Abstract
We describe the first case of venous air embolism during percutaneous tracheostomy performed during continuous renal replacement therapy with citrate anticoagulation. Venous air embolism is a known risk during percutaneous dilatational tracheostomy; concurrent use of extracorporeal circuits may present a compounded risk due to the negative pressure developed by the blood pump. Use of regional citrate anticoagulation, permitting invasive procedures to be performed without discontinuing renal replacement therapy, may lead to an increase in the incidence of venous air embolism during tracheostomy.
Introduction
Regional citrate anticoagulation (RCA) for renal replacement therapy (RRT) is recommended by KDIGO for prolonging filter lifetime and reducing spontaneous filter failure. 1 It is the default anticoagulant in the Intensive Care Unit (ICU) at Southmead Hospital, Bristol. It is common in our ICU to perform invasive procedures without suspending RRT, allowing flexibility with timing, increasing the effective dose delivered and reducing the costs associated with new haemofilter sets. The potential complications of this approach have, however, not yet been clearly illustrated. We report a case where ongoing RRT during percutaneous dilatational tracheostomy (PDT) may have been a factor in the generation of venous air embolism (VAE).
Case report
Consent was provided by the patient for publication of this case report.
A 62-year-old man was ventilated having sustained a crush injury to his chest causing injury to the thoracic spine and paraplegia. He was receiving RRT with RCA for acute kidney injury via a right internal jugular high-flow central venous catheter. PDT was undertaken to facilitate respiratory weaning. The procedure was performed under bronchoscopic guidance by two experienced practitioners, one a consultant. At the time of tracheostomy, RRT had been running well for over 48 h with a net negative fluid balance of 1000 ml each day.
As is our standard practice the patient was positioned 20° head up after induction of general anaesthesia. This position is favoured to aid oxygenation, venous drainage and intra-cranial pressure control. The tracheal tube was withdrawn under direct vision to the level of the vocal cords. Lidocaine with adrenaline was infiltrated subcutaneously prior to needle to skin.
The first puncture, made without skin incision, was assessed as being between cricoid and first tracheal ring; the cannula was withdrawn and the site loosely covered with sterile gauze. A second puncture was made and judged to be between first and second tracheal rings. At this point air bubbles were noted in the arterial access limb of the RRT circuit shortly followed by a fall in blood oxygen saturation (100%–92%) and expired end-tidal carbon dioxide (CO2) (3.9 kPa–2.3 kPa). The end-tidal capnograph had a normal shape and the tracheal tube – checked with the bronchoscope – had not moved. The filter then failed.
There was complete resolution of these changes within 5 min. The procedure was completed uneventfully. A chest radiograph taken post-procedure showed no significant changes, and although there was no active exclusion of a pulmonary embolus, the patient was receiving pharmacological thromboprophylaxis.
Discussion
VAE occurs when atmospheric air enters the circulating vascular system driven by a negative pressure gradient between the right atrium and the point of entry, typically a surgical incision or puncture. 2 One series in neurosurgical patients demonstrated that a gradient of as little as 5 cm H2O was sufficient to aspirate 200 ml of air, 3 and at this gradient a potentially lethal volume of 100 ml could be aspirated through a 14 -g cannula in seconds. 2
The clinical diagnosis of an air embolus can be a difficult one – signs and symptoms are vague and dependant on the patient’s body position, mimicking cardiac, neurological and pulmonary pathologies. 4 The gold standard for detecting VAE is transoesophageal echocardiogram, which can detect volumes of air as small as 0.02 ml.kg−1, with well positioned precordial Doppler providing a non-invasive alternative capable of detecting 0.05 ml.kg−1 of air. Whilst less sensitive, the most ubiquitous monitor that may demonstrate signs of air embolism in the invasively ventilated patient is end-tidal capnography, with the volume of air entrained correlating with the degree of decrease in end-tidal CO2 concentration and duration for which this occurs. 2
Invasive procedures involving the neck have long been known to be high risk for VAE – the first published report of VAE following neck surgery occurred as early as 1818. 5 The first published case of VAE following tracheostomy was in 1958, as a delayed complication of surgical tracheostomy in an infant. 6 Case reports detailing VAE after percutaneous tracheostomy are, however, more difficult to find. There were no reported cases of VAE as a complication in recent randomised trial comparing early and late percutaneous placement of tracheostomy. 7
Haemodialysis was also identified early on as a potential risk factor, due to the use of a blood pump and long extracorporeal circuit; however, despite this the reported occurrence of VAE in this cohort is sparse, 8 with most cases occurring during insertion, removal or disconnection of central venous catheters. 4 This may be due to perfusion being stopped in modern machines when air is sensed by bubble detectors.
