Abstract
Background
This study aims to identify any effect of frailty in altering the risk of death or poor outcome already associated with receipt of organ support on ICU. It also aims to assess the performance of mortality prediction models in frail patients.
Methods
All admissions to a single ICU over 1-year were prospectively allocated a Clinical Frailty Score (CFS). Logistic regression analysis was used to investigate the effect of frailty on death or poor outcome (death/discharge to a medical facility). Logistic regression analysis, area under the Receiver Operator Curve (AUROC) and Brier scores were used to investigate the ability of two mortality prediction models, ICNARC and APACHE II, to predict mortality in frail patients.
Results
Of 849 patients, 700 (82%) patients were not frail, and 149 (18%) were frail. Frailty was associated with a stepwise increase in the odds of death or poor outcome (OR for each point rise of CFS = 1.23 ([1.03–1.47]; p = .024) and 1.32 ([1.17–1.48]; p = <.001) respectively). Renal support conferred the greatest odds of death and poor outcome, followed by respiratory support, then cardiovascular support (which increased the odds of death but not poor outcome). Frailty did not modify the odds already associated with organ support. The mortality prediction models were not modified by frailty (AUROC p = .220 and .437 respectively). Inclusion of frailty into both models improved their accuracy.
Conclusions
Frailty was associated with increased odds of death and poor outcome, but did not modify the risk already associated with organ support. Inclusion of frailty improved mortality prediction models.
Introduction
Clinical frailty describes a multidimensional state of reduced physical, physiological, and cognitive reserve, 1 resulting in greater susceptibility to illness and critical illness. 2 As such recent estimations place the prevalence of frailty on modern Intensive Care Units (ICUs) to be 23–35%.3,4
Data supports the association of frailty with poor outcomes following critical illness4,5. However, it is unknown which organ failures present the greatest risk to patients and whether frailty itself changes the odds of death or discharge destination already associated with the need for organ support. Whilst surviving critical illness represents a fundamental outcome from intensive care admission, the ability to return home (or to a patient’s original residence) is also an important endpoint for many patients and their families. 6 Understanding how frailty impacts on the probability of survival and the discharge destination of patients following the support of different organs will assist clinicians in making appropriate care escalation decisions, and will improve the information clinicians can give to patients and their families about the benefits and risks of intensive care treatment
It is also unknown if the mortality prediction tools commonly used on critical care units in the UK adequately reflect the impact of frailty within their respective mortality models. Commonly used mortality prediction models in the UK include that used by the Intensive Care National Audit and Research Centre (ICNARC), 7 and the Acute Physiology And Chronic Health Evaluation II (APACHE II) 8 used worldwide. Having mortality models which are accurate across the grades of frailty is important when assessing unit performance and when risk stratifying patients in critical care trials.
This study aims to identify whether the association of death or poor outcome (a composite of death and discharge to a medical facility rather than discharge home/prior residence) following different forms of organ support is modified by the presence of frailty. It also aims to examine the performance of the ICNARC and APACHE II models in predicting mortality in frail and non-frail populations.
Methods
Following appropriate consideration by the South-Central Research Ethics committee, ethical approval was not deemed necessary. Heath Research Authority was granted in September 2017.
Clinical frailty score. 11

Data on all admissions between 01/04/2016–31/03/2017 was extracted from the unit database and from sequential analysis of patient notes. This data included: patient demographics, diagnoses, daily organ support data, ICNARC and APACHE II score and risk of death, length of ICU and hospital stay and discharge destination. The ICNARC and APACHE II scores are scoring systems used to calculate the severity of illness of patients on ICU and the predicted ICU mortality. They are calculated by allocating scores for chronic conditions (such as cirrhosis or severe heart failure), age, and acute derangements in the patient’s condition (such as conscious level and physiological parameters). The ICNARC risk of death probability was calculated by dividing the ICNARC score by a denominator published by the creators of the model. The APACHE II risk of death probability is calculated by the model when all indices are entered into it. Discharge options recorded were: home/prior residence, medical facility (which included long term rehabilitation/care facility, health related institution, nursing home or equivalent, hospice or equivalent) non-health related institution or prison. Levels of organ support were defined using the Critical Care Minimum Dataset (CCMD) definitions applicable in the UK 10 ; these definitions are summarised in Table S1. Data was collated in Excel (Microsoft Office 365 Pro Plus, version 1909, Microsoft Corporation).
