Abstract
Introduction:
This study aims to assess the association between mean platelet volume (MPV) and poor outcome in patients with COVID-19.
Methods:
We performed a comprehensive literature search using the PubMed, Embase and Scopus databases with keywords “2019-nCoV” OR “SARS-CoV-2” OR “COVID-19” AND “mean platelet volume” OR “MPV” on 8 July 2021. The primary outcome was composite poor outcome, defined as severe COVID-19 or mortality. The pooled effect estimate was reported as mean differences in terms of MPV between the group with and without outcome.
Results:
There were 17 studies which consist of 4549 patients with COVID-19 were included in this study. The incidence of poor outcome was 25% (20%–30%). Mean MPV was found to be higher in the poor outcome group in compare to no poor outcome group (10.3 ± 1.9 fL vs 9.9 ± 1.7 fL). The mean MPV difference between both group was 0.47 fL [95% CI 0.27, 0.67], p < 0.001; I2: 62.91%, p < 0.001). In the sub-group analysis, patients with severe COVID-19 had higher MPV (mean difference 0.54 fL [95% CI 0.28, 0.80], p < 0.001; I2: 54.84%, p = 0.014). Furthermore, MPV was also higher in the mortality group (mean difference 0.54 fL [95% CI 0.29, 0.80], p = 0.020; I2: 71.11%, p = 0.004). Meta-regression analysis showed that the association between MPV and poor outcome was not affected by age (p = 0.789), gender (p = 0.167), platelets (p = 0.056), white blood cells (p = 0.639), and lymphocytes (p = 0.733).
Conclusion:
This meta-analysis indicated that increased MPV was associated with severity and mortality in patients with COVID-19. Further research is needed to determine the optimum cut-off point.
Introduction
Coronavirus disease 2019 (COVID-19) is currently one of the most prevalent disease in the world. 1 Although most patients have mild symptoms, a significant proportion of patients developed end-organ complications.2–4 Two of the most common and potentially lethal complications are thrombotic- and coagulopathy-associated with inflammation, activation of coagulation pathway, and endothelial cells.5–7 Interest is growing toward a cheap, reliable, and readily available laboratory parameter to diagnose and to predict the outcome of COVID-19 infection.
Platelet is crucially involved in the complex interaction of each step in inflammatory cascade. 8 Platelet activation serves as a response toward pulmonary viral infection and might contribute to detrimental effect of inflammation toward the lung.9,10 During the course of COVID-19, exaggeration of inflammatory response promotes the release of inflammatory cytokines which influence platelet activation and maturation. One of these cytokines is interleukin-6 (IL-6), which is well known to correlate with the severity of disease. 11 IL-6 along with interleukin-1 (IL-1), interleukin-3 (IL-3) and tumor necrosis factor-α (TNF-α) play a significant role in affecting megakaryocyte ploidy through an increased level of thrombopoietin and causing more greater reactive production of megakaryocyte.8,12–14 Therefore, more immature platelets are released into the circulation which eventually will increase MPV. 8
Mean platelet volume (MPV) is one of the routinely ordered laboratory examination which reflects the platelet size and its activation, shows a promising result in predicting mortality in COVID-19.15,16 MPV which reflects platelet size also represent its activation. Platelet size is well known to be influenced by the course and severity of infection therefore it might carry valuable information in determining the prognosis. 17 MPV level is also proven to be a diagnostic and prognostic factor in other settings of infection, such as sepsis, infective endocarditis, pneumonia, brucellosis, cellulitis, and acute pyelonephritis.18–23 This study aims to assess the association between MPV and poor outcome in patients with COVID-19.
Material and methods
This meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guidelines. This meta-analysis is registered in PROSPERO (Blinded for review).
Search strategy and study selection
We performed a comprehensive literature search using the PubMed, Embase and Scopus databases with keywords “2019-nCoV” OR “SARS-CoV-2” OR “COVID-19” AND “mean platelet volume” OR “MPV” 8 July 2021. The PubMed (MEDLINE) search strategy was ((2019-nCoV) OR (SARS-CoV-2) OR (COVID-19)) AND ((mean platelet volume) OR (MPV)). Two independent authors performed title and abstract screening after removal of duplicates. The articles eligibility was then assessed for their eligibility using the inclusion and exclusion criteria.
