Abstract
Anlotinib is a tyrosine kinase inhibitor that targets the vascular endothelial growth factor receptor for the treatment of lung cancer. Pneumothorax is a rare complication of anlotinib treatment. Here, the case of a male patient in his early seventies, with lung cancer combined with emphysema, who developed a pneumothorax during treatment with anlotinib, is described. The patient was admitted to hospital mainly for dyspnoea and was diagnosed with pneumothorax after digital radiography of the chest. The patient's symptoms improved significantly after closed chest drainage, and a repeat chest digital radiography showed a more resolved pneumothorax. The patient had no previous history of pneumothorax. After discontinuation of anlotinib, the latest follow-up chest computed tomography assessment in August 2023 showed no recurrence of pneumothorax, thus, the pneumothorax is presumed to have been associated with anlotinib in this patient. In addition, the authors speculate that emphysema may be a cause of pneumothorax in patients with lung cancer receiving anlotinib treatment. Therefore, clinicians should be alert to the risk of pneumothorax occurrence in patients with emphysema combined with lung cancer who are treated with anlotinib.
Introduction
Lung cancer was reported to account for approximately 18% of cancer deaths worldwide in 2020, far surpassing other cancer types and ranking first in the number of cancer deaths. 1 Despite the fact that most patients are diagnosed at advanced stages, molecularly targeted drugs are still making impressive achievements in lung cancer treatment. Anlotinib is a relatively new tyrosine kinase inhibitor (TKI) developed in China that significantly inhibits angiogenesis and reduces microvascular density to achieve antitumor effects. 2 Based on results of the phase II ALTER 1202 clinical study, 3 anlotinib was approved by China’s National Medical Products Administration in May 2018 for third-line treatment of advanced non-small cell lung cancer, and since then, has been approved for extensive stage small cell lung cancer, soft tissue sarcoma, and other malignancies as a post-line treatment.
