Abstract
We present a case of a woman in her 70 s, on cyclophosphamide for multiple myeloma, who was admitted to critical care with grade III acute kidney injury. Renal replacement therapy with regional citrate anticoagulation was commenced. Shortly thereafter her systemic-ionised calcium levels fell and remained stubbornly low until post-filter calcium return was doubled. Her total-to-ionised calcium ratio gradually increased and so, to avoid further accumulation of citrate, anticoagulation was changed to heparin. Cyclophosphamide, which accumulates in renal failure, is known to interfere with key enzymes involved in the tricarboxylic acid cycle. We postulate that cyclophosphamide interfered with her citrate metabolism, resulting in persistent systemic chelation of calcium.
Keywords
The use of regional citrate anticoagulation (RCA) for renal replacement therapy (RRT) is increasing and with it the incidence of adverse effects. We wish to highlight an interesting patient, in whom we believe that non-excreted cyclophosphamide led to impaired citrate metabolism and persistent chelation of calcium.
Our patient, in her 70 s, with a background of chronic kidney disease and multiple myeloma on bortezomib, cyclophosphamide and dexamethasone, presented to hospital having collapsed. Tumour lysis syndrome was tentatively diagnosed after investigations demonstrated hyperuricaemia, grade III acute kidney injury and hypocalcaemia.
She was transferred to the intensive care unit and RRT with RCA was commenced. Pre-initiation, her corrected calcium (cCa) was 1.95 mmol/L and her ionised calcium (iCa) was 1.01 mmol/L. At 2 h post-commencement, her iCa was measured at 0.8 mmol/L, giving a total-to-ionised calcium (T/I) ratio of 2.44. Thereafter, post-filter calcium replacement was increased incrementally until her iCa returned towards normal (see Figure 1).
A graph comparing total and ionised calcium concentrations with calcium replacement during the period in which our patient received RRT with RCA.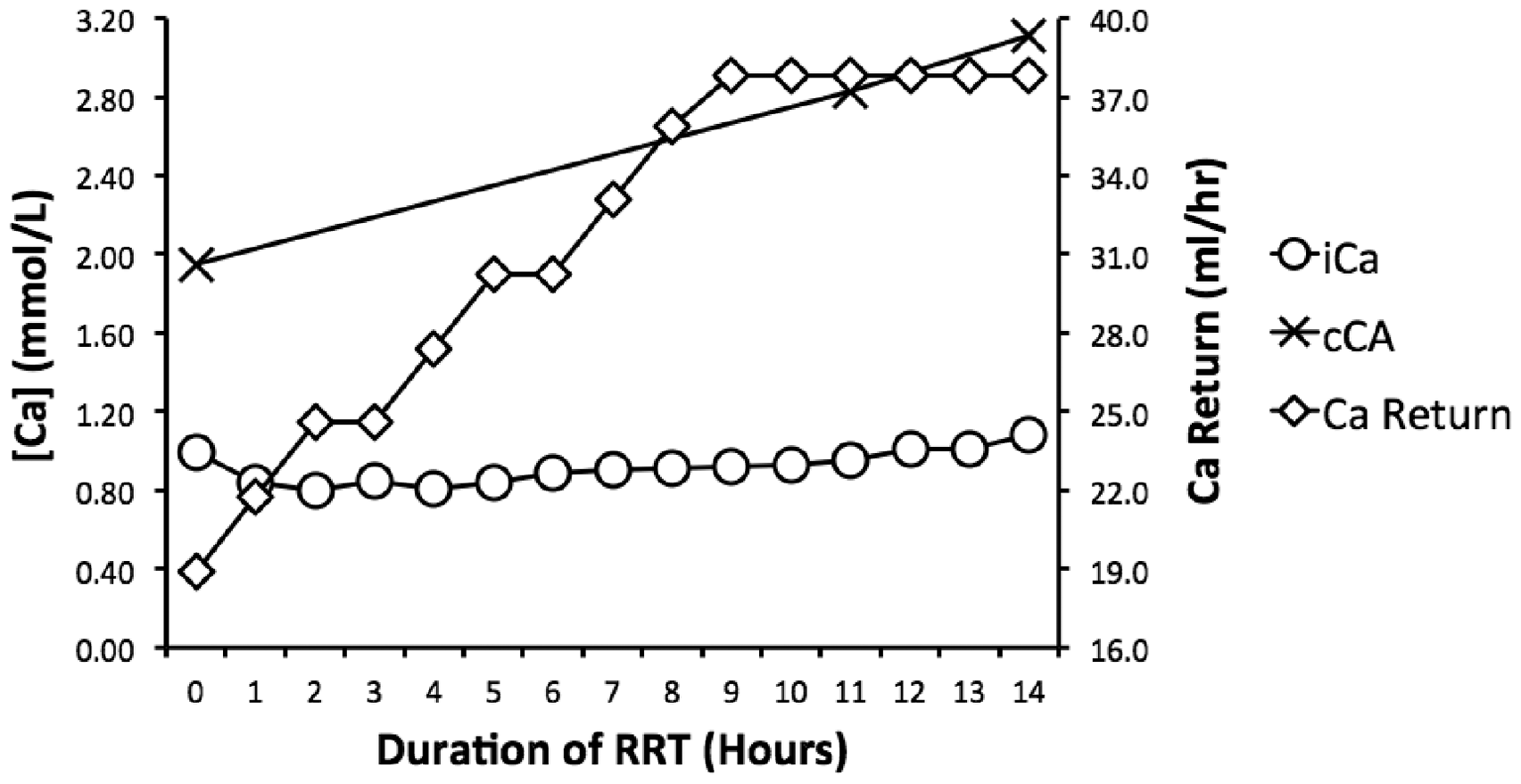
It was felt that citrate was accumulating and therefore, to optimise extracorporeal clearance, the filter blood flow rate was increased. At 14 h post-commencement, her T/I ratio was 2.9 and thus, despite a near-normal iCa, anticoagulation was changed to heparin to avoid further citrate accumulation. Initially, her renal function improved but, despite our best efforts, she developed multi-organ dysfunction and was discharged to palliative care.