Eligibility criteria
Studies were eligible for inclusion if they fulfill all of the following criteria: (1) observational studies reporting patients with confirmed COVID-19 with age ⩾18 years-old, (2) reporting severe COVID-19 or mortality, (3) containing continuous data or dichotomous data on MPV based on the outcome. We did not performed any language restriction due to the possibility of missing a relevant study.
Studies were excluded if they fulfill one of the following criteria: (1) review articles, (2) editorial, (3) non-research letter, (4) case reports, (5) animal studies, (6) and studies in pediatric populations (⩽17 years old).
Outcome
The primary outcome was composite poor outcome, defined as severe COVID-19 or mortality. The pooled effect estimate was reported as mean differences in terms of MPV between the group with and without outcome. Severe COVID-19 was defined as patients that fulfill at least one of the following criteria: (1) respiratory rate ⩾30 breaths per min; (2) oxygen saturation ⩽93% (at rest; (3) ratio of partial pressure of arterial oxygen to fractional concentration of oxygen inspired air (PaO2:fiO2 ratio) ⩽300 mmHg; (4) lung infiltrates >50% of the lung field within 24–48 h; or (5) specific complications, such as septic shock, respiratory failure, and or multiple organ dysfunction or required intensive unit care (ICU care). 24 Mortality was defined as clinically validated death.
Data extraction
Data extraction was performed independently by two authors by using a standardized extraction form. The variables of interest were author, study design, year of publication, age, gender, MPV, platelet count, white blood cell count, lymphocyte count, and the outcome.
Risk of bias assessment
Two independent authors performed risk of bias assessment using the Newcastle-Ottawa Scale (NOS) Discrepancies that arose were resolved by discussion.
Statistical analysis
Der-Simonian Laird random-effects meta-analysis was used to pool the continuous variable and obtain mean differences along its 95% confidence interval (95% CI) regardless of heterogeneity. p-Values below 0.05 indicates statistical significance. All p-values were two-tailed and statistical significance was set at ⩽0.05. We assessed heterogeneity using the Cochran Q test and I2 statistics, and p-values of <0.10 or an I2 value of >50% indicates statistically significant heterogeneity. Subgroup analysis were performed for severity and mortality. The potential for publication bias was assessed using funnel-plot analysis and Egger’s test. Non-parametric trim-and-fill analysis using Linear 0 estimator to impute studies on the right side of the plot. Egger’s test was used to assess the potential for small-study effects quantitatively. Restricted-maximum likelihood random effects meta-regression was performed for age, gender, platelets, white blood cells, and lymphocytes. Meta-analysis was performed using STATA software (Version 16; Stata Corporation, College Station, TX).
Results
Baseline characteristics
In total, there were 4549 patients with COVID-19 from 17 studies that were included in this systematic review and meta-analysis (Figure 1).15,25–40 Baseline characteristics of the included studies are displayed in Table 1. The incidence of poor outcome was 25% (20%–30%). The mean MPV for poor outcome group and without poor outcome group were 10.3 ± 1.9 fL versus 9.9 ± 1.7 fL respectively.