Pneumothorax secondary to lung malignancy is rare, accounting for less than 2% of all pneumothorax cases, and there are no proven features regarding the occurrence of pneumothorax in patients with lung cancer. 4 However, pneumothorax, a rare complication of anlotinib, usually leads to suspension of the drug and may even be life-threatening for patients. 5 The present report describes the case of a patient who had lung cancer combined with emphysema and developed a pneumothorax during treatment with anlotinib. In addition, the risk of pneumothorax in patients with lung cancer combined with emphysema, and the possible mechanisms, are explored.
Case presentation
Written informed consent to treatment was obtained from the patient. All patient details were deidentified for this case report, thus, the patient’s informed consent to publish the case was not considered necessary, and ethics approval was also not required for case report publication. The reporting of this study conforms to CARE guidelines. 6
A male patient in his early seventies, with lung cancer, was admitted to the Department of Medical Oncology, Lixin County People's Hospital of Bozhou City, China, in July 2022, with right shoulder and back pain with dyspnoea for 3 days. Previously, the patient had visited his local hospital in 2019 for intermittent coughing and was diagnosed at that time by the local pathology department with small cell lung cancer, then diagnosed with small cell lung cancer extensive stage (Figure 1). The patient had a history of smoking and emphysema. He received six cycles of chemotherapy at a local hospital between March 2021 and August 2021 with the following specific regimen: carboplatin target area under the curve (AUC) 5 mg/ml/min, day 1, combined with etoposide 100 mg/m2, day 1–3, intravenous (i.v.) drip, every 3 weeks. Cancer progression was assessed by imaging in November 2021 and the patient subsequently received four cycles of chemotherapy at a different hospital between November 2021 and February 2022, according to the following regimen: albumin-bound paclitaxel 260 mg/m2, day 1, i.v. drip, every 3 weeks. Cancer progression was noted on imaging in June 2022 and the treatment regimen was again changed. Two cycles of anlotinib were initiated in June 2022 at the same hospital, according to the following regimen: 12 