Abstract
Background
There is no consensus on the instruments for diagnosis of post-intensive care syndrome (PICS). We present a proposal for a set of outcome measurement instruments of PICS in outpatient care.
Methods
We conducted a three-round, semi-structured consensus-seeking process with medical experts, followed each by exploratory feasibility investigations with intensive care unit survivors (n1 = 5; n2 = 5; n3 = 7). Fourteen participants from nine stakeholder groups participated in the first and second consensus meeting. In the third consensus meeting, a core group of six clinical researchers refined the final outcome measurement instrument set proposal.
Results
We suggest an outcome measurement instrument set used in a two-step process. First step: Screening with brief tests covering PICS domains of (1) mental health (Patient Health Questionnaire-4 (PHQ-4)), (2) cognition (MiniCog, Animal Naming), (3) physical function (Timed Up-and-Go (TUG), handgrip strength), and (4) health-related quality of life (HRQoL) (EQ-5D-5L). Single items measure subjective health before and after the intensive care unit stay. If patients report new or worsened health problems after intensive care unit discharge and show relevant impairment in at least one of the screening tests, a second extended assessment follows: (1) Mental health (Patient Health Questionnaire-8 (PHQ-8), Generalized Anxiety Disorder Scale-7 (GAD-7), Impact of Event Scale – revised (IES-R)); (2) cognition (Repeatable Battery for the Assessment of Neuropsychological Status (RBANS), Trail Making Test (TMT) A and B); (3) physical function (2-Minute Walk Test (2-MWT), handgrip strength, Short Physical Performance Battery (SPPB)); and (4) HRQoL (EQ-5D-5L, 12-Item WHO Disability Assessment Schedule (WHODAS 2.0)).
Conclusions
We propose an outcome measurement instrument set used in a two-step measurement of PICS, combining performance-based and patient-reported outcome measures. First-step screening is brief, free-of-charge, and easily applicable by health care professionals across different sectors. If indicated, specialized healthcare providers can perform the extended, second-step assessment. Usage of the first-step screening of our suggested outcome measurement instrument set in outpatient clinics with subsequent transfer to specialists is recommended for all intensive care unit survivors. This may increase awareness and reduce the burden of PICS.
Trial registration
This study was registered at ClinicalTrials.gov (Identifier: NCT04175236; first posted 22 November 2019).
Keywords
Background
Chances of survival after critical illness have dramatically increased in the last decades, generating a new cohort of intensive care unit (ICU) survivors who suffer from medium- and long-term impairments as a result of ICU care.1–4 Research into these outcomes has become a major target of critical care, as ICU survivors commonly face problems in the three domains of mental health, cognition, and physical function.1,3,5–10 Regarding mental health, ICU survivors frequently suffer from anxiety, post-traumatic stress disorder, and depression.11–13 As to cognition, ICU survivors show impairments in major domains, including memory, attention, verbal fluency, and executive functioning.14–17 ICU survivors are also at higher risk for dementia. 18 Impairments in physical function include exercise limitations and limitations in activities of daily living.19–21 Participants of a pioneering stakeholders' conference in 2010 suggested the umbrella term ‘post-intensive care syndrome’ (PICS) to cover persistent impairments in a variety of outcomes. 6
There is heterogeneity and lack of consensus about appropriate, feasible outcome measures for PICS.8,9,11–14,16,17,19,22–25 A recent systematic review of Robinson et al. concluded that none of the measures of the PICS domains had an adequate evidence base for sufficient measurement quality in survivors of critical illness. 2 Nevertheless, they found promising psychometric results for the Hospital Anxiety and Depression Scale (HADS), 26 the Impact of Event Scale – revised (IES-R), 27 and the EQ-5D.28–31 Needham et al. developed a set of outcome measurement instruments (OMIs) for survivors of acute respiratory failure, consisting of the HADS and IES-R for mental health, and the EQ-5D and/or the Short Form-36, version 2, for HRQoL, but did not achieve a consensus regarding measures of cognition or physical function. 32
Given the dynamic state of evolving PICS research, it seems premature to define PICS core outcome sets (COS) with sophisticated and rigorous methodological COS standards.33–35 Rather, clinical studies should first establish potential OMIs for PICS as a basis for future COS development.33–38 Further, OMIs should fulfill some basic requirement apart from general considerations of validity. Specifically, they should include patient reported as well as objective measurements, their application should be feasible in the vulnerable target population and they should be free of charge. In addition, questions of their implementation across different health care settings need to be explored. For example, Needham et al. discussed a stepwise approach for PICS diagnosis. 6 Screenings might detect potential PICS patients in primary care settings, and an extended assessment by specialized clinicians could confirm screening results for therapy initiation. 39 In this work, we propose a set of OMIs to measure PICS in settings of outpatient healthcare service as well as a feasibility assessment of these OMIs.
Methods
General approach
We conducted a three-round, semi-structured consensus-seeking process, informed by the results of recent systematic reviews and consensus statements on outcomes in ICU survivors. After each consensus meeting, we conducted small pilot studies with samples of survivors from critical illness. The process is illustrated in Figure 1. All patients consented to participate in feasibility testings, and we complied to standards of the Declaration of Helsinki. This work was approved by the Institutional Review Board of the Medizinische Hochschule Brandenburg (record: E-01-20190619) and the Ethics Committee of Charité – Universitätsmedizin Berlin (record: EA4/122/19). The set of OMIs was developed in the context of the study ‘Enhanced Recovery after Intensive Care’ (ERIC) that investigates the impact of a telemedical critical-care intervention on quality of care and functional outcomes (CTN-identifier: NCT03671447). Within ERIC, functional outcomes are assessed using the OMI set proposed in the work at hand.
