Abstract
Background
Intrapulmonary percussive ventilation is used in various clinical settings to promote secretion clearance, reverse or treat atelectasis and improve gas exchange. Despite a few studies reporting the use of intrapulmonary percussive ventilation in critical care, the available data remain insufficient, contributing to weaker evidence toward its effectiveness. Also, there is a paucity of studies evaluating the safety and feasibility of intrapulmonary percussive ventilation application in critical care. This retrospective pilot study has evaluated the safety and feasibility of intrapulmonary percussive ventilation intervention in non-intubated patients admitted to an intensive care unit.
Methods
The medical records of 35 subjects were reviewed, including 22 subjects who received intrapulmonary percussive ventilation intervention and 13 subjects matched for age, sex, and primary diagnosis who received chest physiotherapy. The records were audited for feasibility, safety, changes in oxygen saturation, chest X-ray changes, and intensive care unit length of stay.
Results
A total of 104 treatment sessions (IPV 65 and CPT 39) were delivered to subjects admitted with a range of respiratory conditions in critical care. Subjects completed 97% of IPV sessions. No major adverse events were reported with intrapulmonary percussive ventilation intervention. Intensive care unit length of stay in the intrapulmonary percussive ventilation group was 9.6 ± 6 days, and in the CPT group, it was 11 ± 9 days (p = 0.59). Peripheral oxygen saturation pre to post intervention was 92% ± 4 to 96% ± 4 in IPV group and 95% ± 4 to 95% ± 3 in the CPT group.
Conclusion
Application of intrapulmonary percussive ventilation intervention was feasible and safe in non-ventilated adult patients in critical care.
Keywords
Introduction
Critically ill patients, both ventilated and non-ventilated, are at risk of developing pulmonary complications such as pulmonary atelectasis, pneumonia, and respiratory failure.1,2 The incidence of respiratory failure in non-ventilated patients with pulmonary complications is high, often requiring ventilatory support.3,4 Respiratory impairments, such as excessive airway secretions, poor mucociliary clearance, and ineffective cough, increase the likelihood of extubation failure.5,6 This can consequently result in prolonged ICU stay, associated increased health care cost, and increased mortality.5,7 Physiotherapy aims to assist in preventing or treating pulmonary complications by promoting secretion clearance, improving gas exchange, and restoring or maintaining lung volumes.8,9 The most commonly used chest physiotherapy (CPT) interventions in this patient group include positioning, manual techniques (such as chest percussion and vibrations), deep breathing exercises, active cycle of breathing technique, forced expiratory technique, airway suctioning, manual, and ventilator hyperinflation.4,9 In addition to conventional CPT techniques, various other techniques such as high-frequency chest wall oscillation, continuous positive airway pressure, mechanical insufflation–exsufflation, prescribed nebulized mucolytic medications, and intrapulmonary percussive ventilation (IPV) are also used by physiotherapists to aid secretion clearance, prevent, and/ or reverse pulmonary atelectasis and improve oxygenation.4,9–11
The IPV device was developed in 1979 by Forrest M Bird. 12 The intrapulmonary percussive ventilator is a pneumatic device that delivers mini bursts of sub-tidal positive pressure breaths at high frequency (60–400 cycles/min), which is superimposed on patients breathing cycle. These positive pressure breaths are known to restore or maintain lung volumes and improve alveolar ventilation, whereas the asymmetrical pattern of the flow waves assists in mobilizing pulmonary secretions toward the central airway.10,13,14 Since its inception, IPV has been used either as a stand-alone modality or in conjunction with other CPT techniques for hypoxemia, pulmonary atelectasis, airway clearance, and respiratory acidosis in various clinical settings.15–18 Antonaglia et al. 15 demonstrated that IPV resulted in improved oxygenation and reduced the length of stay compared to CPT in 40 COPD patients admitted to ICU who were being treated with non-invasive ventilation. Similar findings of improved oxygenation and reduced incidence of pneumonia were reported by Clini et al. 19 in 46 tracheostomized patients when IPV intervention was added to CPT compared to CPT alone. Despite a few studies reporting the use of IPV in critical care, the available data remains insufficient, contributing to weaker evidence toward its effectiveness. In addition, there is a lack of studies evaluating the safety and feasibility of IPV intervention in patients admitted to critical care.
