Abstract
Objectives
This retrospective study aimed to investigate and analyze the clinical significance of blood platelets (PLTs) and mean platelet volume (MPV) in patients with ulcerative colitis (UC).
Methods
A total of 132 patients with UC and 208 healthy subjects were enrolled. PLTs, MPV, platelet-large cell rate (P-LCR), C-reactive protein (CRP), fibrinogen (FIB) and D-dimer were analyzed. Correlations were calculated between disease activity/extent of disease and MPV, PLT, and CRP levels in patients with UC.
Results
PLT levels were significantly higher while MPV and P-LCR were lower in patients with UC compared with controls. Disease activity was positive correlated with CRP (r = 0.564) and PLT (r = 0.307) but negatively correlated with MPV (r = −0.351). Extent of disease was positively correlated with CRP (r = 0.312) but showed no correlation with PLTs and MPV. FIB and D-dimer were higher in patients with UC, but the difference in FIB levels was not statistically significant.
Conclusions
PLTs and MPV are potential biomarkers for UC disease activity. These data may help clinicians to attain a more comprehensive judgment of the general condition of patients with UC.
Keywords
Introduction
Ulcerative colitis (UC) is a nonspecific inflammatory bowel disease of unknown etiology. 1 The pathogenesis of UC is multifactorial and is related to genetic predispositions, epithelial barrier defects, dysregulated immune responses, and environmental factors. 2 Current evidence implicates inflammatory responses caused by abnormal intestinal mucosal immunity as key drivers of disease pathogenesis. 3 The most common clinical manifestations are bloody stools and diarrhea. Recently, many studies have demonstrated that patients with inflammatory bowel disease (IBD) may have increased risks of venous thrombosis (VTE). 3
Platelets (PLTs) play a critical role in hemostasis. However, a growing number of studies support their important role as inflammatory amplifiers in chronic inflammatory diseases. 4 Impaired platelet activation may cause persistent mucosal inflammation through P-selectin, CD40-CD40L, and other systems in granulocytes, macrophages, and endothelial cells.5,6 PLTs are activated during the inflammatory process and secrete mediators such as platelet factor 4, platelet activating factor, interleukin-8, and arachidonic acid metabolites.5,7 These mediators deliver activating signals to target cells, including endothelial cells and myelomonocytic cells, resulting in amplification of inflammation. 7
Because IBD is an immune-related disease, several immune-associated markers have been explored including C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), perinuclear anti-neutrophil cytoplasmic antibodies, and cytoplasmic anti-neutrophil cytoplasmic antibodies. 5 Clinical studies found an increase in blood platelet counts and plateletcrit levels, as well as a significant decrease in mean platelet volume (MPV) and platelet distribution width, in patients with UC and Crohn's disease (CD).6,8–10 It has thus been proposed that these changes may serve as new inflammatory biomarkers of IBD.
In this study, we aimed to document changes in blood platelets and their morphological parameters, analyze associations between these factors and manifestations such as disease activity and extent of disease, and examine the potential roles of these factors as biomarkers of UC.
Patients and methods
Patients with UC admitted to The Central Hospital of Wuhan, Tongji Medical College, Huazhong University of Science and Technology (Wuhan, China) from September 2015 to May 2019 were enrolled in the study. Healthy subjects were included as controls. Diagnosis of UC was based on clinical, radiological, endoscopic, and histological criteria. 11 Extent of UC disease was classified by the extent of colonic involvement according to the Montreal classification as follows: ulcerative proctitis (E1); left-sided colitis (E2); or extensive colitis (E3). UC disease activity was determined according to the Mayo score as follows: remission (0–2 points); mild activity (3–5 points); moderate activity (6–10 points); or severe activity (11–12 points). The grouping of patients with UC is summarized in Figure 1.

