Abstract
Background:
Acute kidney injury (AKI) is a serious complication in older patients with hip fractures. However, the prevalence and risk factors for both pre- and postoperative AKI remain poorly characterised. This study aimed to assess AKI prevalence before and after surgery in hip fracture patients and to identify associated clinical and surgical risk factors.
Methods:
This retrospective observational study was conducted on patients admitted with hip fractures throughout 2020. AKI was defined using KDIGO criteria. Data collected included patient demographics, biochemical markers, perioperative details, and mortality outcomes. Statistical analysis compared AKI and non-AKI groups and examined surgical associations.
Results:
A total of 190 patients were included. Preoperative AKI occurred in 2.1%, and postoperative AKI in 19.8%, with an overall AKI prevalence of 21.6%. Patients who developed AKI had significantly higher weight (p = 0.046) and longer hospital stays (p < 0.001). Postoperative AKI was significantly associated with being an inpatient at the time of fracture (p = 0.046). AKI development was also significantly associated with increased mortality (p = 0.001).
Conclusion:
One in five hip fracture patients developed AKI, which was linked to higher mortality. Key risk factors included higher body weight, inpatient fractures, and prolonged admission. Early identification and tailored perioperative care are vital for improving outcomes.
Introduction
Acute kidney injury (AKI) is a clinical syndrome characterised by a sudden decline in renal function, resulting from a wide range of aetiologies. The most common precipitants include hypovolaemia, often due to dehydration or sepsis. Patients with pre-existing intrinsic renal disease or obstructive uropathies are particularly vulnerable to developing AKI (Argyropoulos et al 2019). In the United Kingdom (UK), there are over 600,000 episodes of AKI annually, each costing the National Health Service (NHS) an estimated £5,000, representing a substantial economic burden (Mistry et al 2018, UK Renal Registry 2024). Beyond its immediate impact, AKI increases the risk of progression to chronic kidney disease (CKD), which in turn is associated with further morbidity and increased mortality (Horne et al 2023).
Despite its significant impact, the literature highlights that AKI continues to be under-recognised and inadequately managed in clinical practice (Graham-Brown et al 2024). To address this, standardised classification systems have been adopted and widely accepted by clinicians to aid in the timely identification and management of AKI. The Kidney Disease: Improving Global Outcomes (KDIGO) criteria, based on serum creatinine measurements and urine output, categorises AKI into three stages of severity (Kellum et al 2012). Using established criteria, studies indicate that AKI may be present in up to 60% of patients at the time of hospital admission, with approximately one-third of all inpatients developing AKI during their stay (Argyropoulos et al 2019). Risk factors for AKI appear consistent across age groups, with hypertension, chronic kidney disease (CKD), and diabetes being the most commonly implicated comorbidities (Argyropoulos et al 2019, Wu et al 2020). While these risk factors are prevalent in patients under 65 years of age, their incidence and impact are significantly greater in older populations (Stille et al 2022). As a result, individuals aged 65 and above face a substantially higher risk of developing AKI during hospitalisation (Chang-Panesso 2021, Stille et al 2022).
The incidence of hip fractures is projected to significantly increase in the coming decades as a consequence of an ageing population, with estimates indicating a rise of over 100% within the next 30 years (Harris et al 2024, Murphy et al 2024). As the majority of these fragility fractures require surgical intervention, they place a substantial financial burden on the NHS, with in-hospital costs estimated at approximately £15,000 per patient (Baji et al 2023). Due to the increasing prevalence of osteoporosis with age, most neck of femur (NOF) fractures occur in frail, older individuals (Rantalaiho et al 2019). This overlap with a population already at a high risk of developing AKI highlights the critical need for improved prevention, early recognition, and management strategies to reduce the complications and impact of AKI in the context of hip fractures.