Due to the paucity of data in the critical care setting, we conducted a retrospective study to evaluate the safety and feasibility of IPV application in our ICU. Additional data were also gathered on ICU length of stay to perform a sample size calculation for a proposed randomized controlled trial to evaluate the effectiveness of IPV intervention in critical care.
Methods
Design
A single-centre retrospective pilot study was conducted at Nepean Hospital, New South Wales, Australia, in a 22-bed medical and surgical ICU. The Nepean Blue Mountains Local Health District ethics committee approved the study (Project number: 17-54A). Authors AH and WL searched the medical records. Patients treated with IPV between June 2016 and December 2017 were identified retrospectively from the electronic patient records through keyword filtering such as intrapulmonary percussive ventilation, IPV, MetaNeb, and chest physiotherapy. Patients who received one or more treatment session(s) of IPV intervention were included in the IPV group if they met one or more of the following criteria: (1) fraction of inspired oxygen (FiO2) ≥35%, (2) radiological evidence of pulmonary atelectasis and/or consolidation, (3) signs of respiratory distress (such as SpO2 <90% on room air, respiratory rate ≥25 breaths/min and use of accessory breathing muscles), and (4) evidence of sputum retention (such as moist/productive cough, auscultation, and radiological findings of underlying pulmonary consolidation and/or atelectasis). Exclusion criteria were: (1) receiving invasive mechanical ventilation, (2) intubated within 24 h, (3) receiving FiO2 ≥ 80%, (4) untreated pneumothorax, (5) pneumonectomy patients, (6) post oesophageal surgery patients, (7) facial fractures/injuries/surgeries, (8) pulmonary hemorrhage, and (9) delirious or agitated patients. Electronic medical records were also searched from July 2012 to December 2017 to find a control CPT group matched on primary diagnosis, age (±10 years), and gender. Patients who received one or more CPT session(s) were screened and matched for comparison. Exclusion criteria were: (1) receiving invasive mechanical ventilation, (2) clinical deterioration within 24 h, (3) intubated within 24 h, (4) received IPV treatment, and (5) incomplete CPT session.
Subjects
A total of 35 subjects aged between 16 and 81 years were included in this study: 22 received IPV intervention and 13 matched controls received CPT (Figure 1). Subjects were admitted to ICU with medical and surgical conditions, where more than 50% needed invasive or non-invasive mechanical ventilation prior to receiving IPV intervention (Table 1).