Illustration of the three-round, semi-structured consensus-seeking process.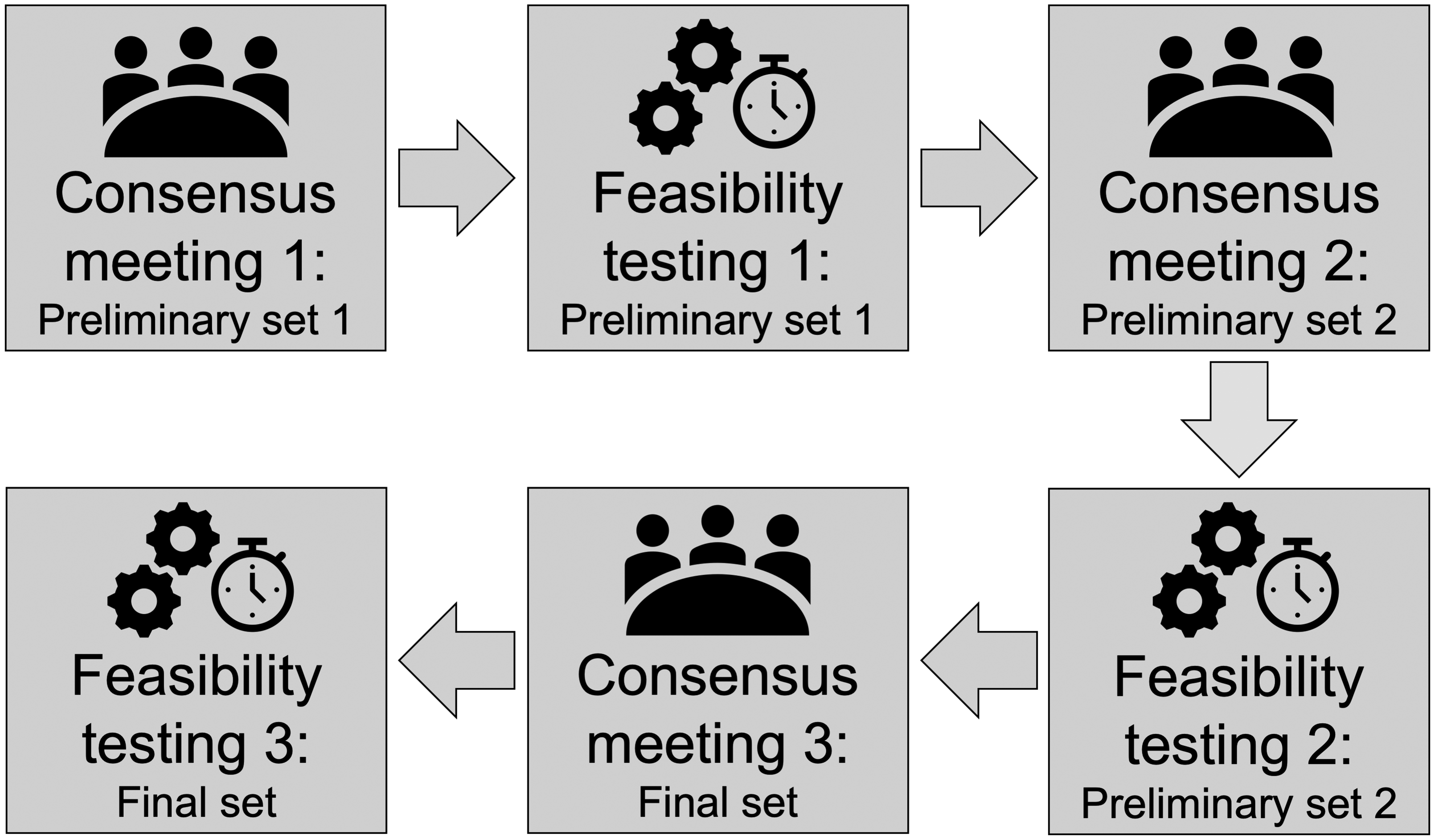
Expert participants
Members of the interdisciplinary, semi-structured consensus meetings comprised nine health care professionals from critical care and anesthesiology, including nursing staff and physicians, a respiratory therapist, a psychotherapist, a sepsis researcher, and a physical therapist. Further, two researchers on health care management, two telehealth researchers, and a clinical psychologist with a focus on outcome and process research participated in the consensus meetings. Half of the participating experts were women. The decision for the final OMI set was made by a core group of six clinical researchers (CDS, BW, CD, JKi, HK and KSt).
Consensus meetings
In preparation of the consensus meetings, two senior physicians (BW, CH) and two senior psychologists (CD, HK) reviewed recent evidence on outcomes in ICU survivors.2,6,8,11–14,32,39 Based on this literature, a preliminary OMI set was chosen and discussed in the consensus meetings. Predefined selection criteria were: (1) free-of-charge, non-commercial application; (2) time for completion of the set not exceeding 20–30 min in total; (3) potentially administrable by a variety of clinical practitioners; (4) validated measurement properties in clinical patient populations, including adults of all age groups; application in and psychometric data from ICU survivorship research were considered beneficial but not necessary; (5) existence of a validated German version.
