Abstract
Introduction
Percutaneous tracheostomy procedures are commonplace in intensive care units worldwide. Haemorrhagic complications, though potentially fatal, are thought to be under-reported. Ultrasonography use has not established itself in these procedures. This study assesses the prevalence and characteristics of pre-tracheal blood vessels, to aid intensivists on potential procedural hazards.
Method
A retrospective, observational study was performed in the UK. Computed tomography angiograms from January 2012 to October 2014 were randomly retrieved and analysed for vessel data.
Results
A total of 343 adult patients were included (mean age: 65 (inter-quartile range 52–79), male: 63%). Forty-one percent of patients demonstrated a vessel overlying percutaneous tracheostomy insertion sites (C6–T1); veins were more common than arteries (69%, p = 0.001). Males were more likely to display veins (78 vs. 53%, p < 0.001).
Discussion
A substantial proportion of patients exhibited pre-tracheal vessels. Front-of-neck ultrasonography has huge potential to identify these. Based on these data, we believe ultrasonography may be an effective screening tool for percutaneous tracheostomy procedures to reduce complications. Further research is required to study outcomes.
Introduction
Percutaneous tracheostomy (PT) has become routine practice in most intensive care units (ICUs). 1 Ultrasonography for venous access and regional nerve blockade is a well-established technology amongst intensivists and anaesthetists. Indeed, the National Institute for Clinical Excellence in 2002 published guidance on the use of ultrasonography for inserting central lines, due to the high failure rate and associated complications including arterial puncture, arterio-venous fistula and pneumothorax. 2 In our experience, bronchoscopic assistance and limited blunt dissection as adjuncts to percutaneous techniques are common practices, whereas ultrasonography screening of neck structures to identify anatomical variations prior to insertion is not.
PT techniques have been shown to be safer than surgical techniques. 3 Fatal haemorrhage from PT is a known but rare complication, although it is thought to be under-reported. 4 One study highlighted injury to blood vessels, which could have possibly been identified using ultrasound. 5 It was considered significant in 24 (5%) of 497 PT procedures performed without ultrasound evaluation prior to tracheal dilatation. Of those 24 patients, a quarter required conversion to surgical tracheostomy. The fourth National Audit Project reported two adverse outcomes in intensive care patients where a tracheostomy tube could not be sited successfully due to ‘difficulty’. 6 There were also two cases of fatal haemorrhage with PT, one occurring on insertion and the other on removal. The report later went on to suggest intensive care patients are of a higher risk category yet no national guidance on use of ultrasound or imaging prior to PT insertion exists.
A case report in the Netherlands identified two mortalities with delayed fatal haemorrhage secondary to PT, one of which died from tracheo-innominate fistula haemorrhage and the other from profuse venous bleeding during tracheal dilatation. 7 Sustic et al. looked at 26 patients, 15 of which were performed on as a blind technique, with 11 undergoing ultrasound guided PT. 8 Cranial misplacement was found in 5 of the 15 blind techniques and in none of the ultrasound guided group. A study published in 2011 looked at 10 ultrasound guided percutaneous tracheal punctures and wire insertions in nine cadavers; eight out of nine wire insertions were successful on first attempt and one on the second 9 . Research in France looking at ultrasound-guided PT was successful with low complication rates in the critically ill obese population. 10
To our knowledge, there is no current evidence within the literature demonstrating that ultrasound screening prior to PT insertion reduces bleeding risk. This study aims to assess the prevalence and characteristics of pre-tracheal blood vessels in the general population. This may help ascertain whether front-of-neck imaging is warranted prior to insertion of PT tubes.
Methods
This was a retrospective, observational study performed at King George’s and Queen’s Hospitals in London and Essex, UK. Data were collected using computed tomography angiography (CTA) to ascertain information regarding neck vasculature. Patient data were taken over the period January 2012 to October 2014. Inclusion criteria included any patient over 16 years old who had received a CTA of the neck. Exclusion criteria consisted of paediatric patients. CTA images were collected from the hospital trust's picture archiving and communication system. These were then examined using the support of a consultant neuro-radiologist.
Collected data consisted of patient demographics (age and sex), presence of a pre-tracheal blood vessel, type of vessel (artery or vein), the predominant running direction of the vessel (horizontal or vertical) and the corresponding spinal level at which the vessel was most prominent.