AKI is a common and largely preventable complication in hip fracture patients, significantly worsening outcomes and increasing mortality risk up to three-fold (Agar et al 2022, Hong et al 2017). The literature highlights a postoperative AKI prevalence in this cohort of approximately 17%, with higher rates observed in older adults (Porter et al 2017, Zhou et al 2021). Existing research has also identified a range of risk factors, broadly classified into patient-related (e.g. age, comorbidities, frailty), surgical/perioperative (e.g. blood loss, surgery duration), and biochemical parameters (e.g. elevated CRP, low albumin, impaired renal function) (Li et al 2021, Zhou et al 2021). However, within the existing literature there remains limited characterisation of preoperative AKI prevalence and investigation into preoperative physiological and admission-related factors that may influence AKI (Haddad et al 2023, Lisitano et al 2023). These include variables such as admission route, length of stay, and preoperative biochemistry markers. During the immediate postoperative period, as recovery and critical care teams are the first to manage fluid balance and monitor renal markers postoperatively, understanding these risk factors is essential for tailoring early, renal protective care (Douville et al 2025). In addition, there is scope to evaluate intraoperative factors and to longitudinally assess established AKI biochemical markers (pre- and postoperatively) in relation to AKI. While the association between AKI and increased mortality is well established, characterising the nature of these associations within the present cohort remains equally important (Bui et al 2024).
The aim of this study was to investigate the preoperative and postoperative prevalence of AKI in patients with hip fractures and to evaluate clinical and surgical factors associated with AKI development. The hypothesis is that there is a significant prevalence of both preoperative and postoperative AKI in patients with hip fractures, and that specific clinical and perioperative factors are associated with AKI development.
Materials and methods
This was a retrospective observational cohort study that included all patients admitted to the Trauma unit of a district general hospital with a diagnosis of NOF fracture during the full calendar year of 2020. This study was exempt from Institutional Review Board (IRB)/Research Ethics Committee (REC) approval as it was conducted as an analysis of the patient population within the host institution’s Trauma and Orthopaedic department, without changing treatment or patient care from accepted standards for any of the patients involved and not aiming to achieve generalisable findings. The study was undertaken in compliance with local clinical governance standards.
Data for all patients were collected using the hospital’s electronic health record system (MediTech version 6, Medical Information Technology Inc., Westwood, Massachusetts, USA). This cohort of hip fracture patients was also evaluated in another study, investigating the mortality rates of hip fracture patients at various timepoints, however the emphasis of the data analysed herewith is entirely different.
AKI was defined according to the KDIGO staging system (Kellum et al 2012), which categorises acute deterioration in renal function into three stages. Stage 1 is defined by a 1.5–1.9 fold increase in serum creatinine levels with a urine output of <0.5ml/kg/hour over a 6 hour period. Stage 2 AKI is defined by a 2.0–2.9 fold increase in serum creatinine with a urine output of <0.5ml/kg/hour over 12 hours, Stage 3 AKI is defined by a > 3.0 fold increase in serum creatinine with a urine output of <0.3ml/kg/hour for 24 hours or anuria for 12 hours. However, if the urine output has not been calculated, AKI is generally staged based on serum creatinine levels alone (Hoste et al 2018). The data variables collected included all parameters and risk factors relevant to the development of AKI in NOF fracture patients who underwent surgical intervention (Li et al 2021, Zhou et al 2021). This included demographic details and pre-admission residence to identify if there is a relationship with preoperative AKI rates. A variety of serum blood test results were collected (pre- and postoperatively) that reflect renal function, inflammatory markers, and albumin levels (Kang et al 2020). Information related to the surgical procedure was also collated, including the type of hip fracture surgery performed, time from admission to surgery, and the grade of all involved clinicians (namely the operating surgeon and anaesthetist), as these are also significant factors. Diabetes and hypertension were not included as separate variables, as their severity and control vary between patients and glycated haemoglobin (HbA1c) or average blood pressure readings are not routinely available in the acute setting. To account for overall comorbidity burden, the anaesthetic assessment of American Society of Anaesthesiologists (ASA) Physical Status Classification System (Fitz-Henry 2011) was used to assess pre-existing patient comorbidities. Other relevant variables collected include nutritional assessment, total length of inpatient hospital stay, and the discharge location. Data were also collected for the overall 30-day, 90-day, and 1-year mortality rate.