Feasibility test and data analysis
After each consensus conference, a convenience sample of survivors from critical illness was chosen to assess feasibility and to qualitatively analyze the experience of survivors and examiners. Patients suffered from critical illness with main diagnoses from surgery, neurology, or internal medicine. They were either treated at Charité – Universitätsmedizin Berlin or Brandenburg Clinic in Bad Belzig. Feasibility testing was performed with five patients after the first consensus conference, five patients after the second consensus conference, and seven patients after the third consensus conference. After each feasibility testing, interviewers and patients reported on their experiences during the assessment with emphasis on completion time, strains the tests imposed on patients, and subjective operability of the set. OMI results were evaluated according to test manuals and cut-off values, and descriptive statistics (median, range, rate of patients exceeding cut-offs) were calculated. Upon completion of the final feasibility testing, four experts (BW, NP, HK, CD) subjectively rated the feasibility of each OMI set based on practicability, applicability, and ability to identify PICS on a scale from 0% (not feasible) to 100% (perfectly feasible).
Results
Consensus meeting outcomes: Preliminary Set 1
The preliminary OMI set included Patient Health Questionnaire-9 (PHQ-9),40,41 Generalized Anxiety Disorder Scale-7 (GAD-7), 40 Impact of Event Scale (IES), 42 MiniCog,43,44 Animal Naming, 45 Timed Up-and-Go (TUG), 46 and EQ-5D-3L.47,48 Two single items assessed current subjective physical and mental health concerns in terms of current distressing problems that are frequent in ICU survivors. Four visual analogue scales (VAS) were presented as health thermometers and measured self-reported subjective physical and mental health on a scale from 0 to 100, each both prior to ICU treatment and in the week prior to the assessment.49–53
Feasibility testing: Preliminary Set 1
In the first feasibility testing, the preliminary OMI set was piloted with five survivors of critical illness. Three of those patients did not consent to publishing their results and one did not complete the test set; therefore, they were excluded from analysis. PHQ-9, GAD-7, MiniCog, Animal Naming, and EQ-5D-3L were easily completed. For the TUG, the patient was unable to walk. The IES as well as the VAS health thermometers were difficult to understand for both patient and health care professional who administered the tests and questionnaires. Completion of the assessment took approximately 40 min.
Consensus meeting outcomes: Preliminary Set 2
Based on results of the first consensus meeting and feasibility testing, the IES was dropped, and the VAS thermometers were replaced by numerical rating scales (NRS) from 0 to 10.
Feasibility testing: Preliminary Set 2
In the second feasibility testing, the revised OMI set was piloted with five survivors of critical illness. One patient did not consent to publishing results and was therefore excluded from analysis. Instruments were completed by patients without any problems. Overall, the assessment took between 30 and 40 min per participant and was deemed too long and extensive by both patients and examining clinical health professionals.
Consensus meeting outcomes: Final set
The core group agreed that the OMI set was still too time-consuming to be used in outpatient, primary health care settings. In addition, the outcome measures were considered not granular enough to differentiate between varying intensities of PICS. Furthermore, there were reliable and valid updates of the EQ-5D47,48 and IES,
65
as well as ultra-short versions of the PHQ and the GAD.40,41 Finally, handgrip strength, as measured with hand-held dynamometry, was considered a valid indicator of muscle strength and physical health. Taking together the abovementioned reasons, members of the expert group decided to divide the OMI set in two steps, with a short screening and an extended assessment. Patients showing new or worsening health problems after ICU and exceeding at least one of the cut-off points in the screening tests participate in a second, more comprehensive PICS assessment. Figure 2 and Table 2 show the questionnaires, tests, and cut-off points of both steps of the final OMI set.