Organ support, frailty and risk of death
Patients were classed as alive if they survived to the point of hospital discharge. Logistic regression analysis was used to investigate any association between frailty and death. It was also used to investigate if frailty modified any association between different forms of organ support (advanced respiratory, advanced cardiac and renal) and death. Regression models were used to investigate the association between death and each form of organ support separately, whereby potential effect modification by level of patient frailty was explored via a frailty score by organ support interaction term. Frailty score and the different forms of organ support were then considered together as exposures in a single regression model which additionally adjusted for requirement for multi-organ support, age and sex.
Organ support, frailty and risk of poor outcome
A similar process was then repeated examining patient outcome rather than death, whereby a poor outcome was defined as death or discharge to a medical facility and good outcome was defined as discharge alive to the patient’s home/prior residence. Logistic regression models were again used to investigate any association between frailty and outcome and whether frailty modified any association between organ support and outcome.
Frailty score was considered as a continuous variable (rather than a categorical variable) in these analyses. The CFS was not considered as an ordered categorical variable, due to small sample sizes which would have resulted in model estimation issues which would lack meaning and from which inferences could not easily be drawn.
Mortality prediction models and frailty
Three approaches were used to investigate whether the ability of ICNARC and APACHE II to predict mortality differed by whether or not participants were frail.
Firstly, logistic regression analysis was used whereby the outcome was death, the exposure was risk score and an exposure by binary frailty status interaction term (and then categorical frailty status interaction term) was included to investigate potential modification of the relationship between risk score and mortality due to frailty status.
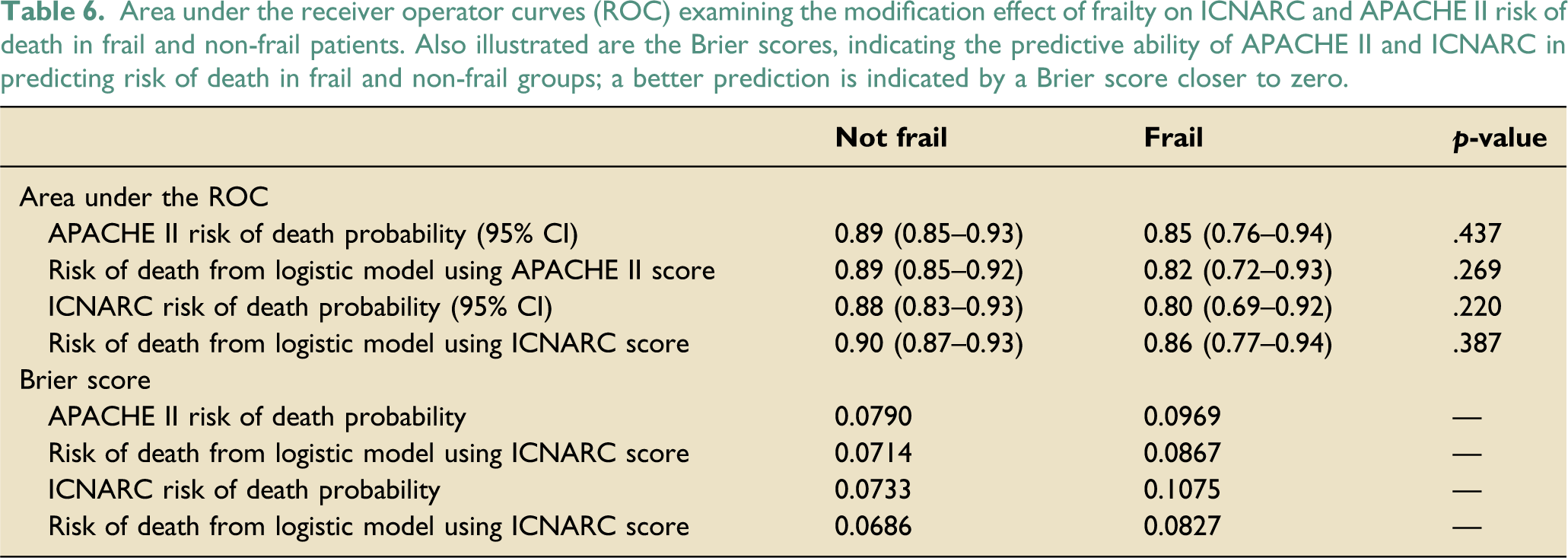
Secondly, Receiver Operating Characteristic (ROC) curves were generated for each of the risk scores separately for frail and non-frail participants and the Area Under the Curve (AUC) compared to investigate if the discriminatory ability of the risk scores differed by frailty status. Similarly, the AUC of the ICNARC and APACHE II risk of death probabilities were also compared between frail and non-frail participants.
