Abstract
Large randomised controlled trials show no benefit of high intensity renal replacement therapy compared to lower intensity regimens. Previous data suggest large variation in practice. This audit evaluated practices in relation to intensity of replacement therapy in critical care units across the Scottish National Health Service over a 28-day period. The mean delivered weight-adjusted effluent flow rates for continuous veno-venous haemofiltration were 29.1 (8.1 SD) ml kg−1 h−1 which was 89% of that prescribed. For continuous veno-venous haemodiafiltration, the mean delivered dose was 41.3 (7.9) ml kg−1 h−1 which was 88.4% of that prescribed. Of the eight patients undergoing intermittent haemodialysis, seven had daily treatments, whilst the eighth had four treatments in five days. The prescription and delivery of renal replacement therapy within Scottish critical care units are routinely performed at an intensity that is higher than necessary. Avoidance of excessive dose could provide important cost savings.
Introduction
The diagnosis of acute kidney injury (AKI) is associated with increased risk of death, hospital length of stay and costs. 1 International studies highlight that approximately 5% of critically ill patients with AKI will require renal replacement therapy (RRT). 2 Within Scotland, the proportion is much higher exceeding 10%. 3 Despite improvements in technology and advances in critical care, mortality and morbidity in the critical care environment remain high. 2
Efforts to improve outcome have focused on several variables including choice of modality, timing of initiation and membrane type, anticoagulation and dialysate buffer used. The most extensively researched area is intensity, which is defined by effluent flow rates in continuous modes or frequency and urea control in intermittent therapies. The first well-conducted study into dose revealed an improved survival with higher effluent flow rates in patients managed by continuous haemofiltration. 4 After conflicting results in various centres,5−9 two large multi-centre, randomised controlled trials have clearly demonstrated that higher intensity RRT does not improve outcome and is associated with an increase in electrolyte abnormalities.10,11 Questionnaires and observational studies performed before and during these trials suggested both uncertainty with regard to dosage12−14 and wide variations in practice.2,15
The present study aimed to describe the practice of RRT in Scottish intensive care units (ICUs) and to compare the dose prescribed and delivered with that in major published literature.
Methods
This prospective, national and observational study of current practice with regard to RRT was conducted over 28 days beginning at 00:00 on 1 September 2012 and ending at 23:59 on 28 September 2012. Dedicated local data collectors were embedded within each of the 21 general ICUs capable of providing RRT in Scotland. Caldicott Guardian approval was granted prior to commencement of the study. In light of the observational nature of the study, the West of Scotland research ethics committee waived the requirement for formal ethics approval.
Every patient admitted to a general ICU requiring RRT was included. Patients were identified on a daily basis, and variables were entered onto predefined data collection sheets. Recorded information, obtained from the 24-h observation charts, case notes and the computerised WardWatcher™ (Critical Care Audit Limited, Ilkley, UK) database included patient demographics, physiological parameters and information relating to conduct of RRT. Patients were categorised by treatment modality.
As with a previous large multi-centre study, dose was based on current clinical practice rather than a theoretical equivalent expression of dose. 15 Thus, dose of continuous modes of RRT (CRRT) was calculated as the effluent flow rates per kilogram body weight per hour. The dose for intermittent RRT was expressed as the regularity of sessions, whilst the mean urea concentration measured after implementation of treatment was used as a surrogate of intensity.
Follow-up of patients was for a maximum of 96 h for continuous modes, and 96 h or four treatment sessions for intermittent modes.
Data were collated centrally and analysed using Microsoft Excel software (Microsoft Corporation, Redmond, WA, USA).
Results
Of the 21 ICUs, 19 provided RRT for a total of 53 patients during the treatment period. All the 21 centres reported complete data capture for the study period. With regard to the mode of RRT, 34 patients underwent continuous veno-venous haemofiltration (CVVH), 11 continuous veno-venous haemodiafiltration (CVVHDF) and eight intermittent haemodialysis (IHD). The different units provided RRT as follows: 10 units performed solely CVVH, five units CVVHDF and two units IHD. One unit provided both CVVH and IHD and a further unit provided both CVVH and CVVHDF.
