Abstract
Lemierre's syndrome is a rare complication of acute pharyngitis characterised by septicaemia with infective thrombophlebitis of the internal jugular vein, most commonly due to Fusobacterium necrophorum. It characteristically affects healthy young adults causing persistent pyrexia and systemic sepsis presenting several days after an initial pharyngitis. Septic emboli seed via the bloodstream to distant sites including the lung, joints, skin, liver, spleen and brain. Prolonged antimicrobial therapy is required and admission to intensive care common. This once rare condition is increasing in incidence but awareness amongst clinicians is low. We present a classic case in a young man who developed multi-organ failure requiring intensive care support and describe the epidemiology, pathophysiology, microbiology, clinical features and management of the disease.
Case report
A previously healthy 28-year-old male presented with a five-day history of sore throat, progressive neck stiffness, malaise, dry cough, pleuritic chest pain and haemoptysis. Clinical examination in the emergency department revealed a pulse rate of 120, respiratory rate 35, oxygen saturation 92% on air and consistent pyrexia of over 39℃. He was admitted to the medical high dependency unit and commenced treatment with intravenous amoxicillin-clavulanic acid and clarithromycin for suspected atypical pneumonia.
He clinically deteriorated over the following 24 h and developed type 1 respiratory failure, requiring intubation, ventilation and transfer to the Intensive Care Unit (ICU). Investigations revealed a white cell count of 13.1 × 109/L, c-reactive protein 430 mg/L, procalcitonin 46.49 ng/mL and a platelet count of 48 × 109/L. Repeat chest radiography showed progressively worsening bilateral lower zone patchy consolidation and pleural effusions.
A clinical picture of a severe inflammatory response, respiratory failure, septic shock, acute kidney injury and mild disseminated intravascular coagulation was attributed to an atypical pneumonia. He required fluid resuscitation and cardiovascular support with noradrenaline. In view of his worsening oxygenation, he was ventilated using airway pressure release ventilation (APRV) with an open lung approach allowing permissive hypercapnia.
On day 2 of ICU admission, peripheral blood cultures grew a Gram negative bacillus identified to be Fusobacterium Necrophorum, a bacterium pathognomonic for Lemierre’s Syndrome. Bedside ultrasound scanning of the internal jugular veins (IJV) suggested the presence of thrombus in the left IJV with no flow seen on colour Doppler (Figure 1). A contrast CT of the neck and thorax confirmed left IJV thrombosis extending into the left brachiocephalic vein. Extensive cavitating septic emboli were seen throughout the lungs bilaterally in addition to changes consistent with acute respiratory distress syndrome and significant hepatosplenomegaly (Figure 2).