Proposal of a set of outcome measurement instruments (OMIs) of post-intensive care syndrome (PICS).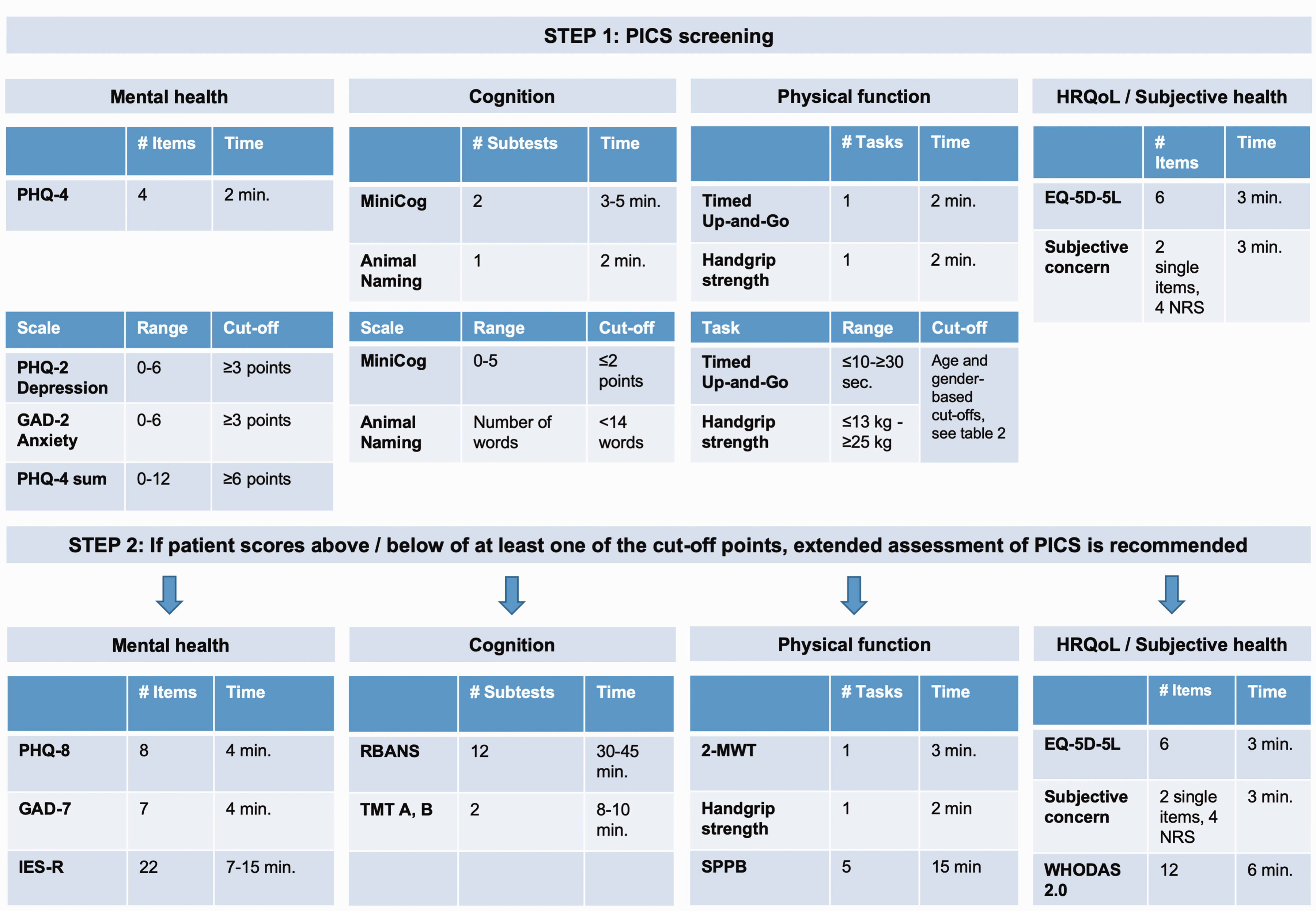
Screening
The screening consists of a combination of brief tests covering PICS domains. After a short instruction, they can be administered by a variety of health care professionals:
Physical function: Timed Up-and-Go (TUG),
46
handgrip strength.
66
New or worsening health problems: Two single items assess whether a patient currently has subjective mental, cognitive, or physical health concerns. Comparing the NRS ratings of subjective physical and mental health indicates whether current health impairments are new problems or worsening of conditions that have already existed prior to ICU admission. The screening takes about 20 min per patient.
Extended assessment
The extended assessment includes more elaborate and comprehensive OMIs and should be administered by experienced staff familiar with PICS patients. The standardized questionnaires and tests are:
Mental health: Patient Health Questionnaire-8 (PHQ-8),40,54 Generalized Anxiety Disorder Scale-7 (GAD-7),
40
Impact of Event Scale – revised (IES-R)
65
Cognition: Repeatable Battery for the Assessment of Neuropsychological Status (RBANS),62,67 Trail Making Test (TMT) A and B
68
Physical function: 2-Minute Walk Test (2-MWT),
57
handgrip strength,
66
Short Physical Performance Battery (SPPB)
69
Health-related quality of life: EQ-5D-5L,47,48 12-Item WHO Disability Assessment Schedule (WHODAS 2.0); versions for self-rating and proxy-rating exist for measuring both patient and caretaker evaluation, respectively.70,71 New or worsening health problems: The interviewer can assess the single items and NRS of subjective mental and physical health for a second time to include the resulting scores in a reassessment of the extent of patients' subjective health concerns. The extended assessment takes between 85 and 110 min per patient.
