Abstract
Continuous renal replacement therapy necessitates the use of anticoagulation. The anticoagulant of choice has traditionally been heparin. Emerging evidence has highlighted the deleterious effects of systemic heparin anticoagulation in the critically ill. Regional citrate anticoagulation has been used as an alternative in the setting of continuous renal replacement therapy. Our retrospective before-and-after cohort study aimed to ascertain if regional citrate anticoagulation is associated with any benefit in terms of circuit longevity, rates of complications, blood transfusion requirements and mortality, when introduced to a large general intensive care unit with a case mix of acute medical patients and acute and elective surgical patients. The switch to regional citrate anticoagulation for continuous renal replacement therapy in our intensive care unit has been associated with a dramatically longer circuit life, with major implications for cost savings in terms of reduced nursing workload. We hope to look at fiscal aspects of the change in protocol in greater depth.
Introduction
Continuous renal replacement therapy (CRRT) is used to support critically ill patients with renal failure. CRRT usually requires anticoagulation as without it circuit life is compromised by circuit clotting. 1 This can cause clinically significant interruptions to the delivery of CRRT and therefore reduce the time during which the patient receives renal support. Heparin is the most commonly used extracorporeal anticoagulant worldwide. 2
Heparin is cheap and it is a drug with which clinicians are familiar. It is however associated with a number of disadvantages the most clinically significant of which is the increased risk of bleeding. 3 Heparin requires frequent laboratory blood sampling which has consequences for the responsiveness and cost of its management. Critically ill patients are at an increased risk of haematological dyscrasias due to sepsis or the systemic inflammatory response. This can manifest as either an increased tendency to bleeding or hypercoagulability and heparin resistance. 4 The response of the critically ill patient to heparin is therefore unpredictable and as such circuit life with heparin anticoagulation is often suboptimal. Consequently, there has been a considerable amount of research in seeking an alternative anticoagulant in the setting of CRRT. Regional citrate anticoagulation (RCA) has emerged as a viable contender. 5
Citrate is well established as the standard anticoagulant for stored blood products. It chelates calcium and prevents activation of coagulation cascades and platelets. The half-life of citrate is approximately 5 min and it is therefore suitable as a regional extracorporeal anticoagulant. Citrate does not cause heparin-induced thrombocytopenia and theoretically carries no increased risk of bleeding. 6 However, there are a number of disadvantages associated with citrate use. Metabolic alkalosis and acidosis, hyper- and hypo-natraemia, and hypocalcaemia have been reported in previous studies, as well as citrate accumulation. 7 In rare cases, cardiac arrest can occur due to rapid accidental infusion of citrate.
Bradford Royal Infirmary (BRI) is a large teaching hospital in West Yorkshire. It has a combined intensive care unit (ICU)/high dependency unit caring for a mixture of medical and acute and elective surgical patients, with approximately 1100 admissions per year of which 500 require level 3 care. Approximately 120 patients require RRT each year.
In April 2015, the BRI ICU switched its default method of anticoagulation from heparin to RCA. This was applied to all patients and henceforth no patient received non-RCA. The method of CRRT remained continuous veno-venous haemodiafiltration and the CRRT machines (Gambro Prismaflex) were not changed beyond the necessary software update. The new renal protocol was based on one provided by Gambro. This protocol employs a dilute citrate solution and mandates regular monitoring of ionised calcium levels within the circuit and the patient. Daily total to ionised calcium ratios are also required in order to detect any untoward effects of citrate accumulation. Training on the changes in protocol was delivered by ICU medical staff and senior nursing staff, with additional support from Gambro.
Our retrospective before-and-after cohort study set out to establish whether the reported benefits of RCA would be applicable to our ICU in a non-trial setting and if it might confer any advantage over existing non-RCA protocols.
Method
Data were collected retrospectively. One hundred consecutive patients were identified from a contemporaneous database which holds details of all patients receiving CRRT at BRI. Our patient cohort included patients who were admitted to ICU between April 2015 and September 2015. In addition to demographic details, this electronic database houses information on the duration of CRRT, number of circuits required per patient and the longevity of each circuit used.
Anticoagulants used in non-RCA cohort.

RCA: regional citrate anticoagulation.
