Abstract
Diarrhoea, defined as > 3 loose or liquid stools per day, affects 9.7–41% of intensive care unit patients, negatively impacting on patient dignity, intensifying nursing workload and increasing morbidity. Its pathogenesis is poorly understood, but infective agents, intensive care unit therapies (such as enteral feed) and critical illness changes in the gut microbiome are thought to play a role. We analysed a consecutive cohort of 3737 patients admitted to a mixed general intensive care unit. Diarrhoea prevalence was lower than previously reported (5.3%), rarely infective in origin (6.5%) and associated with increased length of stay (median (inter-quartile range) 2.3 (1.0–5.0) days vs. 10 days (5.0–22.0), p < 0.001, sub-distribution hazard ratio 0.55 (95% CI 0.48–0.63), p < 0.001) and mortality (9.5% vs. 18.1%, p = 0.005, sub-distribution hazard ratio 1.20 (95% CI 0.79–1.81), p = 0.40), compared to patients without diarrhoea. In addition, 17.1% of patients received laxatives <24 h prior to diarrhoea onset. Further research on diarrhoea's pathogenesis in critical care is required; robust treatment protocols, investigation rationalisation and improved laxative prescribing may reduce its incidence and improve related outcomes.
Introduction
Diarrhoea, defined as > 3 loose or liquid stools per day, 1 affects 9.7–41% of intensive care unit (ICU) patients.2–5 Here, it impacts negatively on patient dignity, mobility and self-care; increases nursing workload; and can cause fluid and electrolyte imbalance, impaired nutritional state and dermal injury. Such factors may contribute to the increased patient length of stay (LOS), cost of care and mortality with which it is associated.6–11
The pathogenesis of diarrhoea in the critically ill is poorly understood, 12 but factors including infection (e.g. Clostridium difficile, norovirus), pancreatic exocrine failure and the administration of feed and/or medications (e.g. laxatives, antibiotics) 13 as well as changes in the gut microbiome, and the structure, function and perfusion of the gut mucosal surface, 14 may play a role.
In 2016, we reported that 12.9% of 9331 consecutive patients admitted to the large, mixed general ICU at a Central London teaching hospital (University College London Hospitals, UCLH) suffered diarrhoea. We also reported that non-infective causes appeared to predominate, with one-fifth of patients receiving laxatives before diarrhoea onset. Finally, we found diarrhoea (compared to no diarrhoea) to be independently associated with increased ICU LOS (14.8 vs. 3.2 days, p < 0.001) and mortality (22.0% vs. 8.7%, p < 0.001). 11
We sought to determine whether these observations were more generally applicable, by studying the ICU patients of a smaller North London district general hospital. The aims of our study were to classify the prevalence of diarrhoea in our patient cohort; describe the proportion of infective cases and its association with laxative and enema use; and finally, to describe the impact of diarrhoea on ICU outcomes, including LOS and mortality.
Methods
We performed a retrospective service evaluation, registered as an audit with the Whittington Hospital NHS Trust. All data analysed were fully anonymised. As such, no ethical approval was required.
The Whittington Hospital is a 360-bed teaching hospital located in North London. Computerised records were retrospectively analysed to identify patients older than 18 years who were admitted to its 15-bed mixed medical/surgical ICU/high dependency unit (HDU) for level 2 or 3 care in the 60 months between 1/2/2013 and 1/2/2018. The Whittington ICU has clearly defined protocols for sending stool samples for microbiological or virological analysis, based on the Bristol Stool Chart scoring system (stool type 6 or 7, of sufficient severity and length (>3 stools/24 h), to be sent to the laboratory). Consistent with our previously published methodology, 11 patients who had a stool sample sent during their admission were judged to have suffered from an episode of diarrhoea (by definition, samples sent represent a severity of diarrhoea which suggests the need for investigation and management. Short-lived episodes of infrequent and/or mildly loose stool were therefore not included). The index stool sample was analysed for each admission, but some patients were admitted to the ICU more than once.
