Abstract
The presented case is one of severe dehydration and acute kidney injury with significant resultant complications that required considerable fluid and electrolyte replacement. Approaches to fluid resuscitation in the context of hypernatraemia and the hyperosmolar state were considered and then judiciously combined to manage a complex case with a successful outcome. Conflicting guidance in this domain is discussed with recommendations for a future management strategy that is tailored to individual patients.
Case presentation
Results pre-operatively on arrival at tertiary referral centre.

Clinical and laboratory data from time of initial hospital presentation.

Patient results over the first few days at his local hospital.

On the third day of the admission, he developed a right hemiplegia (1/5 power in both his right arm and leg). An urgent CT head was performed and it demonstrated a large subdural haemorrhage (SDH) with subarachnoid extension; there was evidence of midline shift and developing hydrocephalus (Figure 1).
CT head scan.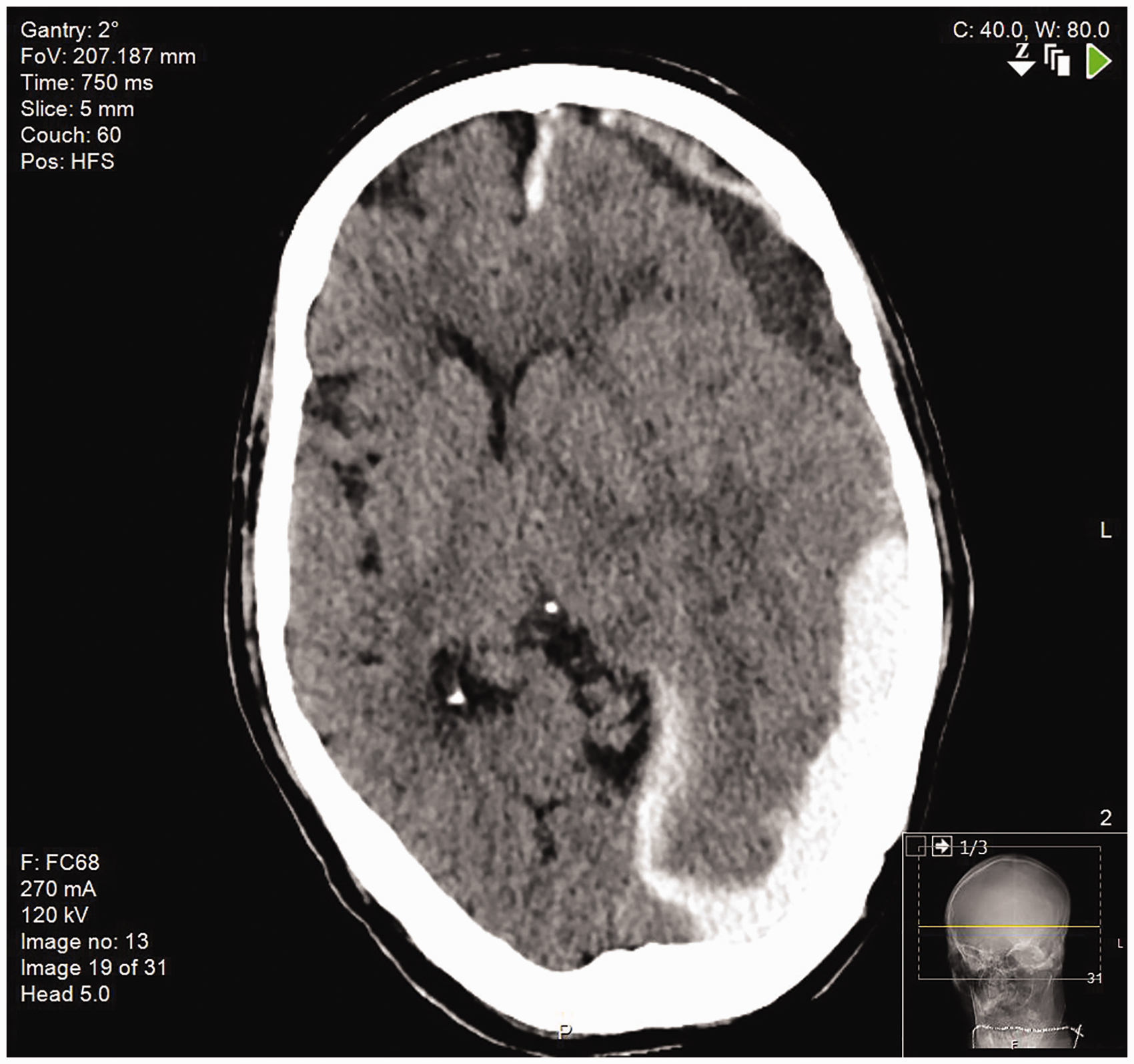
At this point, neurosurgical advice was sought but before transfer could be arranged his condition deteriorated: his GCS fell to 8/15 and he developed a fixed and dilated pupil. A decision was made to provide end of life care, however, over the following 24 h the patient made a significant, spontaneous, neurological recovery; he became alert, was obeying commands and his pupils normalised but he remained dysphasic with a right-sided weakness. Further discussions with the neurosurgical team led to the patient being transferred to our institution for urgent neurosurgery.
