Abstract
Aortic dissections are associated with significant mortality and morbidity, with rapid treatment paramount. They are caused by a tear in the intimal lining of the aorta that extends into the media of the wall. Blood flow through this tear leads to the formation of a false passage bordered by the inner and outer layers of the media. Their diagnosis is challenging, with most deaths caused by aortic dissection diagnosed at post-mortem. Aortic dissections are classified by location and chronicity, with management strategies depending on the nature of the dissection. The Stanford method splits aortic dissections into type A and B, with type A dissections involving the ascending aorta. De Bakey classifies dissections into I, II or III depending on their origin and involvement and degree of extension. The key to diagnosis is early suspicion, appropriate imaging and rapid initiation of treatment. Treatment focuses on initial resuscitation, transfer (if possible and required) to a suitable specialist centre, strict blood pressure and heart rate control and potentially surgical intervention depending on the type and complexity of the dissection. Effective post-operative care is extremely important, with awareness of potential post-operative complications and a multi-disciplinary rehabilitation approach required. In this review article we will discuss the aetiology and classifications of aortic dissection, their diagnosis and treatment principles relevant to critical care. Critical care clinicians play a key part in all these steps, from diagnosis through to post-operative care, and thus a thorough understanding is vital.
Introduction
Aortic dissection is associated with significant mortality and morbidity often requiring immediate intervention.1–3 Its diagnosis can be challenging due to the multiple, and often non-specific, ways it can present. It has an estimated incidence of 6 per 100,000, with >60% of deaths secondary to aortic dissection diagnosed at post-mortem.4,5
An array of tools now exists to help expedite diagnosis of an aortic dissection, however without suspicion of the diagnosis these are of no use.2,6 The management of an acute aortic dissection is dependent on the patient’s condition, the type of dissection and its extension.
In this narrative review we will discuss the classification and aetiology of aortic dissections and their management, concentrating on the role of the critical care team.
Pathophysiology and classification
An aortic dissection describes a tear in the intimal lining of the aorta extending into the media of the wall.1,4,6 Blood flow through this tear leads to the formation of a false passage bordered by the inner and outer layers of the media. Connective tissue diseases such as Marfan or Ehlers-Danlos syndrome increase the risk of dissection due to weakening of the media.4,5 Other causes include atherosclerosis, trauma, inflammatory or infective and iatrogenic conditions.4,5
Aortic dissections can be classified by their location and chronicity, with management strategies differing significantly between them (Figure 1).
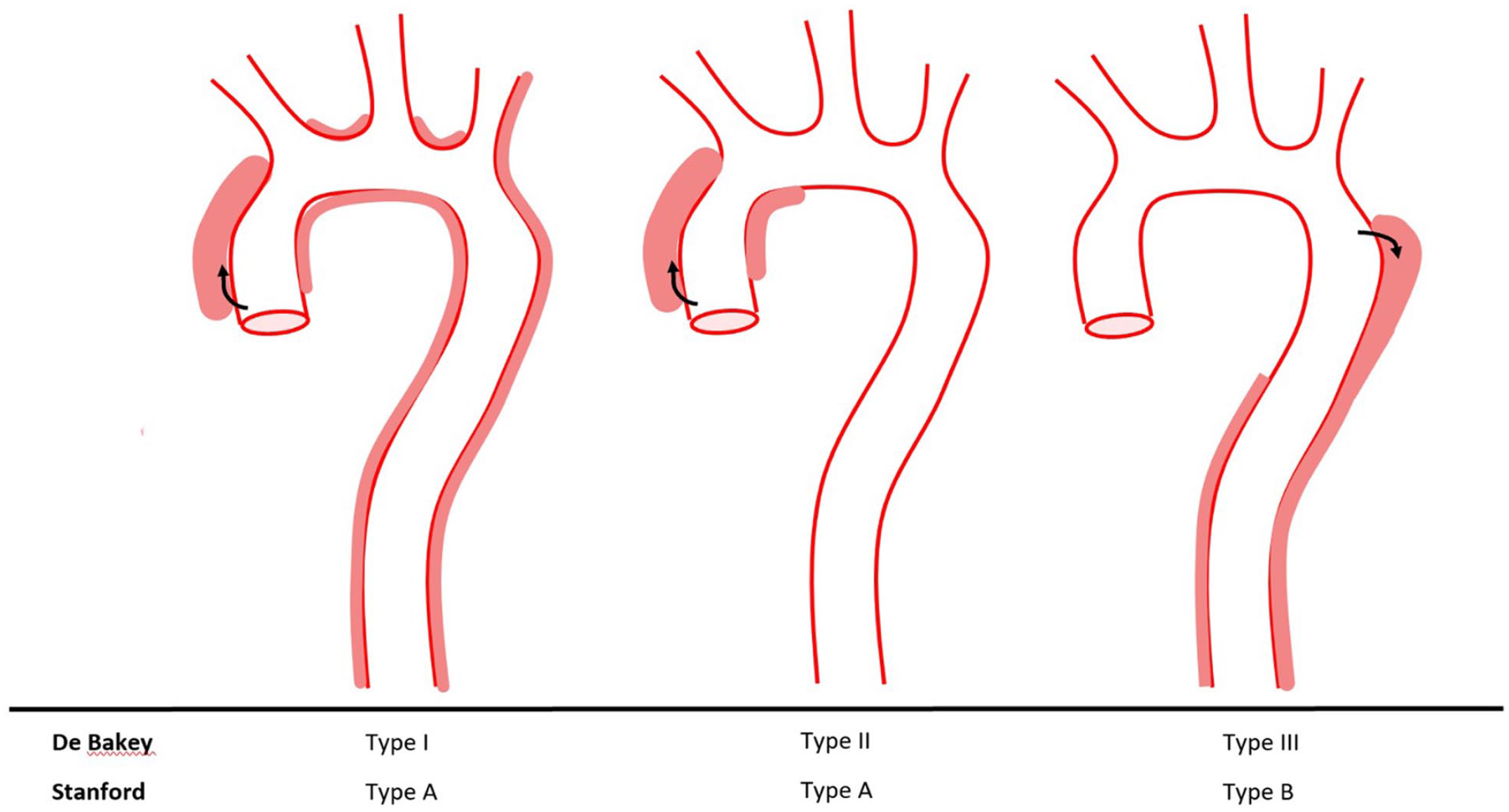
Classification of aortic dissections.