Our local protocol for RRT was updated in April 2015 and heparin was superseded by citrate. The first 50 consecutive patients who received RCA were identified following this change: these cases constituted our RCA group.
A data collection tool was created, trialled and modified. Patients were given anonymised identification numbers in order to maintain confidentiality.
Each case identified from the RRT database was further investigated by interrogation of the electronic patient record (Innovian – Drägerwerk AG & Co.) and web-based access to diagnostic results (Integrated Clinical Environment – Sunquest Information Systems Inc.).
We recorded the following variables:
Reason for ICU admission, e.g. ventilatory support, inotropic support and invasive monitoring; Length of stay in ICU; Mortality; Reasons for discontinuation of CRRT (each individual circuit was analysed); Complications such as bleeding, suspected heparin-induced thrombocytopenia, symptomatic hypocalcaemia, hypotension
8
and citrate accumulation; Blood transfusion requirements at any point following commencement of RRT.
Acute Physiology and Chronic Health Evaluation II (APACHE II) and Intensive Care National Audit and Research Centre (ICNARC) scores were calculated for all cases.
No cases were rejected for incomplete data.
We analysed our data using Stata 13 (StataCorp, 2013). Simple analyses were conducted using the Mann–Whitney U test for numeric data and Fisher’s exact test for proportions.
Results
A comparison of patient demographics between non-RCA and RCA cohorts.
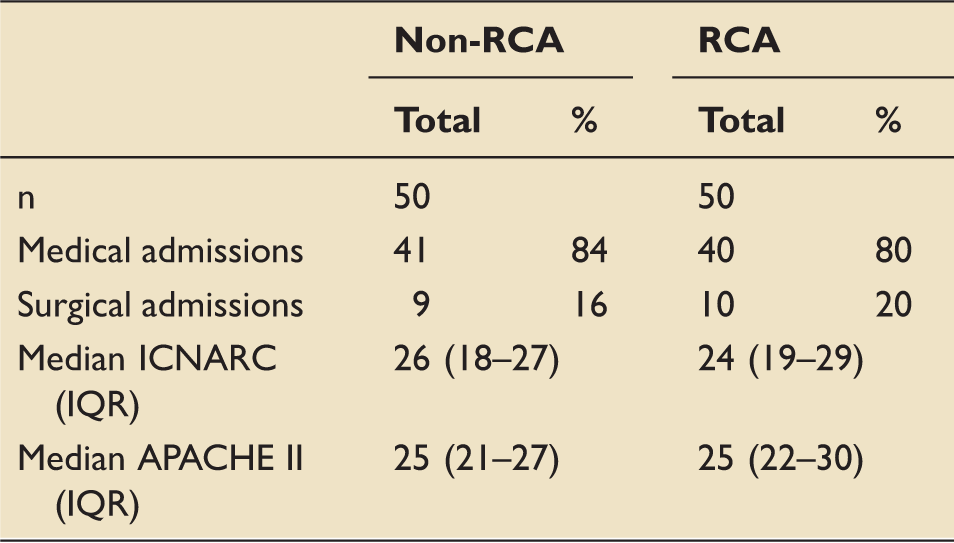
APACHE II: Acute Physiology and Chronic Health Evaluation II; ICNARC: Intensive Care National Audit and Research Centre; IQR: interquartile range; RCA: regional citrate anticoagulation.
A total of 157 circuits were utilised in the non-RCA group. This equates to a mean value of 3.2 circuits per patient over a mean 10.2-day stay in ICU. Thirty-nine fewer circuits were used in the RCA group (n = 118) which is a mean of 2.4 circuits per patient over a mean 11.3-day stay.
Our analysis of circuit life included both elective and acute discontinuations. Examples of elective discontinuations include scenarios in which the patient was moved off ICU for the purposes of treatment or investigation, or when there was a scheduled pause in CRRT. Acute discontinuations were due to circuit clotting, problems with access, bleeding and patient death. We found that the circuit life was non-parametrically distributed. With all circuits included, the median circuit life in the non-RCA group was 12 h (IQR (6–28)) compared with 26 h (IQR (10–52)) in the RCA group. Therefore, use of RCA was associated with a 14 h increase in median circuit life per patient. This was highly statistically significant (p = 0.0002).
