Abstract
The aim of this study was to assess the feasibility and outcome of a neuropsychiatric evaluation protocol intended for adult intensive care unit survivors in a Danish regional hospital, in which a follow-up consultation was conducted 2 months after hospital discharge. Twenty-three participants were able to finalize the neuropsychiatric evaluation, and 20 (87%) among those were detected with neuropsychiatric manifestations, including cognitive impairment (n = 17; 74%) and fatigue (n = 17, 74%). This study finds a high prevalence of neuropsychiatric manifestations and fatigue, and evaluates a follow-up protocol for the ICU patient population.
Background
Patients with critical illness admitted to an intensive care unit (ICU) are at risk of developing post-intensive care syndrome (PICS) in which cognitive impairment have been reported as common late manifestations.1–5 Comorbidity, age, obesity and substance abuse,1–5 pathophysiological factors 5 and ICU-related factors such as mechanical ventilation, conservative fluid administration, delirium and ARDS. 5 are associated with development of PICS. Despite that almost half of ICU patients report of symptoms of fatigue after discharge, 6 assessment of neuropsychiatric manifestations following ICU treatment is rarely performed in a systematic manner.
Here, we evaluate the feasibility and outcome of a neuropsychiatric evaluation programme in which neuropsychiatric manifestations were assessed in ICU survivors.
Methods
Design
During a 12-month prospective study period, patients (⩾18 years old) with a history of ⩾72 h of admission at our combined medical and surgical intensivist/anesthesiologist-staffed ICU at North Denmark Regional Hospital (teaching hospital) were invited to a follow-up consultation 2 months after discharge from the ICU. The 2-month timeframe was chosen in order to give patients enough time to obtain experience from their daily activities. Participant selection and exclusion criteria are shown in Figure 1.
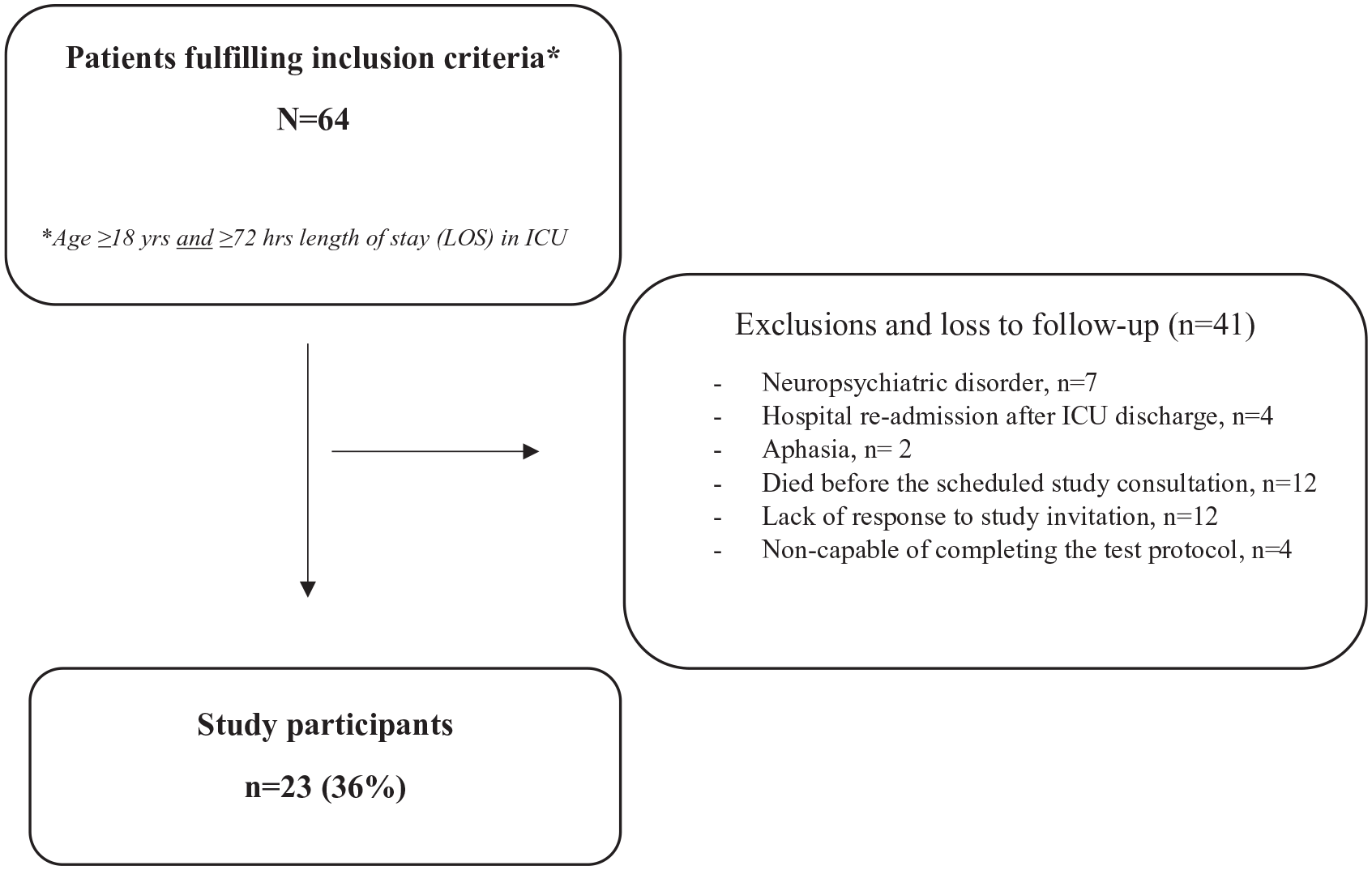
Study enrolment flowchart showing the total number of patients fulfilling the inclusion criteria in the 12-month enrolment period and the final number of study participants.