Stanford classification
This system classifies dissections in relation to their involvement of the ascending aorta:
DeBakey classification
This divides aortic dissection into three types:
◦ 3A – limited to the descending thoracic aorta ◦ 3B – extending below the diaphragm.
Chronicity
Aortic dissections may also be classified by their chronicity as follows:
Diagnosis
Aortic dissections may present in several ways and mimic other pathologies, making diagnosis challenging. A high degree of suspicion is thus required in patients with suggestive features. ‘Think Aorta’ has been recommended as an approach for its early recognition in any patient with severe chest pain by the patient charity ‘Aorta Dissection Awareness UK’. 7 A recent investigation, conducted by the Health Service Investigation Branch (HSIB), highlights many of the challenges faced by clinicians when managing acute aortic dissection. 8
Presentation of a Stanford type A aortic dissection may include severe chest pain (typically worst at onset), syncope, aortic insufficiency, pulse differential, myocardial ischaemia and neurological signs.1,6,9,10 Presentation of a Stanford Type B aortic dissection may include severe chest, back or migrating pain, distal pulse differential, limb pain/claudication, renal insufficiency and high blood pressure.10–13
Rapid diagnosis is vital as treatment is time critical. More than 20% of patients with acute aortic dissections die before reaching hospital, with more than 50% dying before reaching a specialist centre. 14 Aortic dissection mortality increases by 1%–2% per hour after symptom onset. 15
Several investigations can be used to help rule in and out aortic dissections, ranging from bedside tests to magnetic resonance imaging (MRI). 6 Combining a patient’s presentation, risk-profile and the results of relevant investigations are key to making a timely and accurate diagnosis.
Computed tomography
