Abstract
Medication error is the commonest cause of medical error and the consequences can be grave. This integrative review was undertaken to critically appraise recent literature to further define prevalence, most frequently-implicated drugs and effects on patient morbidity and mortality in the critical care environment. Forty studies were compared revealing a markedly heterogeneous data set with significant variability in reported incidence. There is an important differentiation to be made between medication error (incidence 5.1–967 per 1000 patient days) and adverse drug event (incidence 1–96.5 per 1000 patient days) with significant ramifications for patient outcome and cost. The most commonly implicated drugs were cardiovascular, gastrointestinal, antimicrobial and hypoglycaemic agents. Beneficial interventions to reduce such errors include computerised prescribing, education and pharmacist input. The studies described provide insight into suboptimal management in the critical care environment and have implications for the development of specific improvement strategies and future training.
Introduction
Medical errors are increasingly high profile and the focus of both scientific investigation and potential litigation. Medical errors have been found to be the eighth commonest cause of death in the USA. 1 Apart from substantial morbidity and mortality, there are also consequences on clinical governance, resource distribution and a substantial economic burden.
Medication error (ME) is the commonest cause of medical error with previous estimates of incidence of 9.1/1000 patient days. 2 The consequences of ME can range from minor to catastrophic, and cases of ME which have caused patient harm and even death are well reported. As such, the topic has attracted much interest from patient safety groups. 3
Medication error is particularly pertinent in the critical care setting. Patients are vulnerable as a consequence of a reduced physiological reserve, reduced ability to metabolise drugs and alterations in pharmacodynamics. Polypharmacy is common along with prescriptions of drugs to which patients have not been previously exposed. The use of high risk substances and varied routes of administration is more prevalent and these factors occur in the setting of busy and highly pressurised environments.
Medication errors are often cited as being one of the most common sources of error within hospitals2,4,5 but the prevalence of such errors, the most frequently implicated drugs and the effects on patient morbidity and mortality have not been recently systematically reviewed. Previous studies have tended to focus on specific areas of error, e.g. parenteral drug use or prescriptions only,6–8 and investigated only those studies performed in the developed world. 9
Initial review of the literature showed that published studies are very heterogeneous in trial methodology, study population, setting and methods of data presentation. The primary data did not lend themselves to a systematic review with meta-analysis. An integrative review was therefore undertaken to critically appraise the recent literature to determine the prevalence of ME in the critical care environment, the drugs most commonly associated with ME and the consequences of ME.
Methods
An integrative review is a reviewing process summarising available literature to provide a more comprehensive understanding of a clinical problem. It is more structured and rigorous than a narrative review in situations where the primary data do not support the use of a systematic review and meta-analysis. Integrative reviews present the current understanding of a subject and contribute to theory development. They have gained favour in academic nursing journals and represent a process well suited to many areas of current intensive care medical practice.
The integrative review was structured to incorporate the five-step review framework described by Whittemore and Knafl
10
:
Problem identification Literature search Data evaluation Data extraction, synthesis and analysis Presentation of results
Problem identification
This has been described in the introduction. The aim of this integrative review is to answer the following questions:
What is the prevalence of ME in the setting of critical care? What are the drugs most commonly associated with ME in the setting of critical care? What are the consequences of ME in the setting of critical care?
Literature search
Studies published from 1995 to June 2015 were identified using Medline, CINAHL and Embase databases. The following search strategy was used: MeSH terms: Medication Errors, Medical Errors, Pharmaceutical Preparations, Critical Care, Intensive Care, and Intensive Care Units were used in conjunction with title and abstract searches of “medication error”, “medical error”, mistake, “adverse event”, drug, “level 2”, “level two”, “level 3”, “level three”, “critical care”, “intensive care”, “high dependency”, hdu, icu, and “intensive treatment unit”. The amalgamation of the three broad categories of error with drug with critical care gave the final results which were then limited to English Language, Humans and Adults.
A hand search was also performed of personal files and reference lists of relevant review articles. Further, ‘snowballing’ from the reference lists of all relevant full text studies identified from the electronic search was undertaken.
Data evaluation
Relevant studies were identified by title, abstract and finally full text.
Studies were included for review if the following predefined inclusion criteria were met:
Rates of ME reported ME investigated and reported in a critical care setting ME rates reported in isolation rather than in conjunction with other forms of medical error. Studies were excluded for review if the following predefined exclusion criteria were present: Studies outside the setting of critical care or studies combining data from critical care and non-critical care settings Studies of primarily paediatric populations Review articles, correspondence, abstracts, dissertations, and editorials Full text not available in English.