Statistical analysis
All continuous data distributions were evaluated using plotted histograms with fitted curve lines, box-plots, normal Q-Q plots and the Kolmogorov-Smirnov test. All serum blood test data (continuous variables) displayed a skewed distribution, and the appropriate non-parametric statistical tests were used in their analyses. The Mann–Whitney U test was used for between-group statistical analyses and the Wilcoxon Signed Rank test was used for within-group analyses. The Chi-square test for independence was used for the statistical analyses of categorical variables. The level of statistical significance was set at p < 0.05. Statistical analysis was performed using SPSS for Windows version 26.0 (IBM Corp., Armonk, New York).
Results
This study included a total of 190 patients, comprising patients who were admitted to the Trauma unit with a diagnosis of NOF fracture. Table 1 displays the patient demographics. The majority of patients were female (n = 154), with a mean age of 81.9 years and a mean BMI of 23.6. ASA classification showed that 66.3% (n = 126) of patients were classed as ASA grade 3 and above. Three patients were deemed medically unfit for surgery, and so were managed conservatively (i.e. analgesia, bed rest, physiotherapy, and optimisation of medical comorbidities). Postoperative data analysis was undertaken for the 187 patients who underwent hip fracture surgery.
Patient demographics
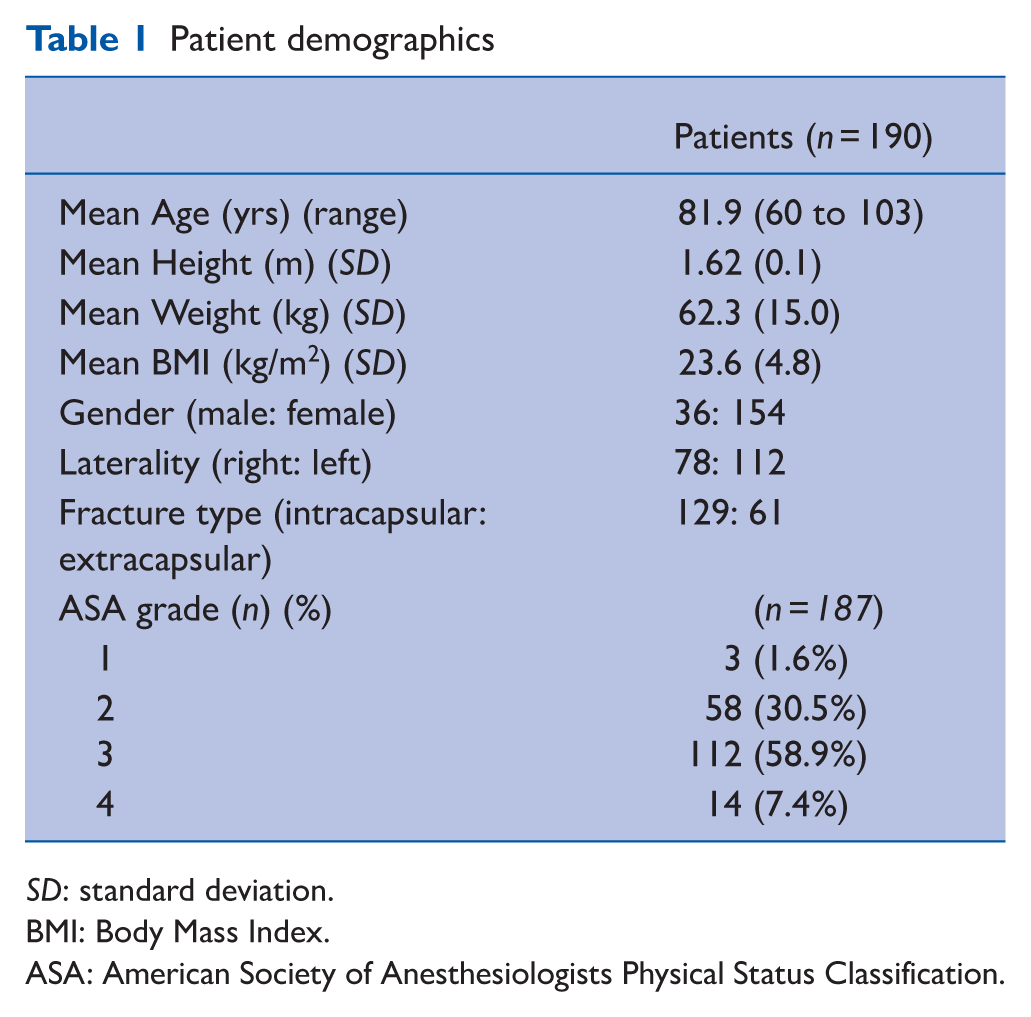
SD: standard deviation.