Post-operatively, he was brought to our Neurosurgical Critical Care Unit (NCCU) for management of his dehydration and recovery from evacuation of sub-dural haematoma. He required intubation for 5 days, during which time his fluid status and biochemistry were carefully corrected; he never required renal replacement therapy.
He was discharged to a neurosurgical ward on day 12 and on day 32 was repatriated to his local hospital. He had, by this stage, made a dramatic neurological recovery with no residual motor deficit or dysphasia, although he remained mildly confused. He was followed up in the neurosurgical outpatient clinic 6 months after repatriation and, by this stage, he was living at home independently, suffering no obvious sequelae of these events. No cause was ever found for the precipitating diarrhoea and vomiting.
Management of the severely dehydrated patient
Appropriate management of the severely dehydrated and uraemic patient is challenging and there is no specific guidance in the literature. Clinicians must use their judgement and discretion when tailoring alternative available guidelines for use in individual patient cases.
Since at least 1949, it has been understood that the mainstays of treatment for uraemia included the meticulous correction of water and electrolyte disturbance, with water balance taking precedence over electrolytes. 1 Today’s clinician has a greater ability to monitor, measure and modify fluid and electrolytes, but this still must be done with caution and an awareness of the potential risks.
There are, broadly, two theoretical approaches one could take to correcting this patient’s severe dehydration and uraemia:
Management of hypernatraemia by estimating and correcting the fluid deficit according to serum sodium levels and managing the patient as per existing management of hypernatraemia guidelines (e.g. Adrogué),
2
or Managing and correcting the hyperosmolar state, but the only available guidance here is the hyperosmolar hyperglycaemic state (HHS) guideline where the hyperosmolar state is secondary to an elevated glucose (although these patients are additionally profoundly dehydrated and may have an elevated sodium too).
3
Hypernatraemia approach (Adrogué)
Formulae hypothetically allow a clinician to calculate a patient’s free water deficit by utilising the extracellular sodium concentration as a surrogate marker of hydration status. Hypotonic solutions (5% dextrose in water, 0.2% sodium chloride in 5% dextrose in water and 0.45% sodium chloride in water) are recommended as the intravenous resuscitation fluids in the accompanying guidance excepting in the presence of ‘frank circulatory compromise’, when ‘isotonic’ solutions such as 0.9% sodium chloride are advised. The theory is that dehydration culminating in a relative water deficit is visible to the clinician via the measured sodium concentration.
Applying the Adrogué formula to this case of hypernatraemia, at the time of arrival to the NCCU, with an estimated patient weight of 65 kg and a plasma sodium concentration of 169 mmol/L:
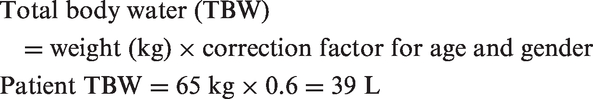
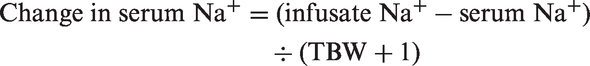
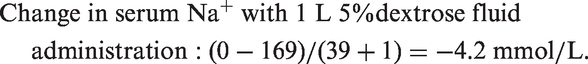
This suggests that after 1 L of 5% dextrose, the expected measured sodium would be approximately 165 mmol/L and to normalise sodium (reference range 135–145 mmol/L), 6 L of 5% dextrose would be required.
