Abstract
Background:
Mechanically ventilated Intensive Care Unit (ICU) patients often require wrist restraints, contributing to immobility and agitation, over-sedation, and delirium. The Exersides® Refraint® (Healthy Design, LLC), a novel restraint alternative, may be safe and facilitate greater mobility than traditional restraints.
Objective:
This National Institutes of Health Small Business Technology Transfer (STTR) Program Grant-funded single-site Phase I feasibility study evaluated Exersides® safety and feasibility in anticipation of a multi-site Phase II randomized controlled trial (RCT).
Methods:
In two academic ICUs, mechanically ventilated adults ⩾25 years old who were non-comatose, required restraints and had an expected stay of ⩾2 days were enrolled to wear Exersides® and traditional wrist restraints for 4 h on day 1, in a randomized order, and in the reverse order on day 2. Main outcomes were Exersides® safety (i.e., patient/clinician lacerations/injuries), feasibility (i.e., ⩾90% of required data collected), and patient/family/clinician feedback.
Results:
Eight patients were enrolled; one no longer required restraints at initiation, yielding seven subjects (median [interquartile range (IQR)] age 65 [55, 70] years, 86% men). All seven wore Exersides®, averaging (SD) 2.5 (1.0) hours per session, with no safety events reported. Across restraint time periods, 92% and 100% of Richmond Agitation-Sedation Scale (RASS) and wrist actigraphy data, respectively, were collected. Feedback was positive (more movement and comfortable than traditional restraints) and constructive (bulky, intimidating to apply).
Conclusions:
This pilot study provided key safety and feasibility data for a Phase II RCT evaluating Exersides® versus traditional wrist restraints. Feedback motivated minor device modifications before RCT initiation.
Introduction
The use of wrist restraints is common in the intensive care unit (ICU) setting, particularly in those requiring mechanical ventilation. Most commonly placed to prevent accidental dislodgment of life-sustaining medical equipment (e.g. self-extubation, removal of lines/drains),1,2 soft wrist restraints have not been shown to prevent self-extubation. 3 Restraints may, in fact, make patient-initiated device removal more likely to occur, thought to be due to increased agitation.3 –6 Restraint use is also associated with adverse patient outcomes, including agitation, delirium, immobility, post-traumatic stress disorder (PTSD), and prolonged ICU length of stay.7 –15 For ICU nurses, restraints confer a sense of security, but are also associated with feelings of guilt, sadness, and moral distress.16,17 The 2018 Society of Critical Care Medicine Pain, Agitation/Sedation, Delirium, Immobility, and Sleep Disruption (PADIS) Clinical Practice Guidelines outline the many unintended and harmful consequences of physical restraint use, and recommend that efforts be taken to reduce their usage in the ICU setting. 18
As an alternative to traditional wrist restraints, a novel device, the Exersides® Refraint® (Healthy Design, Rutland, VT), was developed with the support of a National Institutes of Health Small Business Technology Transfer (STTR) Program Grant. The goal of this device is to allow patients to increase arm mobility without increasing the risk of dislodgement of life-sustaining medical devices. Featuring a soft shoulder strap, adjustable arm tube, clear removable hand shield, and optional straps to secure the device to the bed, Exersides® allows anterior, medial, and lateral arm extension, wrist supination and pronation, and the grasping of objects (i.e. television remote control) while allowing adjustable reduction in elbow flexion (Figure 1). The aim of this Phase I study was to evaluate the preliminary safety and feasibility of Exersides™ in mechanically ventilated patients in an ICU setting.