Computed tomography (CT) remains the first-line imaging for definitive diagnosis of aortic dissection. It is readily available in most hospitals and allows for characterisation of the type, position and size of the dissection alongside identification of potential differential diagnoses (e.g. pulmonary embolism). However, unlike echocardiography, it requires transfer of the patient and thus may not be immediately possible in haemodynamically unstable patients.
The preferred protocol is non-enhanced CT followed by a contrast enhanced CT angiography. 6 To assess the full extension of the dissection the scan should extend from the chest all the way down to the pelvis. 6 The presence of a visible dissection flap has previously been described as the ‘tennis ball’ sign (Figure 2).6,16
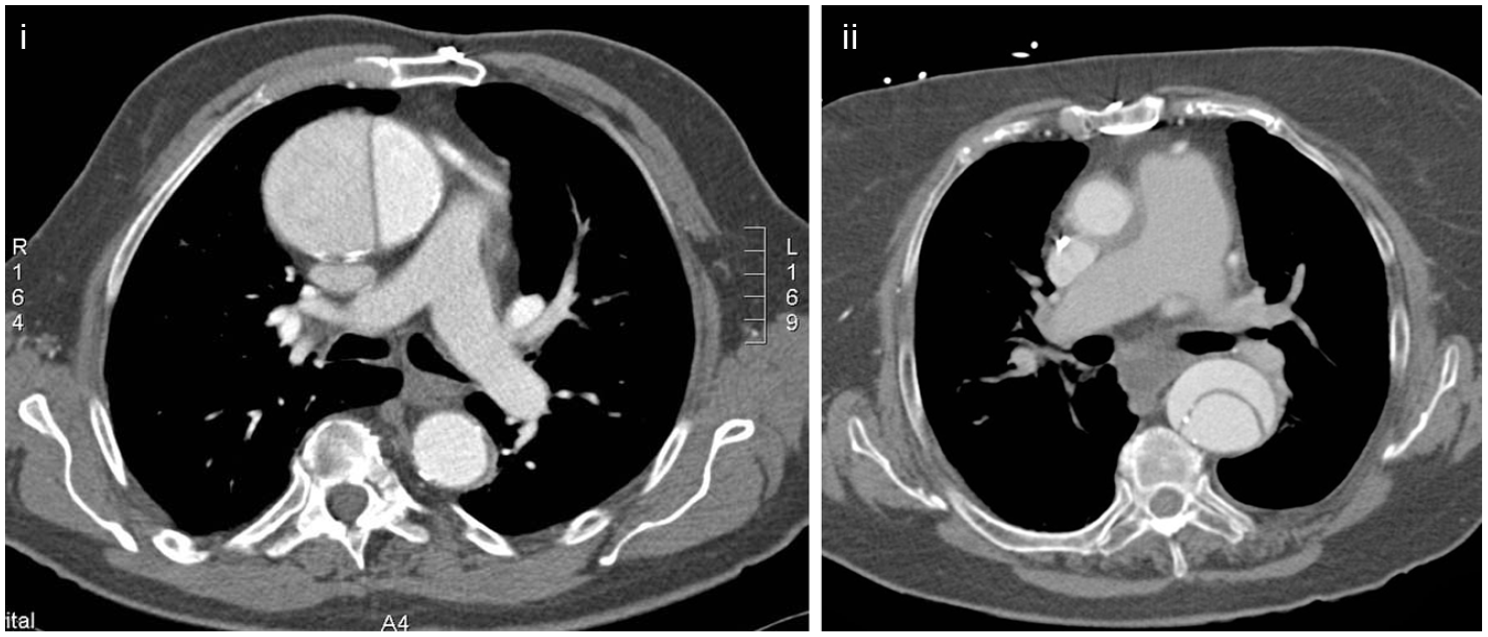
Computed tomographic images demonstrating the ‘tennis ball’ sign in (i) a type A dissection and (ii) a type B dissection.
Point-of-care ultrasound
Echocardiography
Both transthoracic (TTE) and transoesophageal echocardiography (TOE) can be utilised in the diagnosis of acute aortic dissection. 17 Their ability to rapidly diagnosis aortic dissection at the bedside is potentially lifesaving. Transthoracic echocardiography has the advantage of being less invasive than TOE and more readily available. 18 Also, TOE is impractical in a patient with symptoms even if sedation is employed, making it a less favourable imaging modality at the stage of diagnosis. TOE is usually used in the operating room after induction of anaesthesia if surgery is warranted and sometimes in the ICU if the patient is intubated and ventilated.
Both modalities allow for visualisation of the aortic valve, aortic root, ascending aorta and the pericardium (Figure 3).18–20 Use of the suprasternal echocardiographic window also allows for visualisation of the arch of the aorta. Potential echocardiographic findings in aortic dissection are shown in Table 1. 17 It is however important clinicians acknowledge that whilst echocardiography may rule in a dissection, it should not be used to rule one out.
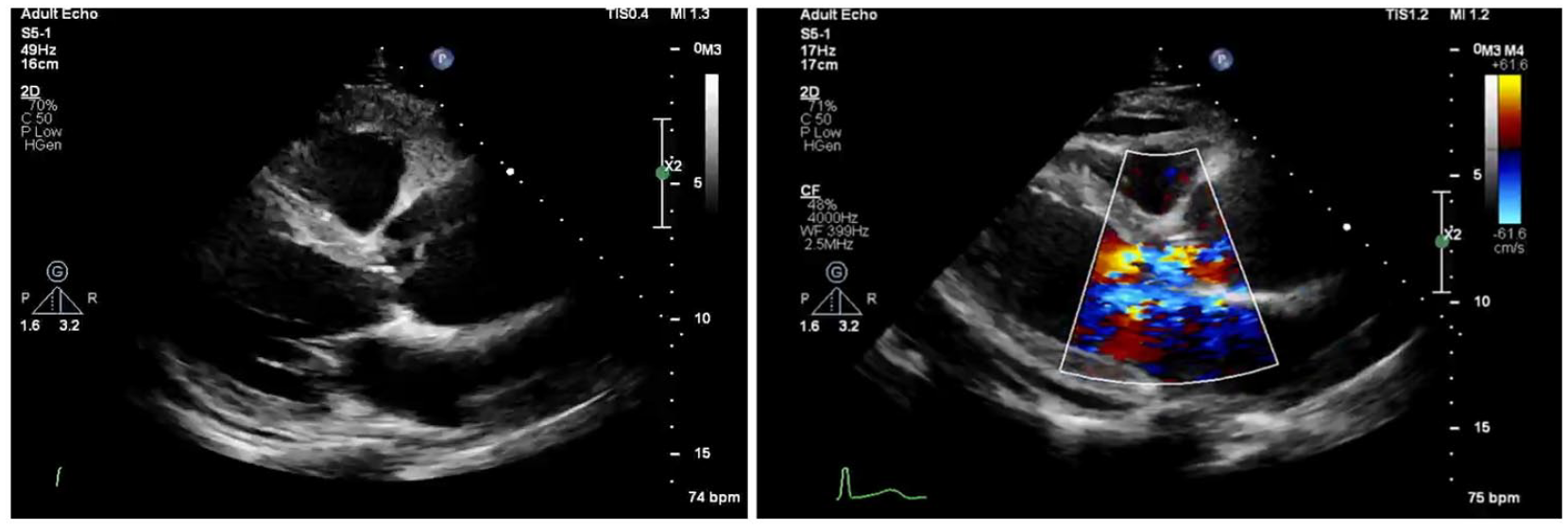
Transthoracic echocardiographic view of a dilated aortic root with an ascending aortic dissection and severe aortic regurgitation.