Abstracts were reviewed separately by two of the authors, and any discrepancy between the decision on whether to include in the final analysis was resolved by discussion. Complete articles were then analysed for type of study, incidence of medication error and type of error investigated, associated adverse events and most commonly implicated drugs.
Definitions
Medication error (ME): any mistake in the prescription, preparation or administration of a drug which does not necessarily cause harm. 11
Adverse drug event (ADE): a medication error where harm occurs. 11
Data extraction, synthesis and analysis
For each study data were extracted to define study characteristics. A qualitative synthesis of the primary data was then undertaken to determine the prevalence and consequences of ME and the most commonly implicated medications.
Presentation of results
Data are presented in narrative and tabular form.
Results
Summary of included studies.

P = prospective; CR = chart review; VR = voluntary reporting; O = observational; I = interventional; CCU = Coronary Critical Care Unit; MICU = Medical Intensive Care Unit; L-T = long-term. Italics = how the errors were scored. PS = physician scored; PhS = pharmacist scored; NS = nurse scored; VT = validated tool. #Study leads. #MDT = multidisciplinary team led; #MDT* = multidisciplinary team led not including physician involvement; #PH = pharmacist led. Others as stated.
The findings of our literature review can be summarised within the following domains: study methodology; units used to report medication error incidence; overall incidence of medication error; severity of medication error; and drugs implicated in medication errors.
Study methodology
Study methodology varied between studies. Data were gathered either prospectively or retrospectively or as part of an interventional study to identify medication error incidence prior to and following an intervention. Errors were identified using three main methodologies: chart review, voluntary reporting or direct observation.
Units used to report medication error incidence
The units used to report medication errors also varied between studies. The most common units used to report medication error incidence were medication errors per 1000 patient days. Other units used included medication errors per 100 admissions, percentage of patients experiencing one or more medication errors, percentage of medication error as a proportion of overall drug administration or medication errors per 100 medication orders.
Overall incidence of error
The incidence of ADE and ME varied widely between studies.
The incidence of ADEs ranged widely from 1 to 96.5 per 1000 patient days37,38 or 1.3 to 21.1 when reported as per 100 admissions.25,29
When data were reported in different units, one study reported the incidence of ADEs as 34.1% of patients 31 and another as 0.16 per 100 medication orders. 24
The incidence of ME ranged from 5.1 to 967 per 1000 patient days,22,44 or when reported as per 100 admissions – 0.88–780.24,26 Percentage of medication error as a proportion of overall drug administration ranged from 1.96 to 69.7%.17,39 One study reported the incidence in units per 100 orders – 10 per 100 orders. 24
Severity of medication error
Severities of ADEs were described in terms of cost, monitoring implications and patient harm. Extra cost per patient varied between $347 16 and $6647. 14 Increased monitoring was reported in only one study as being required after 23.5% of ME. 26 Serious harm (3.39 to 64.8% of ADEs)30,38 and fatal consequences (between 0.45 to 25% total ADEs)24,30 of ADEs were also described.
Drugs implicated in medication errors
Drugs implicated in medication errors (for example, 32% is the mean average for the five studies that documented percentage of their total error being related to cardiovascular drugs.).