Ultrasound image showing thickened, abnormal left internal jugular vein with no flow on Doppler and for comparison the normal right internal jugular vein. Cross section from CT scan showing extensive bilateral septic embolic in addition to changes consistent with acute respiratory distress syndrome.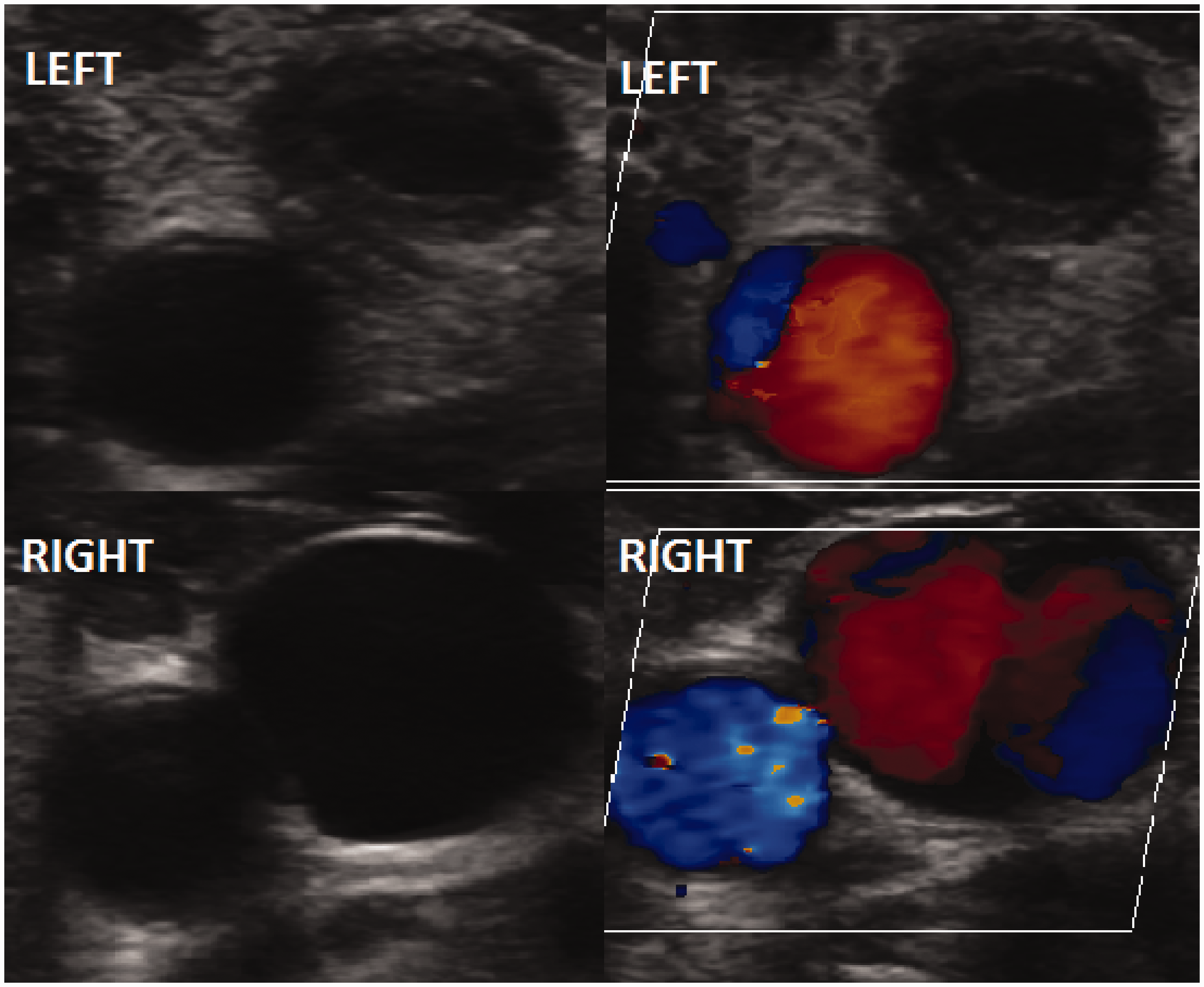
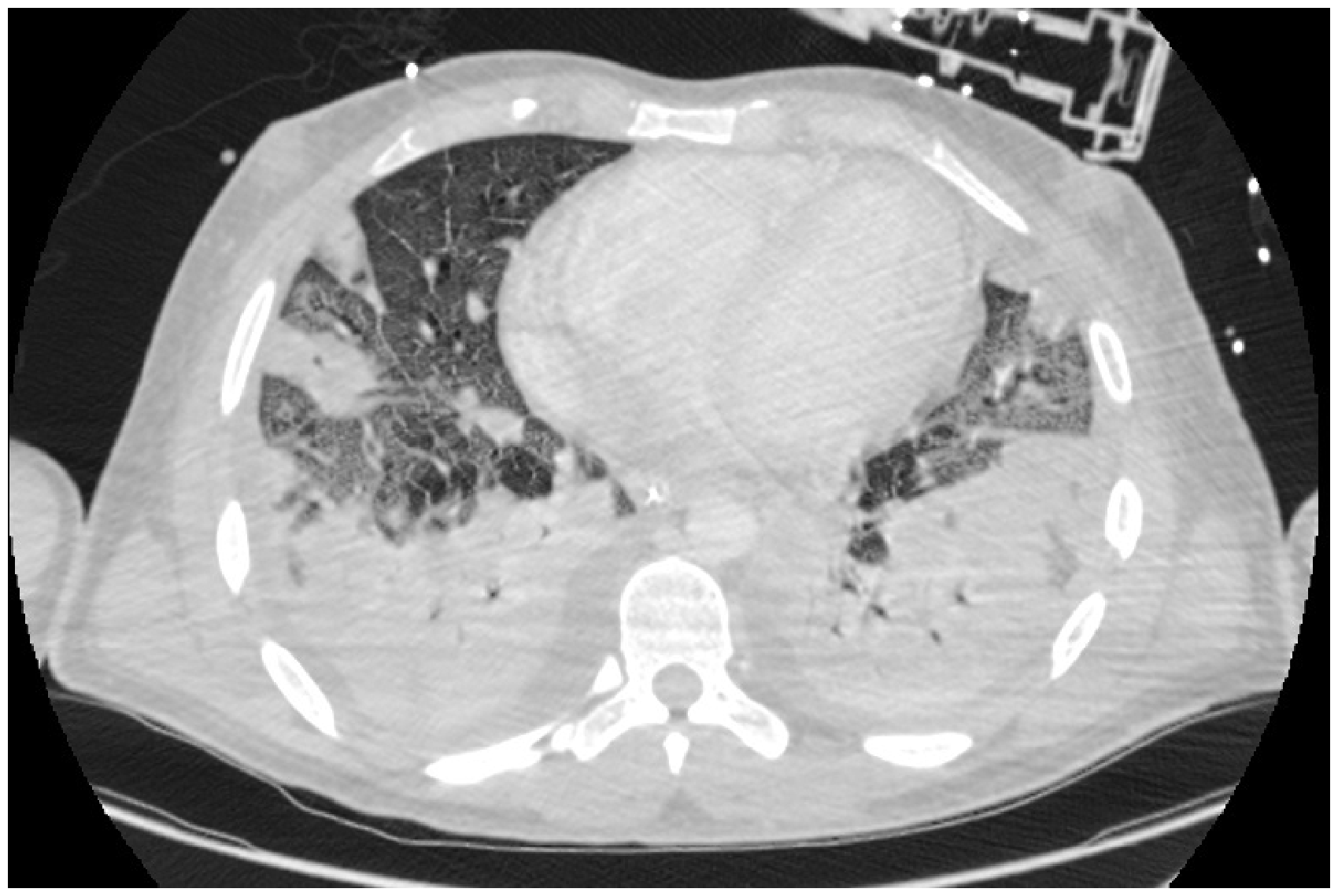
In view of persistent pyrexia > 39℃, antibiotic therapy was escalated to piperacillin-tazobactam, metronidazole and clindamycin. Systemic anticoagulation with low-molecular weight heparin was commenced for the left IJV thrombosis. An intercostal drain was inserted for an enlarging right-sided pleural effusion which proved to be a sterile transudate. Interestingly polymerase chain reaction (PCR) testing for Epstein Barr virus (EBV) was positive but IgM antibodies were negative. This was felt to represent a false positive, a known association between Lemierre's and EBV. A percutaneous tracheostomy was placed on day 16 to facilitate weaning, the patient was decannulated on day 18 and discharged from ICU on day 20.
Clindamycin and benzylpenicillin were continued for a total of six weeks of which four weeks were intravenous therapy. Anticoagulation was continued with rivaroxaban for a total of three months. He was discharged from hospital on day 31.