Echocardiographic findings in aortic dissection.

BSE: British Society of Echocardiography.
Abdominal aorta scanning
Ultrasound examination of the abdomen allows for identification of an aortic dissection extending down to the abdominal aorta. The aorta is examined in transverse and longitudinal section, with typical appearances being the presence of a flap or thin membrane fluttering within the vessel. Colour flow Doppler can also be incorporated to ascertain for the presence of flow through the false lumen. When combined with echocardiography, this technique has been quoted to have a sensitivity of 67%–80% and specificity of 99%–100% for diagnosing aortic dissection. 21 No literature currently exists describing a method to accurately differentiate acute from chronic aortic dissection using ultrasound.
Magnetic resonance imaging
Magnetic resonance imaging (MRI) has a high sensitivity and specificity (98%) for the detection of aortic dissection. 6 The use of MRI is limited by its availability, prolonged scan time, challenges associated with monitoring and contraindication in patients with metal implants such as pacemakers. 22
Electrocardiogram (ECG)
Aortic dissection may result in abnormal ECG findings which can distract the clinician. 23 More than 70% of patients with type A acute aortic dissections have an abnormal ECG. Previous studies have demonstrated type A acute aortic dissections may present with the following:
ST elevation (8%) – this is closely related with direct coronary involvement
ST depression (34%)
T wave changes (21%). 24
Presence of ECG changes are associated with increased incidence of shock and cardiac tamponade. 24 It is thus important aortic dissection remains on the differential list in such cases and is not brushed aside by a tunnel vision pursuit of a myocardial infarction.
Chest X-ray
Traditional teaching emphasises the presence of a widened mediastinum in patients with type A aortic dissection. It is however important to recognise that >20% of patients with type A dissections do not have this finding and is should therefore not be used as a rule out investigation. 2 Cardiomegaly may also be present in cases of pericardial effusion or aortic valve incompetence secondary to aortic dissection. 25
Biochemical markers
Much work has been done to try an identify a biochemical marker of aortic dissection. D-dimer is one such biomarker. A meta-analysis assessing its use in the diagnosis of acute aortic dissection found a d-dimer of ⩾500 ng/ml to be associated with a 97% sensitivity and 56% specificity. This corresponded to a negative predictive value of 96% and positive predictive value of 60%. 26 These results highlight a potential role for d-dimer in identifying patients without an acute dissection. However, the role of biomarkers is significantly limited in the acute scenario, where time can not be afforded to waiting for results and thus they are likely useful only as a rule out tool in a potential chronic presentation. 6