Lindner et al. analysed four different formulae designed to aid the correction and maintenance of serum sodium in a critical care setting through estimation of the free water deficit and found the formulae were not able to accurately guide infusion therapy in hypernatraemic states in the ICU patient. 4
The established and consistent guidance in the literature on managing chronic hypernatraemia (an elevated sodium for >48 h) is of a maximum reduction in sodium concentration of 10 mmol/L (equivalent to an osmolarity of 20 mmol/L) in a 24-h period due to the risks of fluid overload, cerebral oedema or central pontine myelinolysis caused by fluid shifts due to altered tonicity. 5
Hyperosmolar state approach
For practical purposes, osmolality (mosmol/kg) and osmolarity are interchangeable, with the latter being calculable and measured in mmol/L. At the time of presentation to the admitting hospital, our patient had a calculated osmolarity of 428.7 mmol/L (normal range 285–295 mmol/L). On arrival in our NCCU, his calculated osmolarity was 399.6 mmol/L. The equation we have used to calculate this is:
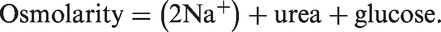
The HHS guideline’s goals of treatment, which make it potentially helpful in this case, are to:
normalise the osmolality; replace fluid and electrolyte losses; (normalise blood glucose levels).
3
It is important to note that the guideline is, of course, designed to guide the rehydration of patients who are hyperosmolar secondary to severe hyperglycaemia. This is important to consider because whilst sodium and glucose are both effective osmoles contributing to tonicity, urea is not, as it may cross the cell membrane freely.
The rate of decline of osmolarity suggested by the HHS guideline is 3–8 mmol/L/h utilising 0.9% saline as the preferred resuscitation fluid. However, the reduction in osmolarity must be balanced against the rate of change in serum sodium level (a maximum change of 10 mmol/L in a 24-h period, as before), the sodium being expected to rise in the first instance as water moves intracellularly. The expected fluid replacement required in the management of HHS is 6–13 L in a 60 kg patient, with 50% being given in the first 12 h.
Our approach
On initial presentation at his local hospital, our patient was moderately hypernatraemic (sodium 152 mmol/L), severely uraemic (urea 118 mmol/L) and hyperosmolar (calculated osmolarity 428 mmol/L), but his effective tonicity if urea is discounted as being an ineffective osmole was not elevated (effective osmolarity 310 mmol/L). He was given fluids (precise details are unknown) and had an initial positive fluid balance but close monitoring and fluid resuscitation stopped when the patient was briefly palliated.
Four days after the initial presentation, on arrival at our institution, he was severely hypernatraemic (sodium 169 mmol/L), severely uraemic (49 mmol/L) and hyperosmolar (calculated osmolality 399 mmol/L), but due to the effectiveness of the components of osmolality, he had a higher effective tonicity (effective osmolarity 350 mmol/L) than on initial presentation to hospital.
We felt it prudent to be somewhat cautious with rehydration post-operatively as his initial period of fluid administration was associated with development of an SDH. We postulated that the SDH occurred through a combination of uraemic platelet dysfunction, a known cause of intracranial haemorrhage, and fluid shifts in less elastic tissue through severe dehydration, in this patient with complex pathophysiology. 6 In addition, the rise in serum sodium, with a reduction in urea but more minimal change in the calculated osmolarity following that first episode of fluid resuscitation (at the local hospital) suggests there was perhaps a dilutional relative hyponatraemia secondary to uraemia initially. Changes in plasma sodium may also have been influenced by the consequent cerebral dysfunction.
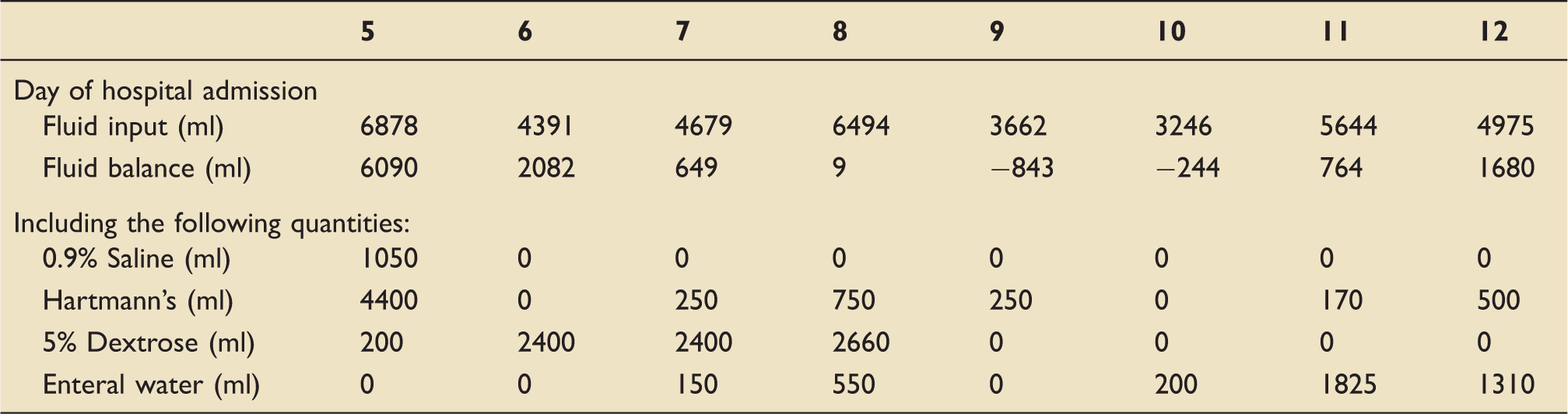
Details of fluids administered during his post-operative recovery phase.