Feasibility testing: Final set
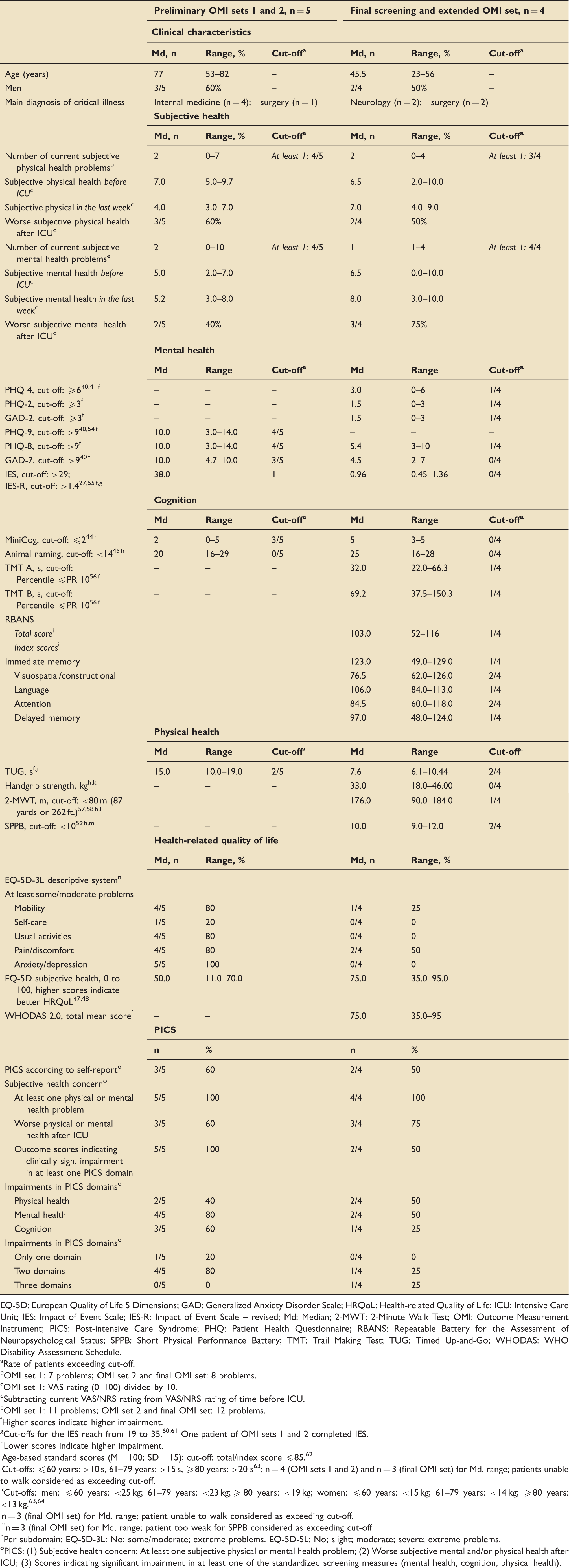
In the third feasibility testing, the screening and the more challenging extended assessment was piloted with seven survivors of critical illness. Two patients did not complete the testing and one did not attend the appointment. Four patients completed the extended assessment of the final OMI set. Patients were able to complete the instruments without any trouble, except one patient who was unable to walk and therefore could not conduct the TUG, the 2-MWT, and the SPPB. Table 1 displays detailed results. In the subjective feasibility rating of each OMI set by four experts (two medical doctors (BW, NP) and two senior psychologists (CD, HK)) on a scale from 0% (not feasible) to 100% (perfectly feasible), median feasibility scores increased from initially 60% for OMI set 1 to 97.5% for the final OMI set (Figure 3).
Expert feasibility rating of OMI sets. Results of nine pilot patients who completed the different versions of the OMI sets. EQ-5D: European Quality of Life 5 Dimensions; GAD: Generalized Anxiety Disorder Scale; HRQoL: Health-related Quality of Life; ICU: Intensive Care Unit; IES: Impact of Event Scale; IES-R: Impact of Event Scale – revised; Md: Median; 2-MWT: 2-Minute Walk Test; OMI: Outcome Measurement Instrument; PICS: Post-intensive Care Syndrome; PHQ: Patient Health Questionnaire; RBANS: Repeatable Battery for the Assessment of Neuropsychological Status; SPPB: Short Physical Performance Battery; TMT: Trail Making Test; TUG: Timed Up-and-Go; WHODAS: WHO Disability Assessment Schedule. Rate of patients exceeding cut-off. OMI set 1: 7 problems; OMI set 2 and final OMI set: 8 problems. OMI set 1: VAS rating (0–100) divided by 10. Subtracting current VAS/NRS rating from VAS/NRS rating of time before ICU. OMI set 1: 11 problems; OMI set 2 and final OMI set: 12 problems. Higher scores indicate higher impairment. Cut-offs for the IES reach from 19 to 35.60,61 One patient of OMI sets 1 and 2 completed IES. Lower scores indicate higher impairment. Age-based standard scores (M = 100; SD = 15); cut-off: total/index score ≤85.