It has been postulated that inflammatory mediators can block circuit pores and therefore cause an increase in transmembrane pressure. This may reduce circuit life. Patients with higher APACHE II scores have greater disease severity and may consequently have a more pronounced inflammatory response. 9 There is a tendency to smaller vasculature in women in comparison to men 10 and this may account for in increased incidence of vascular access issues impeding delivery of CRRT. We therefore used regression analysis to control for APACHE II score and gender. When these two parameters were controlled for we found that RCA was associated with an increase in circuit longevity of 11 h (p < 0.001, 95%CI 5–16 h).
Kaplan–Meier survival analysis demonstrated that half of all circuits in the non-RCA group had failed by 14 h (Figure 1). In the RCA group 50% failure did not occur until 61 h. The survival analysis considered electively discontinued circuits to be censored; however, despite this we noted a ‘drop-off’ in both groups as circuits approached the 72 h expiry limit we use as standard in our ICU. A potential explanation is that circuits which were proving to be problematic close to when they would be discontinued electively were discontinued early rather than staff adopting a troubleshooting strategy.
Kaplan–Meier estimates of circuit survival.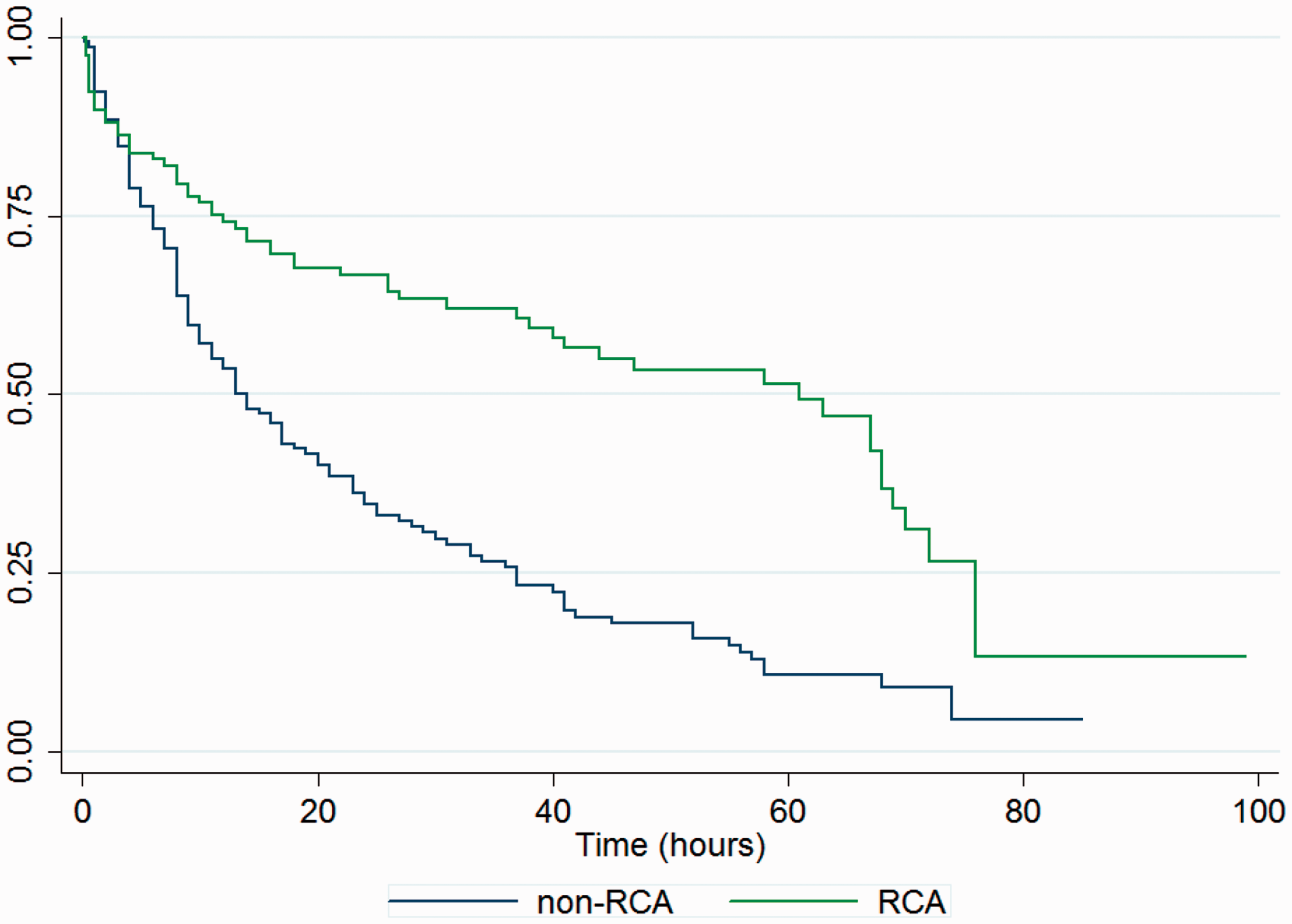
On examination of blood transfusion requirements, 36% of non-RCA patients (n = 18) required blood transfusion compared with 24% of RCA patients (n = 12). This was not statistically significant (p = 0.28).