Flow diagram represents the screening and inclusion process. Baseline characteristics. Baseline characteristics show the demographics and clinical conditions of the subjects. IPV: intrapulmonary percussive ventilation; CPT: chest physiotherapy.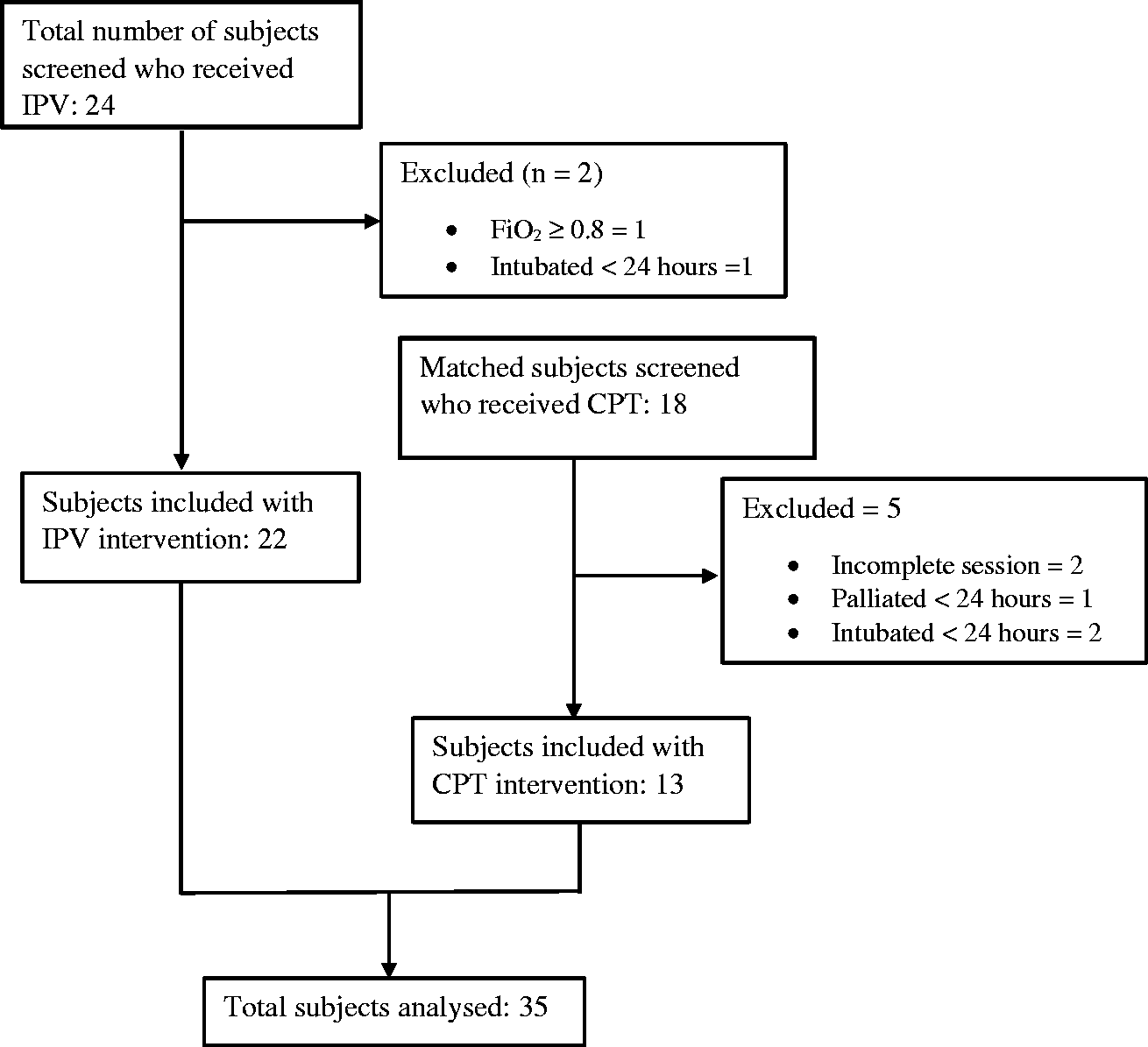
Interventions
IPV intervention
IPV intervention was delivered using the Metaneb® System (Hill-Rom Services, USA; Figure 2). Unlike other conventional IPV devices, this system only has a single port attachment for a disposable patient circuit, which simplifies equipment set-up without the need for internal cleaning or sterilization.
MetaNeb® System (Hill-Rom Services, USA).
IPV intervention included the delivery of small bursts of sub-tidal volume breaths at the rate of 170–230 breaths/min and a frequency of 2.8–3.8 Hz. This device also incorporated an in-line nebulizer for aerosol delivery. Normal saline (NaCl 0.9%) was used as an aerosol for the treatment sessions. IPV treatment was given by physiotherapists with more than 10 years of clinical experience in ICU. A defined IPV treatment protocol was followed during each treatment session. The duration of a treatment session was 10–15 min. Some subjects received IPV treatment twice a day, whereas others received once-daily treatment. A mouthpiece was used to deliver the treatment for those who were able to form a good seal around the mouthpiece, whereas a face mask was used for those who could not maintain an effective seal around the mouthpiece. The airway pressure remained between 10 and 20 cmH2O during the treatment sessions. Most of the subjects were treated in a high sitting position in bed with back supported or in a chair. Two subjects received treatment in a flat side-lying position. Patients in the IPV group did not receive additional CPT intervention. However, they did receive their usual physical activity and mobilization intervention similar to that of the CPT group.
Chest physiotherapy
Standard CPT was given to the subjects in the matched control group. CPT techniques were employed mainly to treat excessive pulmonary secretions and pulmonary atelectasis. For secretion clearance, CPT techniques such as positioning, chest percussion, and vibration, active cycle of breathing technique, forced expiratory technique, and positive expiratory pressure therapy were used. For atelectasis, deep breathing with or without inspiratory hold, incentive spirometer, and positioning were the most commonly used techniques. CPT was given mostly by senior ICU physiotherapists with more than 10 years of experience in ICU and weekend physiotherapists with 1–5 years of experience in ICU. CPT was given once a day to most of the subjects, whereas some subjects were treated twice a day. The duration of a single CPT session lasted for 10–20 min.
Outcome measures
Radiological atelectasis score.