Aortic dissection management
The type of dissection and haemodynamic condition of the patient will have significant impact on the management strategy required, both on the intensive care unit and in the operating theatre.
Initial management
When managing an acute aortic dissection, initial treatment should concentrate on resuscitation and stabilisation of the patient whilst deciding and preparing for definitive intervention (Figure 4). The overriding goal of initial management is to reduce shear forces being placed on the aorta whilst ensuring sufficient organ perfusion.
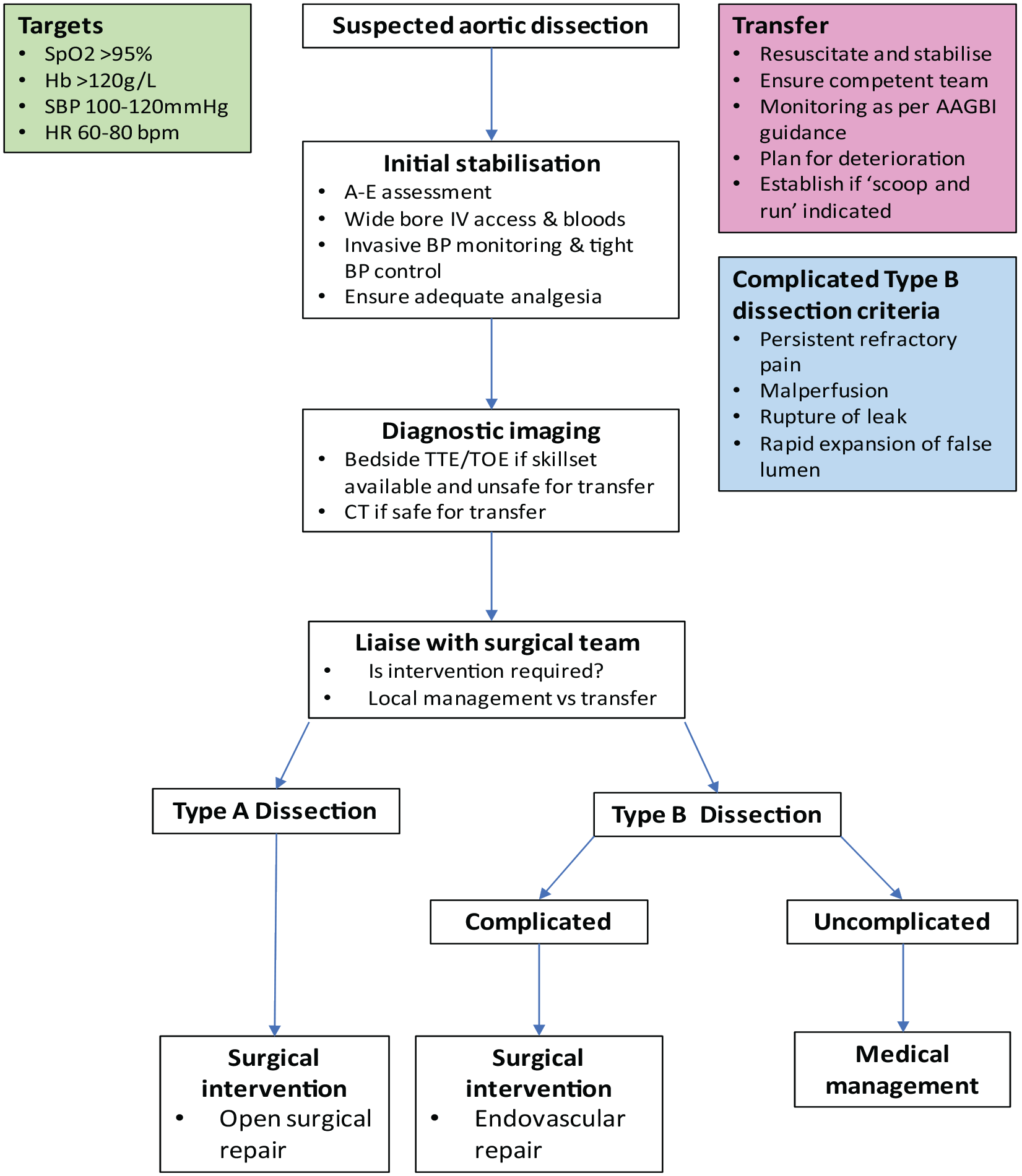
Flow chart for the initial management of an acute aortic dissection.
Blood pressure management
Tight blood pressure control and reduction of the rate of pressure rise during systole is vital to reduce the chances of the dissection propagating further. The shear force within the aorta is defined by the change in pressure over time, the dP/dt. The rate at which an intimal tear propagates is associated to the dP/dt, thus highlighting that simply dropping the blood pressure isn’t sufficient. If you look at Figure 5, the purple line in (i) represents the addition of a vasodilator in isolation. Here, whilst the systolic and diastolic blood pressures have dropped, the dp/dt has increased, secondary to the reflex chronotropic and inotropic response. In image (ii) of Figure 5, the addition of a beta-blocker has lowered the heart rate and thus decreased the dp/dt. In cases where there is a different in blood pressure between limbs, the higher blood pressure should be targeted.
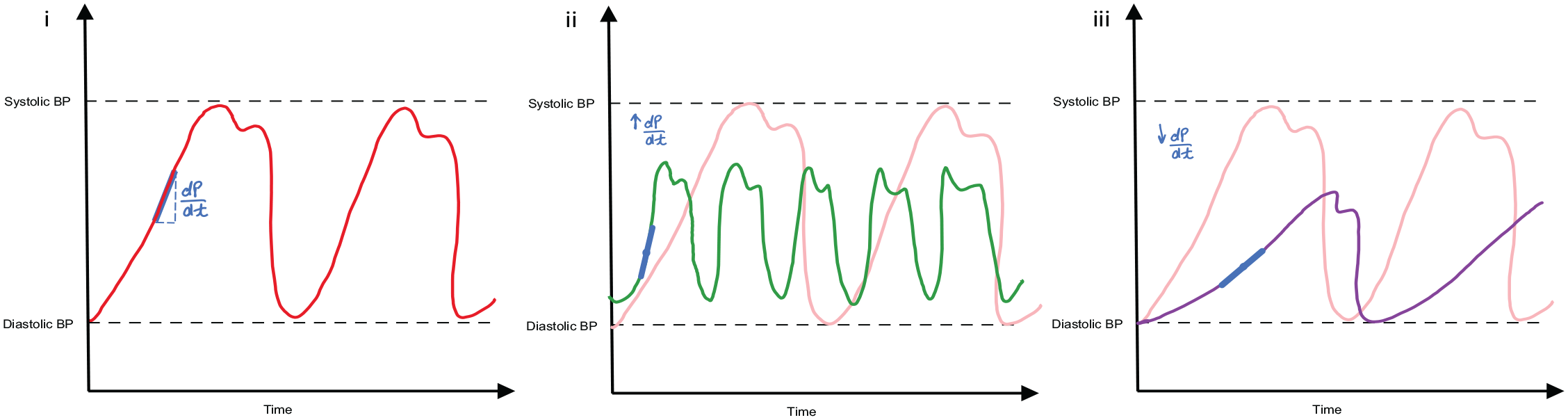
Demonstration of arterial waves forms and the effect of pharmacological agents. (i) Baseline arterial wave form, dp/dt represents the shear force which is a result of the change in pressure over tie; (ii) the background light red line is the baseline arterial wave form, the superimposed green line is the waveform after the use of vasodilators – leading to a reduction in blood pressure (BP) but an increase in heart rate and dP/dt; (iii) again the background light red line is the baseline arterial wave form and the purple line here represents the waveform following beta-blocker treatment – leading to a reduction in BP and heart rate and with this a reduction in dP/dt.