A comparison of complications between non-RCA and RCA cohorts.
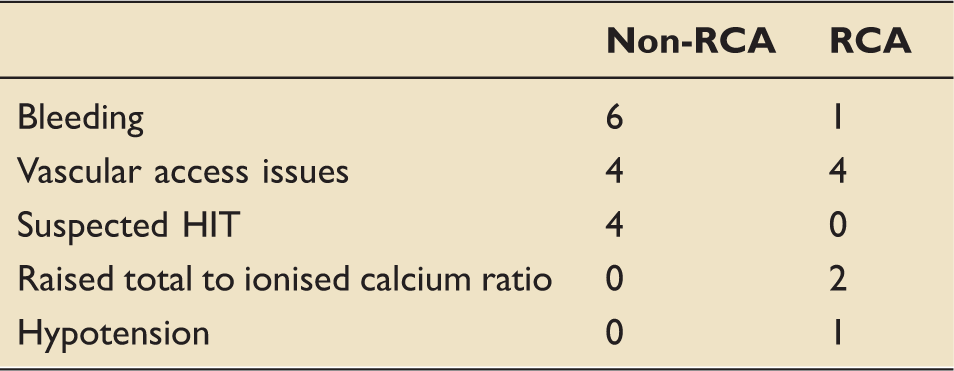
HIT: heparin induced thrombocytopenia; RCA: regional citrate anticoagulation.
Patient mortality in ICU.
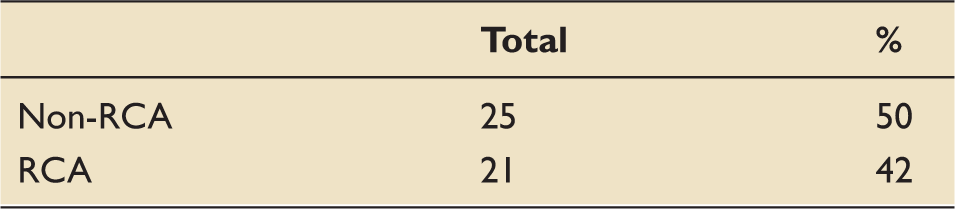
ICU: intensive care unit; RCA: regional citrate anticoagulation.
Cost analysis for seven days of CVVHDF.
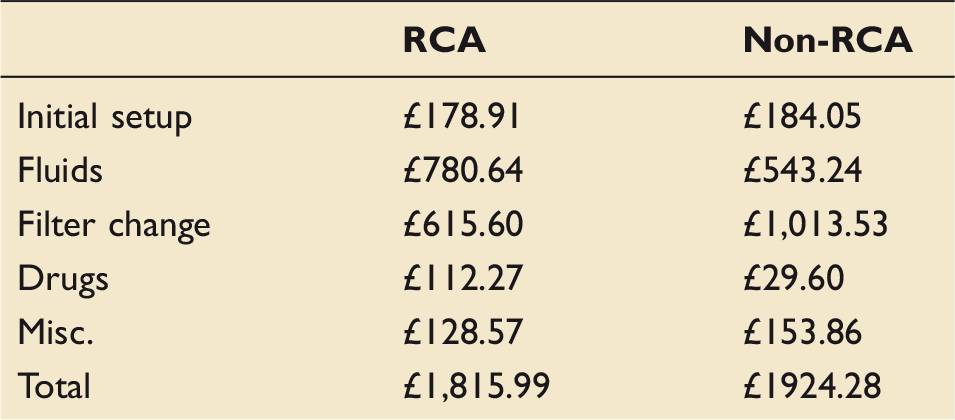
CVVHDF: continuous veno-venous haemodiafiltration; RCA: regional citrate anticoagulation.