The capture of data for RRT sessions is depicted in Figure 1.
Flow diagram illustrating data collection for RRT sessions by mode.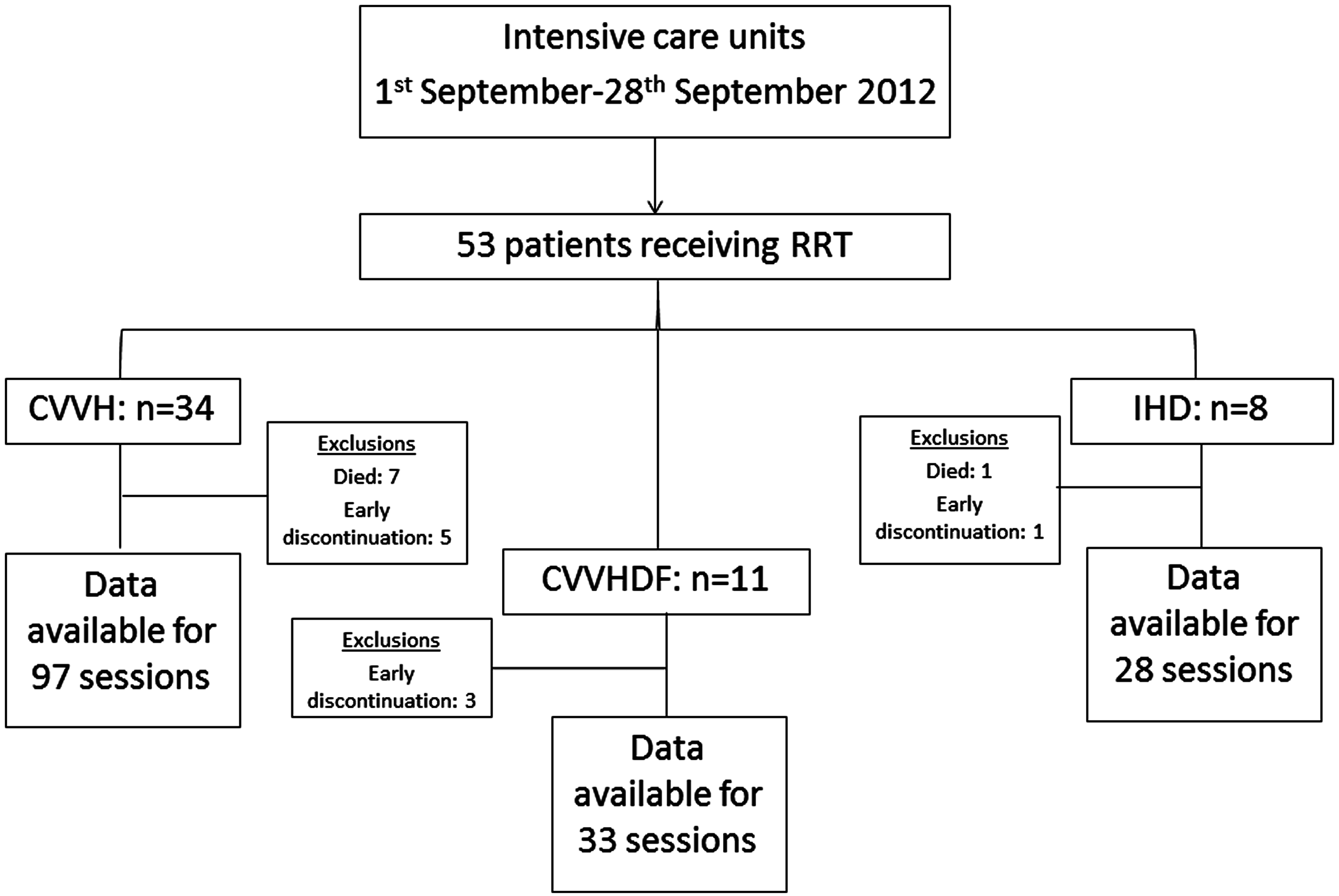
Intensity of RRT was calculated for 97 sessions of CVVH. During the 96 h, seven of our follow-up patients died, and treatment was discontinued early in a further five patients for the following reasons: passing adequate urine volumes, control of hyperkalaemia, control of acidaemia, transfer to the dialysis unit in a patient with known chronic renal failure and repatriation of a patient.