Axial and sagittal images were used to identify all vessels within the neck relative to the larynx, trachea and spine. The different contrast densities allowed differentiation between arterial and venous vasculature relative to the internal carotid artery and internal jugular vein.
We deemed the pre-tracheal region relevant to PT insertion (‘PT-region’) to be the C6–T1 levels. PT insertion varies between the first/second tracheal ring space to the third/fourth tracheal ring space. Traditional teaching conveys C6 as the anatomical level of the cricoid cartilage, however, during neck extension, the trachea is ‘pulled-up’ superiorly; therefore, it would be reasonable to suggest that the first/second tracheal ring space could overlie the C6 level. This was deemed the superior border for the relevant region. Due to its proximity to the jugular notch, the inferior border of the relevant region was deemed the T1 level.
The primary outcome was the prevalence of a pre-tracheal vessel in the ‘PT-region’. Secondary outcomes consisted of the prevalence of the type of vessel (artery or vein), their running direction and their corresponding spinal level.
All relevant data were collected into a data extraction spreadsheet for further analysis. Data analysis was done using Microsoft Excel (Microsoft, 2011). Chi square and Fisher exact tests were used to compare categorical variables, whilst Student t tests were used to compare continuous variables. We defined statistical significance as a p value of < 0.05.
Results
A total of 345 patient CTAs were collected between January 2012 and October 2014. Two paediatric patients were excluded, resulting in a study sample of 343. Sixty-three percent (n = 215) of patients in our cohort were male, 37% (n = 127) were female. Gender data were not available in one patient. Figure 1 below illustrates the age range of the cohort. Mean age was 65, median age was 66 (inter-quartile range (IQR) 52–79) and the mode age interval was 71–80 years old. Ninety-two percent of patients were aged over 40 and 62% were aged over 60.
Age intervals of included patients.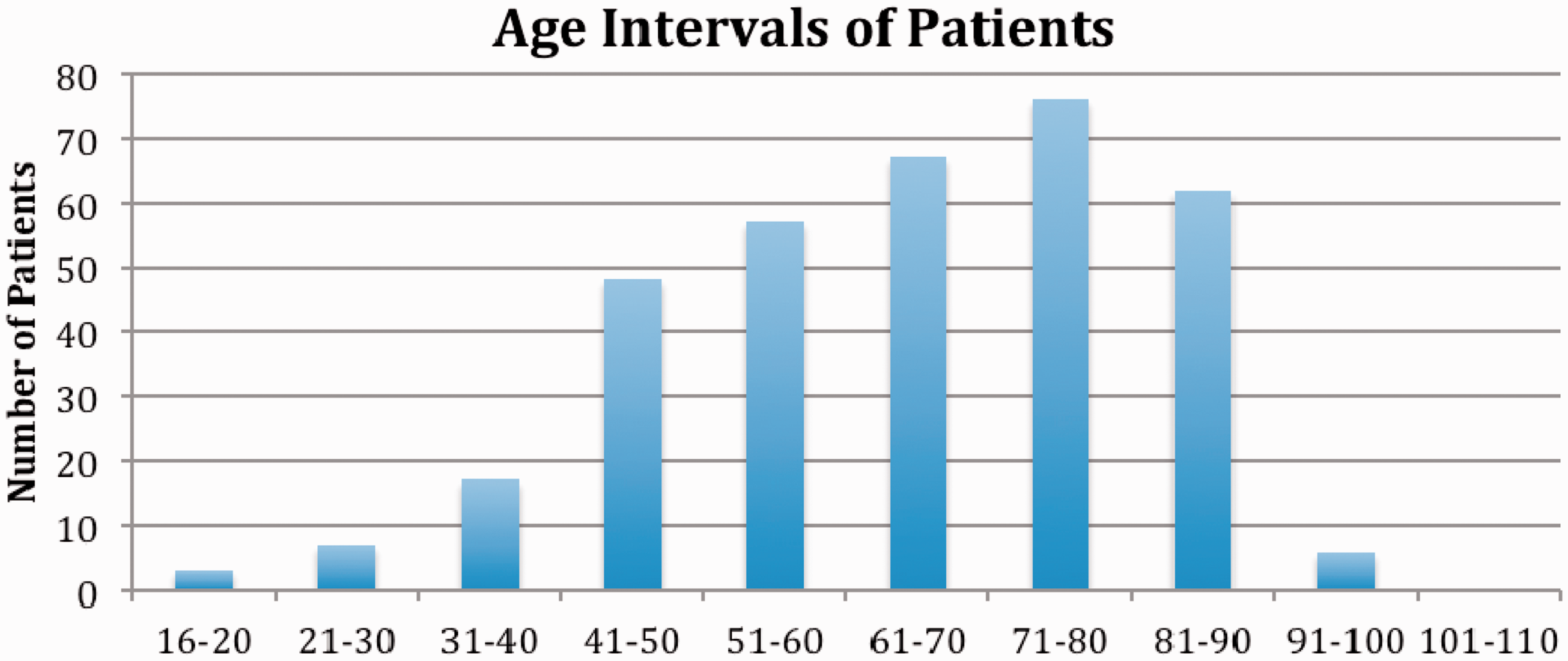
Fifty-five percen (n = 187) of all patients demonstrated a pre-tracheal vessel (artery, n = 56 (16%); vein, 131 (38%)). Of these, 41% (n = 142) demonstrated a vessel within the ‘PT-region’ (Figure 2), with no gender difference (males 64% vs. females 36%, p = 0.65). Mean age (IQR) of patients with and without a pre-tracheal vessel in this region was similar at 65.5 (55.0–77.0) and 67.4 (52.0–79.0), respectively, (p = 0.43) (Table 1). Thirteen percent (n = 44) of all patients demonstrated a pre-tracheal artery and 29% (n = 98) of patients demonstrated a pre-tracheal vein on the angiograms (p = 0.001). There was no difference in ‘PT-region’ vessel presence between genders (64 vs. 36%, p = 0.65). Males were more likely to display a vein than an artery (71 vs. 20, p < 0.001), whilst females were not (27 vs. 24, p = 0.77). Mean age (IQR) of patients with a ‘PT-region’ artery was 59.9 (48.8–72.3), whilst those with a vein were older at 67.8 (61.0–78.0) (p = 0.0012).
Proportions of CTAs demonstrating a ‘PT-region’ vessel. CTA: computed tomography angiography; PT: percutaneous tracheostomy. Table demonstrating demographic and vessel characteristics. IQR: inter-quartile range; PT: percutaneous tracheostomy. Denotes analysis between ‘PT-region’ vessel present and vessel absent groups. Denotes analysis between ‘PT-region’ artery and vein groups. Bold values indicate statistical significance.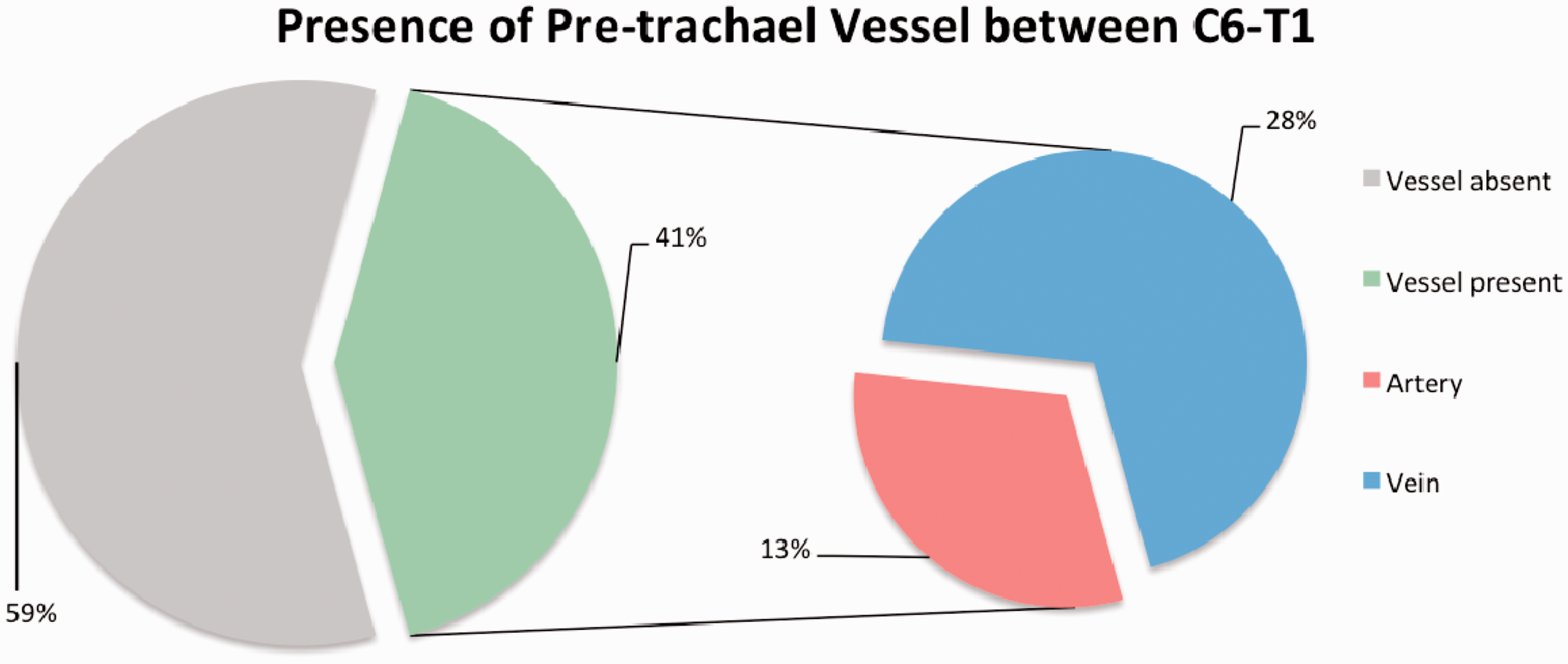