62
Cut-offs: ≤60 years: >10 s, 61–79 years: >15 s, ≥80 years: >20 s
63
; n = 4 (OMI sets 1 and 2) and n = 3 (final OMI set) for Md, range; patients unable to walk considered as exceeding cut-off. Cut-offs: men: ≤60 years: <25 kg; 61–79 years: <23 kg; ≥ 80 years: <19 kg; women: ≤60 years: <15 kg; 61–79 years: <14 kg; ≥80 years: <13 kg.63,64 n = 3 (final OMI set) for Md, range; patient unable to walk considered as exceeding cut-off. n = 3 (final OMI set) for Md, range; patient too weak for SPPB considered as exceeding cut-off. Per subdomain: EQ-5D-3L: No; some/moderate; extreme problems. EQ-5D-5L: No; slight; moderate; severe; extreme problems. PICS: (1) Subjective health concern: At least one subjective physical or mental health problem; (2) Worse subjective mental and/or physical health after ICU; (3) Scores indicating significant impairment in at least one of the standardized screening measures (mental health, cognition, physical health). A two-step set of outcome measurement instruments (OMIs) of post-intensive care syndrome (PICS). Handgrip strength and EQ-5D-5L and should be administered within both screening and extended assessment.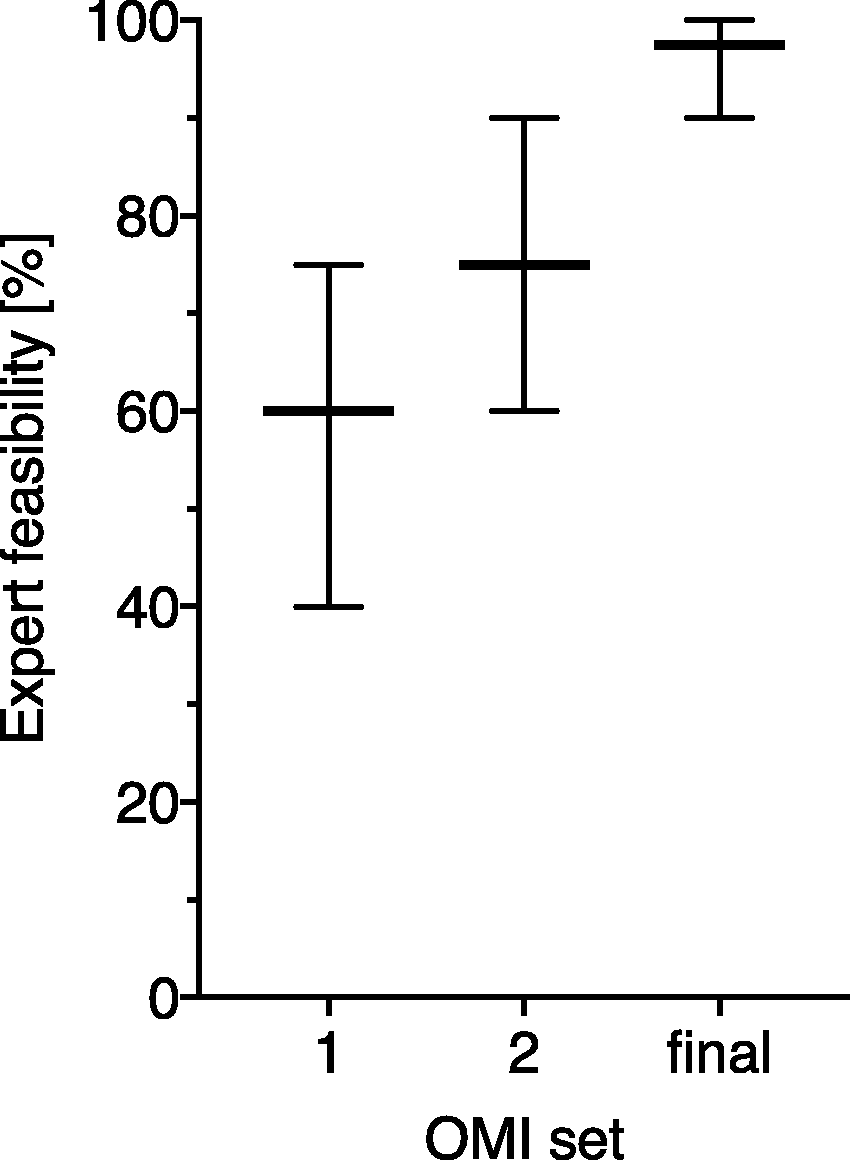

Discussion
Our suggested two-step OMI set presents a combination of performance-based and patient-reported outcome measures for PICS. The standardized screening tools of the first measurement step are brief, applicable by clinical practitioners in outpatient healthcare, and free-of-charge. The instruments are commonly used in clinical research and have validated measurement properties in patient populations. While the screening tests and questionnaires fulfill our defined selection criteria, the instruments of the extended assessment demand staff familiar with PICS patients and more time for completion.
Feasibility and measurement properties of the proposed OMIs
The different scales of the PHQ have proven psychometric quality, and they are frequently used as mental health outcome measures in clinical research.40,41,54,72 Previous or current ICU survivorship research has applied several of the PHQ scales.8,13,73–76 While the ultra-short depression and anxiety screening tool PHQ-4 is part of the proposed PICS screening, we suggest the 7-item Generalized Anxiety Disorder Scale (GAD-7) and the 8-item depression scale (PHQ-8) for the extended assessment. The measurement quality of the PHQ-8 is equivalent to that of the PHQ-9, which has an additional item for thoughts of death and self-harm.40,54 Since this item has not shown to be an accurate screen for suicide ideation, 77 we propose using the PHQ-8.
For the cognitive part of the screening process, we suggest the MiniCog and the verbal fluency test Animal Naming. Both tests are established and psychometrically sound screens for dementia.43–45,78–81 The MiniCog has shown feasibility in the context of preoperative frailty assessment in the elderly 82 and was used in two critical care studies.83,84
Remaining mental health and cognition tests of both screening and extended assessment are well validated in general clinical research and commonly used in ICU survivorship studies. There are investigations of measurement quality in ICU survivors for the IES-R2,8,12,23,32,39 and the EQ-5D,2,8,32,39 which should be used in its current revision EQ-5D-5L.47,48 The psychometric properties of the scales of the WHODAS 2.0 have been investigated in the general population and diverse clinical settings,70,71,85 and results suggest feasibility in critical care settings.86–88 Pivotal ICU survivorship research has applied the RBANS as well as TMT A and B. Unfortunately, to our knowledge, there are no studies elucidating measurement quality of these tools in ICU survivors.9,15,75,89,90 Users should also consider the licensing fees of the RBANS.
