Abstract
We report a case of cardiac arrest secondary to pneumothorax. The patient failed to improve, and further investigation showed an underlying condition predisposing the patient to the development of pneumothoraces – lymphangioleiomyomatosis. This highlights the importance of follow-up and investigation in patients presenting with their first pneumothorax.
Introduction
Pneumothorax is a relatively common occurrence in clinical practice. It can be a potentially life-threatening and life-changing diagnosis. For us, as clinicians, the initial presentation can often be relatively exciting, with an acutely unwell patient requiring urgent clinical procedures. We make a significant difference to a patient’s well-being in a short space of time, often something that we as doctors find satisfying. However, it can be easy to forget about the long-term management of pneumothorax. We believe that our case is important as it highlights that once the patient has stabilized, there is a second very important stage; investigation to either rule out or rule in an underlying cause for their pneumothorax. Our case highlights the importance of further investigation as without computed tomography (CT) scans our patient’s underlying condition would not have been made and subsequent treatment would have been delayed. Although underlying causes for primary pneumothoraces are relatively rare, it is important that we consider them and investigate appropriately to give our patients the highest level of care.
Case presentation
We present a case of a 42-year-old female who presented to the emergency department (ED) in cardiac arrest, following a 2-week history of upper respiratory tract symptoms and a worsening history of shortness of breath over the previous four days. On arrival to the ED, she was asystolic, required 5 min of advanced life support before return of spontaneous circulation (ROSC) was achieved. On review of her chest X-ray (CXR) post ROSC, it was noted she had a large right-sided tension pneumothorax, which was subsequently drained with a wide bore chest drain. She had an uneventful transfer to the Intensive Care Unit and was subsequently extubated after 24 h of ventilation. Post-extubation, her right-sided chest drain continued to bubble and swing and a repeat chest X-ray was requested. This demonstrated a left-sided pneumothorax, which on review of her previous CXR was present but had now enlarged; therefore, a left-sided Seldinger type drain was inserted. Lung fields on the CXR showed no evidence of consolidation.