Discussion
The first report of post anginal septicaemia caused by F. necrophorum was by Courmont and Cade
1
but it was Lemierre
2
who described the distinctive pattern of infection displayed in a case series of 20 ‘anaerobic septicaemias' The appearance and repetition several days after the onset of a sore throat (and particularly of a tonsillar abscess) of severe pyrexial attacks with an initial rigor or still more certainly the occurrence of pulmonary infarcts and arthritic manifestations, constitute a syndrome so characteristic that mistake is almost impossible.
Epidemiology
Lemierre's syndrome was well recognised in the pre-antibiotic era with an associated high mortality, but the introduction of antibiotics in the 1950s led to a rapid drop off in incidence such that it became largely forgotten and very few cases were reported 3 leading to the label of ‘the forgotten disease'. Incidence appears to be increasing during the last three decades and it may not be as uncommon as previously thought; however, its rarity has resulted in a lack of awareness amongst clinicians.
Accurate classification of its incidence is confounded by variable reporting and differing definitions. Two Danish series estimated incidence at 1.5 per million 4 and 14.4 per million, respectively. 5 Reference laboratory surveillance in England and Wales during the 1990s 6 reported an average of 19 cases per year (approximately 0.6 per million) with an increasing trend. There appears to have been a resurgence in cases in the late 1980s and 90s with a marked increase in reporting in the last 10 years. Suggested possible causes for this include a reduction in antibiotic prescribing for sore throats, the dramatic fall in rate of tonsillectomy (60% to 7.5% by the late 1990s), improved microbiological identification of anaerobic bacteria and possibly an increase in availability and resolution of imaging resulting in increasing identification of IJV thrombosis.7,8
Fusobacterium spp. (in particular F. necrophorum) are responsible for the majority of cases but the syndrome has been reported in association with multiple other organisms. In addition, while the original case series by Lemierre described metastatic embolic spread from internal jugular vein (IJV) thrombosis, septic thrombophlebitis in association with F. necrophorum infection has been reported at other sites including iliac, portal and mesenteric veins. 9
In the absence of a formal definition for the syndrome, Riordan
9
proposed:
History of anginal illness or compatible clinical findings. Evidence of either metastatic lung lesions or metastatic lesions in other sites, and Either evidence of internal jugular vein thrombophlebitis or isolation of F. necrophorum or Fusobacterium spp. from blood culture or a normally sterile site.
Based on these criteria, Riordan identified 222 cases in literature from 1970 to 2007, of which 68% isolated F. necrophorum, 17% other fusobacterium spp., 4% other anaerobes (bacteroides, peptococcus and peptostreptococcus), 4% other non anaerobes (streptococcus, proteus and eikenella) and 6% no organism isolated. Cases occurring secondary to staphylococcus aureus (both MSSA and MRSA) have also been reported. 10 It is unclear if these more atypical organisms are truly responsible for causing Lemierre's or whether F. necrophorum may have been present but undetected in these cases due to prior antibiotic therapy or differing culture methods.
It is a disease of previously healthy young adults typically between the ages of 10–35 with a median age of 19 years and a male preponderance (57–66%)9,11 There are no known predisposing factors. In Lemierre's original series, 18 of 20 patients died but even in the era of modern antibiotic therapy and intensive care, the syndrome carries mortality of 5%. Systematic review by Karkos et al. 7 of 114 patients reported 58% of patients required intensive care admission and prolonged hospital stay was the norm (median 25 days). Interestingly disseminated F. necrophorum infection originating from foci in the lower body appears to be a different disease process, described in predominantly elderly patients with abdominal or urogenital malignancy and a correspondingly higher mortality of 26%. 5
Pathogenesis
F. necrophorum is an anaerobic, filamentous, non motile, non spore forming gram negative bacilli. Although commonly described as normal flora of the oropharynx, gastrointestinal and genitourinary tract the microbiological evidence for this is weak 11 and it is probably acquired exogenously. F. necrophorum displays haemagglutination activity, triggering platelet aggregation that can result in thrombocytopenia and disseminated intravascular coagulation. This is presumably the mechanism by which it causes septic thrombus to form in large vessels. Fusobacterium strains also possess a lipopolysaccharide endotoxin similar to some non-anaerobic gram negative bacilli. 12
The primary source of infection is usually pharyngitis, tonsillitis (often with purulent exudate) or peritonsillar abscess. F. necrophorum may be responsible for 10%–20% of all sore throats.7,9 Lemierre's has also been reported following otitis media, mastoiditis, parotitis, sinusitis, orbital cellulitis and dental infections. 11 Thrombophlebitis occurs in the peri-tonsillar veins and spreads through the lateral pharyngeal space to the internal jugular vein. Here platelet aggregation predisposes to the formation of infected thrombus, an ideal anaerobic environment for bacterial replication. Septic emboli seed to the lungs and subsequently to distant organs to form metastatic abscesses.