Abnormalities of serum calcium can occur with RCA and are most commonly due to persistent, systemic chelation from non-metabolised citrate or insufficient post-filter calcium replacement. 1
Citrate accumulation may occur in patients with hepatic failure 1 or who take medications such as anti-retrovirals, isoniazid and metformin. 2 Metabolism of citrate is impaired and it therefore accumulates, resulting in persistent chelation of systemic calcium. The earliest sign of citrate accumulation is an increased T/I ratio. 3 In this case, no other cause was identified and thus cyclophosphamide was deemed the likely offending agent.
Cyclophosphamide is almost exclusively renally excreted, with 20% of the unchanged dose and 33% of the alkylating metabolites appearing in the urine by 24 h. 4 It is likely therefore, in our patient with acute-on-chronic renal failure, that the unchanged compound and metabolites had accumulated prior to commencing RRT.
It has been demonstrated that the cardiac side effects of cyclophosphamide are attributable to inhibition of key enzymes involved in the tricarboxcylic acid (TCA) cycle. 5 In this case, we postulate that due to high concentrations of active metabolites interfering with the TCA cycle, metabolism of returned citrate was impaired, thus leading to systemic accumulation and persistent chelation of calcium.
To summarise, this appears to be the first reported instance of cyclophosphamide interfering with citrate metabolism, resulting in subsequent accumulation in a patient without other discernible cause. The key learning points from this case are the following:
Accumulating citrate results in persistent, systemic chelation of calcium and an increasing T/I ratio; This can usually be mitigated by optimising extracorporeal removal of calcium-citrate complexes and increasing post-filter calcium replacement; Cyclophosphamide can interfere with the TCA cycle and therefore may impair metabolism of citrate.
Consent
Written assent for publication of this letter has been received from the patient’s next of kin.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