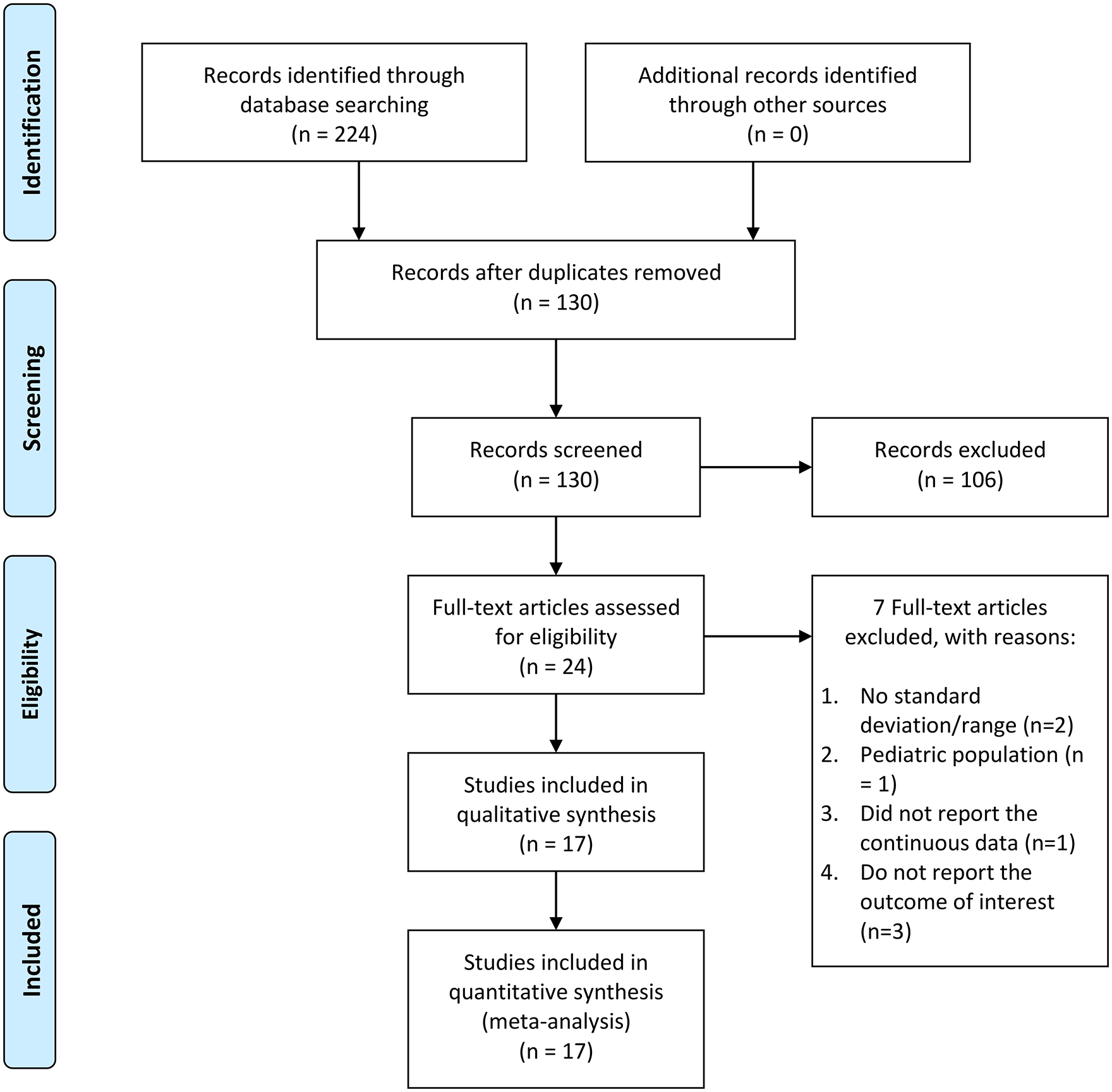
PRISMA flowchart.
Characteristics of the included studies.
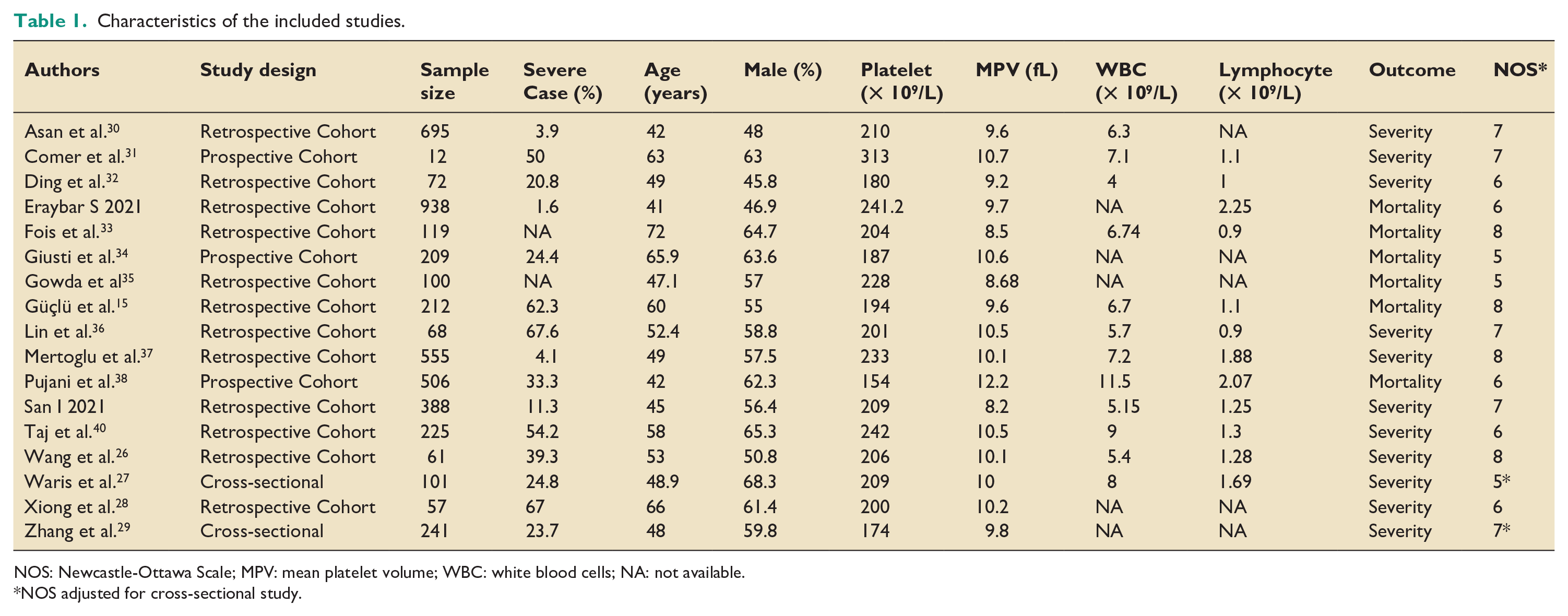
NOS: Newcastle-Ottawa Scale; MPV: mean platelet volume; WBC: white blood cells; NA: not available.
NOS adjusted for cross-sectional study.
Mean platelet volume and poor outcome
COVID-19 patients with poor outcome had higher MPV (mean difference 0.47 fL [95% CI 0.27, 0.67], p < 0.001; I2: 62.91%, p < 0.001) (Figure 2). Patients with severe COVID-19 had higher MPV (mean difference 0.54 fL [95% CI 0.28, 0.80], p < 0.001; I2: 54.84%, p = 0.014). MPV was found to be higher in the mortality group (mean difference 0.54 fL [95% CI 0.29, 0.80], p = 0.020; I2: 71.11%, p = 0.004).
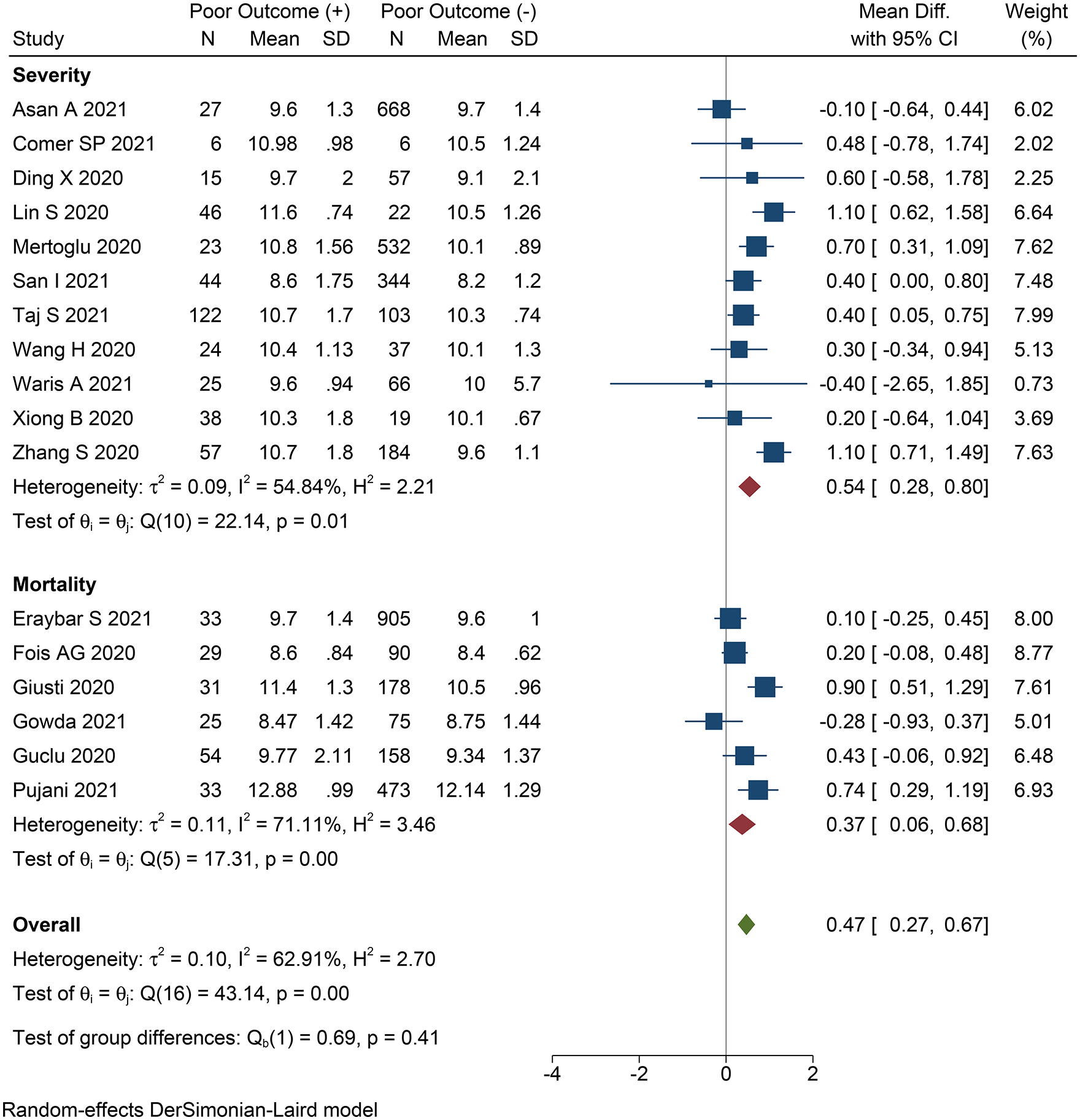
Mean platelet volume and poor outcome.