The single items and NRS of subjective health play a pivotal role to answer if health problems have occurred or worsened after the ICU treatment. Single items, VAS and NRS are frequently used in clinical research and practice, and sufficient measurement properties have been reported for the measurement of pain and general subjective health.49–53 Interestingly, previous data suggest that NRS have better compliance and usability than VAS and verbal rating scales.91,92 There is also established validity of the EQ-5D, which includes a blend of VAS and NRS for the rating of subjective health.47,48 However, the psychometric properties of the single items and NRS for measuring aspects of PICS-related subjective health still have to be investigated in ICU survivors.
For the screening of physical function, we suggest the TUG 46 and handgrip strength dynamometry. 66 The TUG was developed for and used on geriatric patients. 93 Recently, it has also been used in a perioperative setting as a measure of physical decline and recovery. 94 Handgrip strength is one of the basic methods for measuring muscle strength, 95 with normative values for all age groups.63,64 Several studies have assessed both TUG96–98 and handgrip strength dynamometry in ICU survivors.98–103 Some of these investigations also reported promising data on measurement quality of both tests in ICU survivors.96,100–103
For the extended assessment of the physical function, we recommend handgrip strength and two additional physical tests. Handgrip strength, EQ-5D-5L, the NRS, and single items regarding new and worsening health problems are already part of the screening, but a repetition in synopsis with the additional tests allows for a reassessment of the initial impairment. The 2-MWT was developed as a short version of the 6-Minute Walk Test (6-MWT) to account for primarily geriatric patients who are unable to walk for 6 min. 57 Bohannon et al. provide normative values based on a large population-based sample for all age groups above 18 years, but the authors do not report information on health status of the sample. 104 Two studies independently found a mean distance of around 80 m in participants of geriatric inpatient rehabilitation centers 57 and patients with COPD, 58 similar to findings from Connelly et al. 105 In comparison, Chan and Pin reported a mean distance of 75 m in frail elderly suffering from dementia without using walking aids, whereas shorter distances were observed in participants using walking aids. 106 Thus, we conclude that a cut-off of 80 m is feasible for ICU survivors. While the 2-MWT has, to our knowledge, not yet been investigated in ICU survivors, the 6-MWT has been applied in several ICU survivor studies.19,76,96,97,99,102,103 The SPPB consists of three tests on standing balance, walking speed, and standing up from a chair, with a sum score ranging from 0 to 12 points. In a recent meta-analysis, scores below 10 were associated with an increased risk for mortality in elderly. 59 In non-geriatric settings, the SPPB was used in cancer survivors, where it was predictive of survival- and treatment-related complications. 107 Recent ICU survivor studies utilizing the SPPB report favorable results concerning validity.100,101,108,109 Finally, our proposed OMIs for the physical function domain are also supported by feasibility of TUG,110,111 handgrip strength,111–113 6-MWT, 110 and SPPB 114 in patients of acute ICU settings, as well as currently running clinical trials in critical care patients with TUG, 115 handgrip strength115,116 and 2-MWT105 as outcomes.
Implications for clinical research and practice
It is yet unknown which of the outcomes and measurement instruments of PICS domains will prove relevant, reliable, responsive, and valid in future projects to develop a COS for PICS.2,8,32,39 Furthermore, variations in COS, OMIs, as well as definitions and quantification approaches might be appropriate, depending on diverse objectives, scopes, and settings. Different COS and OMIs may be necessary for observational studies versus clinical efficacy trials, and clinical research versus practice. Binary outcomes are needed to determine incidence rates, whereas measurement of change over time and treatment effects may be more successful with several specific continuous outcomes and/or elaborate composite scores.33–38
Here, we present an OMI proposal that is suitable to determine incidence and extent of PICS in settings ranging from primary outpatient healthcare to health centers experienced in PICS. The suggested measures of PICS domains have well-established cut-off points in both the screening and the extended measurement, thereby making an operationalization and quantification of a binary PICS outcome possible. The results may define PICS of at least mild extent: (1) Subjective physical, mental and/or cognitive health concern; (2) lower current subjective mental and/or physical health, as compared with ratings prior to ICU treatment; (3) scores indicating clinically significant impairment in at least one of the screening measures. Yet, the characterization of different grades of PICS severity remains unknown.7,9 In the suggested OMI set, severe PICS might be identified in the extended assessment if results indicate significant impairment in two or three PICS domains, or by substantial impairment in at least one of the more comprehensive PICS measures. In order to determine substantial impairment, more extreme cut-off points are needed, e.g. scores exceeding two standard deviations of a normative sample, as applied in the DSM-5 features of major neurocognitive disorder. 117 The specific scores of EQ-5D-5L and WHODAS 2.0 may contribute to the evaluation of the severity of PICS by investigating to which extent physical, mental, and cognitive health outcomes are related to HRQoL outcomes. A structured approach for the diagnosis of PICS would allow for individually tailored treatments in the conspicuous domains. Tailored treatment concepts might include individual physical therapy, psychotherapy, memory training, or home nursing services.