In the absence of aortic regurgitation, the first line antihypertensive agent is a beta-blocker, targeting a heart rate of 60–80 beats/ min and a systolic blood pressure of 100–120 mmHg. Primary drug choices and example regimens include:
Esmolol – a loading dose of 200 μg/kg/min is given followed by an infusion of 50 μg/kg/min for 4 min. Further loading doses and increased maintenance doses may be used as required.
Labetalol – an initial bolus of approximately 0.25 mg/kg with further boluses at 10 min intervals as required. Once blood pressure is adequately controlled an infusion can be started.
Metoprolol – given as a bolus of up to 5 mg, repeated at 5-min intervals if required up to 15 mg.
Although labetalol is frequently used for blood pressure control in this clinical setting, it should be borne in mind that the drug has an elimination half-life in excess of 4 h and that changes in the rate of infusion will not yield steady-state plasma levels for 16–20 h. Despite labetalol’s popularity it is singularly unsuitable for minute-to-minute blood pressure control.
In patients intolerant of beta-blockers non-dihydropyridine calcium channel blockers such as diltiazem or verapamil are accepted alternatives. 23
The use of vasodilators may also be required to aid rapid blood pressure control. These include:
Sodium nitroprusside (SNP) – given as an infusion titrated to effect at 0.5–1.5 μg/kg/min.
Glyceryl trinitrate (GTN) – given as an infusion titrated to effect at 2–10 mg/h.
Both SNP and GTN may produce a reflex tachycardia that can exacerbate myocardial ischaemia.
Transfer
Patients presenting to non-specialist centres will require urgent transfer to a cardiothoracic centre. Transfer should not be undertaken until the patient has been resuscitated and stabilised, and therapeutic interventions should not be delayed while waiting for transfer. Physiological monitoring should, at a minimum, include electrocardiography, intermittent non-invasive blood pressure measurement and pulse oximetry.8,27 Continuous invasive arterial pressure monitoring should be used in patients receiving antihypertensive drugs.
Patients with acute aortic dissection should be considered critically ill and must be escorted by competent, trained and experienced personnel – typically a doctor and a paramedic or nurse. In determining the timing of transfer, factors such as the distance between the referring and specialist centre, the likely journey time, the availability of suitably trained escort staff and the condition of the patient must be borne in mind. Where transfer by ambulance can be predicted to exceed 90–120 min, or when a sea crossing is required, specialist helicopter or fixed wing aircraft transfer should be considered. The decision of whether to expedite transfer without an appropriate escort or to delay transfer pending the arrival of additional trained staff should be made by a senior doctor. A “scoop and run” philosophy is only appropriate on rare occasions when the urgency of the situation and the need for definitive treatment will limit the time available for stabilisation before transfer. 28 The rationale for expedited transfer should be clearly documented and must include the names and grades of the clinicians involved in the decision-making process.
A plan for dealing with acute deterioration of the patient during transfer should be agreed prior to departure. In the authors’ experience, diversion to an intermediate, non-specialist facility to permit further resuscitation is rarely, if ever, successful.
Copies of all medical records and the results of all relevant investigations should accompany the patient, however this should not delay transfer. The tradition of sending cross-matched packed red cells with the patient has largely been abandoned.
Surgical treatment of acute aortic dissection
Type A aortic dissection
Surgery is the standard treatment for acute type A aortic dissection. The basic principles are to resect the primary tear, establish blood flow in the true lumen and replace the dissected intrapericardial aorta. Surgery should generally be undertaken as soon as possible after diagnosis. 6
The surgical strategies depend on multiple factors:
The anatomy of dissection, which includes, the primary intimal tear location, the aortic dimension, and aortic valve condition.
Patient factors including age, comorbidities, redo surgery, Marfan syndrome or other aortopathy.
Dissection related complications especially malperfusion which may alter the management plan. In the presence of significant malperfusion, especially bowel ischaemia, initial treatment may be endovascular fenestration to restore blood flow in true lumen and then surgery.
Common surgical procedures include:
Replacement of ascending aorta with an interposition tube graft – if the intimal tear is limited to the ascending aorta.
Replacement of the ascending aorta with an interposition tube graft and aortic valve replacement – if the aortic valve is significantly stenosed or regurgitant and not amenable to repair.
Replacement of the ascending aorta with an interposition tube graft and aortic valve repair – if the aortic valve is regurgitant.
Aortic root replacement (with aortic valve replacement or a valve sparing procedure) if the intimal tear extends into the aortic root or there is an aortopathy such as Marfan’s Syndrome.
Replacement of the whole or part of the arch is indicated in addition if the primary tear extends or lies in the aortic arch.