Meta-regression
Meta-regression analysis showed that the association between MPV and poor outcome was not affected by age (p = 0.789) (Figure 3(a)), gender (p = 0.167), platelets (p = 0.056) (Figure 3(b)), white blood cells (p = 0.639), and lymphocytes (p = 0.733).
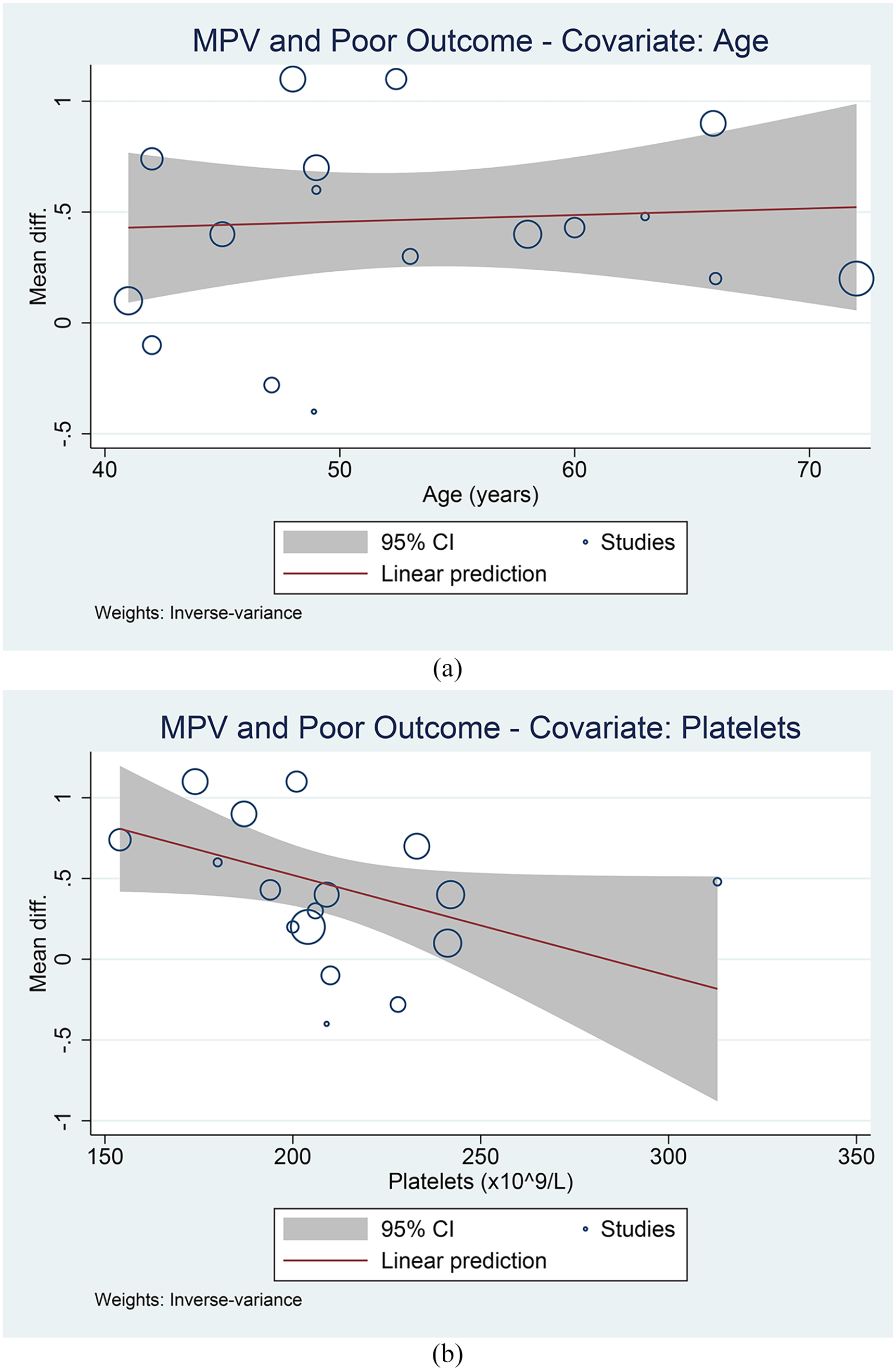
Meta-regression analysis. Covariates: age (a) and platelets (b).
Publication bias
Egger’s test was not significant for small study effects (p = 0.377). Funnel-plot was asymmetrical (Figure 4(a)) and imputation of three studies to the right side of the plot yielded effect estimate mean difference of 0.557 [95% CI 0.350, 0.763] (Figure 4(b)).
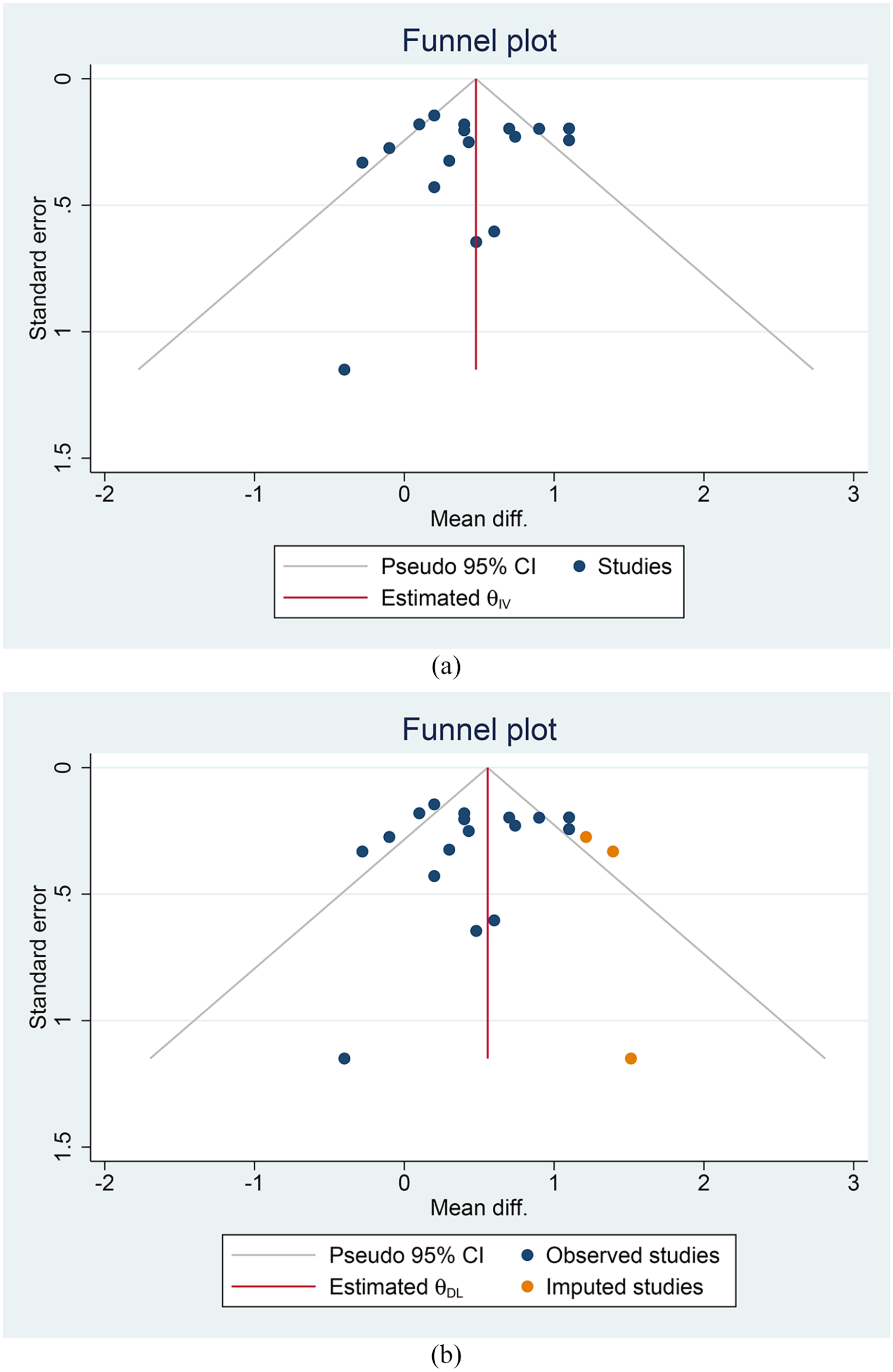
Publication bias. Funnel-plot analysis (a) and non-parametric trim-and-fill analysis (b).
Discussion