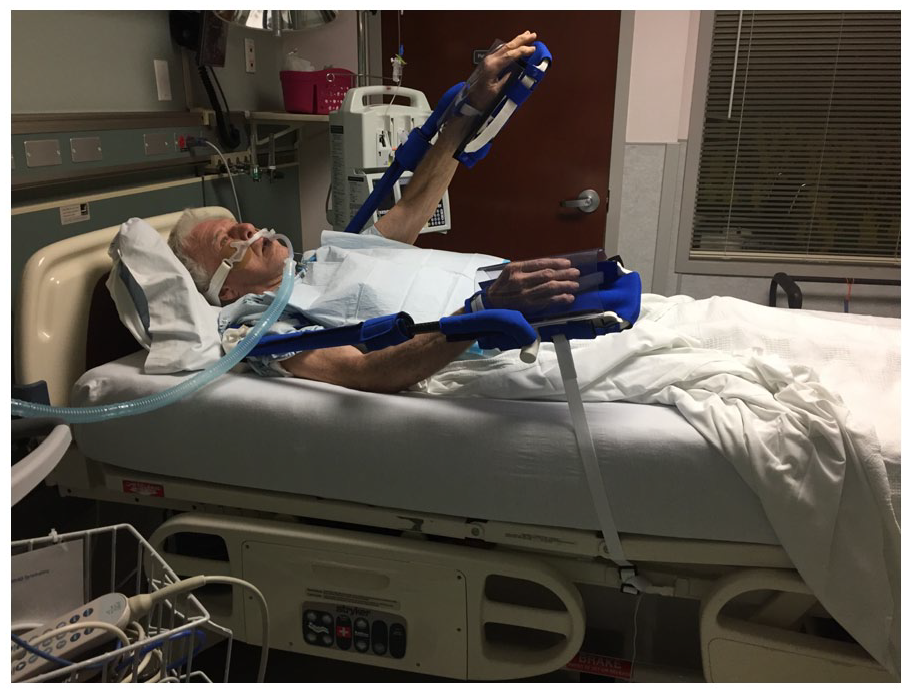
Exersides® Refraint® device on a volunteer acting as a prototypical mechanically ventilated patient. Comprised of a shoulder strap and soft transparent open-ended hand shield connected by an adjustable arm tube, Exersides® facilitates shoulder movement in all directions, wrist supination and pronation, and object grasping while allowing reduction in elbow flexion to prevent entanglement in and dislodgement of medical devices. In addition to the unstrapped maximum-mobility option (pictured above on left arm), Exersides® can be secured to the bed using a stretchable elastic strap (shown on right arm) or a rigid strap (not pictured).
Methods
Study, setting and participants
Supported by an NIH SBIR/STTR (Small Business Innovation Research/Small Business Technology Transfer) award, this study evaluated the novel Exersides® Refraint® following an established two-phase approach: safety/feasibility (SBIR/STTR Phase I) and effectiveness/randomized controlled trial (RCT) (SBIR/STTR Phase II – enrollment completed, analysis pending). In Phase I, we prospectively evaluated the safety and feasibility of Exersides® in critically ill patients at two University of Vermont Medical Center medical ICUs (MICUs), each with 22 beds and a 1:1 or 1:2 nurse-to-patient ratio, depending on the complexity of care.
This Phase I pilot study was designed to evaluate the feasibility of Exersides® and fulfill milestone requirements for progression to the STTR/SBIR Phase II project. As part of pilot study planning, an a priori convenience sample of eight patients was targeted for enrollment. Eligible patients were ⩾25 years old, required mechanical ventilation and bilateral wrist restraints, had a predicted ICU stay of ⩾2 days and were responsive to verbal stimuli. Exclusion criteria included impaired or limited bilateral upper extremity mobility prior to ICU admission, pre-existing severe cognitive impairment or language barrier, neuromuscular blocker infusion, severe skin breakdown on either upper extremity, pregnancy, incarceration, and expected death or withdrawal of life-sustaining treatment within 6 days of enrollment. Study termination could result from the completion of study activities, withdrawal from participation by the subject or their proxy, or the discontinuation of study or restraint orders by the physician. Written informed consent was obtained from next of kin and the study was approved by the University of Vermont Institutional Review Board (IRB).