The Frozen Elephant Trunk technique is a branched graft with stent in situ. The graft is used to replace the aortic arch with reimplantation of supra-aortic vessels and the stent is deployed in the descending aorta. The stent helps to remodel the aorta and can also be used as a landing zone for future endovascular stenting options. There is increasing evidence that this technique lowers the likelihood of future reintervention.
Other associated procedures can also be required, depending on the CT and TOE findings, that is, coronary artery bypass grafting, a mitral valve procedure, etc.
Surgical principles
Initiating cardiopulmonary bypass:
It is essential to establish cardiopulmonary bypass as soon as possible with arterial return into the true lumen. Preferred cannulation strategies include, direct cannulation of the ascending aorta (under TOE guidance), or indirect cannulation of the axillary artery or femoral artery. In an emergency situation, it may also be necessary to cannulate the LV apex or directly cannulate the true lumen after transecting the aorta. Venous drainage is usually through the right atrium but it may be necessary to use femoral cannulation in patients who have undergone cardiac surgery in the past6,29
2. Cerebral protection:
Another important aspect of surgery is cerebral protection. The distal anastomosis on the ascending aorta should be performed without the use of a cross clamp therefore cerebral blood flow will be interrupted. This is also the case if aortic arch replacement is required There are two strategies for cerebral protection during this phase of the operation.
a. Deep hypothermic circulatory arrest. At 20°C, cerebral metabolism and oxygen consumption are approximately 20% of normothermic level. This gives a safe period of circulatory arrest of 30–40 min. This is usually adequate time to carry out a simple distal anastomosis.
b. Antegrade cerebral perfusion. This is used when aortic arch replacement is required. It may be unilateral, achieved by cannulating the right axillary artery and clamping the innominate, left common carotid artery and left subclavian arteries. With a complete Circle of Willis this will provide bilateral cerebral perfusion and is necessary to ensure total cerebral perfusion. Alternatively, directly cannulating the innominate artery and the left common carotid artery provides bilateral antegrade cerebral perfusion. Both techniques are used with a degree of hypothermia that is usually less than with deep hypothermic circulatory arrest. 30 If there is dissection extending into the carotid arteries then antegrade cerebral protection is usually achieved by direct cannulation of the carotid arteries as retrograde circulation through the axillary artery can cause further extension of a dissection.
Spinal drain insertion
Although insertion of a cerebrospinal fluid drain is commonly used prior to elective major thoracic aortic surgery, pressure of time and concern about the adverse impact of impaired coagulation in the presence of acute type A dissection deters many clinicians from using them. The role of a spinal drain in major thoracic or thoracoabdominal aortic surgery is to maintain a stable intrathecal pressure to reduce the risk of paraplegia. It is also used in complex stent procedures in the descending aorta. Reversal of paraplegia, detected after emergence from sedation after surgery using a drain, has been described.
Early post-operative complications
In hospital mortality after surgical repair for type A aortic dissections was 17.4% in data from the National Adult Cardiac Surgery Audit between 2009 and 2018 with a trend to a lower mortality in the more recent years. 31 However, in hospital mortality with medical management remains high at 57% from the international registry of acute aortic dissection (IRAD) data. 15 Multiple organ dysfunction is common after emergency aortic dissection repairs with post operative dialysis rates of about 15%. Stroke occurs in 9%–10% of patients with rates unchanged over this 10-year time period.
As well as the range of complications associated with any cardiac surgical operation there are two specific early complications after aortic dissection repair:
Bleeding due to coagulopathy associated with hypothermia and prolonged cardiopulmonary bypass.
Persistent or new malperfusion. Pre-operative malperfusion may persist after surgery or the alteration of the dynamics of blood flow between the true and false lumens after surgery, may cause new malperfusion. Malperfusion is generally treated with interventional radiological techniques.
Post-operative spinal cord perfusion optimisation
Spinal ischaemia is a significant potential complication following aortic dissection repair. Post-operative management should include neurophysiological and intracranial/intrathecal pressure (ICP) monitoring and CSF drainage for up to 72 h. Neurological assessment should be performed regularly to allow for the timely identification of any deficit.
If a neurological deficit is identified, measures should be instigated to optimise spinal cord perfusion and thus negate its progression. The COPS protocol describes an approach to its management 32 :
◦ If malfunction, then replace drain. ◦ If normal, ensure the patient is flat, with a drain pressure of <5 mmHg, and drain for 7 days.
Type B aortic dissection
The first line of treatment of type B dissection is medical management, control of blood pressure and analgesia. Approximately 90% of type B dissections can be managed in this way in the acute phase. However, intervention is required if the type B dissection is complicated.6,29
Complicated type B aortic dissection is characterised by:
Persistent refractory pain: This indicates risk of impending rupture, bowel or visceral ischaemia and should be considered as one of the early features for intervention.
Malperfusion: In type B dissection, malperfusion may be dynamic or fixed. Depending on the vessel involved the presentation can vary. The patient present with acute abdominal pain, distension or acute limb ischaemia.
Rupture or leak: A leak can usually be identified on a CT scan but may present as persistent, non-resolving pain.