Clinical features
Systemic manifestations of Lemierre’s syndrome. 9
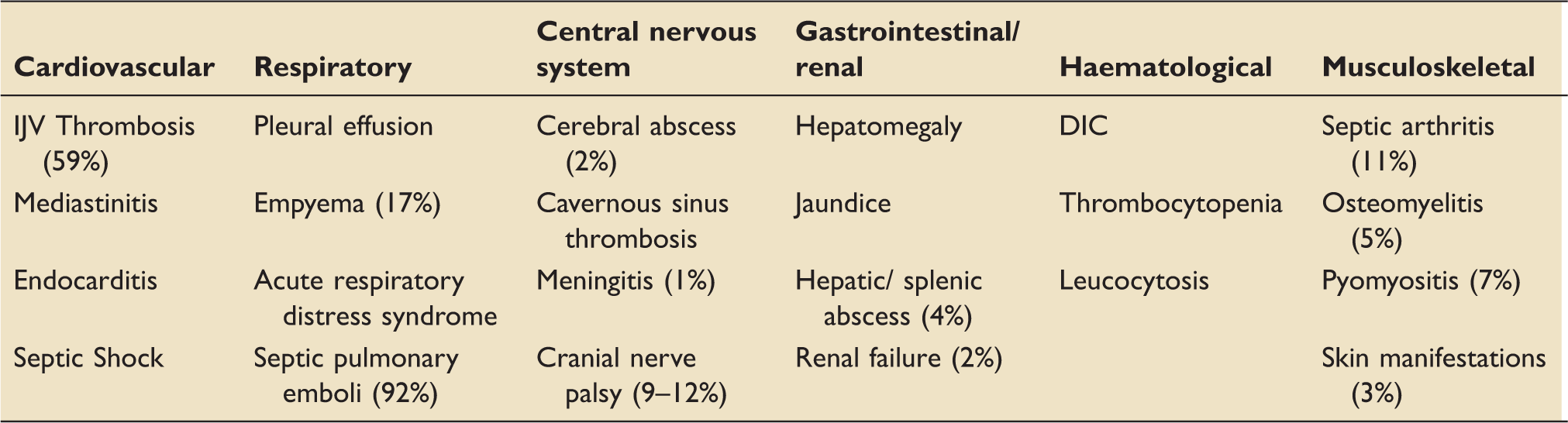
Severity can vary dramatically from a fulminating septicaemia with abscesses at multiple sites to a relatively benign bacteraemia without metastases from which patients recovery rapidly (‘ephemeral bacteraemia'). 13 Karkos' systematic review of 114 cases in 2009 reported that 58% of patients required ICU admission. 7
Diagnosis
Although Lemierre deemed the constellation of symptoms to be ‘so characteristic that mistake is almost impossible', given the rarity of the condition and lack of awareness amongst clinicians, it is often only with the identification of F. necrophorum on blood cultures that the diagnosis is made. Isolation of the bacterium should prompt a search for occult thrombus and concealed abscesses. There may be sub-clinical jaundice and mild disseminated intravascular coagulation (DIC) with thrombocytopenia. IJV thrombus can be diagnosed using ultrasonography, contrast CT or MR angiography. Ultrasonography is readily available and avoids radiation exposure but can miss thrombus above the mandible or below the clavicle, and therefore CT is the investigation of choice. Cross-sectional imaging may also aid localisation of abscesses requiring percutaneous or surgical drainage.
