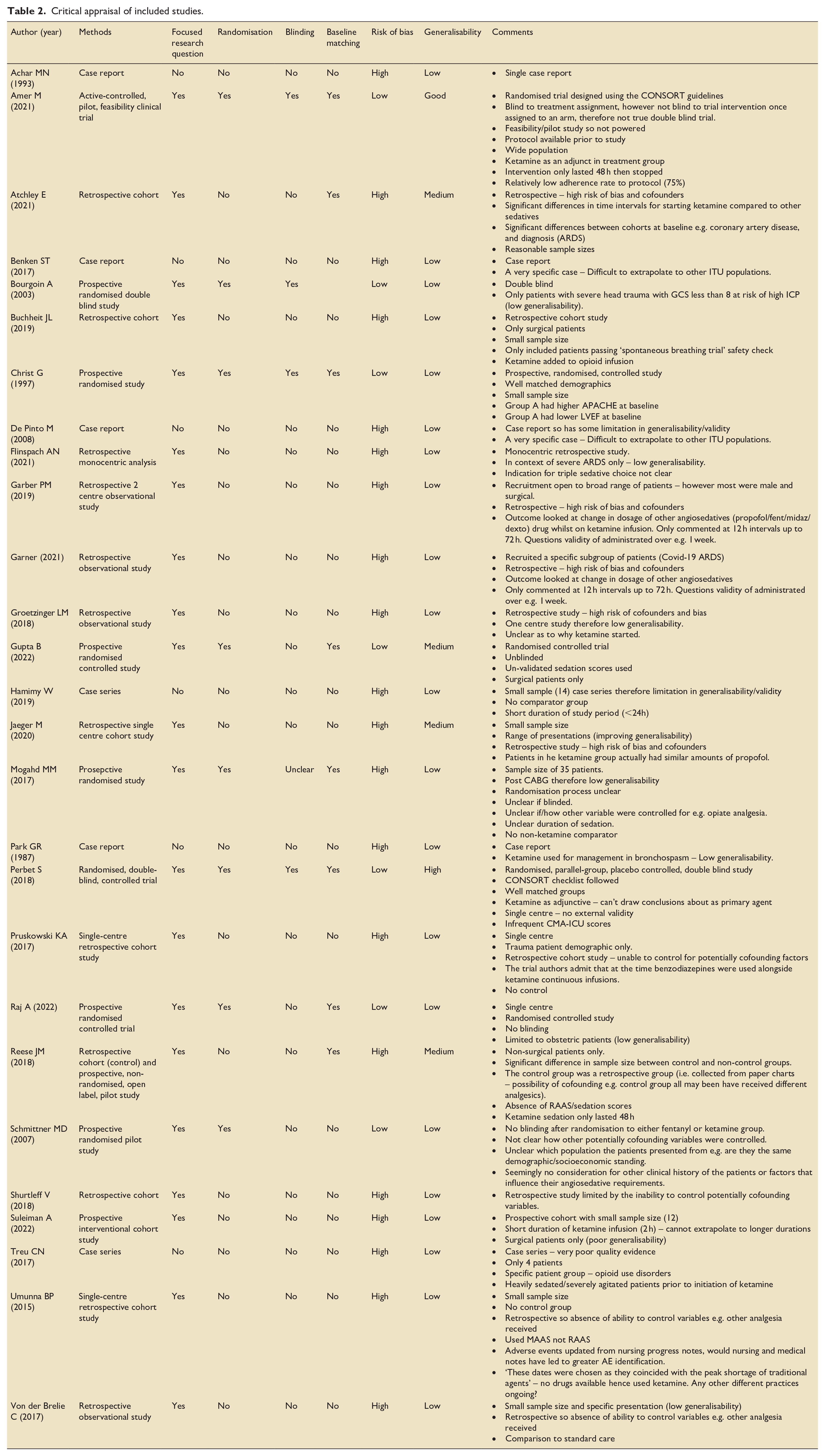
Risk of bias was considered to be high in 20 of the 27 studies (74.1%) commonly due to lack of focused research question and unsatisfactory study designs (retrospective cohorts, lack of blinding, and lack of control groups).
Characteristics
The included studies, country of origin, aims, designs, measures and outcomes, as well as main findings can be found in Table 1.