Thirdly, a Brier Score was calculated for the ICNARC and APACHE II risk of death probabilities, and the model predicted risk of death (from a logistic model using death as the outcome and ICNARC/APACHE score as exposure), for frail and non-frail participants was used to investigate the predictive accuracy of the risk scores. Brier scores range between zero and one; the lower the Brier score is for a set of predictions, the better the predictions are calibrated.
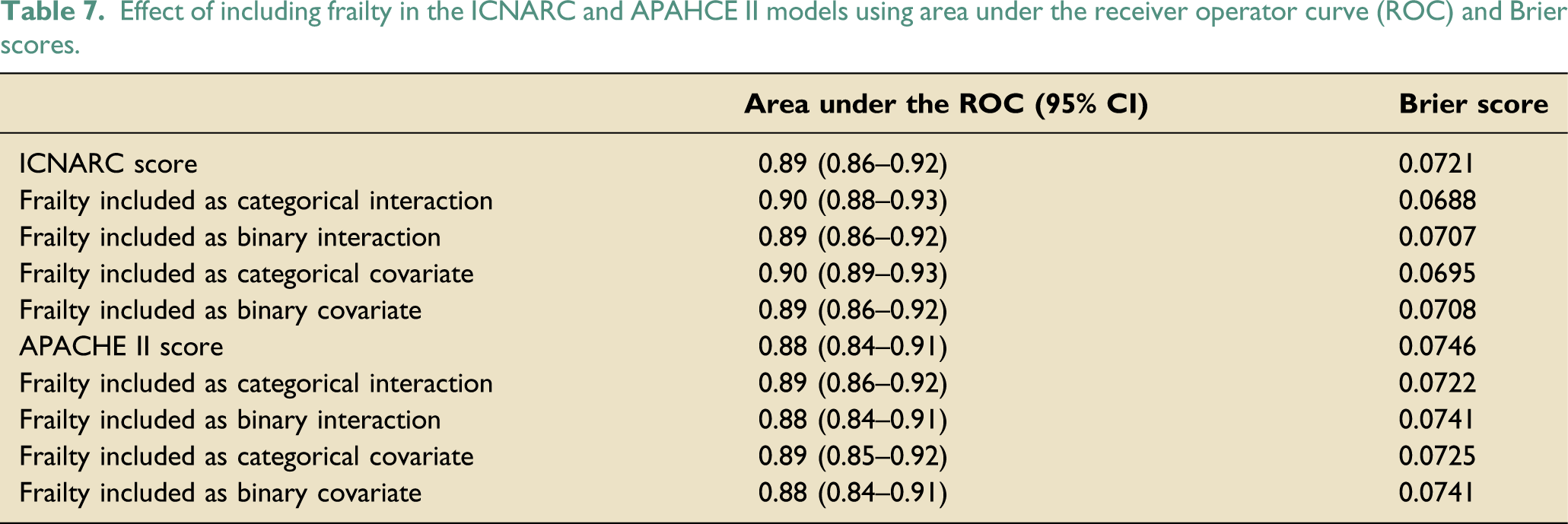
It was then explored whether including frailty in the model may improve the mortality risk prediction. The AUC and Brier scores assessing prediction accuracy of death were calculated and reported from logistic regression models: 1) outcome was death and exposure is ICNARC/APACHE II risk score; model 2) model 1 plus inclusion of categorical frailty status by risk score interaction; model 3) model 1 plus inclusion of a binary frailty status by risk score by interaction term; model 4) model 1 plus inclusion of categorical frailty status as an additional exposure, and; model 5) model 1 plus inclusion of binary frailty status as an additional exposure.
As described above, the data was initially dichotomised (frail vs not frail). The analysis was then repeated (where appropriate) additionally exploring CFS as a categorical variable.
Data was analysed in Stata (version 15.1, Stata Corporation LLC).