Study protocol
Patients were eligible for inclusion if they had orders for unilateral or bilateral wrist restraints, as deemed necessary by the ICU provider team (e.g. to prevent self-extubation, pulling at other tubes and lines, and prevent interference with care), at the time of screening. If enrolled, patients underwent observation during the daytime nursing shift over the course of 2 days, with each day consisting of two 4-h restraint recording periods. The restraint device (Exersides® vs traditional wrist restraint) worn during the first 4-hour period was randomly selected. After the first 4-hour period, the restraint was switched to the other device. For patients with ICU provider orders for restraints on day 2, the order of Exersides® versus traditional wrist restraint was reversed as compared to day 1, again for two 4-h periods during the daytime nursing shift. If Exersides® needed to be removed at any time during the session, they were replaced with traditional soft restraints to comply with the patient’s active order from the ICU provider team.
Outcomes
Safety and adverse events
For this Phase I study, the primary outcome was Exersides® safety, evaluated using a Safety and Compliance form which documented device-related patient pressure ulcers, patient/clinician lacerations, and other patient/clinician injuries. Additional safety and adverse events included patient removal of lines, tubes, or the restraint device itself, and whether the device damaged the bed or other equipment (e.g. ventilator). Device safety was defined, a priori, as an adverse event incidence of <20% (no more than 1 of eight patients).
Feasibility: Actigraphy measurement
Wrist activity was measured using the Philips Actiwatch Spectrum® actigraphy device, a wristwatch-type device containing an accelerometer. The Spectrum® was selected because it is small, lightweight, comfortable, and has been used by the investigators in previous studies.19 –23 Actigraphy recording began after randomization, with Actiwatch devices placed on the wrists where a wrist restraint or Exersides® was present. Activity was logged across 30-second epochs. Actigraphy devices remained in place during the changing of restraint types and were removed after the second 4-hour block or when medically necessary. Actigraphs were placed again the next day for subjects participating in Day 2. Feasibility was defined, a priori, as successful activity measurement across >90% of the Exersides® wear time.
Feasibility: sedation measurement
During the study periods, research staff recorded the Richmond Agitation-Sedation Scale (RASS) Score 24 every hour to measure patient sedation/agitation levels. Feasibility was defined, a priori, as sedation level evaluations completed across >90% of the Exersides® wear time.
Feedback questionnaires
Three surveys were administered to evaluate patient/proxy and staff satisfaction with Exersides®. For each patient, a proxy (e.g. spouse, parent, adult child) was recruited to complete (1) the Quebec User Evaluation of Satisfaction with assistive Technology [QUEST 2.0], 25 a clinical assessment tool designed to evaluate user satisfaction with assistive technology devices and associated services; (2) a Novel Restraint Evaluation Form for Family developed by study team members to evaluate experiences of family members whose loved one required restraints; and (3) semi-structured interview questions. Nurses and physicians completed (1) QUEST 2.0; (2) a Novel Restraint Evaluation Form for Providers evaluating nurse and physician experiences caring for a patient wearing Exersides®; and (3) semi-structured interview questions. Provider questions addressed device durability, ease of use, and whether the device caused patient discomfort. All surveys were obtained during each patient’s 2-day evaluation period.
Data collection and analysis
Demographic and clinical data were collected from the electronic health record and included patient age, gender, race, admission diagnosis category, daily ICU Sequential Organ Failure Assessment (SOFA) scores, the presence of continuous and non-infusion sedative medications, and the occurrence of physical rehabilitation activities.
Descriptive statistics were used to summarize study data. Actigraphy data were summarized across 30-second epochs, and included activity levels, non-zero activity levels, inactivity (zero-activity epochs, indicating motionlessness), and wear time as a proportion of restraint/restraint alternative time. When actigraphy devices were present bilaterally, left and right wrist values were averaged to generate a single activity measure for each 30-second epoch. As this pilot was designed to examine Exersides® safety and feasibility only, P-values were not calculated. Sedation (RASS) and activity (actigraphy) data were stratified by restraint (Exersides® vs traditional) and summarized using Stata version 17.0 (StataCorp LLC, College Station, TX).