Of the 27 articles relating to the use of continuous ketamine infusions for sedation or analgesia during MV, there were nine prospective randomised studies29 –37, one prospective cohort study, 38 , 11 retrospective cohort39 –44 or observational studies,45 –49 and six case reports or case series (Figure 2).50 –55
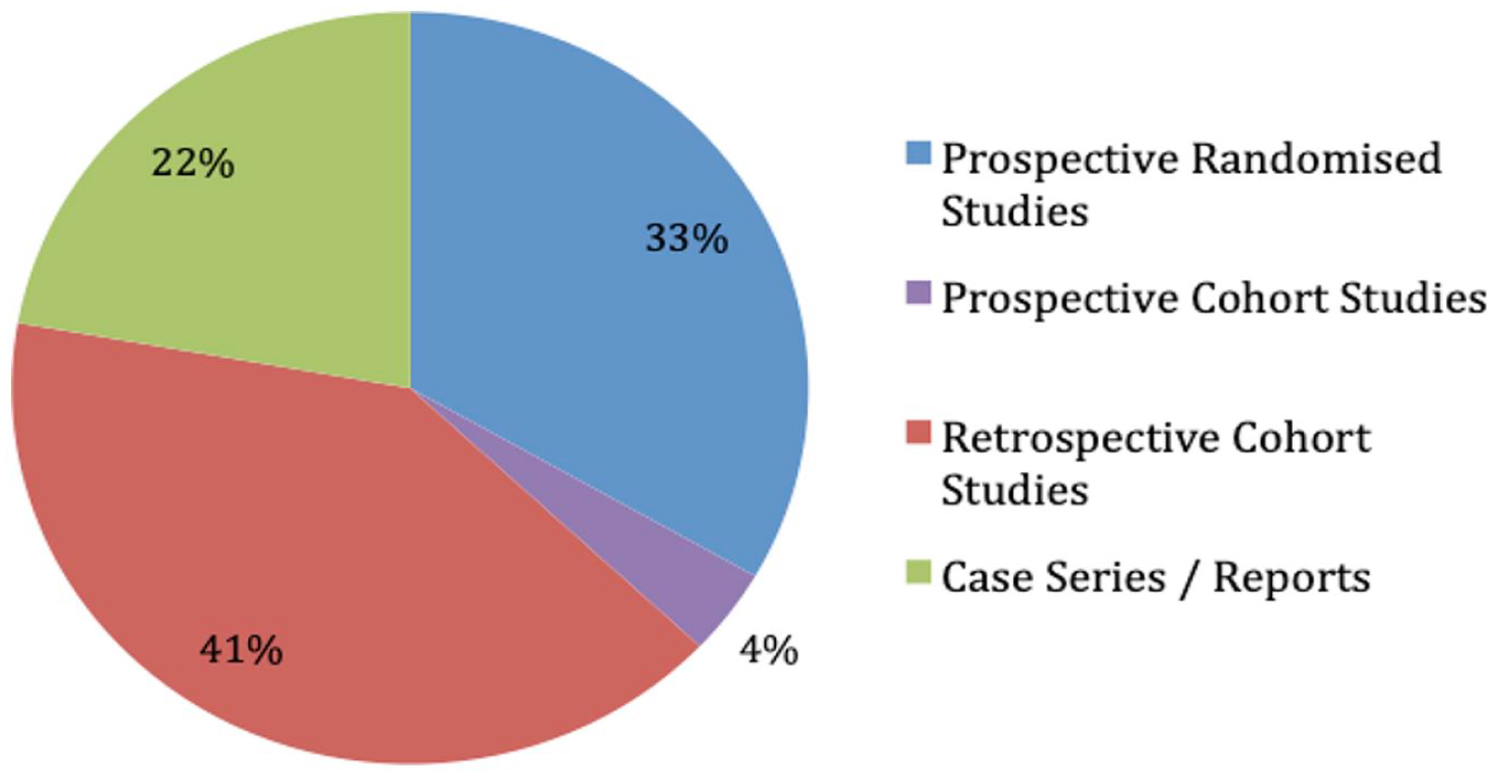
Designs of included studies (%).
A total of 987 participants received continuous ketamine infusions whilst undergoing MV. There were 497 controls or comparators. Sample sizes were generally small with 12 studies using a sample size of less than 30 participants per arm (range
Mechanically ventilated patients were included from the following ICUs: Medical ICU (40.7%),36,41,45,47,50,54,55 Mixed, general, or unspecified ICU (39.0%),29,34,39,43,44,46,48,53 Neurosurgical ICU (8.2%),30,37,49,51 Cardiac ICU (6.7%),31,33 Surgical ICU (2.8%),32,38,40 Obstetric ICU, 35 and Trauma ICU (2.6%).42,52
The most frequent country of origin was the USA (55.5%), other countries included France, Germany, India, and Saudi Arabia, with only one case report coming from the United Kingdom. 54 The majority of studies (77.9%) were published between 2017 and 2022.