Data for 33 sessions of CVVHDF were collected. One patient had a single treatment session before it was realised that the urinary catheter was blocked, whilst another patient required only two sessions to normalise acidaemia following a drug overdose.
Follow-up of the eight patients managed by IHD provided data for 28 sessions of RRT. After a single episode, one patient died and another was discharged to the renal unit after two dialysis sessions.
There was incomplete data to allow calculation of 24-h effluent flow rates for the reasons highlighted and data regarding weight for a further patient were not provided such that no dose could be calculated for a patient undergoing CVVH (this patient died within 9 h of commencing RRT).
Characteristics of patients on commencing RRT subdivided into continuous veno-venous haemofiltration (CVVH), continuous veno-venous haemodiafiltration (CVVHDF) and haemodialysis (HD).
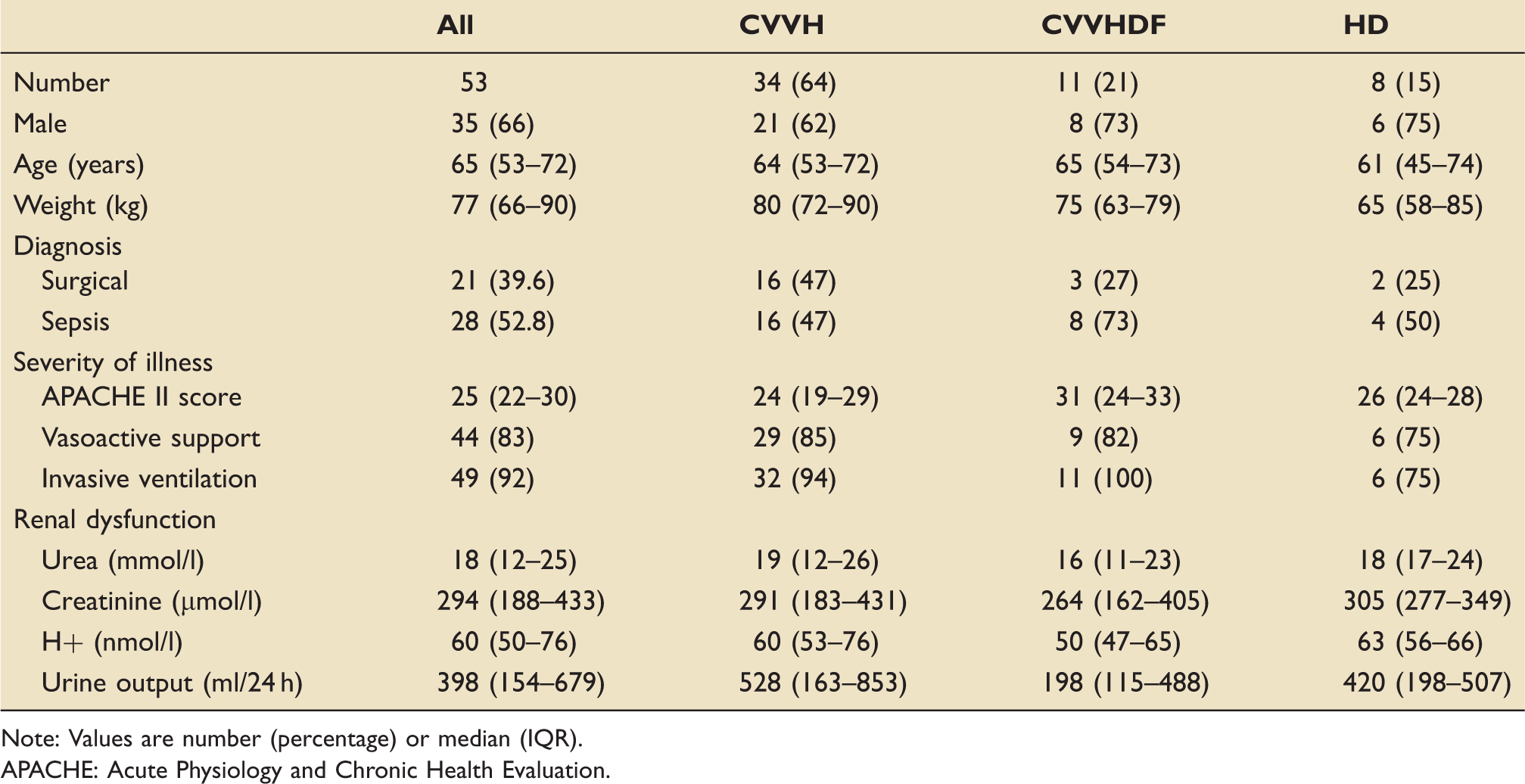
Note: Values are number (percentage) or median (IQR).
APACHE: Acute Physiology and Chronic Health Evaluation.
The mean prescribed weight-adjusted effluent flow rate for the 97 treatment sessions using CVVH was 32.4 (7.1 SD) ml kg−1 h−1. When prescriptions were divided into tertiles: no prescribed sessions were for less than 20 ml kg−1 h−1, 48 sessions (49.5%) were for between 20 ml kg−1 h−1 and 30 ml kg−1 h−1 and 49 sessions (50.5%) were for greater than 30 ml kg−1 h−1. The mean delivered CVVH dose was 29.1 (8.1 SD) ml kg−1 h−1 which was 89% of that prescribed. When delivered sessions were divided: seven sessions (7.2%) provided less than 20 ml kg−1 h−1, 53 sessions (54.6%) provided 20–30 ml kg−1 h−1 and 36 sessions (37.1%) more than 30 ml kg−1 h−1. Replacement fluid was delivered as 54% pre-filter.