mg, day 1–14, orally, every 3 weeks. During a third cycle of anlotinib treatment, in July 2022, the patient was admitted to Lixin County People's Hospital with right shoulder and back pain with dyspnoea for 3 days. On examination, his breath sounds in the right lung were markedly absent on auscultation, and drumming sounds were detected on percussion. In July 2022, digital radiography of the chest showed a right pneumothorax (Figure 2). The patient's pneumothorax was assessed as a grade 3 event, according to the Common Terminology Criteria for Adverse Events (CTCAE), Version 5.0. 7 The patient was instructed to stop taking oral anlotinib, and after treatment by closed chest drainage and oxygenation, a follow-up chest digital radiography 2 days later showed reduced extent of the right pneumothorax (Figure 3). After a further 5 days, follow-up chest computed tomography (CT) showed a reduced extent of the right pneumothorax and bilateral emphysema was observed (Figure 4a). Review of a previous chest CT obtained in June 2022 showed that the patient had no pneumothorax prior to anlotinib treatment (Figure 4b). After treatment with closed chest drainage and oxygenation, and discontinuation of anlotinib, the patient was discharged from the hospital, and the pneumothorax did not recur. Follow-up chest CT in August 2023 showed no recurrence of pneumothorax.
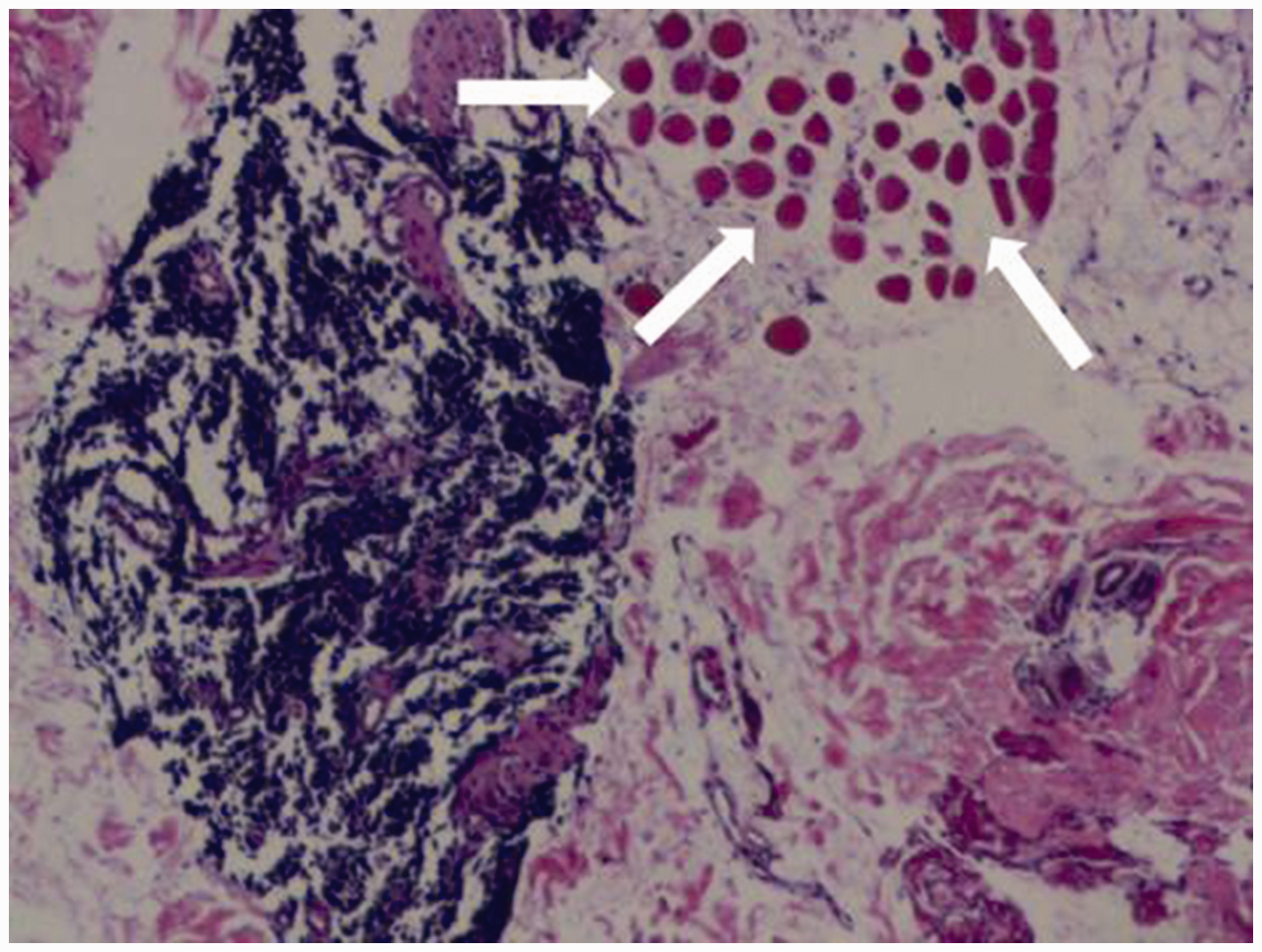
Photomicrograph of haematoxylin and eosin-stained metastatic lymph node puncture biopsy tissue from a 70-year-old male patient showing typical features of small cell carcinoma (white arrows). The tumour cells are smaller than 3 small lymphocytes, have no organ-like structures, are round, oval or spindle shaped, approximate a stripped nucleus and are poorly demarcated (original magnification, ×40).
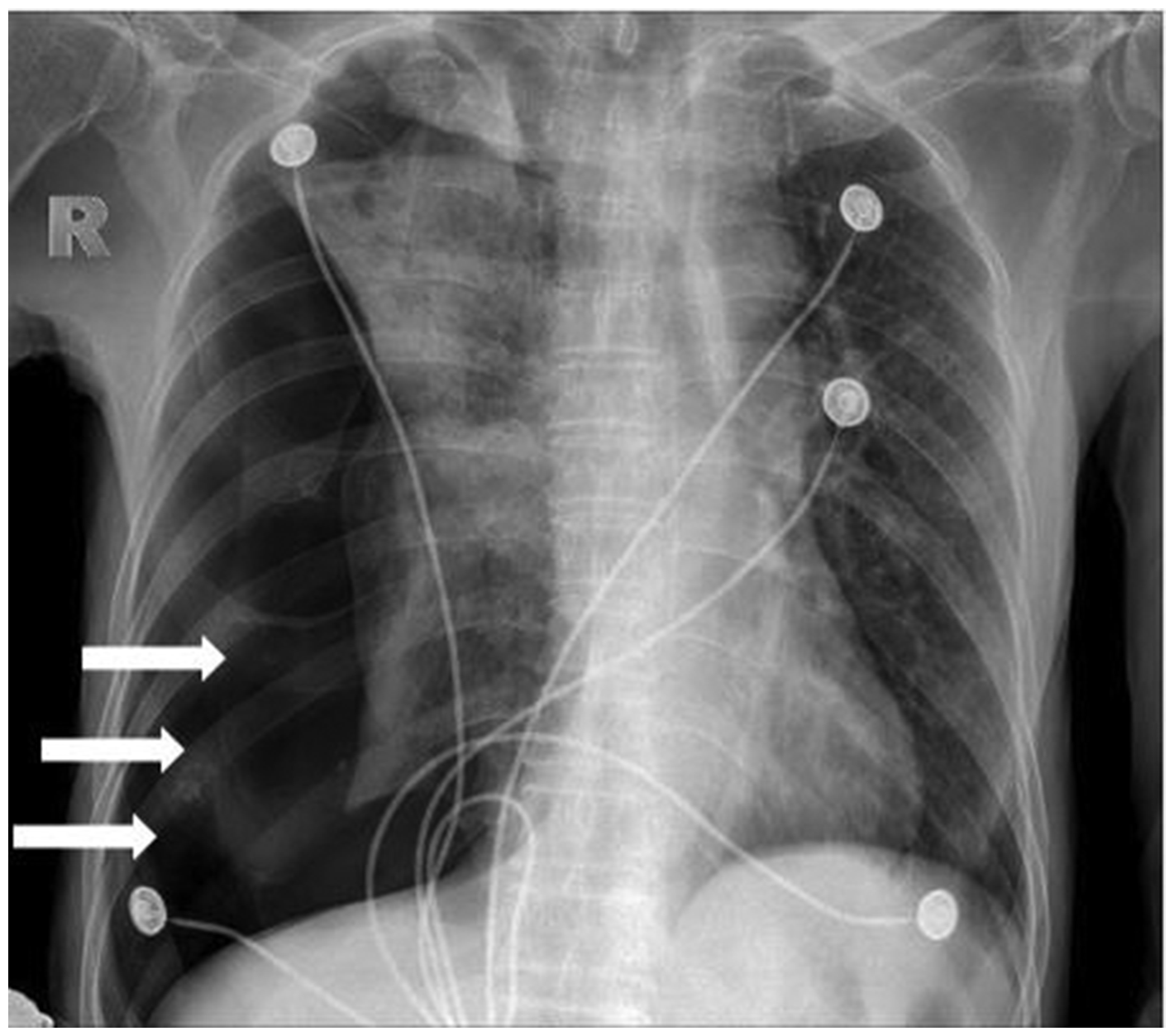
Chest digital radiograph from a 70-year-old male patient with small cell carcinoma showing right pneumothorax (white arrows).
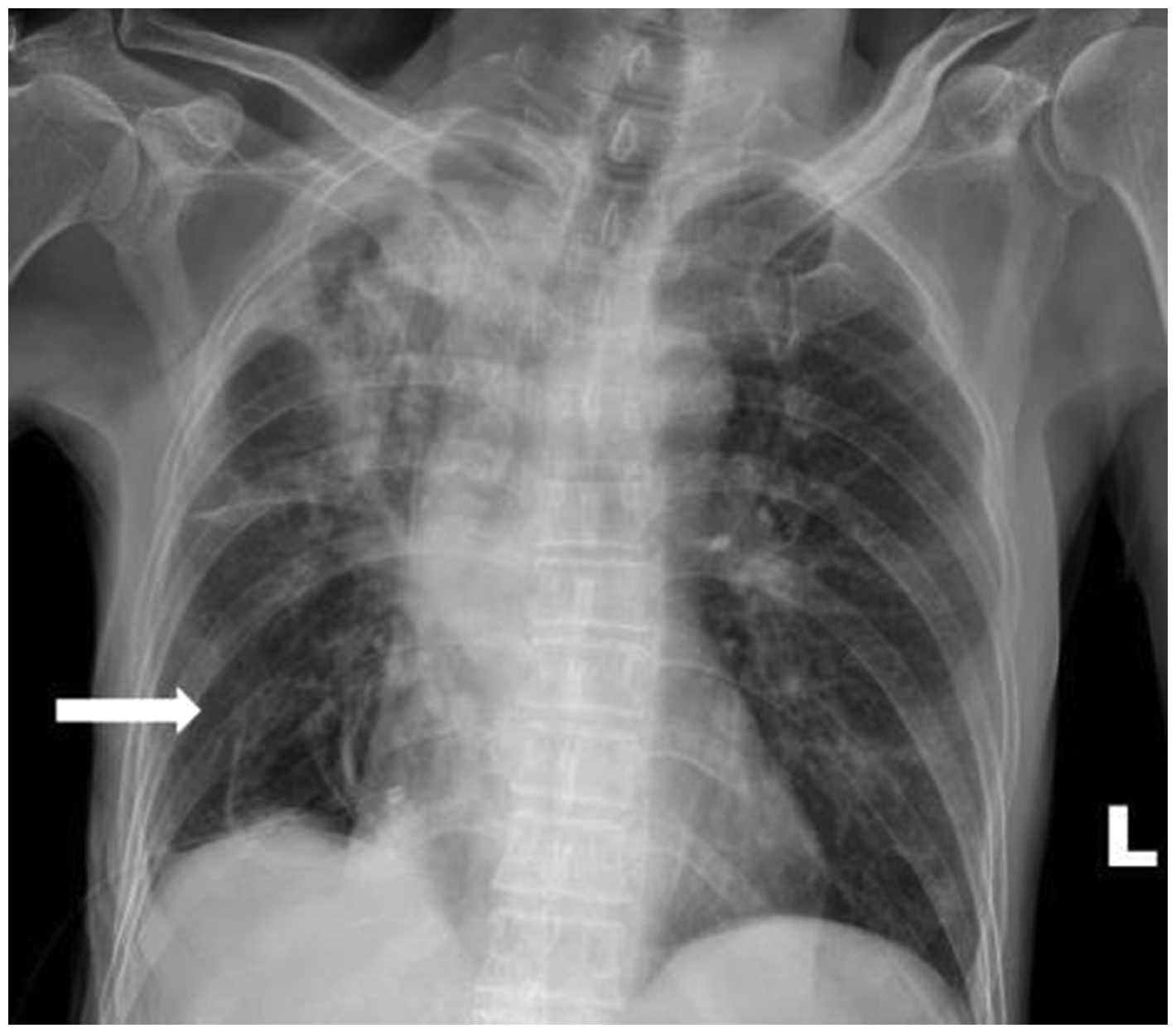
Chest digital radiograph from a 70-year-old male patient with small cell carcinoma showing significant reduction in pneumothorax after treatment (white arrow).