This meta-analysis indicated that increased MPV was associated with severity and mortality in patients with COVID-19.
The mechanisms of platelet alteration during the course of COVID-19 infection are thought to be complex and multifactorial. MPV itself can be considered as a marker of platelet activity as it is associated with aggregation and release of thromboxane A2, platelet factor 4 and b-thromboglobulin. 13 In patient with community acquired pneumonia, Gorelik et al. observed a raised MPV was associated with higher pneumonia severe scores and lower platelet counts on discharge. 41 Severe inflammation was postulated to be the main potential mechanism of increased MPV and MPV elevation may reflect hypercoagulation which is associated with poor outcome. 14
The role of inflammation and increased MPV in COVID-19 could be proposed as following. Firstly, thrombocytopenia might occur as a consequence of direct involvement of coronavirus in the bone marrow. Secondly, there are increased destruction of platelet related to inflammatory response. Thirdly, platelet consumption as a consequence of platelet aggregation in the lung. 42 Eventually all of these mechanisms, in addition to the release of proinflammatory cytokines such as IL-6 which directly acts on megakaryocytes, will stimulate megakaryocyte to produce more platelets to counterbalance the accelerated rate of platelet destruction.8,43 As a result, an increased level of MPV will be found (Figure 5). 44
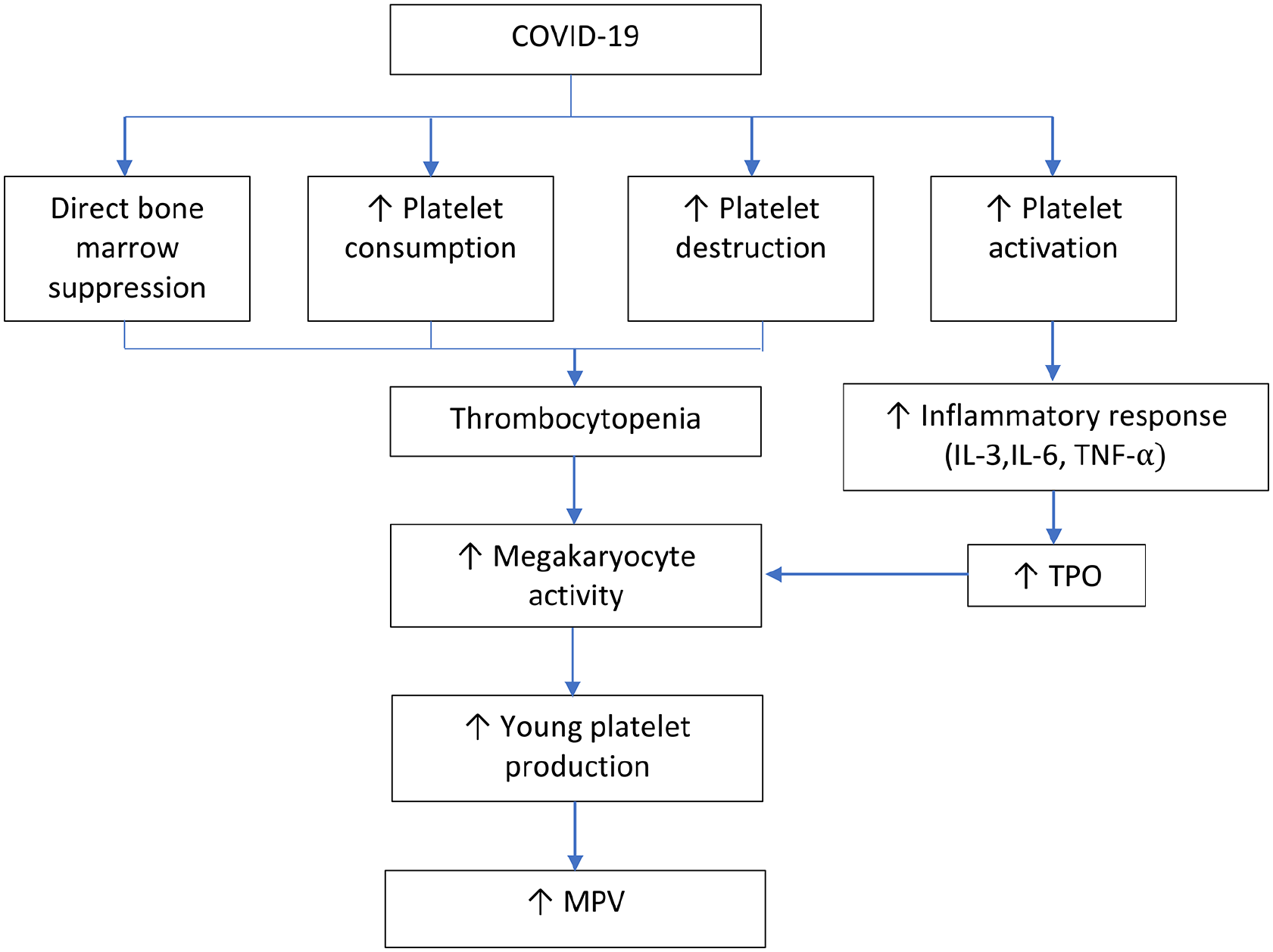
Proposed mechanism of increased MPV in COVID-19 infection.