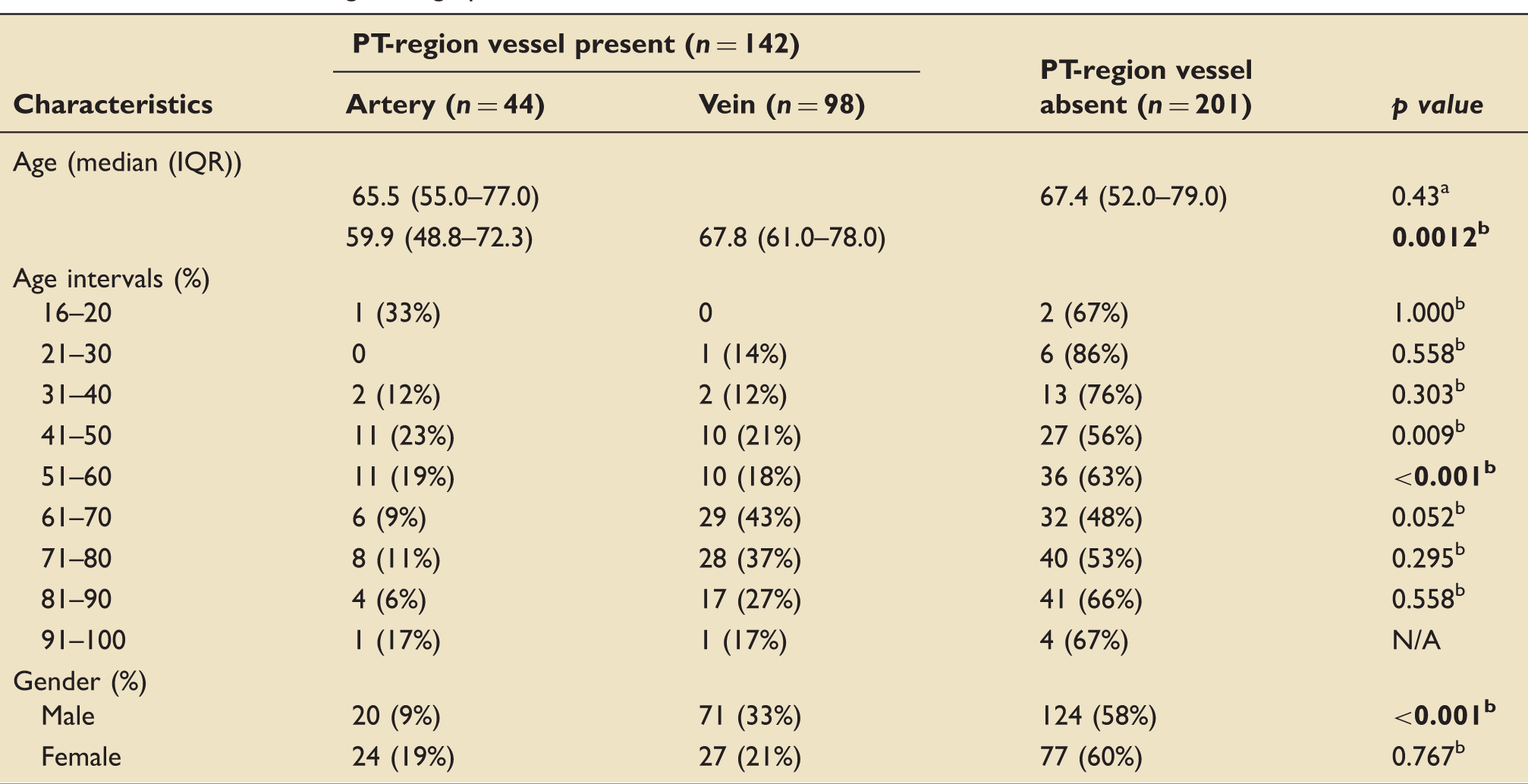
Patient ages were arbitrarily grouped into intervals and ‘PT-region’ vessel data documented (Table 1). Age thresholds were subsequently analysed to assess for statistical significance. Using thresholds of 20, 30, 40, 50, 60, 70, 80 and 90 years, we compared patients younger against patients older than the threshold. The threshold ages were included in the younger group in every comparison. When comparing the presence or absence of a pre-tracheal vessel between the two groups, no threshold reached significance, indicating the varying presence of vessels in the older group compared to younger group were likely due to chance. Subsequently, we compared the presence of an artery or vein in the ‘PT-region’ cohort. The only threshold to reach significance was at 60 years, indicating that the prevalence of veins compared to arteries is higher once patients reach 61 years (p < 0.001).
Table demonstrating vessel height in terms of spinal vertebrae level.
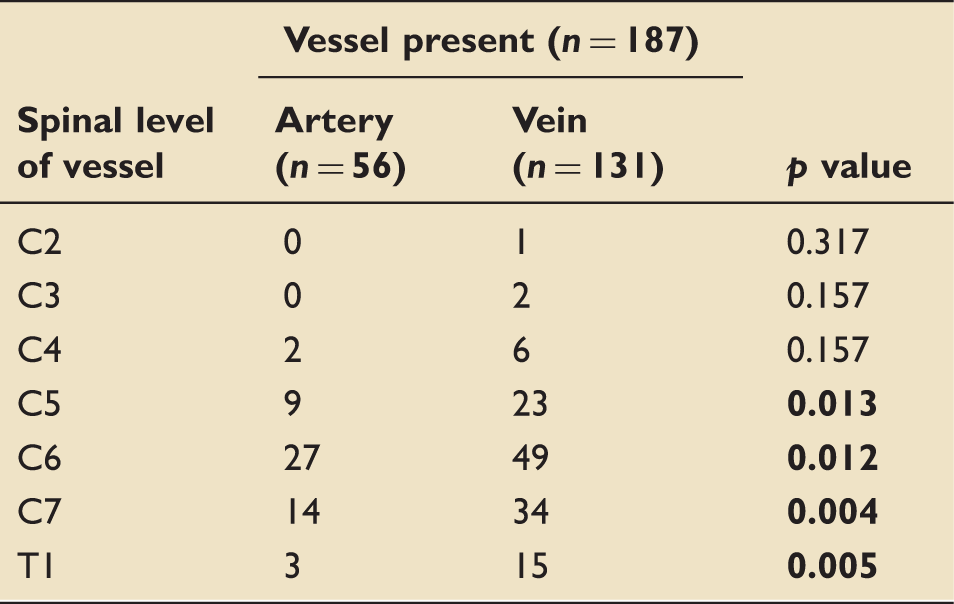
Bold values indicate statistical significance.