Patient data
Patient admission data, background and medical history was extracted from patient medical records.
Neuropsychiatric assessment
A semi-structured interview and neuropsychiatric testing were performed by a neuropsychologist (KK) as part of the follow-consultation protocol. Cognitive function was tested by three index tool components in Weschler Adult Intelligence Scale (WAIS) III 7 ; Working Memory Index (WMI), Processing Speed Index (PSI) and Perceptual Organization Index (POI). The subtests of each component were compared to an adjusted mean of 10 with standard deviation (SD) of 3, where a score <10 indicated lower performance than expected. Presence of fatigue were assessed by the validated Fatigue Severity Scale (FSS) questionnaire. 8 A mean score ⩾4 and was considered indicative of moderate to severe fatigue. Finally, the Major Depression Inventory (MDI) and Anxiety Symptoms Scheme (ASS) were also incorporated in the protocol.
Statistical analysis
Statistical analysis was carried out using Prism GraphPad software 6.0. p-Values ⩽0.05 was considered statistically significant.
Results
Clinical characteristics
Out of 64 patients fulfilling the inclusion criteria, one-third (n = 23) completed the follow-up consultation (Figure 1). Mean age (SD) of the 23 study participants were 64.9 (±8.3) years, and 12 participants (52%) were males. Mean Charlson Comorbidity Index (CCI) of 3.9 (±1.9) and Median Simplified Acute Physiology Score (SAPS) III of 52 (IQR: 46–60). The majority of participants (91%) had been admitted to the ICU due to both surgical and medical gastrointestinal disease (n = 14) and/or respiratory failure (n = 7). Eleven (48%) participants were treated for septic shock. Moreover, 12 participants (52%) were diagnosed with delirium based on a positive outcome of the Confusion Assessment Method of the Intensive Care Unit (CAM-ICU), and 16 participants (70%) had been assisted by mechanical ventilation, and 5 participants (22%) received non-invasive ventilation (NIV) therapy. Six patients (26%) were diagnosed with critical illness polyneuropathy (CIN). Median length of ICU stay (LOS) was 10 days (IQR: 6–18).