Our case is of interest as it suggests that procedures on the neck may be at higher risk of generating VAE when associated with an extracorporeal circuit. This has also been reported by Lother et al., 9 who detail two cases of VAE during PDT whilst patients were receiving extracorporeal membrane oxygenation (ECMO), resulting in failure of the ECMO system and death due to hypoxia. They postulated that the negative pressure generated by the ECMO system through the proximal port of the AVALON ELITE cannula could result in suction of air through the inferior thyroid vein.
Whilst the flow rates are far lower, the Prismaflex system we use for RRT can generate access pressures of −50 to −150 mmHg with blood flow rates of 250 ml·min−1 via the central venous catheter. We believe that a similar effect to that described with ECMO occurred with RRT via a catheter sited appropriately in the SVC, i.e. there was a suction effect from the extracorporeal circuit. The large pressure difference draws a high volume of air through even a small hole in a minor vein (Figure 1).
Venous air embolism during percutaneous tracheostomy whilst patient is undergoing renal replacement therapy.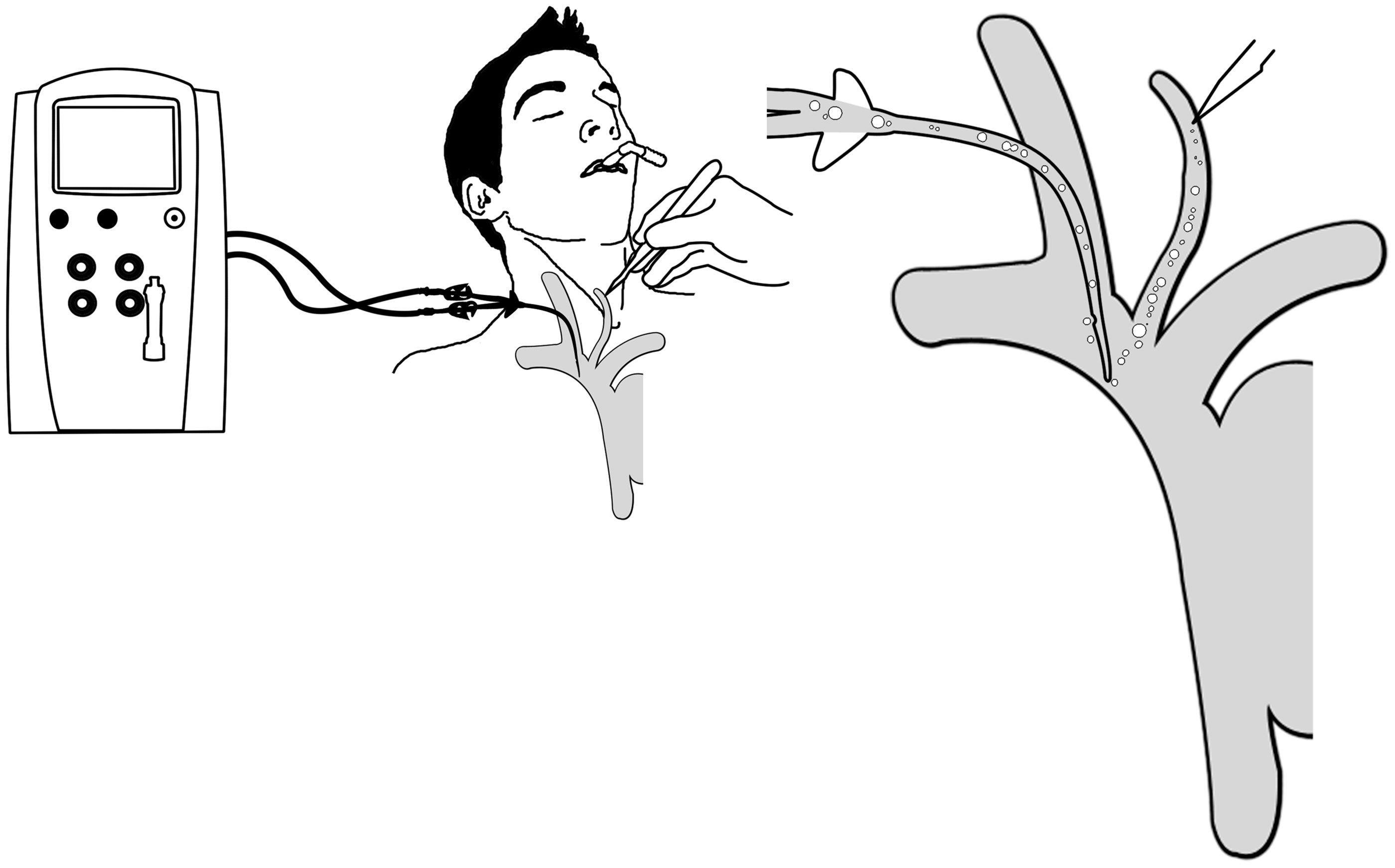
Being aware of the risk of VAE during invasive procedures is vital to minimise risk of VAE development; patient positioning (ideally Trendelenburg) and optimisation of BP to avoid hypotension can both be useful in reducing occurrence of VAE. Post-procedure having a high index of suspicion is vital to enable early recognition and prompt treatment of VAE.
The initial step in the management of VAE once suspected is to reduce any further accumulation of air in the venous system by occluding the surgical point of gas entry. Patient should be placed in the Trendelenburg position to prevent right ventricular outflow tract obstruction. Supplemental oxygen should be provided to all patients who are hypoxaemic or symptomatic. Oxygen administration causes nitrogen washout and hence decreases embolus size. If the patient is not already mechanically ventilated, this should be considered. If this fails hyperbaric oxygen therapy can cause a reduction in air embolus size more rapidly than conventional oxygen therapy, however, requires a specialist centre and must be initiated within 5 h of VAE formation.