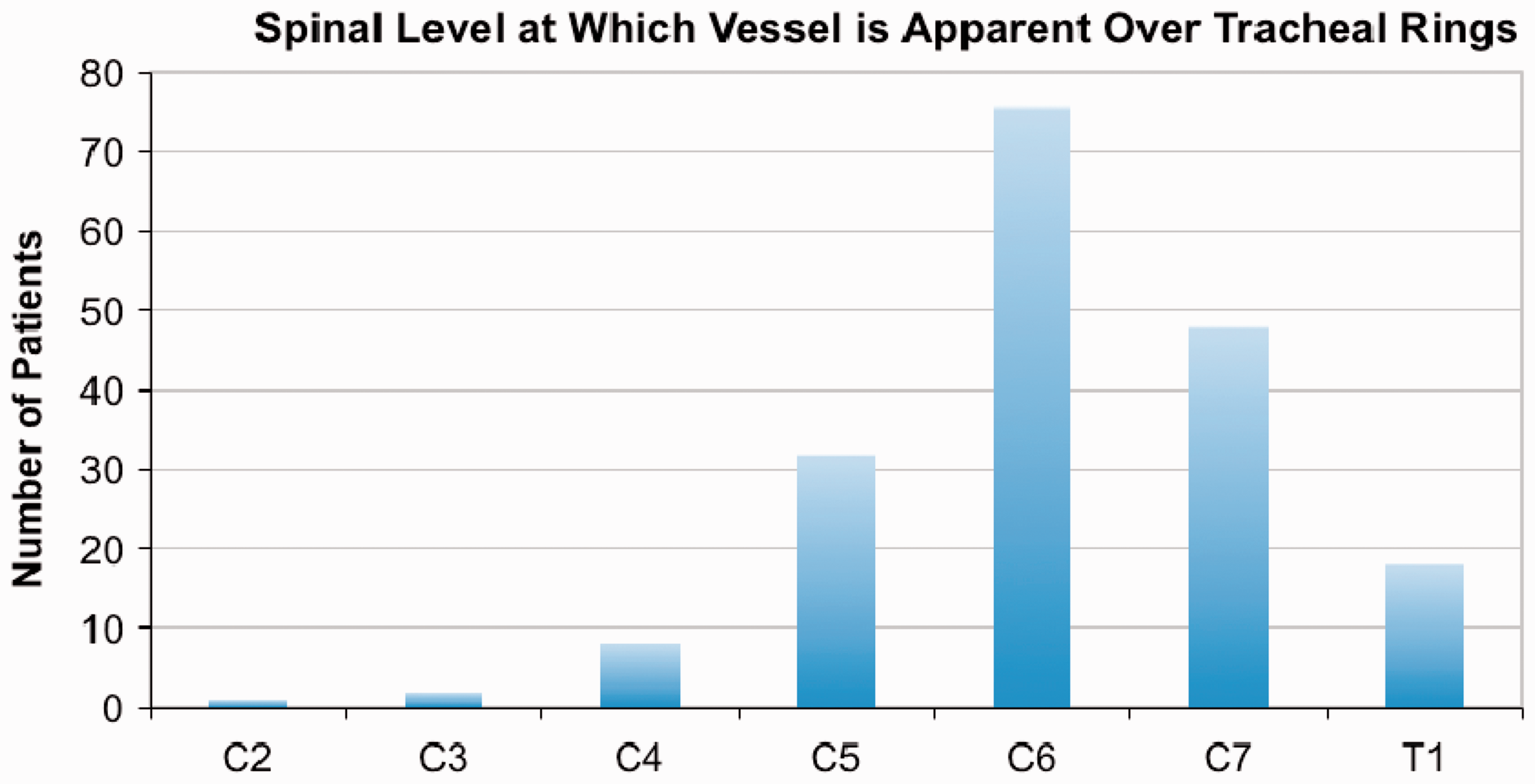
Spinal level at which the aberrant vessels overlay the trachea.
We investigated whether aberrant vessels were directed vertically or horizontally in the first 150 patients of the cohort. Gender data were missing in one patient and the running direction of the vessel was inconclusive in one patient, hence these were excluded. Of the 148 included patients, 66% were male, with mean age of 65 years and median age 66 years (IQR 55.0–77.8). 56.1% (n = 83) demonstrated an aberrant vessel and 46% (n = 68) in the ‘PT-region’; 21% (n = 31) of these displayed an arterial vessel, 25% (n = 37) displayed a venous vessel (p = 0.6). Ninety percent (n = 61) of the aberrant vessels ran vertically and 10% (n = 7) ran horizontally (p < 0.001). Arterial and venous vessels were more likely to run vertically (artery: 28 vs 3, p < 0.001; vein: 33 vs 4, p < 0.001) (Figure 4).