Statistical analysis
Since this was a pilot study with a sample size that looked at feasibility and safety only, hypothesis testing for the differences between the groups could not be performed for all the outcome measures reported. 21 Student's t-test and Chi-squared tests were performed to compare the baseline characteristics of the groups, and other outcomes reported in this study. All descriptive and summary statistics were performed using Program R (RStudio Inc.), Microsoft® Excel program, and SigmaPlot (Systat Software Inc.) software. Results such as physiological variables data are presented as mean ± SD unless otherwise stated.
Results
Patient characteristics
This pilot study included 35 subjects who received a total of 104 treatment sessions of IPV or CPT treatment (IPV = 65 sessions and CPT = 39 sessions) (Table 1). The subjects were between 16 and 81 years of age. These included subjects were admitted to ICU with a range of mixed medical and surgical conditions, with 23% of subjects having hypoxemic respiratory failure and 18% with pneumonia and the rest with impaired respiratory function secondary to mechanical ventilation and ICU stay. A total of 24 subjects received IPV between June 2016 and December 2017. Two subjects were excluded because they did not meet the inclusion criteria (Figure 1). Twenty-two subjects (15 males and 7 females) with a mean ± SD age 60 ± 17 years were included in the IPV group. Ten of these subjects (45%) were initially intubated and ventilated and received IPV therapy post-extubation. The remaining 12 subjects (55%) were not intubated previously, but six of these subjects (27%) had non-invasive ventilation prior to receiving IPV. The average number of IPV treatments given was three sessions per subject.