Chest computed tomography (CT) axial images from a 70-year-old male patient with small cell carcinoma showing: (a) lobule-centred emphysema (white arrows) and pneumothorax (red arrow); and (b) lobular-centred emphysema (white arrows), but no indication of pneumothorax prior to treatment with anlotinib (image obtained in June 2022).
Discussion
Angiogenesis plays a crucial role in tumour growth and metastasis. Therefore, blocking this pathway has become a major direction of research in the clinical treatment of cancer. Anlotinib is a novel oral multitarget receptor TKI that inhibits vascular endothelial growth factor receptor (VEGFR)-1, VEGFR-2, VEGFR-3, mast/stem cell growth factor receptor Kit (c-Kit), hepatocyte growth factor receptor (c-Met), proto-oncogene tyrosine-protein kinase Src (c-Src), platelet-derived growth factor receptor beta (PDGFRβ), receptor tyrosine-protein kinase erbB-2 (HER2), and epidermal growth factor receptor (EGFR). In preclinical studies, anlotinib has been shown to inhibit human lung cancer cell viability and induce apoptosis, which in turn, enhances the cytotoxicity and antiangiogenic effects of anlotinib through the janus activating kinase 2 (JAK2)/ signal transducer and activator of transcription 3 (STAT3)/ vascular endothelial growth factor (VEGF)A signalling pathway. 8 Results of the study by Qin et al. 9 showed that anlotinib may inhibit lymphangiogenesis and lymphatic metastasis by inactivating VEGFR-3 phosphorylation. The ALTER 0303 and ALTER 1202 studies have established the status of anlotinib in the treatment of advanced lung cancer in China.3,10 In previous studies, the most common adverse events of anlotinib were hypertension, elevated thyroid stimulating hormone, hypertriglyceridemia and hand-foot syndrome. However, pneumothorax is a rare complication of anlotinib. 5
The patient reported in the present case had no previous history of pneumothorax, developed pneumothorax during the application of anlotinib, and has not developed pneumothorax since discontinuing anlotinib due to pneumothorax. Therefore, our analysis concluded that the pneumothorax in this patient was caused by anlotinib. In a previous retrospective multicentre study, treatment with anlotinib was permanently discontinued in one patient due to two recurrent pneumothoraces, and the possible mechanism was speculated to be related to subpleural and pleural metastases and cavitary lung lesions after anlotinib treatment. 