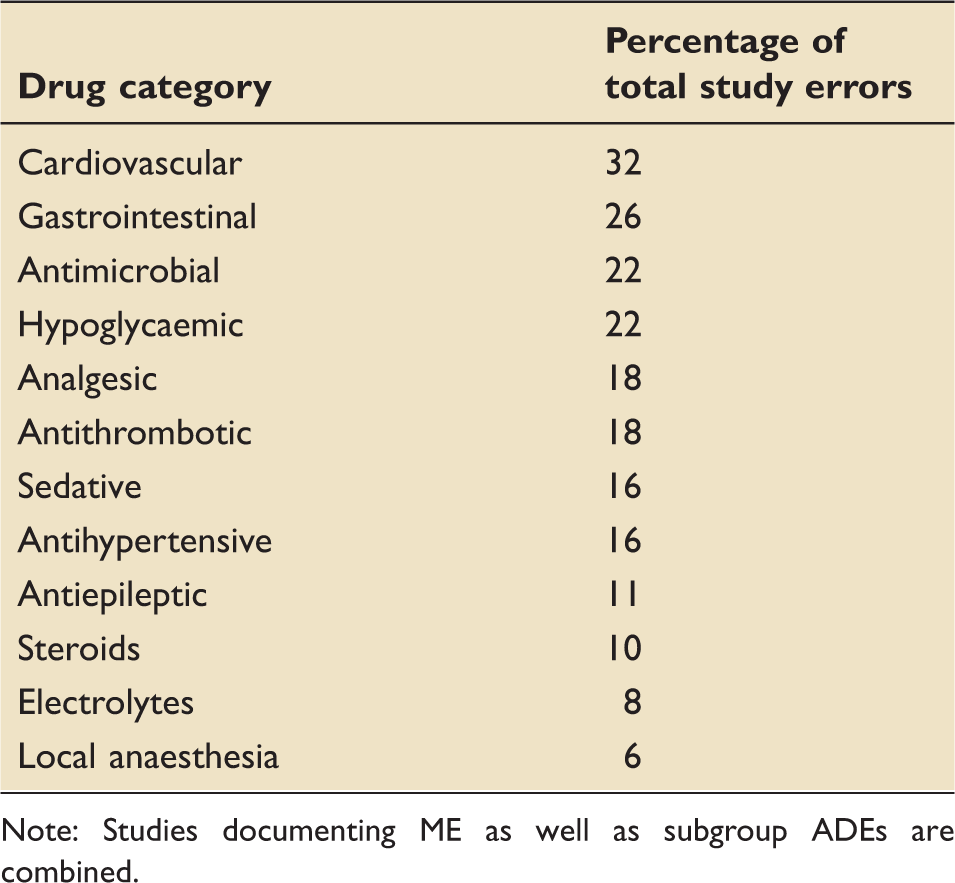
Note: Studies documenting ME as well as subgroup ADEs are combined.
Effect of different interventions
Effect of interventions on ME/ADE.
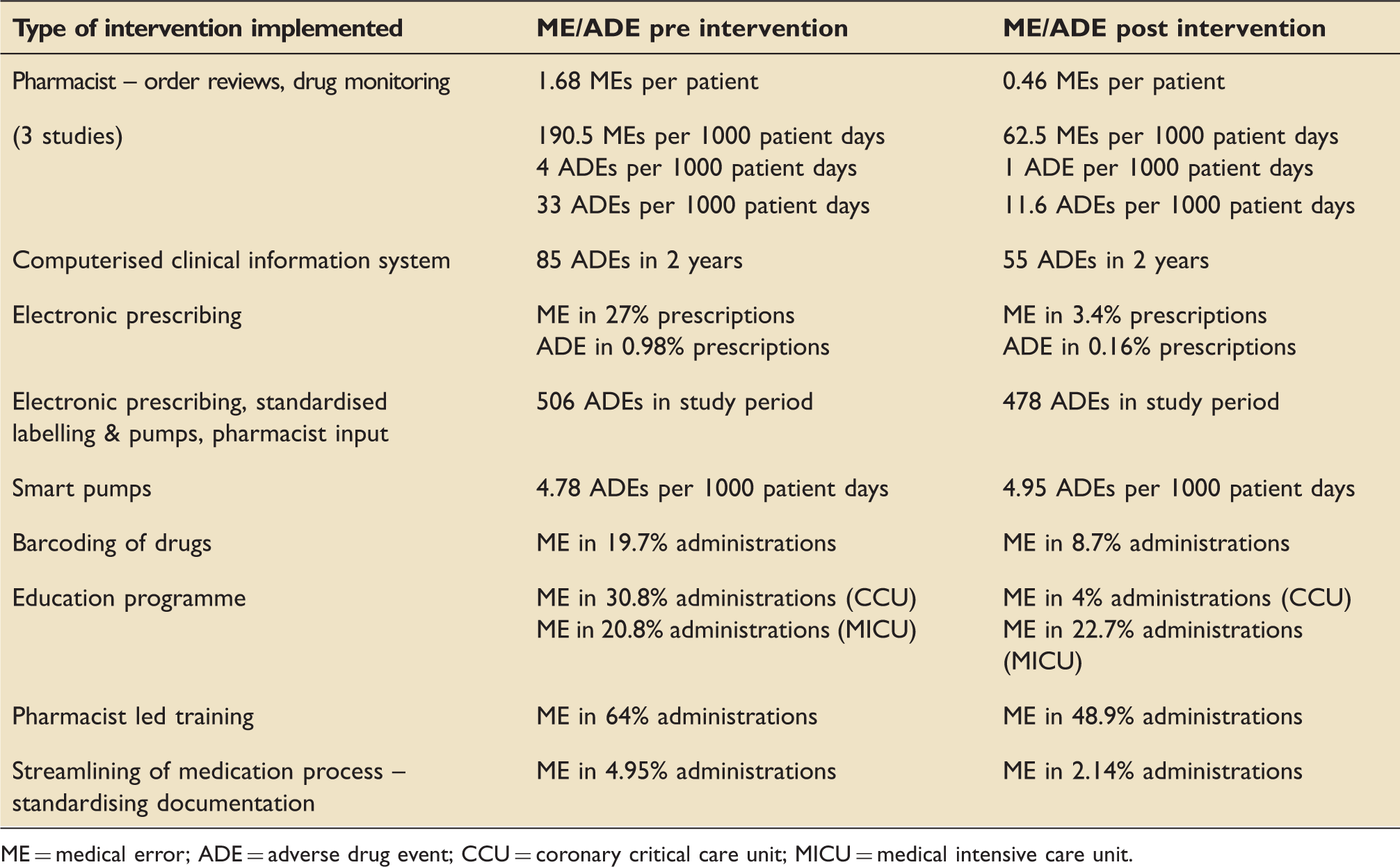
ME = medical error; ADE = adverse drug event; CCU = coronary critical care unit; MICU = medical intensive care unit.
Discussion
The main findings of this integrative review into drug errors in a critical care setting were: forty studies reported the incidence of medication error or adverse drug event in critical care; these errors are common in critical care; there are wide variations in estimates of incidence reflecting diverse methodology of studies; the consequences are often severe; and the drugs implicated are both specific to critical care but also include those which are commonly implicated in non-critical care settings.
Forty studies met the inclusion criteria. Not all studies specifically investigated the incidence of medication error as their primary outcome. Eleven of the studies were interventional studies investigating the impact of an intervention on the incidence of medication errors in critical care.
Medication errors are relatively common (1 to 96.5 per 1000 patient days) in a critical care setting. Polypharmacy is common along with prescriptions of drugs to which patients have not been previously exposed. Medication reconciliation may not be prioritised and the use of high risk substances and varied routes of administration is more prevalent. All these factors occur in the setting of busy and highly pressurised environments.
Perhaps the most interesting finding of this integrative review was the wide variation in nomenclature, quality and methods of data collection which clouds interpretation and comparison of studies, and does not allow for meta-analyses.