Another treatment option is the aspiration of air via venous catheters inserted into the right side of the heart. We believe that in this case the RRT system that caused the entrainment at the same time removed, via the SVC catheter, a proportion of the air entrained. This proved to be sufficient to limit the fall in saturations and end-tidal CO2 and to prevent cardiac compromise. Once the circuit had failed no further air was entrained leading to rapid resolution with no other action needing to be taken. The importance of the RRT circuit is underlined by our subsequent experience performing transthoracic echocardiography during PDT in a patient not receiving RRT: no evidence of VAE was seen (unpublished observations).
Our PDT database includes data from more than 2000 tracheostomies, of which approximately 500 have been performed since the switch to citrate; there are no other documented cases of VAE. This complication has not lead to a change in the practice within our ICU as we believe the benefits of the head up position for oxygenation, airway positioning and ICP control along with the flexibility of timing for the procedure and ability to deliver adequate dialysis dose outweigh the risks of a rare event in VAE. We have encouraged vigilance amongst our PDT operators and educated about this complication, advising the application of saline-soaked gauze to puncture sites. Pre-procedure ultrasound is not a current standard in our unit and we do not believe it would have altered the outcome in this case as no major vessel was injured and there was evidence of no pre- or post-VAE bleeding.
Conclusion
We describe a case of VAE during percutaneous tracheostomy performed whilst on continuous RRT with citrate anticoagulation. VAE is a known risk during PDT; concurrent use of extracorporeal circuits may present a compounded risk. Use of RCA, permitting invasive procedures to be performed without discontinuing RRT, may lead to an increase in the incidence of VAE during tracheostomy.
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