Results
During the 1-year study, 868 patients were admitted to the ICU and frailty data was captured prospectively in 849 (98%) admissions. The 19 patients with no frailty status were excluded from further analysis. Of the 849 patients, 700 (82%) patients were not frail and 149 (18%) patients were frail. The distribution of frailty across all admissions is illustrated in Figure 1. Patient characteristics are displayed in Table 2. Distribution of patients by frailty score. Patient characteristics.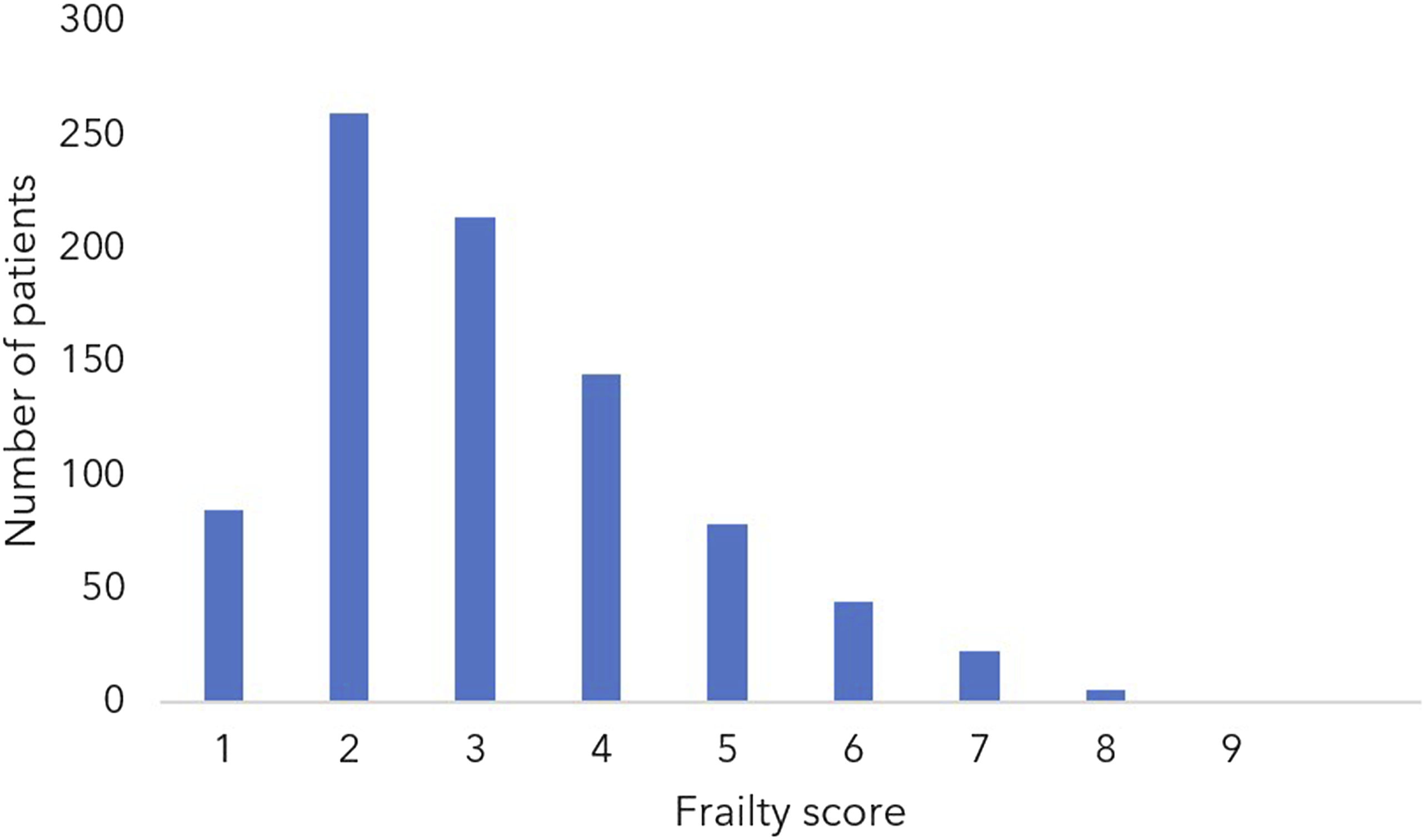
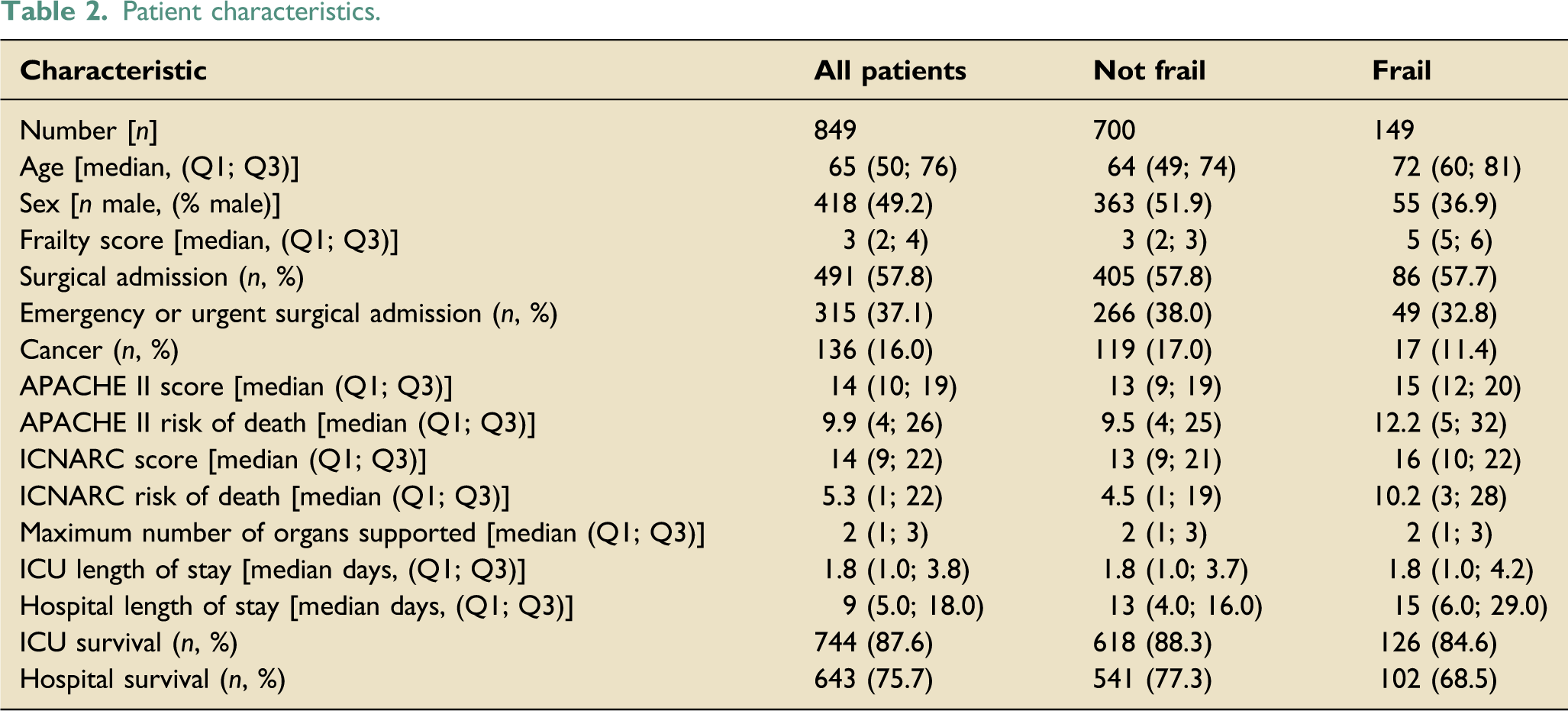
Organ support, frailty and risk of death
Organ support requirements in frail and non-frail patients.
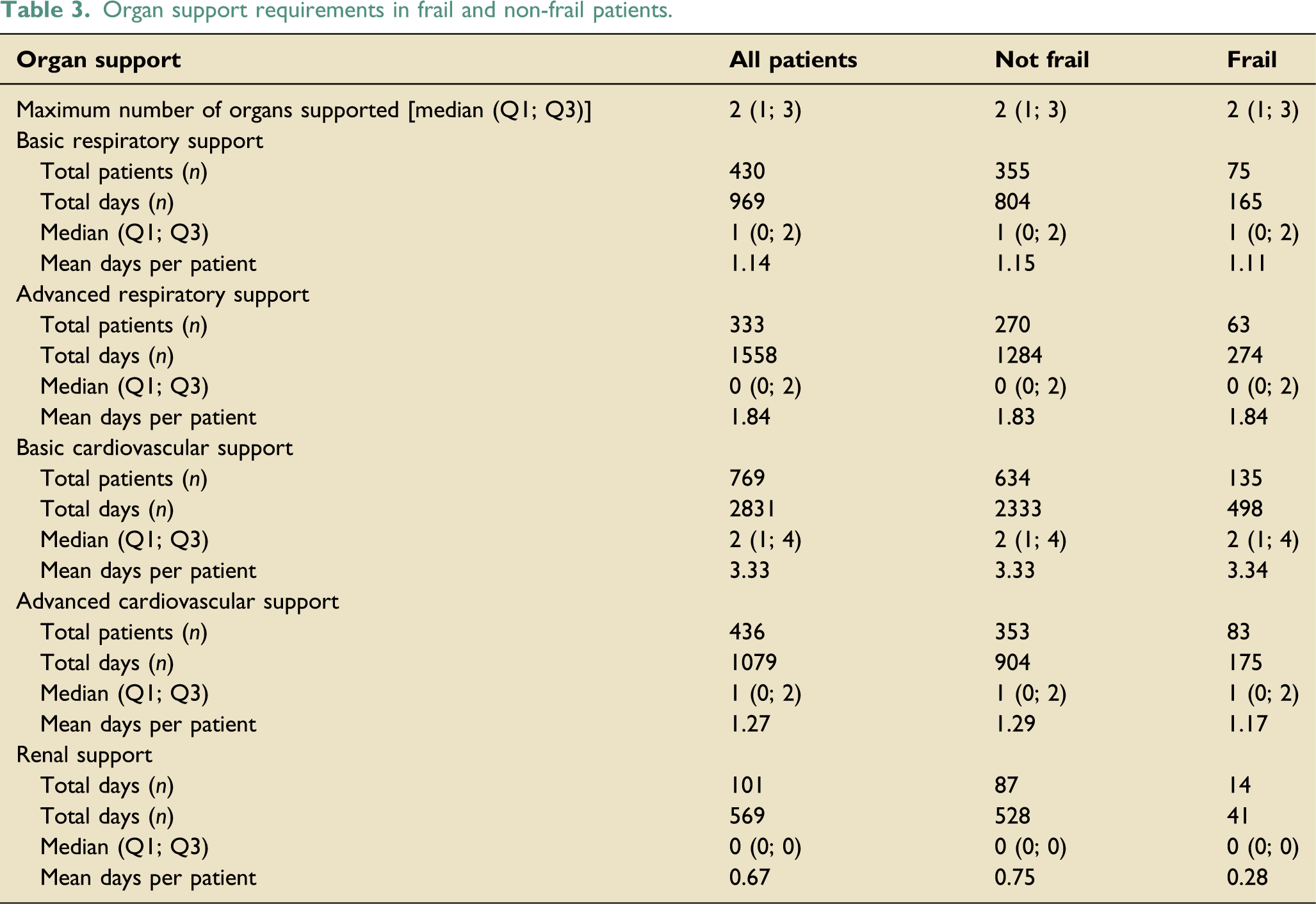
The impact of frailty on the odds ratio of death or poor outcome (a combination of death and discharge to a medical facility), illustrating the adjusted and unadjusted odds that a one unit increase in frailty score brings. The unadjusted model uses frailty as the covariate.
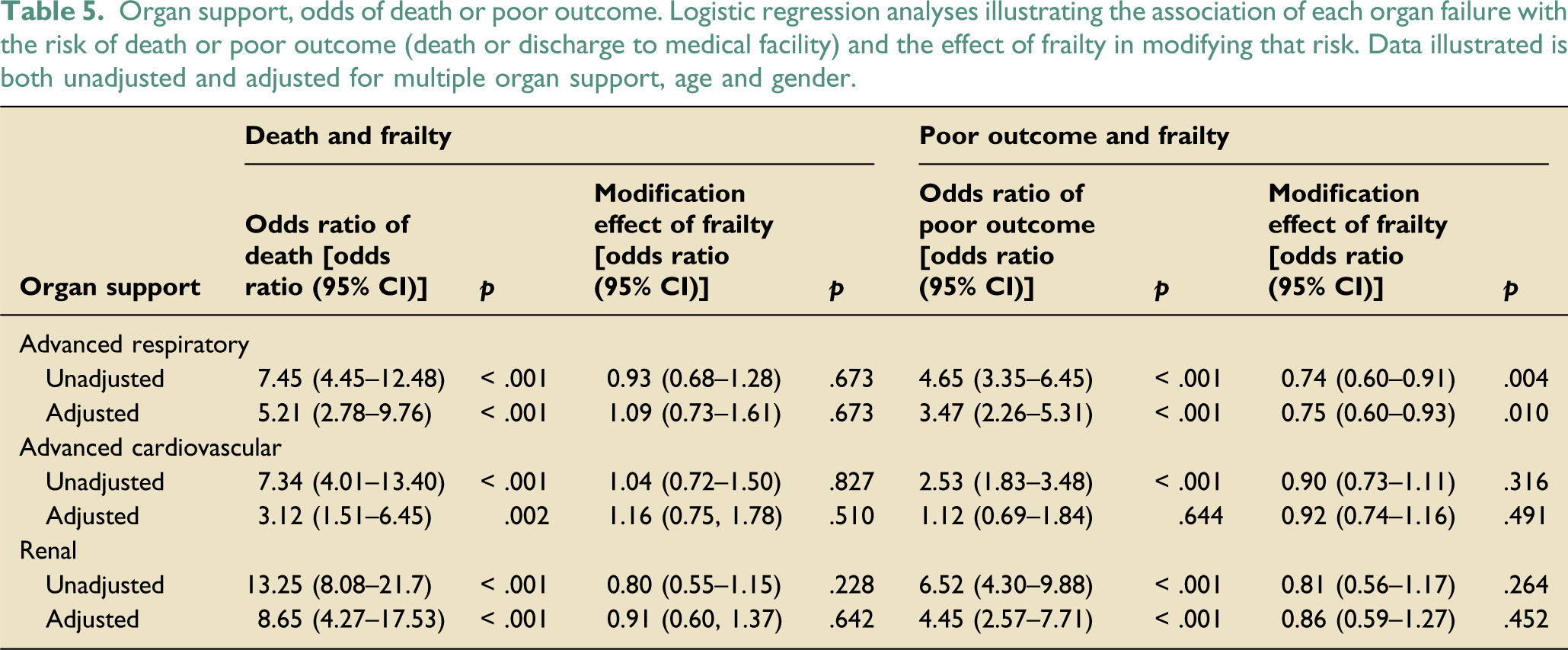
Organ support, odds of death or poor outcome. Logistic regression analyses illustrating the association of each organ failure with the risk of death or poor outcome (death or discharge to medical facility) and the effect of frailty in modifying that risk. Data illustrated is both unadjusted and adjusted for multiple organ support, age and gender.