Stool samples were analysed by microscopy and culture (from February 2013 to June 2017), and bacterial polymerase chain reaction (PCR) from July 2017 onwards, reflecting changes in Trust laboratory practice. Based on patient presentation and symptoms, samples were also analysed for C. difficile toxin A and B using an immunoassay enzyme, C. difficile glutamate dehydrogenase antigen and Norovirus 1 and 2. The presence of C. difficile antigen indicates the existence of a potentially toxin-producing organism which is likely associated with disease; therefore, samples with the presence of toxin positive and toxin negative/antigen positive were classified as positive infectious samples; these are known to have good sensitivity and specificity. 15
Demographic data, including age, sex, admission category (medical or surgical), and Acute Physiology and Chronic Health Evaluation (APACHE) II score, were collected. Laxatives (including lactulose, senna, macrogol, sodium docusate and ispaghula husk), enemas (phosphate) and suppositories (glycerol) received during admission were also documented. Measures of outcome including LOS and mortality were recorded.
Patient data were extracted from the ICU database and stool data from the hospital laboratory software (ICE Anglia).
Statistical analysis
Data were compared between patients suffering from diarrhoea, and those not suffering diarrhoea. Differences between groups were assessed using Kruskal–Wallis test for continuous data and chi-square test or Fisher's exact test for categorical data. Time to event data would usually be analysed using survival methods such as the Kaplan–Meier method to handle censored data. In the analysis of LOS, for individuals who die prior to discharge, the assumption of future event occurrence in the Kaplan–Meier estimate is violated. Estimators based on treating mortality as a competing risk have been shown to have more clinically relevant interpretations compared to other commonly used to approaches to handle this type of data. 16 Similarly, patients discharged from hospital are likely to have a lower or higher risk of death than average, violating the assumption of non-informative censoring, and standard survival methods are no longer valid. 17 In order to compare time to discharge or mortality between groups, we have therefore performed a competing risk analysis; competing risks are present when a patient is at risk of more than one mutually exclusive event and the occurrence of one will prevent the other event from happening. A sub-distribution hazard ratio (SHR) was calculated for the discharge rate for each group relative to the no diarrhoea group. The competing risks model was fitted using the Fine and Gray approach. 18 As some patients had multiple admissions, a robust cluster estimator was used to relax the assumption that observations are independent. Data were analysed using Microsoft Excel 2010 and Stata Version 14 (StataCorp, Texas).
Results
Diarrhoea prevalence
Between 1/2/2013 and 1/2/2018, a total of 3737 patients were admitted to the Whittington Hospital ICU, median age (inter-quartile range (IQR) 64 (48–77), 1912 (51.1%) male, 1328 (35.5%) from surgical admissions, median APACHE II score 14 (IQR 9–19). Diarrhoea was found in 199 admissions (5.3%).
Patients without diarrhoea had a median age (IQR) of 64 (48–78), 1811 were male (51.5%), 1285 (36.3%) were surgical admissions, with a median APACHE II score of 14 (IQR 9–19). In comparison, patients with diarrhoea were younger, with a median age (IQR) of 63 (50–74), p = 0.40 and a smaller proportion were male (101 (50.7%), p = 0.89). Only 43 (21.6%), p = 0.17 were surgical patients, and their median APACHE II score was higher (18 (IQR 14–24), p < 0.0001).
Infective aetiology
Of the 199 admissions associated with diarrhoea, stool sample analysis suggested an infective aetiology in 13 (6.5%) (see Figure 1): 1/175 (0.006%) of samples sent for bacterial microscopy and culture/PCR, 12/175 (6.9%) of stool sample sent for C. difficile and 0/2 (0%) of samples sent for virological analysis. The single positive stool microscopy and culture was for Campylobacter.