Our patient had minimal medical history. She had had surgery on her sinus in 2011 for recurrent sinusitis, but no other surgical history. She had a history of seasonal hay fever causing upper airway symptoms. She had occasional migraines, for which she took sumatriptan.
She had worked as an insurance broker, with no occupational exposure to substances common to lung disease. She had smoked seven cigarettes a day since being a teenager, consumed very minimal alcohol and had an unlimited exercise tolerance. She had not been pregnant, had stopped microgynon due to migraines and had used the depot injection for four years; prior to her admission, she was on no contraception. Her only significant family history was her father had Churg Strauss Syndrome and died at an early age from bowel cancer.
Clinical course and management
Following chest drain insertion, both drains continued to bubble and swing and her case was discussed with the Cardiothoracic team who advised for the drains to be placed on suction. A high-resolution computed tomography (HRCT) scan was performed which revealed the presence of extensive thin-walled cysts bilaterally. This appearance was characteristic of lymphangioleiomyomatosis (LAM) (Figure 1).
CT thorax – showing formation of multiple cysts consistent with the diagnosis of LAM.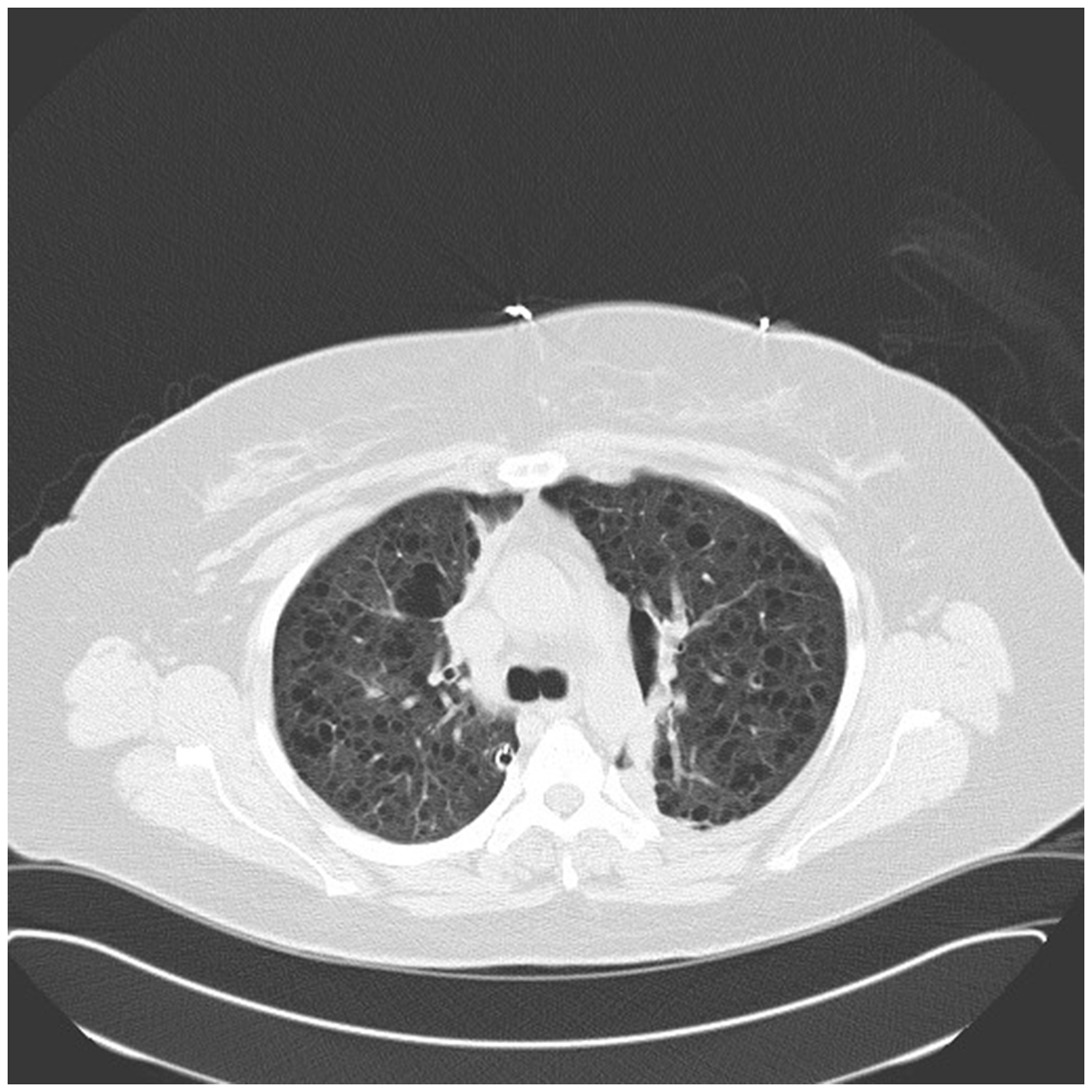
Her case was duly discussed with the National Centre for LAM in Nottingham who advised re-referral to the Cardiothoracic team and for the chest drains to remain in situ as there was a high risk of pneumothorax reoccurrence.
In accordance with the plan from the LAM centre, our patient underwent a CT abdomen to look for associated features of LAM such as angiomyolipoma and lymphatic disease, of which there was no evidence. She was referred to Cardiothoracics who performed a video-assisted thorascopic surgery (VATS) procedure on the right lung, took a lung biopsy which histologically confirmed LAM and performed pleurodesis as per recommendation from the LAM centre. The plan was for further VATS procedure on the left side; however, post surgery, it was noted the patient had developed a new left-sided pneumothorax and hence wide bore chest drain was inserted and talc pleurodesis was performed.
The patient was discharged home after an uneventful post operative recovery and is awaiting follow-up with the local respiratory team and with the National LAM Centre in Nottingham.