Intervention and comparators
Of the 27 studies included, 11 (40.7%) investigated the use of ketamine as a primary sedative,30 –33,35 –37,39,41,44,53 the remainder used ketamine as an adjunct to pre-existing sedative or analgesic therapies. Eleven (40.7%) directly compared ketamine primary sedation or adjunctive ketamine to non-ketamine regimes.29 –31,33,36,37,39,41,43,45,49 One study compared the addition of ketamine to placebo. 34
Doses and durations
Ketamine was administered as a continuous infusion in all studies. Doses across included studies (Table 1) varied greatly with a minimum reported infusion dose of 0.036mg/kg/h, 52 and a maximum of 6.42 mg/kg/h. 30 The majority of studies used a dose range under 1 mg/kg/h with 12 authors reporting an average dose or dose range under 0.5 mg/kg/h. Seven studies used a range between 0.5 and 1 mg/kg/h, three used between 1 and 2 mg/kg/h, and four authors reported doses over 2 mg/kg/h. Only one author did not report the doses of ketamine. 49
Duration of continuous ketamine infusions also varied significantly across studies (Table 1), with some study designs stipulating a maximum duration, and others continuing until cessation of MV (or death of patient). The minimum reported duration was 2h 38 and the maximum was 36 days (864 h). 49 Six studies reported durations of infusions under 24 h, nine reported durations between 24 and 72 h, and ten studies reported durations over 72 h.
Reported outcomes and findings
The primary and secondary outcome measures, and significant findings relating to the review question are reported in Table 1 in the appendix.
The most commonly reported primary outcome was the effect of ketamine on dose or rate of other concomitant sedatives or analgesics (25.9%).33,34,40,42,46 –48 Other primary outcomes included sedation efficacy,32,35,41,53 safety of ketamine infusions,44,49 effects on neurological parameters e.g. intra-cerebral pressure,30,37 cardiovascular effects for example, blood pressure and vasopressor requirements,31,36,39,47 respiratory parameters for example, gas exchange and airway pressures,38,50 feasibility data, 29 and effect on delirium. 43 Commonly reported secondary outcomes included cardiovascular parameters, vasopressor requirements, ICU length of stay, ventilator free days/duration of MV, adverse events, and mortality. None of the studies reported long-term patient-centred outcomes such as quality of life nor the incidence, severity, or duration of depression or PTSD.
Concomitant infusions
Seven of 27 studies (25.9%) investigated the effect of initiating ketamine infusions on the rate of concomitant analgesic and sedatives,33,34,40,42,46 –48 this was reported as secondary outcome in a further 12 studies (44.4%).
Initiation of ketamine was associated with a decrease in propofol doses in six studies40,42,43,46
–48 and a decrease in alternative sedatives (e.g. dexmedetomidine) in two studies.46,48 Benzodiazepine requirements (intermittent or continuous) were reduced in two studies36,41, opioid requirements were also significantly lower in three studies.33,36,40 However, in some studies, there was no difference reported in the dose of concomitant sedatives
47
or opioid requirements.34,47 One study reported an increase in dexmedetomidine doses following initiation of ketamine
42
and another reported an increase in opioid requirements.
47
These inconsistencies were sometimes present even within studies. Pruskowski et al,
42
for example, found that ketamine infusions led to a significant reduction in propofol (35vs 22.8 mcg/kg/min;
Sedation efficacy
Only four studies (14.8%) investigated the efficacy of sedation as a primary outcome,32,35,41,53 however assessment of sedation using a validated scoring system (e.g. Richmond Agitation Sedation Scale (RASS) or Riker Sedation-Agitation Scale (SAS)) were reported as secondary outcomes in 11 additional studies (40.7%).
Three authors reported sedation scores when comparing a ketamine-based sedation to a non-ketamine sedation regimen. Amer et al
29
reported more target RASS scores at 24 h (67.5%vs 52.4%) and 48 h (73.5%vs 66.7%) when comparing ketamine and fentanyl to ‘standard care’. Gupta et al
32
found sedation scores were higher in the ketamine-dexmedetomidine group compared to dexmedetomidine alone (
Additional studies reported an improvement in the time study participants spent within the RASS target range (7.1% pre-ketamine to 25% post-ketamine;
Only one study, conducted by Shurtleff et al,
43
reported lower median percentage RASS scores at target with adjunctive ketamine compared to standard care alone (70%vs 84%;
Cardiovascular outcomes
Many investigators reported on the effect of ketamine on haemodynamics for example, mean arterial pressure (MAP), blood pressure (BP), or heart rate, as well as vasopressor requirements.
Several authors reported improved haemodynamics, including Christ et al
31
who demonstrated a significant increase in MAP at 24 h (68 ±11mmHg to 77 ±14mmHg;
These results were inconsistent, however, with some investigators such as Amer et al,
29
Groetzinger et al,
48
and Umunna et al
44
reporting no significant differences in vasopressor requirements or haemodynamics. Whilst Bourgoin et al
30
reported no significant difference in MAP, they found significantly less fluid was administered in the ketamine group (429 ± 405ml vs 992 ± 703ml;
Respiratory outcomes
Suleiman et al 38 demonstrated improved respiratory dynamics including inspiratory airflow, increased minute ventilation, and tidal volumes, as well as improved work of breathing. Achar et al 50 reported improved gas exchange and bronchodilation following the addition of ketamine for refractory asthma.