ME is increasingly accepted to mean any mistake in the prescription, preparation or administration of a drug which does not necessarily cause harm in contrast to ADE that refers to a medication error where harm occurs. 11 One of the sentinel studies in this field remains that of Bates et al. 2 who emphasised the importance of consistent definition and explains the term ADE as, for example, a patient with first degree heart block inappropriately receiving a beta blocker resulting in complete heart block requiring pacing. This is in contrast to a potential ADE synonymous with ME which refers to, for example, a patient with a documented allergy receiving said drug but not reacting. 2 Some studies investigate the so-called preventable ADEs as decided by an ICU pharmacist and physician, 37 whilst others define minor, intercepted and serious medication prescription errors (MPE). 7 In contrast, medication error was also described as any deviation in drug preparation and administration from the doctors’ prescription, hospital policies or manufacturer’s instructions such as diluted in saline as opposed to water. 8 The variations in definition are numerous and are not all listed here. Review of the two extremes of reported ME/potential ADE per 1000 patient days included one study that reported only 5.1 MEs per 1000 patient days despite documenting anything from delayed dose of a drug to accidental intravenous administration of planned intrapleural streptokinase. 44 Incident outcome was not separately differentiated between other non-drug-related incidents that occurred. At the other end of the spectrum with the most MEs of 967 per 1000 patient days was a study that included an extensive array of ‘discrepancies’ such as prescription omissions such as “route” not written next to a prescribed drug. 20 This may partly explain why their results were disproportionately higher than the other studies analysed. Clearly, the frequency of prescribing errors would be expected to be higher than that of actual error or harm to the patient.