11 The previous retrospective study investigated the use of anlotinib in patients with pulmonary metastases from osteosarcoma, 11 however, the patient in the present case was diagnosed with primary lung cancer and no cavitary lung lesions or pleural metastases were observed on chest CT, suggesting that the described mechanism may not be the cause of pneumothorax in the present case. Patients with primary lung tumours very rarely develop pneumothorax during treatment. However, pneumothorax appears to occur more frequently in patients with sarcoma metastases, particularly osteosarcoma, than in patients with primary lung cancer. 12 This may be related to the rupture of necrotic tumour nodules, necrosis of subpleural metastases, direct tumour invasion into the pleura, and a compromised tumour-related haemostatic valve mechanism during targeted therapy or chemotherapy for lung metastases.13–15 The abovementioned cause was not necessarily the reason why the present patient developed a pneumothorax. The patient reported in the present case had primary lung cancer combined with emphysema, and developed pneumothorax after receiving targeted therapy for lung cancer. Thus, we speculate that emphysema may have been involved in the pathophysiology of pneumothorax formation after targeted therapy in this patient. Disruption of the alveolar septa attached to the outer walls of the small airways is reported to be associated with wall remodelling and luminal narrowing of these airways in emphysema. 16 The alveolar septa belong to the interstitium of the lung, which contains a dense network of continuous-type capillaries. The mechanism of anlotinib is to significantly inhibit angiogenesis and reduce microvascular density, thus, it may be speculated that pneumothorax probably occurred in the present patient due to the combined effect of the reduction of microvascular density by anlotinib and the destruction of the alveolar septum by emphysema. Of course, this is only speculation based on the mechanism of action of anlotinib and the pathology of emphysema formation, and these factors do not prove such a