Study flow diagram.
Data on PLTs, MPV, platelet-large cell rate (P-LCR), fibrinogen (FIB), D-dimer, and CRP at admission were collected from electronic medical records. All patients underwent colonoscopy during hospitalization.
This retrospective study was conducted according to principles laid out in the Declaration of Helsinki and was approved by the Ethical Committee of The Central Hospital of Wuhan, Tongji Medical College, Huazhong University of Science and Technology (approval number: 2021004). Verbal informed consent was obtained from patients and healthy subjects or their legal guardians. All patient details were de-identified.
Statistical analysis
All statistical analyses were conducted using SPSS 17.0 software (SPSS Inc., Chicago, IL, USA). Measurement data were summarized as mean ± standard deviation (SD). Differences between and among groups were assessed using the Student’s t-test and one-way ANOVA, respectively. Categorical data were summarized as counts (%). Differences between categorical variables were assessed using the Chi-square test. Pearson’s correlation coefficients and Spearman’s correlation coefficients were used to analyze correlations between CRP, PLTs, MPV and disease activity or extent of disease. Correlation coefficients (r) were reported as follows: 0 < r < 1, positive correlation; −1 < r < 0, negative correlation; and r = 0, no correlation. Values of P < 0.05 were considered statistically significant.
Results
Basic and clinical parameters of the study population
A total of 132 patients with UC and 208 healthy subjects were enrolled in the study. Among the 132 patients with UC, 16 had ulcerative proctitis (E1), 35 had left-side colitis (E2), and 81 had extensive colitis (E3). Five patients were in remission, 43 had mild disease activity, 74 had moderate disease activity, and 10 had severe disease activity.
The characteristics of the study population, including demographic information, PLT counts and PLT morphological parameters, are summarized in Table 1. There was no difference in age or gender distribution between patients with UC and controls. The mean (± SD) ages of patients with UC and controls were 49.42 ± 14.57 years and 51.66 ± 10.10 years, respectively. Overall, 60 (45.5%) of patients with UC and 110 (52.9%) of controls were women. Compared with controls, the PLT counts of patients with UC were significantly higher (265.390 ± 106.040 × 109/L vs. 219.850 ± 51.324 × 109/L) (P < 0.01). MPV (9.289 ± 1.615 fL vs. 9.910 ± 1.620 fL) and P-LCR (23.670 ± 8.220% vs. 28.204 ± 8.248%) were significantly lower in patients with UC compared with controls (P < 0.01).
Demographic characteristics, PLT counts, and PLT morphological parameters of patients with UC and healthy controls.

Data are shown as mean ± standard deviation or n (%).
PLT, platelet; UC, ulcerative colitis; MPV, mean platelet volume; P-LCR, platelet-large cell rate.
Correlations between disease activity and CRP, PLTs, and MPV in patients with UC
Levels of CRP, PLT and MPV in patients with different UC disease activities are shown in Table 2 and Figure 2. The mean levels of CRP and PLTs in patients with severe UC disease activity were 4.743 ± 3.100 mg/dL and 346.100 ± 84.125 × 109/L, respectively, remarkably higher than levels in the other three groups (P < 0.01). By contrast, the mean level of MPV in patients with severe UC disease activity was 8.343 ± 1.691 fL, significantly lower than in the other three groups (P < 0.01). Table 3 shows correlations between disease activity and CRP, PLT, and MPV levels in patients with UC. Disease activity was positively correlated with CRP (r = 0.564, P < 0.001) and PLTs (r = 0.307, P < 0.001) but negatively correlated with MPV (r = −0.351, P < 0.001). The absolute values of the correlation coefficients, r, for PLT and MPV were lower than that of CRP.
CRP, PLTs, and MPV in patients with different UC disease activity.

Data are shown as mean ± standard deviation (SD).
CRP, C-reactive protein; PLT, platelet; UC, ulcerative colitis; MPV, mean platelet volume.

Histogram of C-reactive protein (CRP) (a), platelets (PLTs) (b) and mean platelet volume (MPV) (c) in patients with different UC disease activity (remission, mild, moderate, or severe). Data were expressed as mean ± standard deviation (SD). **P < 0.01 vs. remission group.
Correlations between UC disease activity and CRP, PLTs, and MPV.