Diarrhoea in the Whittington intensive care unit.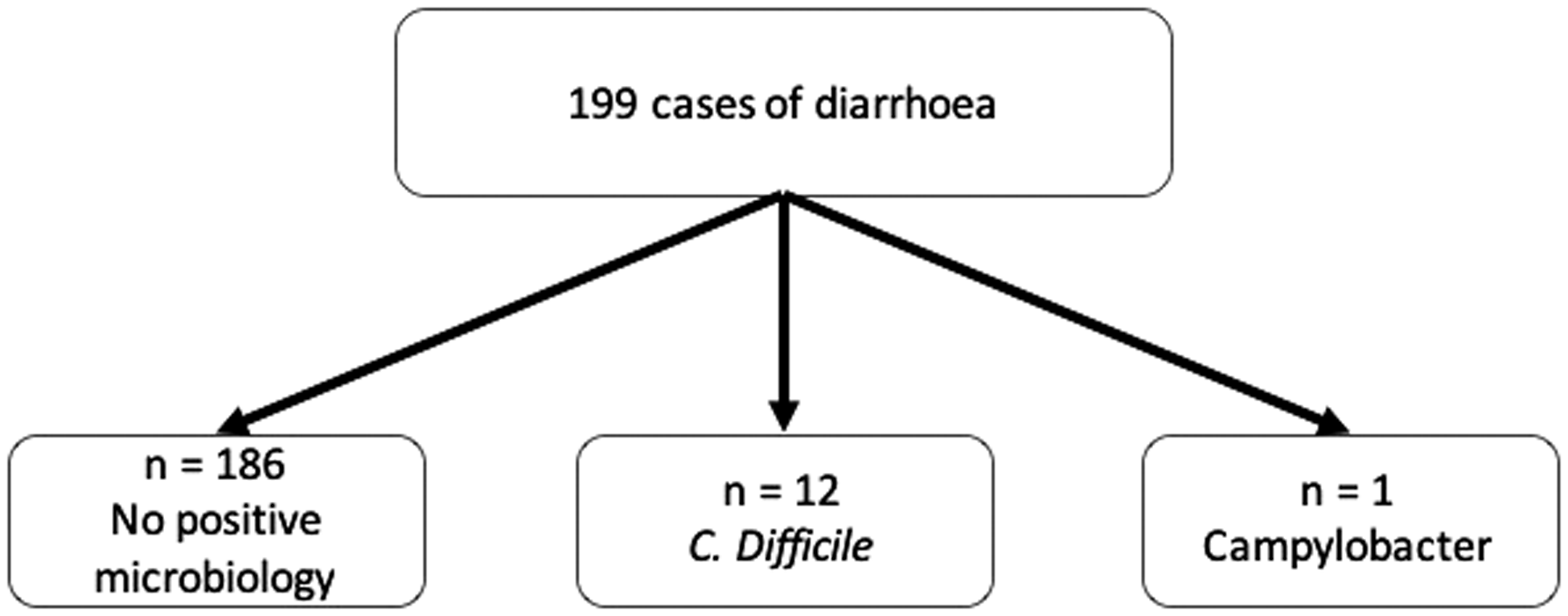
Of the 12 positive C. difficile samples, 10/12 (83.3%) were antigen positive and 2/12 (16.7%) were toxin positive. C. difficile was the most common infective agent, occurring in 12 admissions and 11 individual patients (median age (IQR) 76 (51–78), p = 0.42, 67% male, 67% medical admissions). Three patients were post-operative, with the most common medical reason for admission being sepsis (5/9).
No patient tested positive for more than one pathogen in the same sample.
Laxatives, suppositories and enemas
Of the 3727 ICU patient admissions, 1005 (26.9%) received laxatives during their stay. Of 199 diarrhoea cases, 34/199 (17.1%) received laxatives < 24 h prior to onset of diarrhoea, while 3/12 (25%) of C. difficile cases received laxatives within 24 h of a positive sample.
Laxatives administration.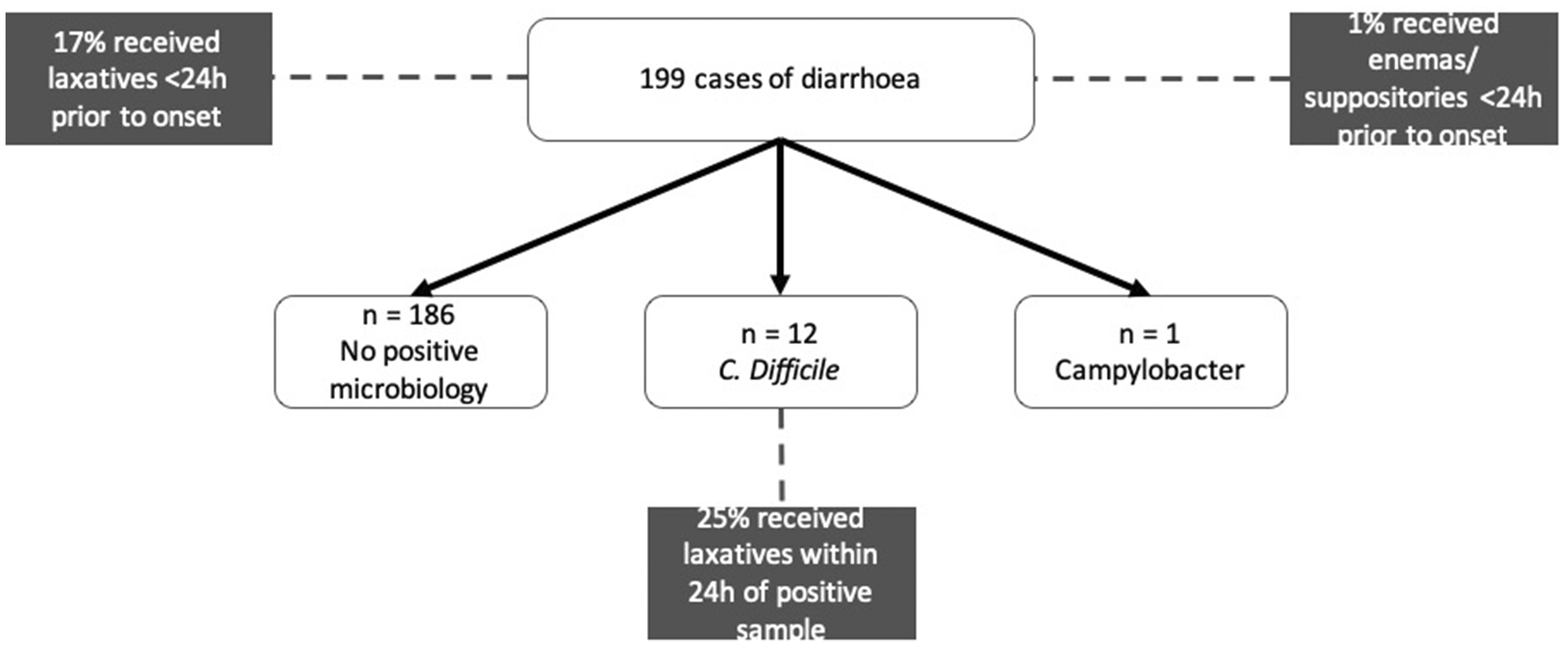
Of the 3727 ICU admissions, 279 (7.5%) received enemas/suppositories during their stay. Of the 199 diarrhoea cases, 2/199 (1%) received enemas/suppositories < 24 h prior to its onset (see Figure 2).
Clinical outcomes