cause. However, future basic trials may be needed to demonstrate whether there is a synergistic effect between these two factors in the formation of pneumothorax. Previous studies have only reported the number of people presenting with pneumothorax,3,5,10 without further analysis of the mechanisms of pneumothorax development or the clinical characteristics of the patients. Therefore, it remains unclear whether pneumothorax is more likely to occur with anlotinib in patients with lung cancer combined with emphysema. Based on our speculation about the occurrence of pneumothorax in the present case, the use of anlotinib in patients with lung cancer combined with emphysema requires vigilance for the occurrence of pneumothorax.
The results of the present study may be limited by several factors. In particular, the case report study design makes it difficult to evaluate the overall characteristics of pneumothorax in patients treated with anlotinib for lung cancer combined with emphysema. Thus, further studies are required to clarify the present results.
In conclusion, clinicians need to be aware that the occurrence of pneumothorax is possible during the use of anlotinib to treat patients with lung cancer combined with emphysema. Therefore, it is necessary to monitor patients closely in order to detect pneumothorax early and avoid life-threatening accidents.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231208923 - Supplemental material for Case report of severe pneumothorax due to lung cancer treated with anlotinib
Supplemental material, sj-pdf-1-imr-10.1177_03000605231208923 for Case report of severe pneumothorax due to lung cancer treated with anlotinib by Haowei Lu, Feiyang Li, Yong Yang and Dong Zhao in Journal of International Medical Research
Footnotes
Acknowledgements
We thank our colleagues, Lihong Liu and Jingjing Sun, who helped with this manuscript. We thank the anonymous reviewers and the editor for their valuable comments.
Author contributions
Haowei Lu and Feiyang Li wrote the original manuscript. Dong Zhao helped design the project. Yong Yang supervised the manuscript. All authors reviewed the final version.
Data accessibility
The dataset used and/or analysed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