Eighteen matched subjects were screened for the CPT group. Five subjects were excluded because they did not meet the inclusion criteria (Figure 1). Included subjects in the CPT group (68 ± 10 years) were similar in baseline characteristics to that of the IPV group (p = 0.16); however, the average APACHE III score was lower in IPV group compared to CPT group (59 ± 27 vs. 72 ± 20; p = 0.11) (Table 1). Eight subjects in the CPT group (61%) were initially intubated and ventilated and only one subject received NIV treatment. These subjects also received an average of three CPT sessions.
Feasibility and safety
Inspection of medical records showed that 65 (out of 67) IPV treatment sessions were completed. One subject refused treatment due to fatigue, and the other subject could not participate due to drowsiness. During the 65 IPV sessions, none of the subjects requested discontinuation of treatment, which shows good compliance. There were no reports of discomfort or poor treatment tolerance by the subjects. The average number of completed sessions by each subject was similar in the IPV and CPT group (three sessions per patient), showing that application and completion rates with IPV intervention were similar to CPT. Additionally, records did not reveal any change in treatment plans by the therapists due to IPV equipment or patient circuit unavailability. Technical difficulties during IPV device setup or treatment delivery were not mentioned in the records.
Feasibility and safety of IPV intervention.
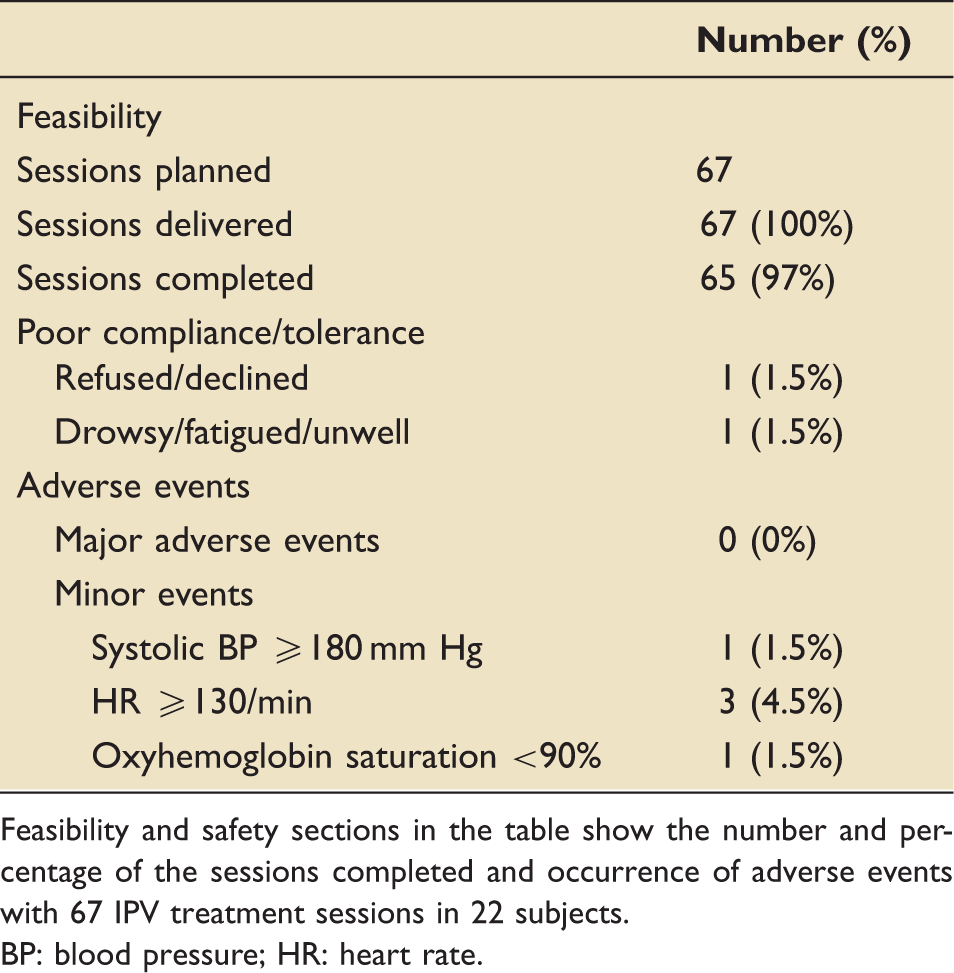
Feasibility and safety sections in the table show the number and percentage of the sessions completed and occurrence of adverse events with 67 IPV treatment sessions in 22 subjects.
BP: blood pressure; HR: heart rate.
Oxygenation and oxygen requirement