Neurological outcomes
Three authors investigated effects of ketamine on intracranial pressure. All found no significant increases, changes, or variations in ICP30,37,49 and Suleiman et al
38
reported Ketamine increased beta-gamma electroencephalogram power compared to baseline (
Delirium
Perbert et al
34
found a reduced incidence of delirium in their ketamine cohort (21%vs 37%;
Safety and adverse events
Over half of authors (51.9%) commented on the safety of ketamine; 85.7% of these authors reported no significant adverse reactions or events29,32,33,35,40,41,44,46,48,52 –54 and the absence of emergence or psychomimetic reactions was specifically reported in four studies.29,40,52,54
Treu et al
55
reported increased oral secretions, and Umunna et al
44
reported a 6.7% incidence of tachyarrhythmia but with an overall incidence of adverse events similar to more commonly used sedatives. Atchley et al
39
reported a longer duration of MV (247.7vs 137.3 h;
Discussion
Summary
In this thorough and comprehensive review of the literature we report 27 publications relating to the use of continuous ketamine infusions as an analgesic or sedative for mechanically ventilated patients on ICU.
There was a paucity of high-quality evidence with very few well-designed prospective studies that use patient-centred primary endpoints with only seven studies (25.9%) prospectively comparing ketamine as a primary sedative to alternative regimens.
We were unable to establish any studies that investigated the role of ketamine on the incidence, severity, or duration of depression or PTSD following ICU admission, indicating a potential target for future well-designed prospective clinical trials.
However, despite the lack of well-designed, well-powered studies, this review did provide reassurance around the safety of ketamine sedation with low numbers of ADRs or SAEs reported. Reports of reduced incidence of delirium with no reports of emergence reactions go some way to alleviate apprehensions around unwanted psychomimetic phenomena.
The low incidence of adverse effects was not restricted to particularly low-dose or short-duration regimes, with doses and durations of ketamine ranging significantly across studies.
The reported findings suggest a range of potential patient benefits, including: improved sedation and pain scores, reduced concomitant sedative infusions, reduced opiate requirement, and haemodynamic stability. However these results must be interpreted with caution given the poor quality of included studies and small sample sizes. Additionally, the retrospective nature of many of the studies increases the risk of recall bias, inclusion bias, and reporting bias.
Whilst this was not a dedicated review of the evidence surrounding the relationship between ketamine and ICP, there were three studies that reported the effect of ketamine on intracranial pressure. Interestingly, none of the studies reported a significant increase, change, or variation in ICP. This may help allay the concerns of clinicians with regard to the safety of ketamine in patients with raised ICP or brain injury, which is still regarded as a contraindication to ketamine use.
Questions remain with regard to the overall generalisability of the results presented in this scoping review as many of the studies were conducted in specific patient populations for example, post-cardiac surgery or traumatic brain injury.
Conclusions
This review highlights the lack of robust, high-quality evidence for the use of continuous ketamine infusions as a primary sedation on ICU for patients undergoing MV, however the data available indicates that continuous ketamine infusions on ICU appear safe and well tolerated and may convey some benefits to patients at the doses and durations investigated.
The multiple gaps in the literature identified in this review need to be addressed through a large, prospective randomised controlled trial comparing ketamine as primary sedation to a well-matched control group that incorporates patient centred outcomes and endpoints. Currently no such trials are registered on clinicaltrials.gov.
Limitations
This scoping review was developed using the PRISMA-ScR checklist, and a protocol was publicaly available prior to commencing the review.
This review was limited by the studies available: 10 studies were excluded for not being available in either full-text form or English, which may have potentially excluded some important articles.
Footnotes
Appendices
Data extraction tool.
|
|
|---|
| Title |
| Objectives |
| Question |
| Population |
| Concept |
| Context |
|
|
| Citation |
| Author(s) |
| Year of Publication |
| Origin/country of origin |
| Aims/purpose |
| Population and sample size |
| Methodology /methods |
| Intervention type/comparator |
| Outcomes |
| Key findings relating to scoping question |
Appendix B – Search Strategy and Terms
Preliminary PubMed search terms: “(Ketamine) AND (Intensive Care) AND ((Sedation) OR (Analgesia))”
Subsequent searches of PubMed, Embase, Ovid, Scopus, and Web of Science using words / phrases and index terms from papers retrieved during initial search.
Additional sources were identified through searching reference lists of identified reports and review articles.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