CRP, C-reactive protein; PLT, platelet; UC, ulcerative colitis; MPV, mean platelet volume.
Correlations between extent of disease and CRP, PLTs, and MPV in patients with UC
Table 4 and Figure 3 show levels of CRP, PLT and MPV in patients with different extent of UC disease. The mean level of CRP in the E3 group was 2.181 ± 2.580 mg/dL, remarkably higher than in the other two groups (P < 0.05). There was no significant difference in PLT and MPV levels among patients with difference extents of UC disease. Table 5 shows correlations between extent of disease and CRP, PLTs, and MPV in patients with UC. Extent of disease was positively correlated with CRP (r = 0.312, P < 0.001). There was no evident correlation between extent of UC disease and PLTs or MPV.
CRP, PLTs, and MPV in patients with different extents of UC disease.
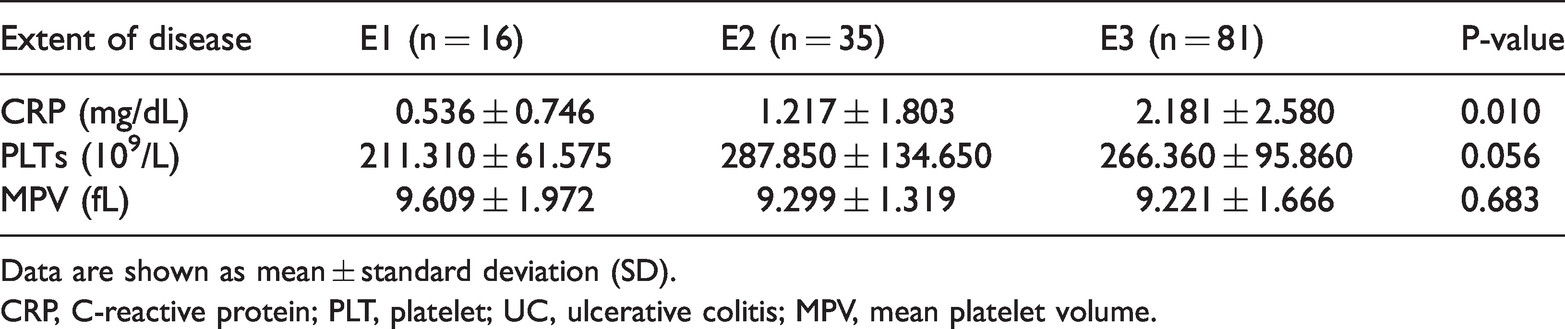
Data are shown as mean ± standard deviation (SD).
CRP, C-reactive protein; PLT, platelet; UC, ulcerative colitis; MPV, mean platelet volume.

Histogram of C-reactive protein (CRP) (a), platelets (PLTs) (b) and mean platelet volume (MPV) (c) in patients with different extent of UC disease (E1, E2 or E3). Data were expressed as mean ± standard deviation (SD). **P < 0.01 vs. E1 group, ***P < 0.001 vs. E1 group.
Correlations between extent of UC disease and CRP, PLTs, and MPV.

CRP, C-reactive protein; PLT, platelet; UC, ulcerative colitis; MPV, mean platelet volume.
Comparison of FIB and D-dimer levels between patients with UC and controls
FIB was tested in the entire study population at admission, while D-dimer was tested in only a few patients. Levels of FIB and D-dimer in patients with UC and controls are shown in Table 6. There was no significant difference in FIB between patients with UC and controls. Compared with controls, D-dimer levels were elevated in patients with UC (0.699 ± 0.448 µg/mL vs. 0.181 ± 0.094 µg/mL) (P < 0.05).
Comparison of FIB and D-dimer levels between patients with UC and healthy controls.