Closed-ended Likert-type survey results were presented as relative frequencies. Comments from open-ended and semi-structured survey questions were aggregated by question.
Results
Demographics
ICU-admitted patients were screened from October 2, 2018 to April 8, 2019. Consent was obtained for eight eligible patients who were enrolled and randomized. One patient subsequently did not require restraints by the time of device initiation and therefore did not participate in the study, yielding seven patients reported herein. The median (interquartile range [IQR]) age was 65 (55, 70) years and 1 (14%) patient was female (Table 1). The median (IQR) admission APACHE II score was 21 (18, 30), with three (42%) patients having a respiratory diagnosis. No patients underwent physical therapy (PT) or occupational therapy (OT) while enrolled in this study.
Patient characteristics (N = 7 patients).

ICU: Intensive Care Unit; IQR: Interquartile Range; RASS: Richmond Agitation-Sedation Scale; APACHE II: Acute Physiology and Chronic Health Evaluation II; SOFA: Sequential Organ Failure Assessment; PT: Physical Therapy; OT: Occupational Therapy.
Calculated once daily using data from the patient’s electronic health record. Score calculation involved use of the worst values for each parameter across each 24-h period (00:00-23:59).
Continuous infusion received during the study day (00:00–23:59).
Restraint wear times and safety
Over the study period, it was feasible for Exersides® to be worn by all seven patients. Across the study, Exersides® wear time totaled 22.9 h, with a mean (SD) total wear time of 2.5 (1.0) hours per 4-h data collection session (Table 2) and a maximum wear time of 3.8 h. On study days 1 and 2, seven and two patients wore Exersides®, respectively, totaling 17.6 h and 5.4 h, with mean (SD) wear times of 2.5 (1.2) and 2.7 (0.7) hours per session. One patient declined left limb placement for unclear reasons, instead wearing a traditional wrist restraint on the left. The patient wore Exersides® on the right for only 0.4 h on day 1, after which their participation in the study was discontinued. In addition to Exersides® feasibility, zero device-related safety concerns (e.g. patient/staff injuries, restraint/equipment damage, self-extubation) occurred.