Median ICU LOS was 2.5 days (IQR 1.1–5.5 days) and mortality 11% (412/3737). When compared to those without diarrhoea, patients admitted with diarrhoea experienced greater median ICU LOS (median (IQR) 2.3 (1.0–5.0) days vs. 10 days (5.0–22.0), p < 0.001) and greater ICU mortality (9.5% (356/3727) vs. 18.1% (36/199)), p = 0.005).
Median ICU LOS for C. difficile patients was 11.5 days (IQR 2.0–14.3). Five of 12 (42%) of patients with either C. difficile toxin or antigen positive samples died during their admission. None of the C. difficile toxin positive patient (0/2) died during their ICU admission.
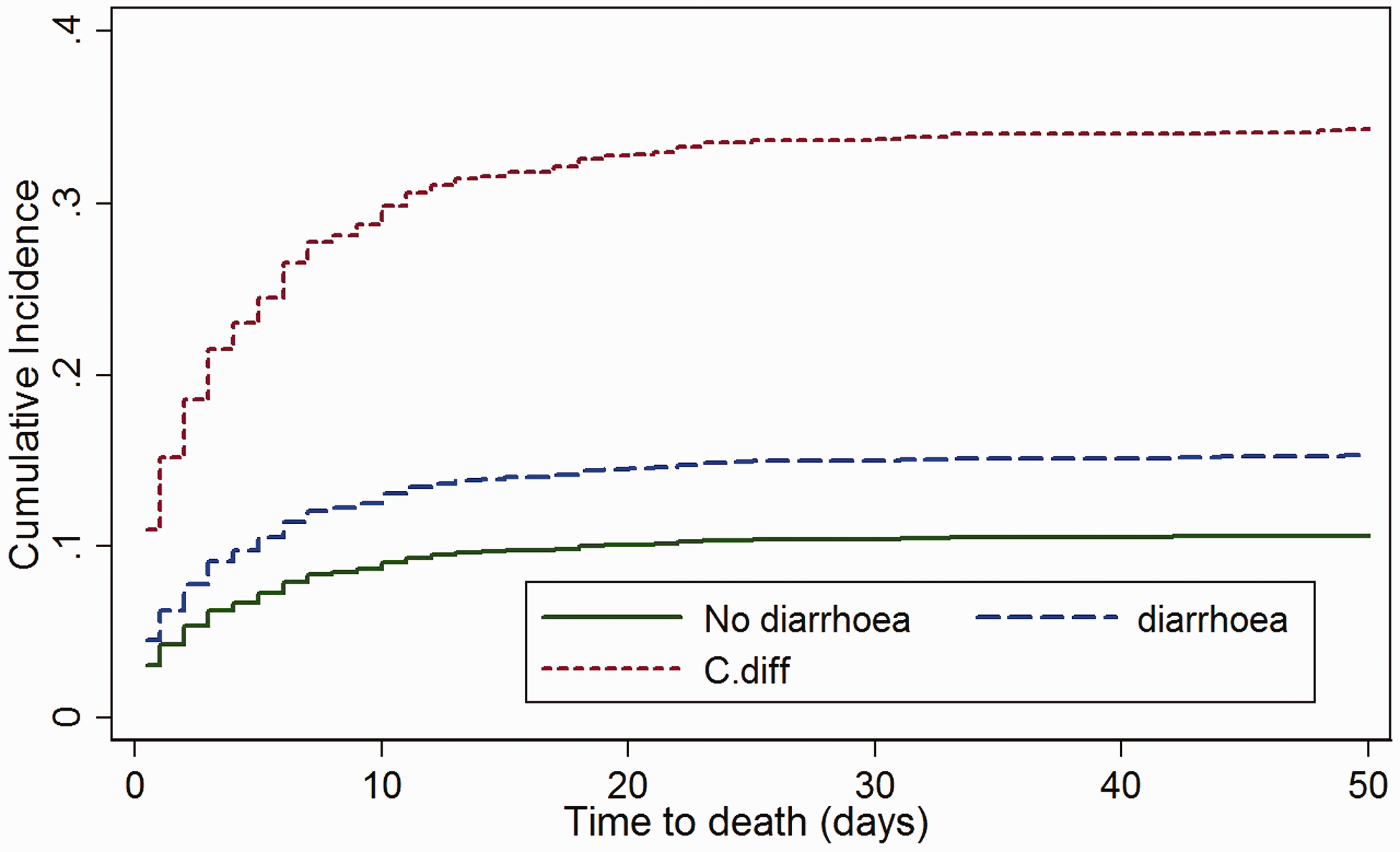
As patients who died had shorter LOS than if they were discharged, despite a worse outcome, mortality needs to be modelled as a competing risk. The cumulative incidence function for discharge (see Figure 3) gives the cumulative proportion of patients discharged over time while accounting for the fact that patients may die before discharge. The sub-distribution hazard ratio (SHR) gives the discharge rate for each group relative to the no diarrhoea group, given that the subject has not died (see Figure 4).