Subgroup of 148 CTAs; the presence and running direction of an aberrant vessel in the ‘PT-region’. CTA: computed tomography angiography; PT: percutaneous tracheostomy.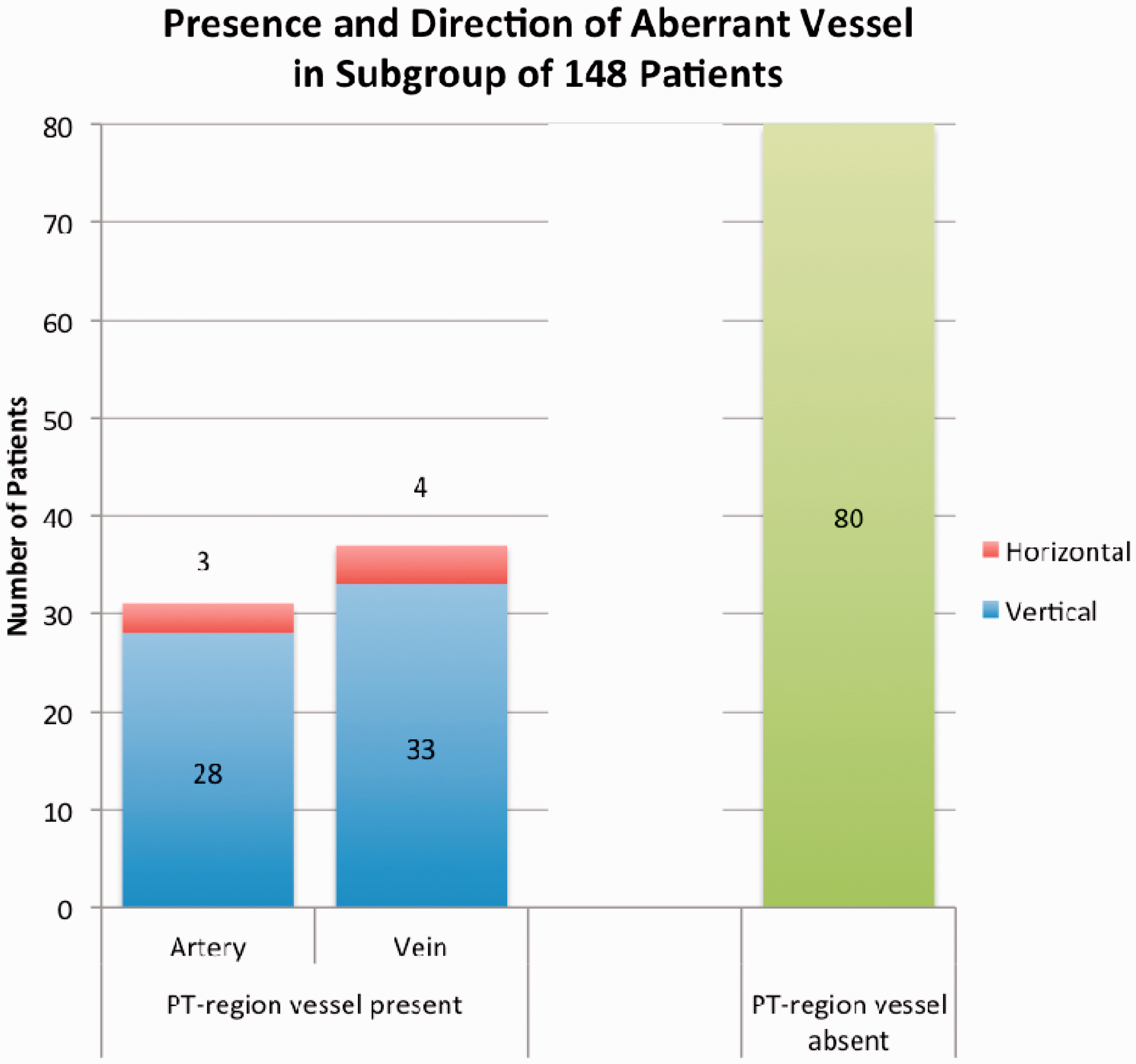
Discussion
In our study, we found a substantial 41% of our cohort exhibited a pre-tracheal vessel in the PT-region (C6–T1 levels). There was no prevalence difference identified between males and females (64 vs. 36%, p = 0.65). Veins were significantly more prevalent than arteries (69 vs. 31%, p = 0.001); females were more likely to display arteries compared to males (47 vs. 22%, p = 0.002). In our cohort, those with a pre-tracheal vein tended to be older than those with arteries (mean 67.8 vs. 59.9 years). This seemed to be skewed towards those between 61 and 90 years of age, however, it is likely that the number of patients in other age groups were insufficient to detect significance. We also demonstrated that vertical front-of-neck vessels were significantly more prevalent than horizontal ones in a subgroup of 148 patients (90 vs 10%, p < 0.001).