The interaction between Epstein Barr virus (EBV) and Lemierre's syndrome is complex and can cause diagnostic uncertainty, particularly in early infection. Riordan identified 23 cases with the association, which was also seen in our patient. 9 Infection with EBV may precede and predispose to Lemierre's – possibly by a transient depression of T cell immunity. In addition, fusobacterium can cause false positive results in EBV rapid tests and in mycoplasma pneumoniae PCR.
Anti-microbial therapy
F. necrophorum is generally sensitive to penicillins, cephalosporins, metronidazole, clindamycin, tetracyclines and chloramphenicol. 14 Production of beta-lactamases is uncommon but has been reported (2% resistance according to Brazier et al. 6 ) Despite in vitro penicillin sensitivity, it seems relatively ineffective as a sole agent. 8 The organism is almost exclusively sensitive to metronidazole but monotherapy is not recommended due to severity of infection and common co-existent of other non-susceptible streptococci . Clindamycin has only weak bactericidal or bacteriostatic activity but may be more effective for treatment of lung abscess. 15 F. necrophorum is commonly resistant to erythromycin (15–22%), 9 intrinsically resistant to gentamicin and quinolones have poor activity. No trials exist to determine the ideal antibiotic therapy but the combination of metronidazole with a penicillin is generally accepted.
The response to antibiotics tends to be remarkably slow, even when the organism is fully sensitive. Median time from initiation of antimicrobial therapy to resolution of pyrexia has been reported between 8 and 12 days. 8 This may be due to poor antibiotic penetration into infected thrombus or necrotic abscesses which provide ideal anaerobic conditions for bacterial growth. Viable F. necrophorum can be found in necrotic abscess formations after several days to weeks of intravenous antibiotics. 14 Given the potential for deep seated infection and abscesses at multiple sites, the duration of antibiotic therapy is necessarily lengthy. Most reports describe six weeks of treatment with a varying degree of intravenous vs. oral therapy depending on the severity of the case. The presence of necrotic tissue, parapharyngeal or mediastinal abscess or empyema is likely to require surgical drainage to achieve source control.
Role of anticoagulation
This is a controversial area as evidence is limited and most patients make a good recovery without anticoagulation. 12 There are no controlled prospective studies in Lemierre's but there is some limited evidence that anticoagulation speeds resolution of pelvic vein septic thrombophlebitis. 16 Proposed benefits of anticoagulation include preventing retrograde clot extension from the internal jugular into the sigmoid or cavernous sinuses and limiting ongoing seeding of septic pulmonary thromboemboli. Some authors 14 have counseled against routine anticoagulation due to possible risk of extending the infection. In view of the paucity of data, this must be considered on a case by case basis.
Role of vascular intervention
There are a number of reports of internal jugular vein ligation or excision in the treatment of Lemierre's, particularly in the pre-antibiotic era. With modern antimicrobial therapy, operative intervention is rare and probably only indicated in cases with uncontrolled ongoing septic embolisation or clot extension despite maximal medical therapy. 17
Conclusion
Lemierre's syndrome is a rare complication of bacterial pharyngitis which affects young healthy individuals and causes extensive morbidity and potential mortality. Rationalisation of community antibiotic prescribing has led to an observed increase in incidence. An awareness of the condition amongst clinicians is crucial to make the diagnosis, which will be aided by bacteriological isolation of F. necrophorum. Identification should prompt a thorough search for occult thrombus and distant septic foci. Prolonged antibiotic therapy with a course of a beta-lactam antibiotic combined with metronidazole is advised. The role of anticoagulation is controversial.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