Restraint, sedation/delirium, and activity data (N = 7 patients).
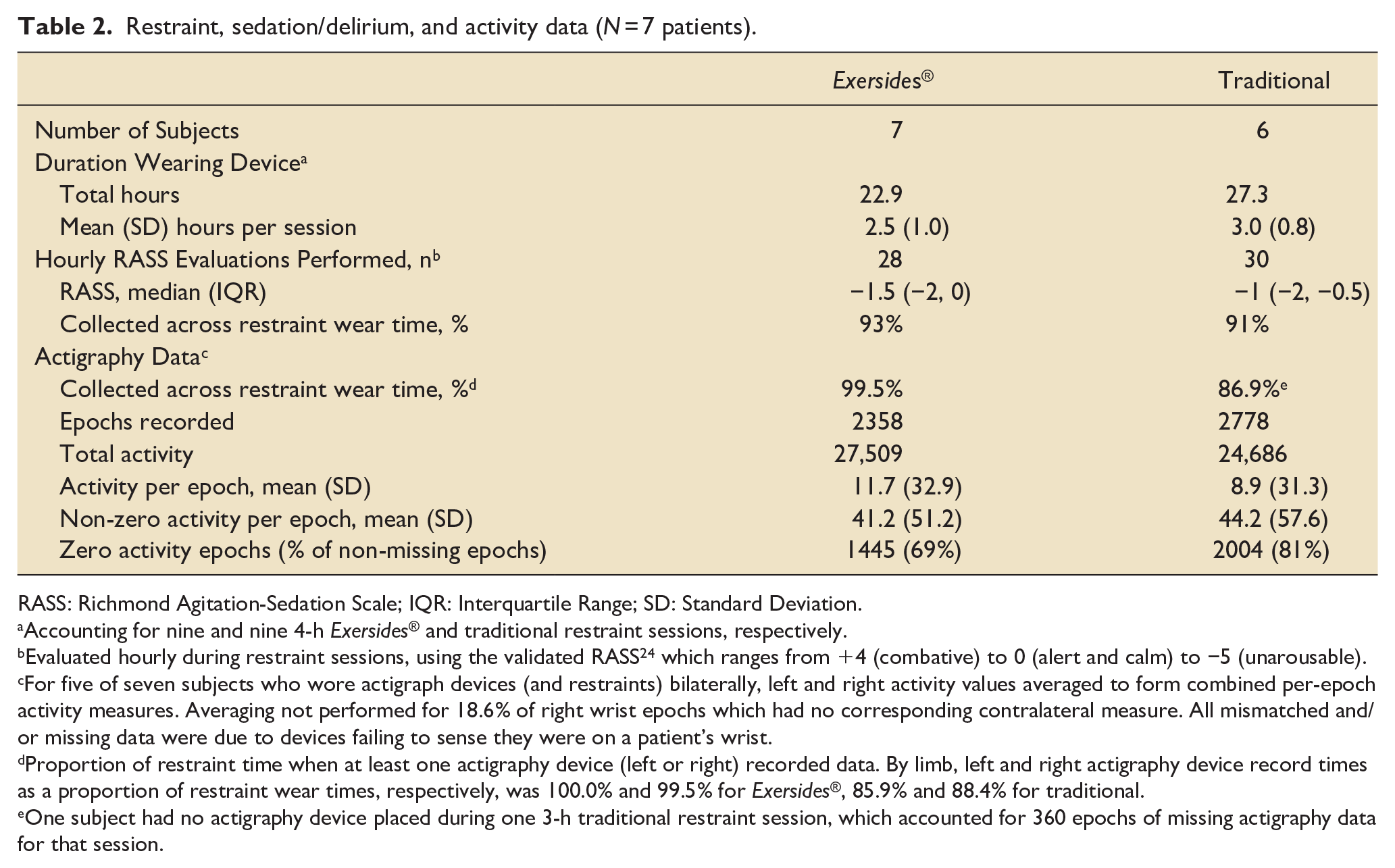
RASS: Richmond Agitation-Sedation Scale; IQR: Interquartile Range; SD: Standard Deviation.
Accounting for nine and nine 4-h Exersides® and traditional restraint sessions, respectively.
Evaluated hourly during restraint sessions, using the validated RASS 24 which ranges from +4 (combative) to 0 (alert and calm) to −5 (unarousable).
For five of seven subjects who wore actigraph devices (and restraints) bilaterally, left and right activity values averaged to form combined per-epoch activity measures. Averaging not performed for 18.6% of right wrist epochs which had no corresponding contralateral measure. All mismatched and/or missing data were due to devices failing to sense they were on a patient’s wrist.
Proportion of restraint time when at least one actigraphy device (left or right) recorded data. By limb, left and right actigraphy device record times as a proportion of restraint wear times, respectively, was 100.0% and 99.5% for Exersides®, 85.9% and 88.4% for traditional.
One subject had no actigraphy device placed during one 3-h traditional restraint session, which accounted for 360 epochs of missing actigraphy data for that session.
Traditional soft wrist restraints were worn by six patients, totaling 27.3 h, with a mean (SD) wear time of 3.0 (0.8) hours per session (Table 2). On days 1 and 2, five and four patients wore the traditional soft restraints, respectively, totally 15.0 and 12.3h with mean (SD) wear times of 3.0 (1.0) and 3.1 (0.6) hours per session. Notably, on study day 1, two of the seven patients who wore Exersides® did not have data collected while wearing traditional restraints; one (mentioned above) terminated study activities early, and the other shifted traditional restraint data collection to day 2 due to a late start on day 1. Two of 4 patients who wore the traditional restraint on study day 2 did not wear Exersides®; one was the subject whose traditional restraint day 1 was shifted to day 2, and the other was extubated and their restraint order discontinued.