Pre and post intervention SpO2 and FiO2 values were averaged daily from day 1 to 4 in the IPV group (Figure 3(a) and 3(c)). Similarly, pre and post SpO2 and FiO2 values were averaged daily from day 1 to 4 in the CPT group (Figure 3(b) and 3(d)). A change in SpO2 from day 1 to 4 of 4% (92 ± 4% to 96 ± 4%) was recorded in the IPV group with a simultaneous change in average FiO2 from 45 ± 12% to 42 ± 10%. In CPT group, the average SpO2 from day 1 to 4 (95 ± 3% to 95 ± 3%) was also recorded with average FiO2 change from 37 ± 14% to 36 ± 14%. There was a steady increase in baseline (pre-IPV) SpO2 over the period of four days in the IPV group (Figure 3(a)), whereas this trend was not observed in the CPT group (Figure 3(b)).
Change (mean ± SD) in oxygenation (SpO2 and FiO2) pre and post IPV and CPT from day 1 to 4. Figures on the left-hand side (a and b) show changes in SpO2 pre and 1-h post IPV and CPT intervention. Figures on the right-hand side (c and d) show changes in FiO2 pre and 1-h post IPV and CPT intervention.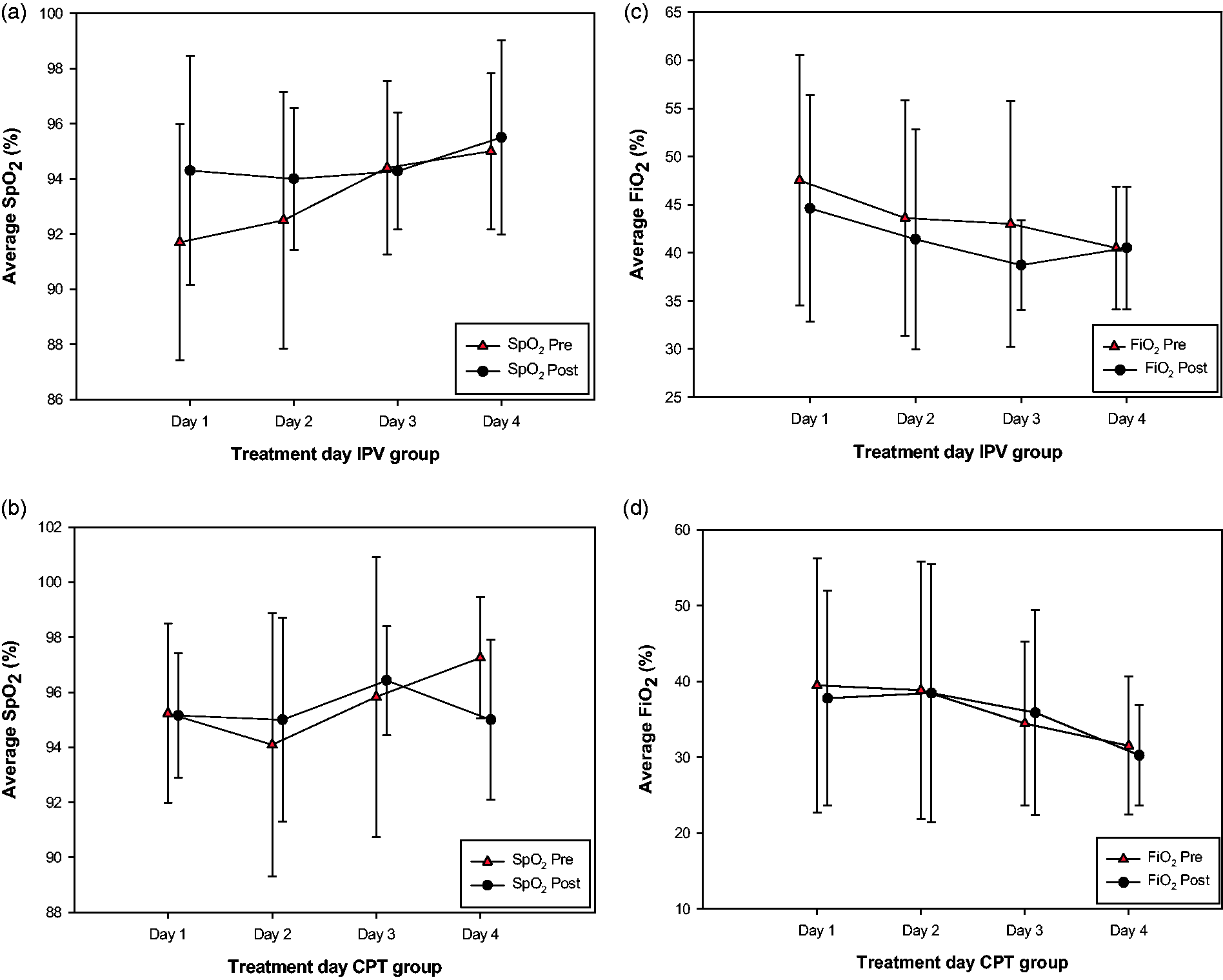
There was a decline in average FiO2 requirement in the IPV group over the first three days from pre to post IPV intervention. Both IPV and CPT groups, however, showed a similar trend in baseline FiO2 requirement over a period of four days (Figure 3(c) and (d)).
Radiological changes
Chest radiographs were available for 17 out of 22 subjects (77 %) in the IPV group and 9 out of 13 subjects (69%) in the CPT group. The overall RAS value in IPV and CPT groups recorded a lower score post-intervention (Figure 4). In the IPV group, 10 out of 17 patients (59%) had lower scores, and in CPT group, three out of nine subjects (33%) had lower chest X-ray scores post-intervention (p < 0.001).