Cumulative discharge incidence with death as a competing risk (plotted over first 50 days). The rate of discharge is decreased by 45% (SHR = 0.55 (95% CI 0.49–0.62), p < 0.001) in those with diarrhoea and by 65% (SHR = 0.35, (95% CI 0.21–.60), p < 0.001) in those with C. difficile. The result remained significant after adjustment for APACHE II score (SHR 0.55 (95% CI 0.48–0.63), p < 0.001 and SHR 0.43 (0.27–0.70), p = 0.001 for diarrhoea and C. difficile, respectively). Cumulative ICU mortality incidence with discharge as a competing risk (plotted over first 50 days). The mortality rate is increased by 47% (SHR = 1.47 (95% CI 1.01–2.15), p = 0.044) in those with diarrhoea and by 273% (SHR = 3.73 (95% CI 1.46–9.52), p = 0.006) in those with C. difficile. After adjustment for APACHE II score, the effect for those with diarrhoea was no longer significant (SHR 1.20 (95% CI 0.79–1.81), p = 0.40), but the effect remained significant for those with C. difficile (SHR 2.71 (1.07–6.84), p = 0.035).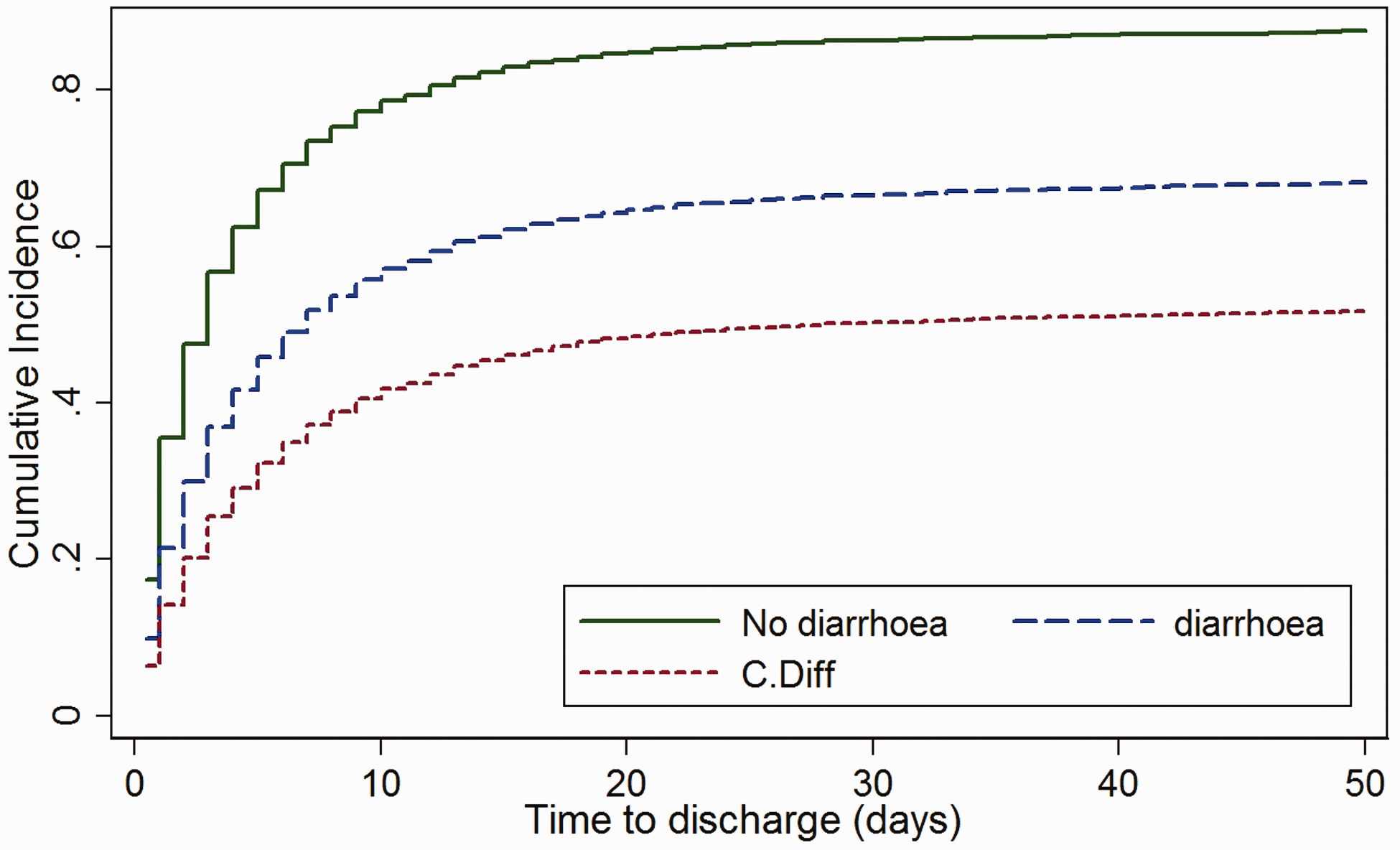
Discussion
We found diarrhoea to be common, affecting nearly 1 in 20 admissions to a small district general ICU/HDU. It was associated with greater ICU LOS (2.3 (1.0–5.0) days vs. 10 days (5.0–22.0), p < 0.001), which persisted after adjusting for illness severity. Diarrhoea was associated with increased mortality (9.5% (356/3727) vs. 18.1% (36/199), p = 0.005); however, the effect was no longer significant when adjusted for illness severity. Infective aetiology was uncommon (<7% of cases) and use of laxatives prior to diarrhoea onset was common (17.1%).
The prevalence of diarrhoea in our unit was lower than the 12.9% which we had previously reported in a large central London ICU (UCLH), 11 and others report a prevalence of 9.7–41%.2–5 This may reflect differences in case-mix: for instance, the proportion of surgical patients was greater at UCLH (66.9% of patients enrolled in the UCH study were surgical, 11 in comparison with 35.5% at the Whittington), necessitating increased enteral feeding, which has been shown to be associated with diarrhoea. 4 Median APACHE II scores also differed in the two studies (14 at the Whittington vs. 16 at UCLH, p < 0.05). Our diagnosis of diarrhoea was dependent on a stool sample being sent to the laboratory: whilst this was the same criterion which we had used before, 11 the prevalence of milder cases may have been underestimated when compared to the studies of others.