Limitations and future directions
Although the number of participants in the consensus process was comparatively low, feedback was comprehensive and partially similar. Nevertheless, future studies should test the proposed OMI set on a larger number of patients and nourish further discussions. We covered only the first stages of an OMI set development project, and thereby established the foundation to apply rigorous methodological standards of COS research in subsequent endeavors.33–38 The study has important strengths. The OMI set is a pragmatic set for daily clinical practice and can be applied across health sectors and in different medical fields, thus contributing to consistent and standardized patient care. For the final OMI set, we selected instruments that have shown robust evidence of measurement quality. Further, most of the selected instruments have demonstrated feasibility in samples of critical care survivors. Since all OMIs currently applied in ICU survivorship research lack elaborate data on measurement quality in this population, 2 further applications of OMIs in clinical PICS studies should contribute to research on reliability and validity.118,119 Necessary data include repeated measurements and assessments of clinical outcomes to determine aspects of content, construct, and criterion validity, by using for example a detailed history and physical examination by clinicians, psychiatric interviews, or biological data.
Conclusions
To summarize our key message, we propose a two-step OMI set for PICS measurement, which combines performance-based and patient-reported outcome measures. The initial screening is brief, but assesses the relevant PICS domains cognition, mental health, physical function, and HRQoL. The subsequent extended assessment is more time consuming and utilizes more elaborate tests for an in-depth assessment of the PICS domains, allowing for tailored therapies. Our proposal fulfils essential criteria for the application in clinical settings ranging from primary outpatient healthcare to health centers experienced in PICS. Future studies should investigate the measurement properties of different OMI sets to identify PICS and examine ways to measure severity of PICS. Aside from the methodological issues, we are convinced that a PICS screening test in outpatient settings followed by a transfer to specialists would possibly increase awareness as well as accessibility and, thus, reduce the burden of PICS.
Footnotes
Acknowledgements
This article is endorsed by the working group ‘Intensive care and critical illness’ of the German Network Health Care Research (Deutsches Netzwerk Versorgungforschung e.V.). Further, the authors thank the ERIC study team for the excellent, perpetual support at all stages of this work. Particularly, Dr Christine Adrion and Professor Dr Ulrich Mansmann from the Institute for Medical Information Processing, Biometry, and Epidemiology of Ludwig-Maximilians-University Munich (LMU) have provided constant support.
Availability of data and materials
The datasets generated during the current study are available from the corresponding author on reasonable request.
Authors' contributions
CDS, BW, HK and CD designed the study. BW, CD, SR and NP collected the data. HK analyzed and interpreted the patient data. HK, BW and NP drafted the article. CDS, CD, JKi, SP, JKr, JJG, KSt, TG, KSc, SR, CH, JC, RB, UM, MG, SWC, and CA revised and commented on the article. All authors read and approved the final version of the article.
Consent for publication
All authors gave their consent for publication.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CDS reports grants from Gemeinsamer Bundesausschuss (G-BA)/Federal Joint Committee, during the conduct of the study; grants from Aridis Pharmaceutical Inc., grants from B Braun Melsungen AG, grants from Drägerwerk AG & Co KGaA, grants from Grünenthal GmbH, grants from Infectopharm GmbH, grants from Sedana Medical Ltd, grants from Deutsche Forschungsgemeinschaft/German Research Society, grants from Deutsches Zentrum für Luft- und Raumfahrt e.V. (DLR)/German Aerospace Center, grants from Einstein Stiftung Berlin/Einstein Foundation Berlin, grants from the European Society of Anaesthesiology, grants from Gemeinsamer Bundesausschuss (G-BA)/Federal Joint Committee, grants from Inneruniversitäre Forschungsförderung/Inner University Grants, grants from Projektträger im DLR/Project Management Agency, grants from Stifterverband/Non-Profit Society Promoting Science and Education, grants from WHOCC, grants from Baxter Deutschland GmbH, grants from Biotest AG, grants from Cytosorbents Europe GmbH, grants from Edwards Lifesciences Germany GmbH, grants from Fresenius Medical Care, grants from Grünenthal GmbH, grants from Masimo Europe Ltd, grants from Medtronic GmbH, grants from Pfizer Pharma PFE GmbH, personal fees from Georg Thieme Verlag, grants from Dr. F. Köhler Chemie GmbH, grants from Sintetica GmbH, grants from the European Commission, grants from Stifterverband für die Deutsche Wissenschaft e.V./Philips, grants from Stiftung Charité, outside the submitted work; in addition, CDS has a patent 10 2014 215 211.9 pending, a patent Application No. PCT/EP2015/067730 pending to Graft Gesellschaft von Architekten mbH, and a patent Application No. PCT/EP2015/067731 pending to Graft Gesellschaft von Architekten mbH. HK, NP, TG, SR, JJG, KSc, KSt, JC, JKr, JKi, CD, SP, CA, RB, UM, SWC and MG declare that there is no conflict of interest. CH reports grants to her institution from the German Federal Ministry of Education and Research (BMBF) via the Center for Sepsis Control and Care Jena, the German Innovations Funds/Federal Joint Committee, and the European Society of Intensive Care Medicine. BW reports personal fees from Orion Pharma Ltd. and personal fees from Dr. F. Köhler Chemie GmbH, outside the submitted work.
Ethics approval and consent to participate
This work was approved by the Institutional Review Board of the Medizinische Hochschule Brandenburg (record: E-01-20190619) and the Ethics Committee of Charité – Universitätsmedizin Berlin (record: EA4/122/19). Informed consent was obtained from all individual participants included in the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was composed in the context of a project supported by the Gemeinsamer Bundesausschuss (G-BA)/Federal Joint Committee of the Federal Republic of Germany (Grant-ID: 01NVF16011). The funders had no role in in the design of the study as well as collection, analysis, and interpretation of data and in writing the article.