Rapid aortic expansion/dilatation: To assess for this a repeat CT is done 48 h after diagnosis and earlier if any signs of complications are present.
If any of these features are present, then intervention is indicated. Typically, this would be an endovascular intervention, placing a covered stent over the entry tear in the descending aorta. To ensure an adequate proximal landing zone it may be necessary to occlude the left subclavian artery.6,29
Deciding when not to operate
All type A dissection cases needs to be discussed with cardiac centres as the decision not to operate is a complex process. Multiple factors are considered in the decision process including patients age, frailty, previous cardiac surgery, dementia, and malignancy. Stroke or malperfusion is not a contraindication for surgery.
Post-operative care
Analgesia
The extent of the operation means significant analgesia is required to ensure the patient can mobilise and ventilate adequately. Multimodal analgesia should be instigated. Patients may benefit from epidural catheter insertion, but it is important to maintain a balance between sensory and motor block so analgesia can be maintained whilst permitting mobilisation.
Therapist input
The main post-operative physiotherapy priorities are prevention of secondary respiratory complications, early rehabilitation, non-pharmacological management of delirium and education.
Respiratory
Post-operative pulmonary complications (PPCS) are frequently reported following aortic dissection repair. Between 15% and 55% of patients develop acute respiratory dysfunction post-operatively, leading to an increased number of days requiring mechanical ventilation and longer ICU length of stay (LoS).33,34 Physiotherapy aims to reduce the incidence of PPCs by:
Increasing lung volume
Removing secretions
Reducing the work of breathing. 35
The beneficial effects of early mobilisation in mitigating PPCs following cardiac surgery have been well established and are considered standard practice within cardiothoracic critical care. 36 Early mobilisation should be the first line post-operative rehab approach unless contraindicated. Where early mobilisation is not permitted further strategies can be utilised including optimisation of patient positioning, mechanical insufflation/exsufflation and non-invasive ventilation.
Rehabilitation
In elective cases, patients are ordinarily reviewed in a pre-assessment clinic where pre-surgical functional status can be determined. The use of outcome measures such as the clinical frailty scores aids identification of patients at higher risk of reduced functional ability post-operatively. 37 For non-elective cases this is often not possible and communication with the patients next of kin and family are vital to establish functional baseline. Family and friends play an important role in the rehabilitation process and should be provided with regular updates and involved in rehabilitation as much as possible with consideration of the patients’ wishes.38,39
Rehabilitation in critical care is provided following comprehensive individual assessment by the physiotherapist, with the formulation of a rehabilitation plan and patient orientated goals. 38 Exercises in critical care include sitting on the edge of the bed, sitting out of bed using an appropriate method of transfer and walking with assistance or aid. More advanced rehabilitation is administered once the patient improves to standing and walking unaided or new neurology has been identified. 40
Assessment of patients in critical care occurs within 24 h of admission and thus physiotherapists are in prime position to detect any new neurology symptoms. Spinal cord infarct (SCI) is a rare and serious complication following aortic dissection repair and occurs every 1 in 130 patients. 41 The management of a patient with SCI following surgical repair will differ from the management of a postoperative dissection, this has not been discussed in detail here.
Post-operative cerebral complications (PCC) are common post-operatively and include new onset stroke, syncope, delirium and coma. 42 Where PCCs have been identified, individualised rehabilitation plans and patient orientated goals are set alongside specialist referral. Physiotherapists play a key role in the non-pharmacological management of delirium, this includes:
early mobilisation
daily orientation
engagement with family members
spontaneous awakening and breathing trials
sleep promotion
daily assessments of delirium using tools such as the confusion assessment method. 39
Patient education
In addition to respiratory care and rehabilitation, physiotherapists promote healthy lifestyle changes through education and referral to appropriate services. This includes smoking cessation and providing guidance on exercise and lifestyle choices to promote recovery and prevent further complications post-operatively.
Discharge from critical care
Management in intensive care is required while organ dysfunction persists. Once organ dysfunction has resolved or resolved sufficiently to be managed outside a critical care environment patients may be safely managed in a ward environment. However, prior to discharge a multi-professional plan should be developed to guide further care. This should involve cardiac surgery, critical care, cardiology, physiotherapy and occupational therapy; with many patients also requiring radiology, neurology and renal medicine to be involved. Discharge destination should be determined by the needs of the patient with some patients needing ongoing dialysis for acute kidney injury. Such plans must involve patients and their next of kin in what is sometimes complex decision making balancing the need of the patient to be close to home with their clinical needs. Patients with aortic dissection will need lifelong follow up as they are at risk of further aneurysmal complications and will require effective blood pressure control typically under 120/80 mmHg to reduce that risk.
Summary
Aortic dissection represents a condition with significant mortality and morbidity, resulting in more than 2000 deaths per year in the UK. Successful treatment relies on rapid diagnosis, strict blood pressure control, minimal delays to definitive treatment and MDT input. Critical care clinicians play a key role in the aortic pathway and thus a thorough understanding of their management is vital.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GC is a trustee of The Aortic Dissection Charitable Trust; LF is an Associate Editor for the Journal of the Intensive Care Society and a member of The Aortic Dissection Charitable Trusts education team.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