Organ support, frailty and risk of poor outcome
There was strong evidence of an association between frailty and outcome. Increasing frailty was associated with greater odds of poor outcome (death or discharge to a medical facility) rather than good outcome (discharge to home/prior residence). The odds ratio of a poor outcome associated with each single point increase in frailty score was 1.32 (95% CI 1.17–1.48, p = <.001) in multivariable analysis as illustrated in Table 4 (correcting for advanced respiratory support, advanced cardiovascular support, renal support, age, sex and multi-organ support).
The odds ratio of poor outcome associated with the support of each organ and the modification effect of frailty is illustrated in Table 5. Following adjustment using the indices described above, there was strong evidence for a positive association between respiratory and renal support and poor outcome, with renal support again conferring the greatest increase in odds (OR 3.47 and 4.45 respectively; p = <.001 for both terms). However, following adjustment using the indices described above, there was no evidence of an association between the receipt of advanced cardiovascular support and poor outcome (OR = 1.12 95% CI 0.69–1.84; p = .644).
There was again no evidence in unadjusted or adjusted models using the indices described above that frailty modified the outcome of patients receiving cardiovascular or renal support (p values ranging from 0.264 to 0.452). However, there was evidence in both unadjusted and adjusted models that the association of poor outcome with advanced respiratory support was reduced with increasing frailty (interaction term for adjusted model OR = 0.75 95% CI 0.60–0.93, p = .010).
Mortality prediction models and frailty
At the time of hospital discharge, there were 159 deaths in the non-frail group and 47 deaths in the frail group, making hospital mortality 22.7% and 31.5% respectively and 24.2% overall. Predicted risk of death was higher in the frail group.
Area under the receiver operator curves (ROC) examining the modification effect of frailty on ICNARC and APACHE II risk of death in frail and non-frail patients. Also illustrated are the Brier scores, indicating the predictive ability of APACHE II and ICNARC in predicting risk of death in frail and non-frail groups; a better prediction is indicated by a Brier score closer to zero.
Effect of including frailty in the ICNARC and APAHCE II models using area under the receiver operator curve (ROC) and Brier scores.
Discussion
This is the first study to examine the effect that frailty has on the odds of death and discharge destination imparted by the requirement to support the respiratory, cardiovascular or renal systems on the ICU. It is also the first study to assess the accuracy of APACHE II and ICNARC models in predicting death in the frail population. Four key findings stand out. First, increasing frailty was associated with increasing odds of death or poor outcome (defined as death or discharge to a medical facility). Second, the greatest increase in odds of death or poor outcome were associated with renal support, followed by advanced respiratory support, with advanced cardiovascular support only conferring greater odds of death but not of poor outcome following adjustment. Third, frailty did not modify the odds of death or poor outcome already imparted by the requirement to support these organs, with the possible exception of the association of advanced respiratory support and poor outcome. Finally, we found that the ICNARC and APACHE II models reliably predicted mortality in the frail and non-frail population, but that the performance of both models could be improved by the inclusion of frailty.