The majority of our patient group were elderly; likely representing the current intensive care patient cohort in the UK and in Western medicine. 11 We presume the reason for our cohort’s advanced age was due to age-related diseases necessitating CTA.
A substantially higher proportion of our cohort displayed a venous aberrant vessel (69 vs. 31%, p = 0.001). Pre-tracheal arterial haemorrhage can be torrential, potentially leading to death. Pre-tracheal venous ooze tends to be less acute, however, can be more troublesome to diagnose and manage, arguably leading to more blood loss than arterial haemorrhage. We feel that these data should not be taken lightly in the context of a procedure that is performed commonly in ICUs worldwide.
Interestingly, our data illustrated that most of vessels run vertically (superior–inferior), however, this was based on a smaller subgroup of our cohort, potentially limiting its validity. This may have implications in the context of PT insertion in deciding whether a vertical or horizontal incision is beneficial prior to inserting the tissue dilator.
We found this an interesting study to investigate in the context of ever-improving technology and striving for better patient outcomes and improved patient safety. It is already known that PTs are associated with high intra-operative complications3,12–20 and bronchoscopic guidance has shown to reduce the incidence of these complications.21–23 Any tool that would allow further reduction in complications would be invaluable. Ultrasound imaging is rapidly becoming more utilised within the healthcare environment and more staff are becoming familiar and skilled in its use.
We acknowledge certain limitations of this study. First, our patient cohort does not necessarily represent critically ill patients. We have no data regarding the diagnoses of our cohort or whether they were admitted into the ICU, however, we have no reason to believe that our cohort would display any anatomical differences to critically ill patients. It could be argued that patients requiring CTAs in the first instance are being investigated for atherosclerotic disease, and therefore have potential for collateral vessels such as the pre-tracheal area. Differing haemodynamics and presence of tissue oedema in the critically ill may alter vessel prominence and make tracheostomy insertion more difficult. This reinforces the proposal to use vessel imaging as a screening tool.
A further limitation is the lack of pre-tracheal vessel diameter measurements. This may have implications when deciding which tracheostomy technique to use; clinicians may continue to use percutaneous versus partial or fully surgical techniques after weighing up the risks of small overlying vessels. Finally, our subgroup analysis on vessel direction was limited to 150 patients, which could potentiate a selection bias. This was minimised by selecting the first 150 block of patients in our study period. Regardless, the subgroup’s age and proportions of pre-tracheal arteries/veins were similar to the overall cohort.
We acknowledge the low reported rates of haemorrhage from PT insertion in the literature, which may bring doubt over the relevance of our data. Yet as mentioned, it is believed that bleeding events are under-reported. To reinforce the importance of these data, most published data report fatal bleeding events but not bleeding events that have morbidity significance.
During the commencement of this study, we have been surprised at how prevalent pre-tracheal vessels are. This has alerted us to the potential dangers of percutaneous procedures. Despite evidence of its superior safety profile versus open surgical techniques, we propose that ultrasonography should be adopted as a screening tool for the presence of pre-tracheal vessels prior to tracheal puncture. This may allow us to better stratify our tracheostomy patients into three groups to reduce bleeding; fully percutaneous, limited blunt dissection with percutaneous and open surgical methods. Moreover, screening may influence a safer site for tracheal puncture. Our study has identified pre-tracheal vessel incidence and characteristics. The next research step would require outcome evaluation of ultrasound-influenced tracheostomy insertion techniques.
As previously stated, incidents occurring during tracheostomy insertion on the ICU are likely to be under-reported and tools to reduce complications from bleeding should be pursued. Based on our evidence, we believe that neck ultrasound should be implemented to screen for front-of-neck blood vessels prior to PT insertion, however, further research using this tool to evaluate outcome data is required.
Footnotes
Authors’ note
All work was carried out in Queen’s Hospital, Romford.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