Data are shown as mean ± standard deviation (SD).
UC, ulcerative colitis; FIB, fibrinogen.
Discussion
Previous studies demonstrated that PLTs both regulated coagulation and thrombosis and were involved in the occurrence and development of intestinal inflammation in IBD.4,12 Our study found that in patients with UC, PLTs were significantly elevated compared with controls. This indirectly suggests that intestinal inflammatory reactions stimulate PLTs, activating and increasing the PLT count.
The mechanisms underlying the abnormal increase in PLT count in patients with IBD are not well understood. It has been shown that the survival time of PLTs does not change significantly following inflammation of the colon, 13 suggesting the increased PLT counts may be related to increased platelet production. Of note, thrombopoietin levels in patients with IBD were significantly higher than those of the general population; this hormone is a key factor in platelet formation.14,15 Although increased PLT count is associated with IBD disease activity, it is not an independent predictor of thrombotic events in patients with IBD, as many other diseases, such as malignancies, can also cause increases in PLTs. 4
Our study found that levels of PLTs in patients with UC were higher than those of controls, whereas MPV and P-LCR levels were lower. Some studies have suggested that decreased MPV is closely related to increased blood PLT counts in patients with IBD, indicating that increasing PLT production results from large platelets in the body being consumed in large quantities. However, some studies also suggest that large platelets are metabolically and enzymatically more active, 16 and that PLTs are heavily activated in IBD to form aggregates, resulting in depletion of larger PLTs. 17 This would explain why MPV and P-LCR were significantly lower in patients with UC compared with controls in our study. These results differ from those of a study by Gawrońska et al. 16 These authors also found that PLTs were higher in patients with UC compared with a control group, whereas MPV and P-LCR were lower, but the differences were not statistically significant. These discrepancies may have been related to the limited number of patients with UC in their study (N = 32).
MPV is an easily determined parameter in the clinic, and is believed to provide important information on the course and prognosis of many pathological conditions including cardiovascular and respiratory diseases, rheumatoid arthritis, IBD, diabetes, and neoplastic diseases. 18 MPV in patients with active IBD was lower than in patients who achieved remission. 19 However, Ozturk et al. 8 suggested that MPV decreased to a greater extent in patients with remissive UC. Yuksel et al. 19 found that MPV was negatively correlated with disease activity in patients with UC, consistent with our results. The authors reported that MPV could differentiate patients with active and remissive UC, and found that the overall accuracy of MPV in identifying patients with active UC was higher than CRP or ESR. They concluded that MPV was the best indicator for evaluating disease activity in patients with UC. 19 In our study, we also found that MPV was negatively correlated with disease activity in patients with UC. However, we found that the correlation between CRP and UC disease activity was higher than that between disease activity and MPV or PLTs. Moreover, CRP was also correlated with extent of disease. Thus, CRP may be a useful biomarker of UC disease activity and extent of disease. Therefore, our data suggest that MPV may play an auxiliary role in assessing the condition of patients with UC. MPV has some disadvantages when compared with the traditional inflammatory marker, CRP.
IBD mainly affects the digestive tract, but extraintestinal manifestations also occur. These include bone disease, skin disease, and eye injury as well as involvement of the coagulation and hemostatic systems, the anticoagulation system and the fibrinolytic system, potentially leading to thrombotic events. 3 The risk of VTE in patients with IBD was two to three times higher than in control subjects. 20 Several studies found that patients with UC had a higher incidence of VTE than patients with CD.21,22 In our study, D-dimer levels were assessed in only a few patients. We found that FIB and D-dimer levels were elevated in patients with UC compared with controls. Although the difference in FIB was not statistically significant, these findings suggest that UC may be associated with a hypercoagulable state. The limited D-dimer data collected in the patients with UC studied here suggests that clinicians may generally underemphasize this parameter. Therefore, D-dimer screening should not be ignored in patients with UC, especially in younger patients who may have a higher risk of blood hypercoagulability. 23
This study was limited by its retrospective design and small sample size. In addition, the study did not include many patients in remission, and we did not perform logistic regression to further assess whether PLT and MPV may be predictive of UC diagnosis.
In conclusion, PLT and MPV may reflect disease activity in patients with UC, providing clinicians with an alternative way to assess patients' general condition beyond traditional inflammatory markers (e.g., CRP and ESR). Because platelet detection is easy and cheap, these biomarkers may be usefully applied to patients with UC as they undergo outpatient follow-up.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This project was supported by grants from the Natural Science Foundation of China (grant number: 81400578), the Ministry of Education Doctoral Fund (grant number: 20130142120096), and the Wuhan City Health Bureau Project (grant number: WX13A07).