For CVVHDF, the mean prescribed effluent flow rates for 33 sessions was 46.7 (7.9 SD) ml kg−1 h−1. Two units provided RRT-adjusted prescription to ideal body weight based on height which accounted for four patients. A further unit prescribed an exchange rate of 35 ml kg−1 h−1 and used additional dialysate in order to normalise biochemistry. All prescribed sessions were for greater than 30 ml kg−1 h−1. The mean delivered dose was 41.3 (7.9 SD) ml kg−1 h−1 which was 88.4% of prescribed. No sessions delivered effluent flow rates less than 20 ml kg−1 h−1, two sessions (6%) provided 20–30 ml kg−1 h−1 and 31 (94%) provided greater than 30 ml kg−1 h−1. Delivery of replacement fluid was 38.8% pre-filter.
During CVVH, 32/97 (33%) sessions were interrupted with a median downtime of 4 h (range 1–19 h). There were interruptions in 20 of 33 (61%) CVVHDF sessions leading to a median downtime of 4 h (range 1–10 h). Documented causes for downtime for all CRRT sessions were as follows: filter clotting 25 (48% of total), access problems 13 (25%), a combination of clotting and access issues 3 (6%), transfer out of unit 4 (8%), elective change of filter 2 (4%). Reasons for filter downtime were unspecified in five cases (9%). The mean time off the filter when a transfer was undertaken was 6.75 h. When a new dialysis line was inserted on four occasions, it took a mean 10.75 h to restart therapy.
Characteristics of haemodialysis sessions and control of urea concentration and daily fluid balance after commencement of treatment for individual patients.