The desired outcome from this study was to generate some data which would provide greater understanding of the role that frailty plays in the outcome from organ support on the ICU and the reliability of the mortality prediction tools when applied to the frail population. It is valuable information for clinicians, patients and relatives in understanding the benefits and risks of organ support on the ICU. It is also valuable when benchmarking units and risk stratification for research.
Our finding that frail patients are more likely to die from a critical illness is consistent with previous data.3,6,7 However, where previous studies have presented a dichotomised odds of death in frail or non-frail patients, our study identifies the odds ratio of death associated with each point rise in frailty score, thereby giving a clearer indication of the risk of death across the spectrum of frailty. Previous authors have also noted an association of frailty with greater disability following critical illness11,12 the need for nursing support following discharge home, 13 and a reduction in the odds of discharge home. 3 However, this is the first study which examines the impact that frailty has on the outcome from different forms of organ support and therefore it offers greater granularity on the understanding of the interplay between the two. For example, the fact that advanced cardiovascular support did not alter the odds of poor outcome may be of interest where the benefits and risks of isolated cardiovascular support were being considered for certain groups of patients (for example patients with gram negative urosepsis).
One finding stands out as being inconsistent with these data and warrants further discussion. In patients receiving advanced respiratory support, we found evidence of reduction in the odds of poor outcome in patients who were frail compared with patients who were not frail. It is unlikely that frailty confers a biological advantage in improving outcome from advanced respiratory support and this finding may represent a multiplicity finding whereby multiple hypothesis tests have produced a positive result by chance alone or it may be a chance finding alone given the size of our dataset.
Only two previous studies have examined the role that frailty may play in modifying the accuracy of the APACHE II mortality prediction model on ICU.14,15 Both studies describe benefit in the inclusion of frailty within the APACHE II model, raising the area under to the ROC to 0.88 (for all critically ill admissions in Australia and New Zealand) and 0.82 (for critically ill postoperative gastrointestinal surgical patients in Spain) respectively. This was very similar to the area under the ROC following inclusion of frailty in our study (0.89). The inclusion of frailty within mortality prediction models should therefore be an area of ongoing research in the UK, using larger data sets. 16
Limitations
This study has several limitations. The CFS is a useful bedside scoring system but there is a degree of subjectivity in allocating patients a score. This is a single centre, observational UK study where data collected prospectively was analysed retrospectively, with a significant skew of patients in frailty groups 1–5. However, the rates of frailty seen in our study align closely with another UK study (which has reported frailty rates of 18.7% 17 ), and our ICU admission characteristics (such as age, co-morbidity and functional status prior to admission) are similar to those of other units across the UK, 18 therefore in the absence of national frailty data on ICU admission, we can reasonably assume that our patient population is not significantly different to others within UK critical care practice. Finally, the number of deaths and renal support days in the frail group were relatively low, and this may have hampered our ability to draw accurate conclusions of the impact of renal support and the modelling accuracy in the frail group.
Conclusions
Frailty was associated with increased odds of death and poor outcome in ICU patients, but did not modify the risk already associated with the receipt of organ support. Mortality prediction models, whilst accurate in frail and non-frail populations, could be improved by the inclusion of frailty.
Supplemental Material
Supplemental Material - The impact of frailty on death, discharge destination and modelling accuracy in patients receiving organ support on the intensive care unit
Supplemental Material for The impact of frailty on death, discharge destination and modelling accuracy in patients receiving organ support on the intensive care unit by Andy Georgiou, Nicholas Turner, Alfredo Serrano Ruiz, Harry Wadman, Emma Saunsbury, Stephen Laver and Rob Maybin in Journal of the Intensive Care Society
Supplemental Material
Supplemental Material - The impact of frailty on death, discharge destination and modelling accuracy in patients receiving organ support on the intensive care unit
Supplemental Material for The impact of frailty on death, discharge destination and modelling accuracy in patients receiving organ support on the intensive care unit by Andy Georgiou, Nicholas Turner, Alfredo Serrano Ruiz, Harry Wadman, Emma Saunsbury, Stephen Laver and Rob Maybin in Journal of the Intensive Care Society
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grant from the Ernest Leach Fund and the National Institute of Academic Anaesthesia.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