Neuropsychiatric outcomes
Overall, 20 participants (87%) had neuropsychiatric manifestations, including cognitive impairment (n = 17; 74%) and fatigue (n = 17, 74%). Mean values (SD) for WMI, PSI and POI were 6.9 (±1.4), 6.2 (±3.1) and 9.2 (±2.6), respectively. Mean FSS score in the study population was 5.0 ± 1.13 (range 2.8–6.9). Five (22%) and three (13%) participants were observed with mild to severe depression and anxiety disorder, respectively (Table 1).
Summarized results from the follow-up interview. Prevalence of abnormal test scores, as well as test results in subgroups are presented.
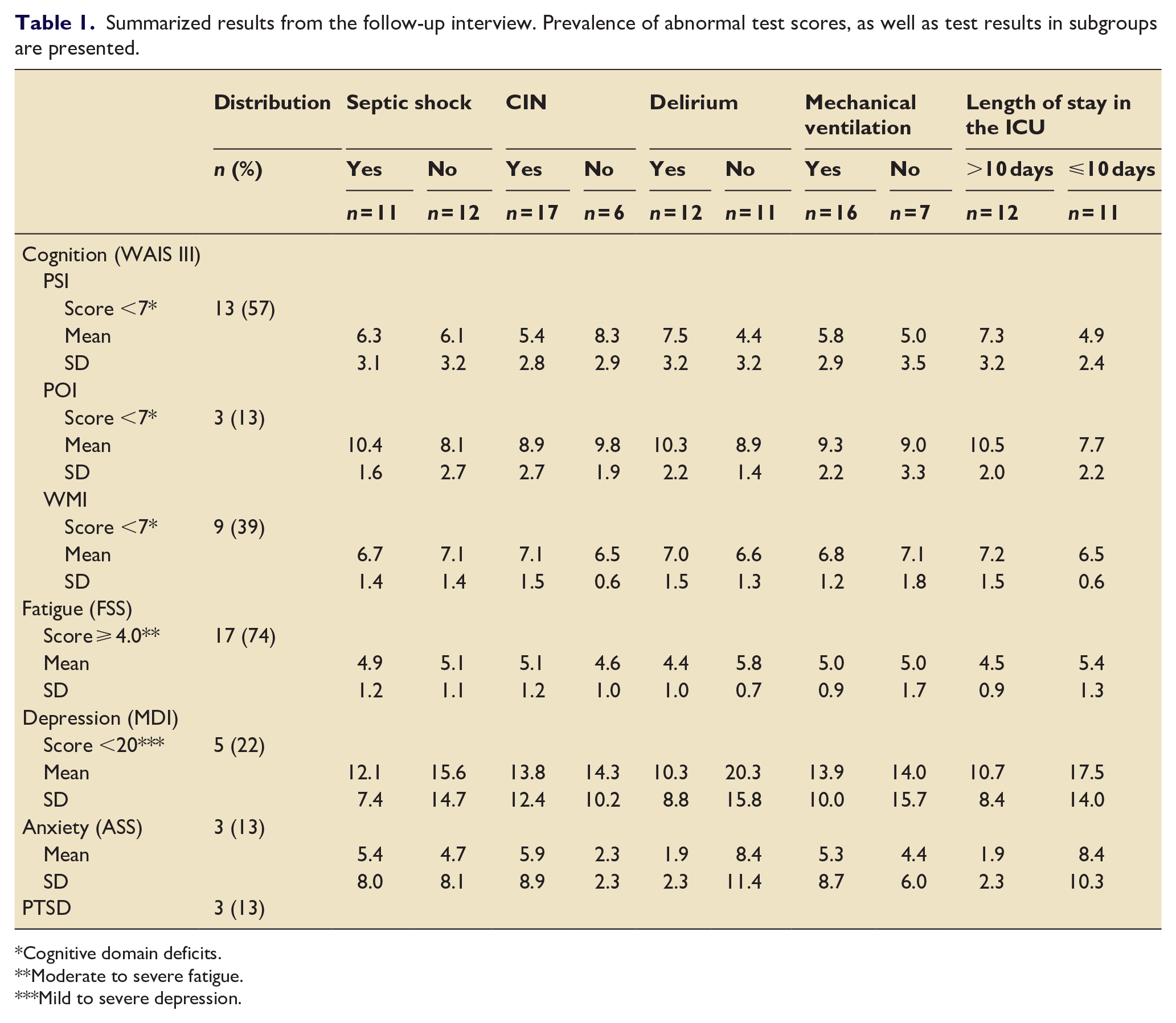
Cognitive domain deficits.