Discussion
LAM is a rare disease of the lungs and lymphatic system which occurs almost exclusively in females, usually in those of child bearing age. The diagnosis of sporadic LAM is given to those patients whose condition is not associated with tuberous sclerosis. There are around 1300 known patients with LAM in North America and around seven patients a year are diagnosed with the condition in the UK. 1
The underlying pathology is that the lungs and lymphatic system are infiltrated by LAM cells. LAM cells are atypical smooth muscle cells which undergo exaggerated proliferation. This triggers a cascade ultimately leading to degradation of the extra cellular matrix culminating in the formation of multiple small cysts within the lungs. 2 The role of oestrogen is unclear but it is thought to play a part in the pathological process as the condition does not occur prior to menarche, is rare after the menopause and gets worse during pregnancy. 3
The average age of onset is at 39, but it can take up to two years for formal diagnosis to be made, so that the average age of diagnosis is 41. The most common present symptom is progressive dyspnoea but over one-third of patients present with spontaneous pneumothoraces as the presenting symptom. Fifty percent of patients diagnosed with LAM will go on to develop a pneumothorax, secondary to rupture of a lung cyst. 4
The diagnostic criteria for LAM are based on large series review and expert opinion. The diagnosis takes into account both radiological and pathological findings. This allows the patient to fall into one of three LAM categories: definite, probable and possible LAM. In order for the diagnosis to be confirmed, other cystic lung diseases need to be excluded. As there is an association of LAM with tuberous sclerosis, patients require investigation to exclude this. This takes the form of a detailed history and examination looking for the hall marks of tuberous sclerosis. 4
LAM is usually a progressive disease associated with deterioration in lung function. Patients who have chest symptoms which are progressive should have regular follow-up with associated lung function tests in order to determine further interventions. Those without symptoms who remain stable for one year do not necessarily require follow-up. Due to the high risk of pneumothoraces, patient education in the signs and symptoms is important to allow them to present for medical attention. Other patient education topics include: pregnancy, avoidance of oestrogen containing medications, air travel, vaccination and osteoporosis prevention. 5
Pneumothorax and recurrent pneumothoraces are a frequent complication of LAM. The European Respiratory Society (ERS) task force states that in patients with LAM, conservative treatment of pneumothoraces is associated with a higher rate of recurrence, than if these patients are managed with pleurodesis via a chest tube or surgical intervention. They recommend that management of pneumothoraces should be carried out jointly between surgeons and respiratory physicians. In patients who have a second pneumothorax, surgical treatment is advised based on their clinical condition. The aim of pleurodesis is to prevent the development of further pneumothoraces.
A potential long-term treatment for LAM is lung transplant. If a patient has undergone pleurodesis as treatment for a pneumothorax, this makes transplant a more difficult procedure, but does not rule it out as a definitive treatment. These patients should be managed at a transplant centre with experience of pleurodesis and transplant. 4
Evidence on the medical management of patients with LAM is limited. Patients should avoid the use exogenous oestrogen as this can promote the progression of pulmonary LAM. Inhaled bronchodilators should be trialed in those patients with airflow obstruction but only 25% of patients will respond; if there is no improvement, then the trial should be stopped. There are a number of ongoing clinical trials into the response of LAM patients to mTOR inhibitors, but no general consensus has been reached yet. The task force recommend LAM patients to take part in trials and the use of mTOR inhibitors is assessed on a case by case benefit. 4
Patients with severe respiratory disease can be considered for lung transplantation. Currently, they make up 1.1% of recipients. The task force were unable to make firm recommendations due to the small number of patients who undergo transplant, but most had severe airway obstruction. Transplantation is often complicated due to adhesions. Boehler et al. 6 reported that the majority of these adhesions were in patients who had not had previous surgery and therefore was due to the disease itself. A study in Japan showed good survival post transplantation at five years, with a survival rate of 73.7%. It should be noted that this is significantly higher than other studies. 7
Our case is unusual as there are no recorded cases of the initial presentation of LAM occurring as cardiac arrest. There is one reported case of a patient presenting for the first time with life-threatening spontaneous tension pneumothorax. This was in a 26-year-old female who was known to have renal angiomyolipoma and LAM was diagnosed following a chest CT. 8 There is a second case of a known asthmatic who presented with presumed life-threatening asthma and was found to have bilateral pneumothoraces. Subsequent investigation diagnosed LAM. 9
The majority of cases reported show patients presenting with chest pain, pneumothorax being diagnosed (the patient remaining stable), the pneumothorax failing to completely resolve and the patient having a CT scan which has then shown findings consistent with LAM. There are a number of unusual presentations of LAM; presentation as cardiac tamponade secondary to a chylous pericardial effusion, 10 first presentation as chyloperitoneum and chylothorax 11 and masked by pregnancy and presenting after stillbirth of a term foetus. 12
Our case highlights the importance of considering an underlying cause for a patient presenting with a pneumothorax, particularly if this is a second pneumothorax. It stresses the importance of any patient presenting with a pneumothorax having the appropriate follow-up with a respiratory physician to rule out underlying lung pathology. The ERS guidelines recommend that any patient presenting with their third pneumothorax should have an HRCT performed, this is particularly important in the high-risk patients for LAM, e.g. nonsmoking female, but they did not achieve an overall consensus on this and it was not based on good quality evidence. They do not recommend that HRCT is performed for patients presenting with a first time pneumothorax to specifically look for LAM. 4
A high index of suspicion for underlying disease in patients presenting with a spontaneous pneumothorax is required. This is particularly important in the LAM population as it has been shown that those patients who present with a spontaneous pneumothorax will have a further 2.5 pneumothoraces 13 before they undergo investigation into why they are having recurrent pnuemothoraces which subsequently diagnoses them with LAM and enables treatment of the condition. This is concerning as 50% of patients with LAM will suffer from a pneumothorax at some point in their disease, which has the potential to be life threatening.