Sedation and activity measurements
While wearing restraints or Exersides®, 92% of hourly RASS 24 assessments were performed on all seven patients. Across Exersides® and traditional restraint wear time, respectively, 93% and 91% of assessments were completed, with median (IQR) scores of −1.5 (−2, 0) and -1 (−2, −0.5) (Table 2).
Of the enrolled subjects who wore restraint devices, 7 (100%) and 4 (57%) contributed actigraphy data on days 1 and 2, respectively. Two patients contributed right-wrist actigraphy data only, one being the subject mentioned above who declined Exersides® placement the left limb and a second whose left-arm actigraphy device malfunctioned. Including these and left-right averaged activity data, 5136 30-second actigraphy epochs were recorded; 2358 and 2778 epochs were recorded in the presence of Exersides® and traditional restraints, respectively (Table 2). Mean (SD) activity levels were 10.2 (32.1) movements per epoch, with 11.7 (32.9) and 8.9 (31.3) movements per epoch while wearing Exersides® versus traditional restraints, respectively. The proportions of zero-activity epochs were 69% and 81% when wearing Exersides® and the traditional restraint, respectively, and when removing zeroes, mean non-zero activity levels were 41.2 (51.2) and 44.2 (57.6) (Table 2). Overall, 74% of epochs containing paired left- and right-wrist data were zero. Actigraphy data were collected during 92.3% of the total restraint wear time, and during 99.5% and 86.9% of the time the patients were wearing Exersides® and traditional restraints, respectively (Table 2).
Survey results
The proxies of four enrolled patients completed the Exersides® QUEST 2.0 satisfaction form, with 100% agreeing or strongly agreeing that the restraint alternative was comfortable, safe, and effective for preventing self-removal of medical tubes. Three of 4 (75%) agreed or strongly agreed that the novel restraint alternative was preferable and more humane than soft wrist restraints. Free-text proxy comments regarding Exersides® included “help in recovery,” “good concept,” “preferred [over] soft wrist,” and “really like [Exersides®]” (
Of the 10 nurses and physicians completing the satisfaction form, 6 (60%) agreed or strongly agreed that the Exersides® device was easy to place and remove, 7 (70%) agreed or strongly agreed that it was safe for staff, 7 (70%) agreed or felt neutral about the restraint alternative being easy to adjust, 9 (90%) agreed or strongly agreed that it was effective at preventing self-extubation, 7 (70%) agreed or were neutral to Exersides® being preferable to traditional wrist restraints, and 9 (90%) were neutral to the statement that Exersides® is more humane than traditional restraints. Nurses and physicians “love the increased safety from self-extubation” but felt Exersides® “appeared clunky” and “complicated” (eTable).
Between nurses, physicians, and patient proxies, a total of 10 QUEST 2.0 surveys were obtained, with 8 (80%) having a QUEST score of “more or less,” “quite,” or “very” satisfied with the device.
Finally, eight nurses and one physician completed the semi-structured Exersides® questionnaire. Favorable comments about Exersides® were that it “[was] more effective to prevent self-extubat[ion],” “provided mobility,” and “increased safety.” Four of eight nurses felt patient arm movements while wearing Exersides® were non-purposeful movements related to agitation rather than “normal” mobility. Regarding ease of use, favorable comments included that the restraint “[did not] have to be ‘tied down’ to the bed” and could “quickly hold the patient’s arm down”; however, nurses found Exersides® “bulky,” “intimidating,” and “cumbersome” and requiring “a long time to put on” with straps “[appearing] a little challenging.” Nurses suggested making Exersides® smaller and lighter.
Discussion
This Phase I feasibility study in seven critically ill, mechanically ventilated ICU patients demonstrated that the Exersides® novel restraint alternative was safe, feasible to place, and well tolerated. Both Exersides® and traditional wrist restraints were each worn for more than 20 h in total by patients over one or 2 days, with actigraphy epochs recorded for 99% of the time that the Exersides® were worn, exceeding the 90% feasibility target. No device-related safety concerns were observed. Finally, semi-structured interviews of ICU patient family member proxies and providers mainly pointed to concerns about the Exersides® device being bulky and cumbersome. This feedback was valuable in refining the Exersides® device for evaluation in the Phase II randomized controlled trial (RCT).