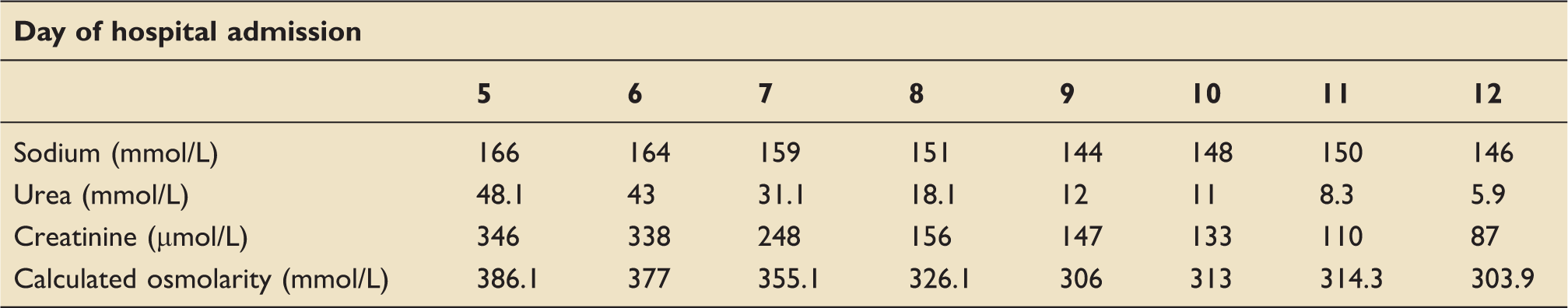
Biochemistry result normalisation during his post-operative recovery phase. Monitoring of biochemistry on fluid resuscitation took place multiple times during the initial post-operative period, but daily results are tabulated here.
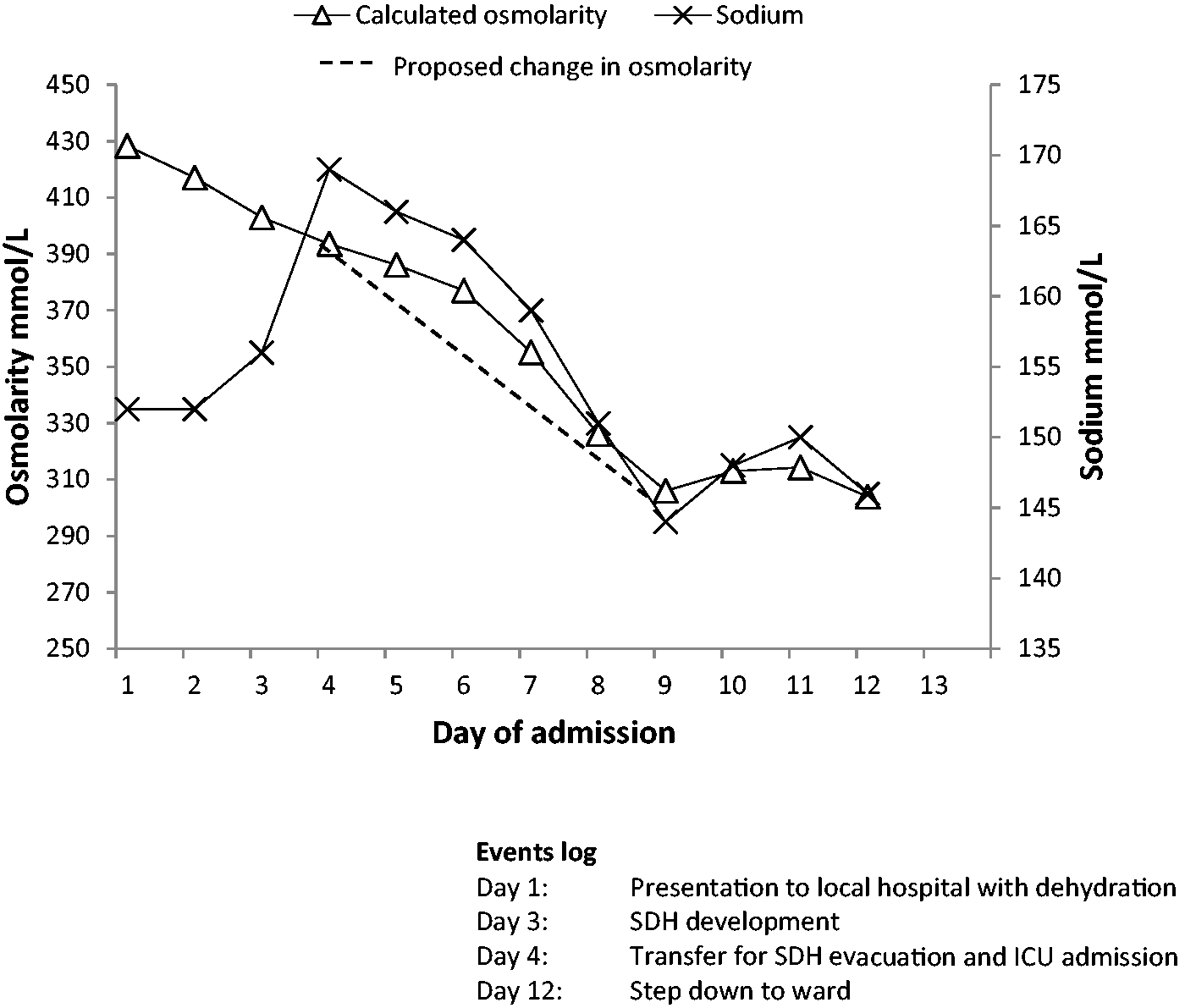
Changes in plasma sodium and osmolarity with rehydration.
Initially, large quantities of isotonic fluids were used for rehydration with frequent blood sampling targeting a daily reduction in calculated osmolarity of 20 mmol/L. Following the initial rehydration, hypotonic solutions were able to be utilised safely and, once the biochemistry results were near reference ranges, enteral water was used. Intravenous fluids given are itemised, with the remainder of fluid input comprising medications and nasogastric feed.
The patient required regular blood sampling to assess the serum biochemical changes in response to fluid type and volume, with close fluid balance monitoring. Normal sodium, urea and creatinine test results were not seen until day 12 of admission, at which point the patient had a cumulative positive fluid balance of 16 L (40% estimated TBW). Calculated osmolarity at this point was 303.9 mmol/L.
When comparing his actual cumulative fluid balance with the initial estimate of free water required from the Adrogué calculation method (6 L), it can be seen how inaccurate that method can be. The standard fluid quantities required to resuscitate a patient with HHS, however are more suggestive of the quantities of fluid that may be required (up to 13 L) in a patient with hypernatraemia secondary to dehydration through hyperglycaemia. Additionally, the Adrogué method promotes the use of hypotonic solutions and advises against isotonic solutions; in direct conflict, the HHS guidelines advocate the use of isotonic rather than hypotonic fluids, although both caution against rapid changes in plasma sodium.
Conclusion
The presented case is one of severe dehydration and AKI that required considerable fluid and electrolyte replacement. Approaches to fluid resuscitation in the context of hypernatraemia and the hyperosmolar state were considered. These were judiciously combined to manage a complex case of severe dehydration and biochemical disarray with a successful outcome.
It may be that we were overly cautious in the speed of correction of this patient’s biochemistry, but in addition to estimating how much fluid may be required to resuscitate a dehydrated patient, this case also serves as a reminder of the potential consequences of dehydration, including hyperviscosity, AKI and uraemic platelet dysfunction; difficulties prognosticating for individuals when the literature provides information regarding cohorts; and the amazing resilience of some patients.
We suggest a reconsideration of the approach to correcting hypernatraemia in patients with chronic hypernatraemia (>48 h) and dehydration, as attempting to calculate the free water deficit in critically ill patients is highly inaccurate. We suggest individualisation of resuscitation regimes for patients based on osmolarity, monitoring and reviewing the response to fluid administration whilst maintaining the clear guidance on safe parameters for changes in sodium concentrations. Our recommendation draws significant parallels with the current guidance on the management of HHS.
Footnotes
Consent
Published with the written consent of the patient.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