Change in RASs pre and post IPV and CPT. Graphs on the left (RAS pre-IPV and RAS post-IPV) show a shift toward lower scores from pre to post-IPV intervention, whereas graphs on the right-hand side (RAS pre-CPT and RAS post-CPT) show a relatively smaller shift toward lower scores.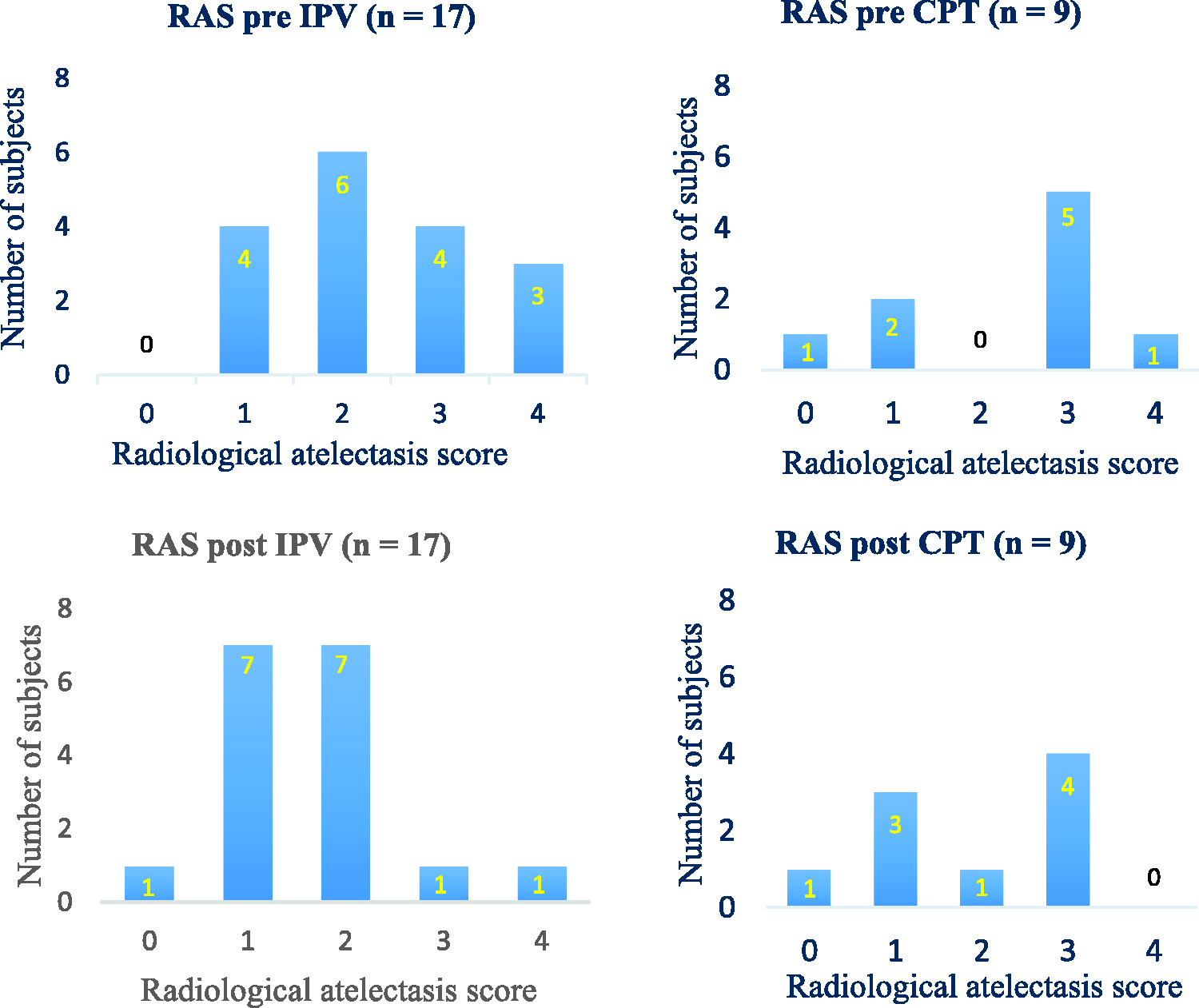
Length of stay
The average ICU length of stay in the IPV group was 9.6 (SD ± 5.9) days, and in the CPT group, it was 11.1 (SD ± 9.3) days (p = 0.59). The shortest ICU stay recorded in the IPV group was three days and two days in the CPT group, whereas the longest ICU stay was 21 days in the IPV group as compared to 39 days in the CPT group.
Discussion
This retrospective pilot study investigated the safety and feasibility of IPV intervention in spontaneously breathing non-ventilated subjects admitted to critical care. This study confirmed that the application of IPV intervention was feasible and safe in non-ventilated patients in critical care. The treatment completion rate for IPV intervention was high amongst the subjects demonstrating good compliance with treatment. There were no records of any adverse events during or immediately after the IPV intervention that would warrant treatment discontinuation.
To our knowledge, there are not many studies that have evaluated the safety and feasibility of IPV intervention in a critical care setting; however, some studies have reported the patient's preferences. A survey in acutely ill cystic fibrosis patients revealed that “ease of use” and “overall preference” for IPV therapy were as good as CPT. 22 Being a retrospective study, we could not evaluate the subject's preferences and acceptance of IPV intervention; however, we found that subjects in both groups completed an equal number of treatment sessions showing high compliance rates without any reports of perceived discomfort.