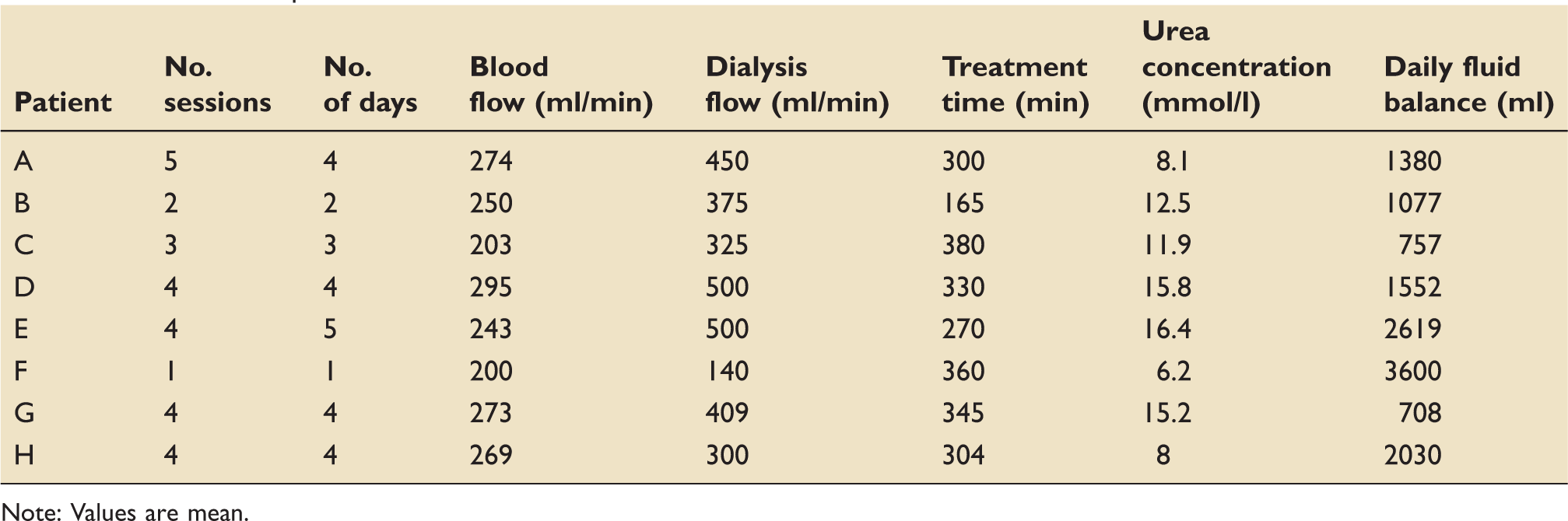
Note: Values are mean.
The primary documented access sites used for insertion of dialysis lines for all modes of RRT were as follows: 19 (36%) femoral, 17 (33%) LIJ, 12 (23%) RIJ and four (8%) subclavian veins. Documentation of replacement for two LIJ lines was into the RIJ vein and a femoral vein. Heparin and prostacyclin were used for anticoagulation. Heparin was used for 69 (71%), 27 (82%) and 12 (44%) sessions of CVVH, CVVHDF and IHD, respectively. No anticoagulation was used in 22 (23%), six (18%) and 15 (56%) sessions of CVVH, CVVHDF and IHD, respectively. Prostacyclin was used during six (6%) sessions of CVVH.
Discussion
This national prospective audit describes the conduct of management of RRT in relation to both prescription and delivery of RRT. Because of the observational nature of this study no formal conclusions can be drawn; however, the study does provide an insight into prescribing practices and highlights RRT dose delivered across a spectrum of general intensive care units.