In 93.5% of cases, no enteric infection could be identified (none viral, C. difficile in 6% and other bacterial infection in 0.5%). These data are similar to those at UCLH (prevalence of pathological stool samples 6.5% vs. 9.2%; and proportion with non-C. difficile bacterial infection 0.5% at both sites). However, the proportion of cases with C. difficile infection was lower (6% vs. 9.3%) as were the proportion with viral infection (0% vs. 5.2%). These differences might partly reflect the smaller sample size of the Whittington study (despite recording data for a year longer, just under 6000 fewer admissions were included in our study), and improved antibiotic stewardship as awareness of the deleterious effects of C. difficile becomes more apparent. This is in contrast to reports in the literature, which report increased rates of C. difficile infection.19,20 The comparatively low number of samples sent for virological analysis (two samples) is a reflection of laboratory policy; diarrhoea samples are not routinely tested for norovirus or rotavirus. Samples are run on the basis of clinical suspicion.
Overall, 26.9% of admissions received laxatives – a greater proportion than we reported at UCLH (17.5%). This difference might be accounted for, in part, by laxatives being a part of an electronic software ‘prescribing bundle’ on the unit (listed as PRN, or ‘as required’ medications, prescribed to patients on admission to the ICU). Some 17.1% of patients received laxatives prior to diarrhoea onset, compared to 26.1% of non-diarrhoea cases (p < 0.05).
Whilst fewer total patients received laxatives prior to diarrhoea onset when compared to the UCLH data (17.1% vs. 20.2%) (and fewer received enemas or suppositories (1% vs. 11.4%) < 24 h prior to diarrhoea onset), these data taken together suggest that laxatives use may be a contributory factor in diarrhoea's aetiology. Laxatives are regularly prescribed in ICU as constipation is common and well recognised to have negative clinical effects. 21 Although laxative prophylaxis does not form part of prescribing guidelines, they are often prescribed as part of an ICU bundle to prevent this complication, along with electrolyte replacement, analgesia and anti-emetics. Care bundles are recognised to be beneficial in treating, for example, sepsis. 22 However, their role in managing constipation is yet to be elucidated, and our data, and that of other colleagues,11,23 suggest that they perhaps cause more harm than benefit. We note that more of our C. difficile patients (25% vs. 13.4%) received laxatives within 24 h of a positive sample than noted in our previous study; however, our sample size of 12 patients is perhaps too small to draw definitive comment.
Patient LOS was greater in those affected by diarrhoea than in those unaffected (10 days (5.0–22.0) vs. 2.3 days (1.0–5.0), p < 0.001). This finding matches with that we made at UCLH (9.5 days (4.6–20.2) vs. 1.7 days (0.9–3.2), p < 0.001). The rate of discharge at the Whittington was decreased by 45% (SHR 0.55 (0.49–0.62), p < 0.001) in those with diarrhoea compared to those without. This must be caveated by acknowledging patients with longer ICU admissions are likely to be more unwell, therefore more likely to receive interventions such as antibiotics and to suffer from critical illness gut changes, and thus more likely to develop diarrhoea. However, when adjusted for illness severity in this study (APACHE II score), the result remained significant (SHR 0.55 (0.48–0.63), p < 0.001). Furthermore, we had previously shown that this association persists after adjustment for confounding factors and allowing for the fact that patients discharged earlier have less time to develop diarrhoea by fitting diarrhoea as a time-dependent co-variate. 11 This suggests a possible causal role for the presence of diarrhoea itself in increasing LOS. Such a role is plausible, as diarrhoea may impair nutrient intake, contribute to dermal injury and to fluid and electrolyte imbalance and thus increase dependency on higher levels of nursing care. It may also be that the presence of diarrhoea impeded discharge to an open ward, given perceived infection control considerations (it is Trust policy for severe diarrhoea cases to be isolated in a side room, which are in short supply); however, the large difference in LOS between diarrhoea and no diarrhoea is unlikely to be explained by this delay alone.