Being a pilot study, we did not intend to perform any statistical testing for any of the outcomes to determine its significance. Our observation of changes in oxygenation in the IPV group demonstrated immediate (1 h post) and sustained (up to four days) effects of IPV therapy. Vargas et al. 18 reported a similar average increase in SpO2 in 25 COPD patients using IPV that were flow limited post-extubation. Furthermore, our observation of steady change in SpO2 and FiO2 from day 1 to 4 in the IPV group was similar to the findings of a randomized trial in 46 tracheostomized patients that reported an improvement in oxygenation in the first five days when IPV intervention was combined with CPT compared to CPT alone. 23
Secretion clearance is one of the main outcomes of IPV intervention where asymmetrical mechanical waves are known to alter sputum rheology by causing mucolysis and subsequently propelling the mucous toward the proximal airways.7,24,25 A recent bench-top study tested and demonstrated the efficacy of IPV in clearing airway secretions by using an artificial lung model that replicated spontaneously breathing “normal” adult humans. This study by Fernandez-Restrepo et al. 7 supported the efficacy of IPV in airway clearance in non-intubated patients. Being a retrospective study, we could not quantify sputum yield; however, we evaluated the alveolar recruitment by scoring the chest radiographs using RAS. Deakins and Chatburn 26 reported a significant improvement in RAS in 12 mechanically ventilated patients post IPV intervention compared to CPT. They showed that the median atelectasis score remained unchanged after six days in the CPT group, whereas in the IPV group, the score improved just after two days.
Additionally, the observed differences in average length of stay in IPV and CPT group were similar to Antonaglia et al. 15 and Vargas et al., 12 where they reported a reduction in length of ICU stay by 2 and 1.1 days respectively, in non-intubated COPD patients treated with IPV, compared to CPT intervention. However, in our study, we suspect that the observed differences in average length of stay in IPV and CPT group could be due to the discrepancy in the APACHE III score, with a lower score in the IPV group, introducing the possibility of a type I error (i.e. observing a difference where no difference exists).
Overall, although the main intention of the current study was to look at the safety and feasibility and not evaluate the effectiveness of IPV intervention, many additional observations of this study appear to corroborate previous IPV studies in acute and critical care settings. However, the sample size in this pilot study was not adequate to reach any statistical significance in our clinical outcomes.
To our knowledge, the effects of IPV intervention have not been evaluated in the general critical care population. An adequately powered randomized control trial will be conducted to evaluate the effectiveness of IPV intervention in the general critical care population.
Limitations
Being a retrospective pilot study, there are some limitations. We could not record the therapist's time to set up the IPV device nor could we assess their experiences with setting up and delivering IPV intervention. Similarly, the subject's experience and their preference could not be assessed to determine the acceptability and tolerability of IPV intervention. A prospective study to assess these outcomes is needed. In addition to this, there might be information bias due to inconsistencies with measurements of physiological variables such as SpO2 and FiO2. Furthermore, the use of nebulized isotonic saline may have contributed to airway clearance in the IPV group. Last, despite our attempts to match, the sample size in the groups remained unequal, with differences in the APACHE III score.
Conclusion
In summary, this pilot study found that the application of IPV intervention was feasible and safe in spontaneously breathing non-intubated subjects in critical care. The treatment completion rate was high showing good compliance with IPV intervention. There is a need for an adequately powered randomized controlled trial to further evaluate the effects of IPV intervention in a non-intubated population in critical care.
Implications for clinical practice
Application of intrapulmonary percussive ventilation (IPV) intervention is feasible in spontaneously breathing, non-ventilated adult patients in critical care. The treatment completion rate of intrapulmonary percussive ventilation intervention is as good as chest physiotherapy. Intrapulmonary percussive ventilation intervention is safe in spontaneously breathing, non-ventilated adult patients in critical care.
Supplemental Material
INC909704 Supplemental Material - Supplemental material for Feasibility and safety of intrapulmonary percussive ventilation in spontaneously breathing, non-ventilated patients in critical care: A retrospective pilot study
Supplemental material, INC909704 Supplemental Material for Feasibility and safety of intrapulmonary percussive ventilation in spontaneously breathing, non-ventilated patients in critical care: A retrospective pilot study by Anwar Hassan, Maree Milross, William Lai, Deepa Shetty, Jennifer Alison and Stephen Huang in Journal of the Intensive Care Society
Footnotes
Authors' contribution
Anwar Hassan: Study design, ethics approval, data collection, data analysis, manuscript preparation, and submission
Stephen Huang: Study design, ethics approval, data analysis, and manuscript preparation
Maree Milross: Study design, data analysis, and manuscript preparation
William Lai: Data collection and analysis, and manuscript preparation
Jennifer Alison: Study design, data analysis, and manuscript preparation
Deepa Shetty: Data analysis and manuscript preparation
Acknowledgements
We thank Mr Hemant Kumar and Mrs Jaya Kumari for their assistance with electronic records and the data retrieval process.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