Our study shows that RRT dose is prescribed at high intensity for all modalities. Despite significant interruptions, the delivered dose of CRRT in real-life compares favourably with large multi-centre studies performed in a research setting. Filter downtime is predominantly the result of either filter clotting or poor access. Transfer out of the unit or requirement for a new vascular access line significantly prolongs downtime. Overall, the delivered dose of CRRT achieved is higher than evidence suggests is necessary. This may have implications with regard to cost and nursing workload. We found that the dose of IHD is not routinely prescribed despite guidelines suggesting that this should be the case. 16 The intensity of IHD as defined by frequency is high. Measured mean urea concentrations after commencement of treatment suggest that the dose delivery compares favourably with previous studies.5,9
In 2000, Ronco published the first major study evaluating dose of RRT in relation to outcome in a single-centre study. 4 Patients who were assigned to receive CVVH with effluent volumes of 20 ml kg−1 h−1 and post-filter lactate based buffer replacement had significantly higher mortality when compared with patients receiving CVVH receiving doses of 35 or 45 ml kg−1 h−1. However, more recently, two large multi-centre randomised, controlled trials concluded that there is no mortality benefit to increasing intensity of RRT10,11 which was confirmed by a large prospective multi-centre observational study. 15
There are several possible reasons for the high prescribed dose of CRRT found in our study. First, current knowledge may not be contemporaneous and thus, prescription by a majority is still aiming for the targets set by Ronco. Second, prescribers may fear inadequate RRT as a result of uncompensated circuit downtime. In addition, higher targets may be set in order to compensate for pre-dilution or because of concerns that evidence for CVVHDF is not transferable to CVVH.
Studies performed prior to the Acute Renal Failure Trial Network (ATN) study and Randomized Evaluation of Normal versus Augmented Level (RENAL) Replacement Therapy study revealed uncertainty amongst intensivists about dose prescription 12 and that prescription practices were inconsistent 13 and without attention to patient’s body weight. 14
It is established that delivered dose does not achieve that targeted ranging from 63% of that prescribed in a single-centre retrospective review 17 to greater than 83% in more recent RCTs.8,10,11 Analysis of circuit downtime has revealed that the median period of inactivity varies from 3 h to over 5 h per day.18,19 The most common causes for interruption being clotting, need for radiological procedures, time spent in surgery and requirement for catheter replacement 19 which concurs with our study. It is legitimate to compensate for this since authors have argued that doses lower than 20 ml kg−1 h−1 could still be associated with worse outcomes; 20 however, this observational study encompassing non-self-selecting-units shows that delivered doses of CRRT are comparable with that achieved in the research environment and thus these fears are unfounded.
In our study, a significant proportion of replacement fluids were delivered pre-filter: 54% and 38.8% for CVVH and CVVHDF, respectively. It is established that pre-filter administration of replacement fluid will decrease clearance by about 15–20%; 21 however, it should be noted that evidence from the ATN 10 study relates to the predominant use of pre-dilution replacement, whilst another study has shown longer filter life with comparable control of urea and creatinine levels using pre-dilution replacement. 22 Thus, the available evidence would suggest the lower doses achieved in larger studies would remain safe irrespective of variations in proportion of replacement fluids. In terms of mode of CRRT, Ricci et al. 23 have shown no difference in solute clearances using either convection or diffusion such that effluent rates comparing CVVHDF and CVVH can be considered equitable.
For CRRT, patient’s weight is of importance when calculating intensity of RRT. Indeed, the patients with the lowest ‘dose’ of CRRT in the DO-RE-MI 15 study were approximately 33% heavier than those with the highest dose. Our study is of further interest, therefore, in that when adjusted body weight (ideal body weight +0.4 (actual weight − ideal body weight) is calculated for 26 CVVH and 10 CVVHDF patients where details of patient height were available, the median weights were 73.5 kg (IQR: 66–79.6) and 73 kg (58.1–76.3), respectively, representing delivered intensities of 32.6 ml kg−1 h−1 (9.9 SD) and 43.3 ml kg−1 h−1 (8.6 SD). This perhaps gives further credence to the conclusion that prescription of RRT intensity can safely be reduced.
This study confirmed that most causes for interruptions to therapy are as a result of filter clotting and poor access. Heparin was the primary anticoagulant used though recent international guidelines advocate the use of regional citrate anticoagulation 16 based on evidence for improved filter longevity and potentially reduced mortality.24,25 Regarding access, nearly 40% of dialysis lines were positioned in either the LIJ vein or a subclavian vein. The Cathedia study found lower rates of dysfunction in catheters placed in the RIJ, and femoral veins compared with the LIJ site. 26 Attention to both anticoagulation and line site may thus reduce interruptions.