MPV might show a significant role in the clinical setting as demonstrated in several studies. Güçlü et al. reported that difference between MPV and oxygen saturation during admission, first and third day had showed a significant result in predicting mortality. 15 Another study that was conducted by Gumus et al. also demonstrated the utilization MPV level in predicting COVID-19 in asymptomatic children. 17 Therefore, MPV might serve as an auxiliary test in detecting COVID-19 infection and predicting COVID-19 mortality. 45
Limitation
Most of the included studies were retrospective studies and did not report the status of their missing data. Most of the studies did not report cut-off points for MPV, thus meta-analysis could not be performed. To be clinically more useful, an optimal cut-off point should be determined in the future studies.
Conclusion
This meta-analysis indicated that increased MPV was associated with severity and mortality in patients with COVID-19. Further research is needed to determine the optimum cut-off point.
Footnotes
Contributorship statement
AFMZZ, CSS, and RP were involved in the conceptualization and design of the manuscript. AFMZZ, CSS, AW, WMR, and RP participated in data curation and investigation. RP performed formal and statistical analysis. AFMZZ, CSS, and WMR drafted the manuscript. AFMZZ and RP review and edited the manuscript.
Data availability
Data are available on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Not Applicable.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not Applicable.
Protocol registration number
PROSPERO CRD42021266701