Notably, average activity levels of patients tended to be higher when wearing Exersides® versus traditional restraints, and patients wearing Exersides® tended to have a lower percentage of zero-activity epochs, a marker of inactivity. Agitation and sedation levels, as determined by the RASS, were similar when patients wore Exersides® as compared to traditional restraints. While Phase I was underpowered to evaluate these outcomes via multivariable models, Phase II is adequately powered to detect differences in upper extremity activity when comparing Exersides® and traditional restraints. Having met key safety and feasibility criteria and greater than 90% of RASS assessments and actigraphy recordings completed, this Phase I study provides essential feasibility data to support initiation of the multi-site, within-patient crossover Phase II RCT evaluating Exersides® versus traditional restraints in mechanically ventilated patients (NCT04067622).
Limitations
This Phase I study had many strengths, but some notable limitations exist. First, the small sample size limited the precision of results and generalizability, which will be addressed in the larger Phase II multi-center study. Second, actigraphy cannot distinguish between directed and agitated motion. To address this limitation, the Phase II RCT includes behavioral mapping of subject movements to distinguish between different types of motion. Third, it was decided that delirium would be important to evaluate moving forward, so the subsequent Phase II protocol included multiple Confusion Assessment Method for the ICU (CAM-ICU) assessments while enrolled patients were wearing both novel and traditional restraints. Fourth, since this pilot, Exersides® has undergone several upgrades based, in part, on bedside experiences and feedback received during this pilot study. Improvements include use of a lower weight material for the Arm Tube, improving Hand Shield usability with an improved transparent material and easier-to-attach clips, and design adjustments to the shoulder attachment straps and clips, making device placement much easier and requiring fewer adjustments. As this Phase I study supported device safety and feasibility and yielded mostly positive feedback, we hypothesize that studies involving newer Exersides® versions will motivate further improvements to improve device tolerability and comfort. Finally, while these Phase I data suggest increased patient mobility with Exersides®, this study was underpowered to evaluate this association. The multi-site Phase II RCT is adequately powered for rigorous activity analyses, as measured using wrist actigraphy, and will also evaluate important clinical variables such as delirium, agitation, and sedative medication use.
Conclusion
The use of wrist restraints in the ICU is associated with adverse patient outcomes, presenting an opportunity for improvement with novel devices. This Phase I study evaluated the safety and feasibility of the Exersides Refraint® which was developed to allow increased patient mobility without increasing their risk of dislodging life-sustaining medical devices. The Exersides® device was found to be feasible for use and subsequent evaluation in the ICU, as indicated by wear time, activity levels, and agitation/sedation levels, with no observed safety or adverse events. Feedback from healthcare providers and patient proxies motivated device refinement, which will be evaluated as part of a Phase II multi-center crossover RCT comparing Exersides® to traditional wrist restraints.
Footnotes
Acknowledgements
The authors thank Prema Menon, MD, PhD for her assistance with study preparation, and Emilee Elfsten, BS for her assistance with data analysis.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MTP is the inventor of the Exersides® Refraint® and founder of Healthy Design Ltd. Co. which sells the device. JHTB has consulted with Healthy Design, Ltd. Co. MTP provided technical input regarding use of the Exersides® Refraint®. MTP and JHTB were not involved with collection, analysis, or interpretation of the data. The remaining authors have no financial or other conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is supported by a Small Business Technology Transfer (STTR) Program Grant from the National Institutes of Health/National Institute on Aging (R42AG059451). BBK is supported by a Paul B. Beeson Career Development Award through the NIH/NIA (K76AG059936). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Consent
The authors confirm that consent has been given for the publication of images contained within this manuscript.