It is recommended that dose of IHD be prescribed and delivery assessed as a manifestation of Kt/V 16 ; however, the standard practice in all the three units providing this mode of RRT in Scotland is not to calculate this. This is consistent with the aforementioned studies on IHD.5,9 There are several plausible arguments for not calculating Kt/V which include urea kinetics in the critically ill, 27 the fact that calculated Kt/V may be equivalent despite improved solute removal when dialysis sessions are lengthened 28 and that urea may not be the most important toxin measured. 29 Because of these issues and the fact that this study was observational, Kt/V was not calculated, though measured mean urea concentrations taken after commencement of treatment suggest that dose delivery compares favourably with published studies.5,9 There may be potential to reduce frequency of dialysis sessions in Scottish units though our study does not provide adequate evidence to support this. Fear of excessive positive fluid balance may drive the desire to continue high-frequency dialysis sessions in Scottish critical care units. One of the many criticisms of the study by Schiffl et al. 5 was that significant differences in fluid removal may have impacted outcome. Indeed, the Program to Improve Care in Acute Renal Disease study found that survival at dialysis cessation is proportional to the degree of fluid accumulation. 30
In a post hoc analysis of the BEST Kidney Study, it was estimated that reducing effluent flow rates would lead to significant cost savings. 31 Based on the results of our study, an approximate cost saving of £20.09 per day might be expected for patients managed by CVVH, and £63.21 per day for patients managed with CVVHDF if effluent flow rates of CRRT of 22.5 ml kg−1 h−1 were achieved.
The major strength of our study is that it was performed prospectively across an entire nation’s healthcare system. Furthermore, it informs of practice in all units providing RRT in comparison to other studies in which units with an interest in RRT may have self-selected to participate. There are potential weaknesses. It is difficult to extrapolate management across a year from relatively small numbers followed over a short time frame. Though we have calculated filter ‘downtime’ we were unable to assess filter lifespan due to the short follow-up period of 96 h. There was awareness of the intention to audit aspects of RRT, and thus, there was potential for bias though blinding of the timing of the study should have minimised this risk. Weaknesses specific to RRT practices relate to the data collected. Our audit did not describe the use of protocols locally, nor adherence to such protocols. With small numbers, it is difficult to ascertain whether or not higher targets were set for individual circumstances such as septic shock, profound hyperkalaemia or acidaemia which may have inadvertently skewed the results relating to prescription. This has implications for those wishing to audit their own unit’s performances. Last, though the large randomised controlled trials did not reveal benefit of higher intensity RRT, data cannot be extrapolated to individual patients and prescription must be individualised for each patient and may need to be adjusted to achieve adequate metabolic, electrolyte and fluid homeostasis. 32
In conclusion, the prescription and delivery of RRT within Scottish critical care units are routinely performed at an intensity that is higher than necessary. The reasons for this cannot be determined from an observational audit. Avoidance of excessive dose could provide important cost savings.
Footnotes
Acknowledgements
The authors thank Robert Docking, David Griffith and Laura Strachan on the Scottish Intensive Care Society Trainees Committee. They also thank John Allan, Pauline Austin, Yvonne Bramma, Andrew Clarkin and Ally Gibson who acted as ‘linkmen’ and Ahmad Abu-Arafeh, Tom Anderson, Ryan Campbell, Arlene Connelly, Andrew Conway Morris, Chris Farley, Catriona Felderhof, Roger Foy, Tessa Housden, Adam Livingston, Katherine Livingstone, Scott McRitchie, Andrew Nath, George Oomen, Erin Owen-Choa, Alistair Partridge, Alyson Primrose, John Rae, Vinoth Sankar, Gemma Scotland, Kate Slade and Liane Tait who acted as local co-ordinators for data collection.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