Costs were obtained from our procurement department. Due to the commercially sensitive nature of publishing precise cost figures all numbers have been scaled to an assumed cost of £10 for a 5 l bag of Prismocitrate. Prisomocitrate is included in ‘fluids’ rather than ‘drugs’ in the below table as it acts as both an anticoagulant and a haemofiltration replacement fluid.
These results demonstrate an over 10% cost saving from our previous practice, before nursing time is considered.
Discussion
Having analysed our results, we are confident that both groups in our retrospective before-and-after cohort study were well matched in terms of illness as is indicated by their similar APACHE II and ICNARC physiology scores. Furthermore, the case mix of surgical and medical patients was also comparable. We are thus assured that the comparison between groups is ‘like for like’.
Our most striking finding in terms of both statistical and clinical significance is the increase in circuit life associated with RCA use. We feel that a 14 h increase in median circuit life per patient represents an important improvement in patient care. Whilst we are aware that in the critically ill patient there may be other factors which hamper RRT delivery, the ability to continuously administer 60 h of uninterrupted therapy in more than half of circuits in the RCA group is clearly advantageous. It should be noted that the citrate data represents the real-world clinical implementation of a new protocol and system. Familiarity and protocol refinement will hopefully improve circuit life over time.
The 14 h increase in median circuit life was of similar magnitude to other studies, 11 but may understate the true effect of the change due to our inclusion of elective discontinuations in the analysis. This was due to the fact that simple exclusion of electively discontinued circuits would result in bias due to a far greater number of RCA circuits being excluded, as well as an apparent tendency of troublesome circuits nearing an elective discontinuation to be marked as failed. The survival analysis is able to account for the first of these issues correctly, by considering the circuits as ‘censored’.
The primary benefits of RCA are clinical but the potential for cost savings in terms of decreased circuit set requirements and fewer nursing hours spent in the stop–start process of changing circuits cannot be ignored.
A full cost analysis is not possible from the data we collected but we have undertaken a crude assessment to support the ongoing business case for RCA. Using a number of approximations and the circuit survival data produces a cost reduction of just over 5% using RCA. This extrapolates to a recurring saving of over £9000/year for our unit and does not factor in nursing time costs. This figure is admittedly imprecise but may help guide other ICUs considering switching to RCA.
Our analysis of circuit complications was crude and a more detailed exploration into this domain may have yielded pertinent results. For example, we identified those cases in which there were vascular access issues but did not probe further to ascertain what those issues were. We acknowledge that this was a somewhat superficial approach. Those complications that have been recorded are not necessarily associated with RRT and may have occurred independently. We were unable to demonstrate a statistically significant reduction in complications associated with RCA.
The same can be said of blood transfusions – we did not look at the clinical context in which the blood transfusion was administered. We focused solely on whether or not the patient required a transfusion during the time at which they received RRT. Although we observed fewer patients requiring transfusion in the RCA group, our study was not powered to determine if this was a genuine effect of RCA.
We limited our analysis of mortality to deaths in ICU – the mortality figures derived were similar across both groups and no statistically significant difference was revealed.
Our study has clearly demonstrated a local benefit in circuit longevity in association with RCA in our ICU. We found no evidence for harm: we did not find evidence for an increased risk of complications, blood transfusion or mortality in ICU, and there may have even been a decrease. However, our study was not powered to fully answer this question.
In the current climate of austerity in the National Health Service, we wish to further explore whether or not our findings correlate with a reduction in expenditure. This will involve a detailed analysis of the cost of RRT in each group. We hope that a by-product of our change in anticoagulant is a saving in resources and a reduction in nursing workload.
In summary, RCA has delivered a significantly improved circuit life when compared to heparin. We postulate that this will deliver patient care, nursing workload and cost improvements.
Footnotes
Acknowledgements
The authors acknowledge the help received from Sister Julie Partridge who kindly shared her RRT database with us.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