Patients with C. difficile also had a greater LOS compared to those without diarrhoea (11.5 days (2.0–14.3) vs. 2.3 days (1.0–5.0)) and had a decreased rate of discharge by 65% (SHR 0.35 (0.21–0.60), p < 0.001) (an association which persisted after adjusting for illness severity; discharge was decreased by 57% in the C. difficile group following adjustment (SHR 0.43 (0.27–0.70), p = 0.001)). This matched findings that were made at UCLH (17.4 days (8.4–32.3) vs. 1.7 days (0.9–3.2)), and this association is reflected in the literature;24,25 it suggests that diarrhoea caused by C. difficile infection may also have a causal role in increasing LOS, for the reasons explored above.
Similarly, crude mortality was greater in patients affected by diarrhoea than in those without (18.1% vs. 9.5%%, p = 0.005). These data match those identified at UCLH (22.0% vs. 8.7%, p < 0.001). The mortality risk for patients suffering from diarrhoea in ICU was one-and-a-half times greater (SHR 1.47 (1.01–2.15), p = 0.044) than patients not suffering diarrhoea at any time point during their ICU admission. However, when adjusted for APACHE II score, the result was no longer significant (SHR 1.20 (0.79–1.81), p = 0.40), illustrating that the effect on mortality may, in part, be due to illness severity. Patients suffering from C. difficile also had higher crude mortality compared to those without diarrhoea (41.7% vs. 9.5%), which matched the results from the patient population at UCLH (28.0% vs. 8.7%), and is similar to other studies that observe outcomes from this infection in ICU patients. 5 The mortality risk for patients suffering from C. difficile is over three-and-a-half times greater (SHR 3.73 (1.46–9.52), p = 0.006) than those not suffering from diarrhoea. When adjusted for APACHE II score, the mortality risk was reduced to just over two-and-a-half times greater in the C. difficile group (SHR 2.71 (1.07–6.84), p = 0.035). Such a role in mortality is plausible, as diarrhoea impairs nutrient intake through the enteral route, leading to malnutrition, which has been shown to be an independent risk factor for in-hospital mortality. 26 Moreover, as mentioned previously, diarrhoea can impair fluid and electrolyte balance, and cause dermal injury, which can further impact on mortality.
There were several limitations to this study. As previously stated, the prevalence of diarrhoea may have been higher than reported as the diagnosis of diarrhoea depended on a stool sample being sent to the laboratory. However, robust ICU protocols exist to ensure that stool samples are sent promptly if an infective cause is suspected. Analysis of electronic records did not allow us to adjust for potential iatrogenic confounders, such as antibiotic administration and enteral feeding, which have been shown to be independent risk factors for diarrhoea. 27 Moreover, we could not determine any relationships of diarrhoea with patient-specific variables, such as the level of organ support, co-morbidities or reason for admission. We were unable to assess the impact of small and large bowel function, as this was an observational rather than mechanistic study. Despite studying data covering a full five-year period, the number of diarrhoea cases was relatively small. A large-scale prospective study would help clarify the impact of such factors in diarrhoea pathogenesis. Doing so might identify factors amenable to intervention, thus mitigating the financial, morbidity and mortality burdens associated with ICU diarrhoea.
In the meantime, robust and regular review of patient's gastrointestinal status, with prompt recognition, investigation and treatment of diarrhoea in light of its association with worsening patient outcomes, should be performed. At the Whittington, a trial of removing laxatives from the prescribing bundle, and protocolised care to ensure appropriate identification of the cause of diarrhoea, management of reversible aetiology and appropriate laxative prescribing, have been suggested as possible ways to reduce the burden of this condition. The low diagnostic yield of laboratory investigations for diarrhoea suggest that rationalising of investigations could help reduce the financial cost to the department; indeed, Manthey et al. suggest that testing for enteric pathogens other than C. difficile in ICU should be avoided and is only reasonable when diarrhoea commenced <48 h after hospital admission. 28
In summary, diarrhoea has a prevalence of 5.3% in the Whittington ICU, less than that seen in previous studies. The infective burden is low, suggesting another aetiological cause for diarrhoea in the critically unwell. Over 17% of our patients with diarrhoea received laxatives in the 24 h prior to its onset, indicating a need to rationalise prescribing. There is an association with increased LOS and patient mortality in patients with diarrhoea, which is increased further in patients with C. difficile.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received no external funding, but was supported by the National Institute for Health Research University College London Hospitals Biomedical Research Centre.