The differing definitions and methodology are reflected in the wide range of estimates for the incidence of ME and ADE reported by this review. The variation in reported incidence of ADE from 1 to 96.5 per 1000 patient days may reflect that the former study 37 used a prospective interventional study looking at impact of pharmacist intervention into medical prescribing, whereas the latter 38 used chart review and incorporated adverse drug reactions (i.e. those that are unavoidable such as anaphylaxis) into their ADE figures. Furthermore, various studies examined a single aspect of drug administration6,14,18,19,28,40 such as parenteral medications only; solely missed doses, 17 or a prescriptions-only study design. 37
Eleven of the studies analysed incorporated pre- and post-intervention data (see Table 3). As far as conclusions can be drawn from the array of data, it seems that significant benefit arises from computerised prescribing, education and the welcome involvement of our pharmacist colleagues. The only intervention that appeared to cause increased patient risk was due to the so-called smart pumps, i.e. pumps that alert users when programmed to deliver duplicate infusions or doses out-with defined ranges. The proposed explanation was that the pumps led to greater identification of potential errors before harm resulted. 28
Chart review was the most commonly used method of identifying error with 20 of the 40 studies analysed using this method, employing unblinded nurse investigators, pharmacists and physicians. Fourteen used voluntary reporting methods by nurses and pharmacists which could lead to under-estimation of the incidence of ME. Thirteen used a direct observational method by independent pharmacy students, pharmacists, nurses or study physicians. Only one study used direct observation in conjunction with reporting in the per 1000 patient day format. 27 It might follow that using this method might result in a higher reported incidence of ADEs and MEs and apart from one other study 38 this was the case with 37.8 ADEs per 1000 patient days. Even using independent observational reporting the incidence of medication error still varied widely and was reported between 7.6 and 69.7% of administrations: 7.6% referred to potential opportunities for occurrence of error per patient while 69.7% referred to errors as a proportion of doses given.30,39
Severity of consequences resulting from an ADE was concerning, ranging from cost and increased monitoring implications, through serious harm (3.39–64.8% of ADEs) to fatal in between 0.45 and 25% total ADEs. Patients in critical care are particularly vulnerable as a consequence of a reduced physiological reserve, reduced ability to metabolise drugs and alterations in pharmacodynamics. New medications are introduced with potential for interaction and pre-existing medications may be omitted due to other clinical priorities.
Drugs commonly implicated in this review were antibiotics, sedatives, analgesics, anticoagulants, hypoglycaemics, antiepileptics, gastrointestinal and cardiovascular medications. These medications are implicated in medication errors in wider patient populations and have attracted interest from patient safety groups. 3 The reasons for this are beyond the scope of this review, but may be due to the frequent use of these medications or may be inherent to them or their administration process. The question remains whether certain drugs are inherently more prone to error, or indeed harm, or whether it is increased usage of particular drugs or their prescription that is more problematic.
This review covers recent literature on medication error and adverse drug events in a more diverse setting than previously published. Errors are common with potentially severe consequences, and research into this area requires standardised definitions and ideas regarding data collection to allow us to make more meaningful conclusions. A number of interventions have indicated improvement in service such as computer-aided drug ordering systems, training, standardised practices, and of vital importance the daily presence and input of critical care pharmacists as supported by the recent PROTECTED-UK study. 46 Other practices that may warrant investigation into whether they can reduce error include colour coding of drug packaging and labels, bar-coding, standardised storage, double checking prior to administration (widely performed but with little evidence base), techniques to minimise interruptions (e.g. “Do Not Disturb, Drug Round” tabards for nurses – perhaps more relevant in the ward environment), and checklists. We recommend a further large-scale national prospective observational trial using set definitions as per the Institute of Medicine and clarified by Bates2,11 to help us progress our knowledge and hence our safety mechanisms in this fundamental area. By understanding the depth of the problem in the intensive care setting, and realising where the risks are greatest, we can endeavour to improve our practice and future training.
Conclusion
Drug errors in critical care are common and have serious deleterious effects on morbidity and mortality. This review incorporated a specific integrative review framework that identified a particularly heterogeneous range of research methods and nomenclature in this area making data interpretation difficult. There is an important differentiation to be made between medication error (incidence 5.1–967 per 1000 patient days) and adverse drug event (incidence 1–96.5 per 1000 patient days) with significant ramifications for patient outcome and cost. The most commonly implicated drug groups include cardiovascular, gastrointestinal, antimicrobial, hypoglycaemic and analgesic agents. The studies described in this review provide insight into suboptimal areas of patient care in the critical care environment and have implications for the development of specific improvement strategies and future training.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