Thirty-three percent of patients diagnosed with LAM will present as a pneumothorax and in a young person they are more likely to be managed as a primary pneumothorax, leading to conservative management and discharge without further investigation.
The definition of pneumothorax is air in the pleural cavity. According to the British Thoracic Society (BTS) guidelines, when the term was first described, the majority of pneumothoraces occurred in patients secondary to tuberculosis; however, it was acknowledged that some patients with healthy lung tissue could also develop a pneumothorax. This led to the classification of pneumothoraces as either primary or secondary. A secondary pneumothorax is always associated with an underlying lung disease. Secondary pneumothorax is associated with higher morbidity and mortality than primary spontaneous pneumothorax (PSP). PSP is associated with smoking and taller stature, the latter causing a wider pressure gradient which predisposes patients to the formation of pleural plebs which can rupture. 14 The European Respiratory Society (ESR) suggest that there is potential for a change in the classification of pneumothorax, as patients with PSP who have further investigation are found to have subtle changes which would reclassify their initial pneumothorax as a secondary pneumothorax. 15
The management of a pneumothorax is dependent on:
Primary or secondary – patients with underlying lung disease tolerate a pneumothorax less well than those with healthy lungs. Clinical symptoms – if a patient is symptomatic, definitive management is required. Size – size viewed on the CXR does not correlate with the severity of symptoms. The size is only important in determining how quickly the pneumothorax will resolve.
In patients who are well and present with a small primary pneumothorax they can be considered for observation and follow-up as an outpatient.
Patients presenting with a PSP who are symptomatic or have a larger pneumothorax should initially undergo needle aspiration. If the patient has a successful needle aspiration and is asymptomatic then they can be considered for discharge with early follow-up. Patient education is vital informing them of the risk of reoccurrence particular in regard to flying, diving and smoking cessation. They should be encouraged to seek medical help if they develop further symptoms of chest pain or shortness of breath. If needle aspiration fails then they should proceed to insertion of a chest drain. These patients require admittance to hospital for observation to ensure that the pneumothorax resolves. Once the decision to admit the patient has been made, early referral to a respiratory physician for appropriate management is required.
There are no guidelines on the further investigation for patients presenting with a PSP. CT is highlighted as the gold standard for the detection of pneumothoraces and size estimation; it will also pick up underlying lung disease. In practice, CT scanning is not widely used apart from when there is diagnostic uncertainty. BTS guidelines recommend CT scanning for uncertain or complex cases whilst the ESR guidelines state that the majority of patients presenting with pneumothorax will not require CT, only if it is complicated or if underlying lung disease is suspected. They do not clarify or provide suggestions on when to consider underlying lung disease. In their recommendations they state there does need to be further research into the potential of genetic testing and thoracic HRCT scanning of all cases of PSP. 15
The BTS guidelines state that the following groups of patients should be referred for a thoracic surgical opinion; if the patient has a persistent air leak or the lung fails to expand then discussion with a thoracic surgeon is recommended. Other indications for referral include synchronous bilateral spontaneous pneumothorax, first contralateral pneumothorax and a second ipsilateral pneumothorax. 16 There is no mention of the need for investigation prior to referral and which patients would benefit from further imaging.
Patients who are over the age of 50 and have a significant smoking history should be treated as a secondary pneumothorax. The main difference in these patients compared to primary pneumothorax is that there is a much lower threshold for chest drain insertion and needle aspiration should only be performed if the patient is asymptomatic. Patients with secondary pneumothorax tolerate the pneumothorax less well due to their underlying lung disease. These patients will usually require admission, normally to a respiratory ward to allow access to a respiratory physician who can manage both their pneumothorax and their underlying lung disease. In patients with a secondary pneumothorax, definitive management should be considered and this is usually in the form of pleurodesis. This can be medical or surgical; the latter has a lower rate of reoccurrence of pneumothorax.
Although a rare condition, we feel that the important learning point from this case is not to think that every patient who presents with a pneumothorax has LAM, but to consider underlying secondary causes in patients who present with unexplained pneumothorax, particularly multiple events. This also raises the question of should there be further clarification as to which patients with PSP are followed up by respiratory clinicians and need further imaging with HRCT.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