Moderate to severe fatigue.
Mild to severe depression.
Evaluation of follow-up interviewing
Twenthy-three patients out of 35 ICU survivors participated in the follow-up interview, whereas 12 patients did not respond to invitation, and 4 did not fully complete the tests, reaching a participation rate of 66% and a completion rate of 83%.
Discussion
This study reports a high presence of late neuropsychiatric manifestations in ICU survivors in line with other studies.1–6,9 In particular, cognitive impairment and fatigue are prevalent manifestations. Knowledge about the neuropsychiatric status of the participants prior to ICU admission was sparse or virtually non-existent, and therefore it is not possible to make a valid causality evaluation regarding the origin of the neuropsychiatric manifestations. However, of notice our study population was performing worse in the WAIS III cognitive test, when compared to a demographically matched background population. 10
A limitation in this study is the small number and proportions of participants, in addition to the lack of a control group. Additionally, loss to follow-up due to post-ICU mortality, incomplete test or lack of response to study invitation may skew our results, since we suspect that cognitive impairment is a direct factor leading to patients not participating or completing the interview. This indicates that a follow-up interview should be planned carefully with regards to timeframe and method.
In conclusion, the study has demonstrated that assessment of neuropsychiatric manifestations is feasible and that fatigue and cognitive impairment is high in prevalence. Future research should involve larger study populations and aim to identify risk exposures and risk patient subgroups in order to implement early focused rehabilitation before hospital discharge.
Footnotes
Acknowledgements
We would like to acknowledge Rikke Borregaard, Åse Taber, Maibritt Rishøj and Malgorzata Beata Pawlowicz-Dvorzanska for their dedicated support in enrolment of study participants.
Abbreviations
ARDS: Acute Respiratory Distress Syndrome, ASS: Anxiety Symptoms Scheme, CAM-ICU: Confusion Assessment Method for Intensive Care Unit, CCI: Charlson Comorbidity Index, CIN: Critical Illness Neuropathy, FSS: Fatigue Severity Scale, ICU: Intensive Care Unit, IQR: Interquartile range, LOS: Length of Stay, MDI: Major Depression Inventory, NIV: Non-Invasive Ventilation, PICS: Post-Intensive Care Syndrome, PTSD: Post Traumatic Stress Disorder, POI: Perceptual Organization Index, PSI: Processing Speed Index, RLS: Restless Leg Syndrome, SAPS: Simplified Acute Physiology Score, SD: Standard Deviation, WAIS: Weschler Adult Intelligence Scale, WMI: Working Memory Index.
Author contributions
MK, TO, PL, KK, HA and SF designed study: MK, KK and HA did data collection. SF, PL and KK did data analysis. SF, PL, MK and TO drafted manuscript. All authors approved the manuscript before submission, and gave written consent before submission
Availability of data and materials
Data and materials can be accessed and reviewed by contacting the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
The study was conducted as part of a clinical quality control programme at the North Denmark Region Hospital. Hence, approval from the North Denmark Region Committee on Health Research Ethics was not required, whereas the study was reported to the Danish Data Protection Agency. Trial registration was not considered relevant.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Marie Pedersen and Jensine Heiberg Foundation provided financial support to the study.