BMI: Body Mass Index.
ASA: American Society of Anesthesiologists Physical Status Classification.
Table 2 presents the AKI data. The prevalence of AKI (including all KDIGO AKI stages) preoperatively was 2.1% (4/190 patients), 19.8% (37/187 patients) postoperatively and the overall prevalence of AKI in this study cohort was 21.6% (41/190 patients).
Prevalence of acute kidney injury
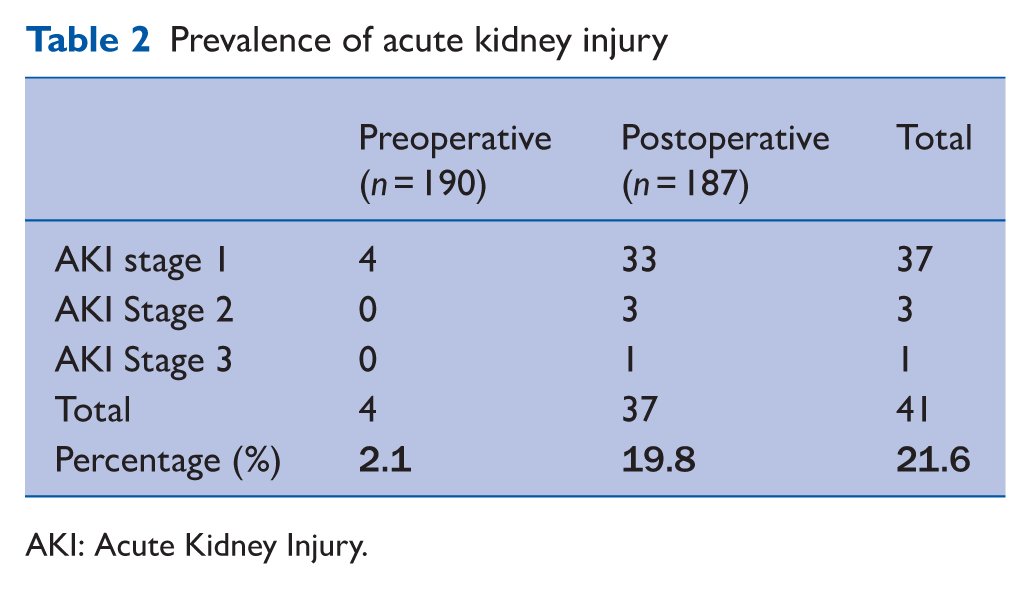
AKI: Acute Kidney Injury.
Table 3 displays blood test data pre- and postoperatively for both the AKI subgroup and entire cohort. There was a significant deterioration in renal function among patients who developed AKI, with increased serum urea (p < 0.001), creatinine (p < 0.001), potassium (p = 0.032), and CRP (p < 0.001), and decreased eGFR (p < 0.001) and albumin levels (p < 0.001). Across the whole cohort, postoperative CRP levels increased significantly (p < 0.001), and albumin levels decreased (p < 0.001).
Longitudinal blood test analysis of AKI subgroup (n = 37) and entire cohort (n = 190)
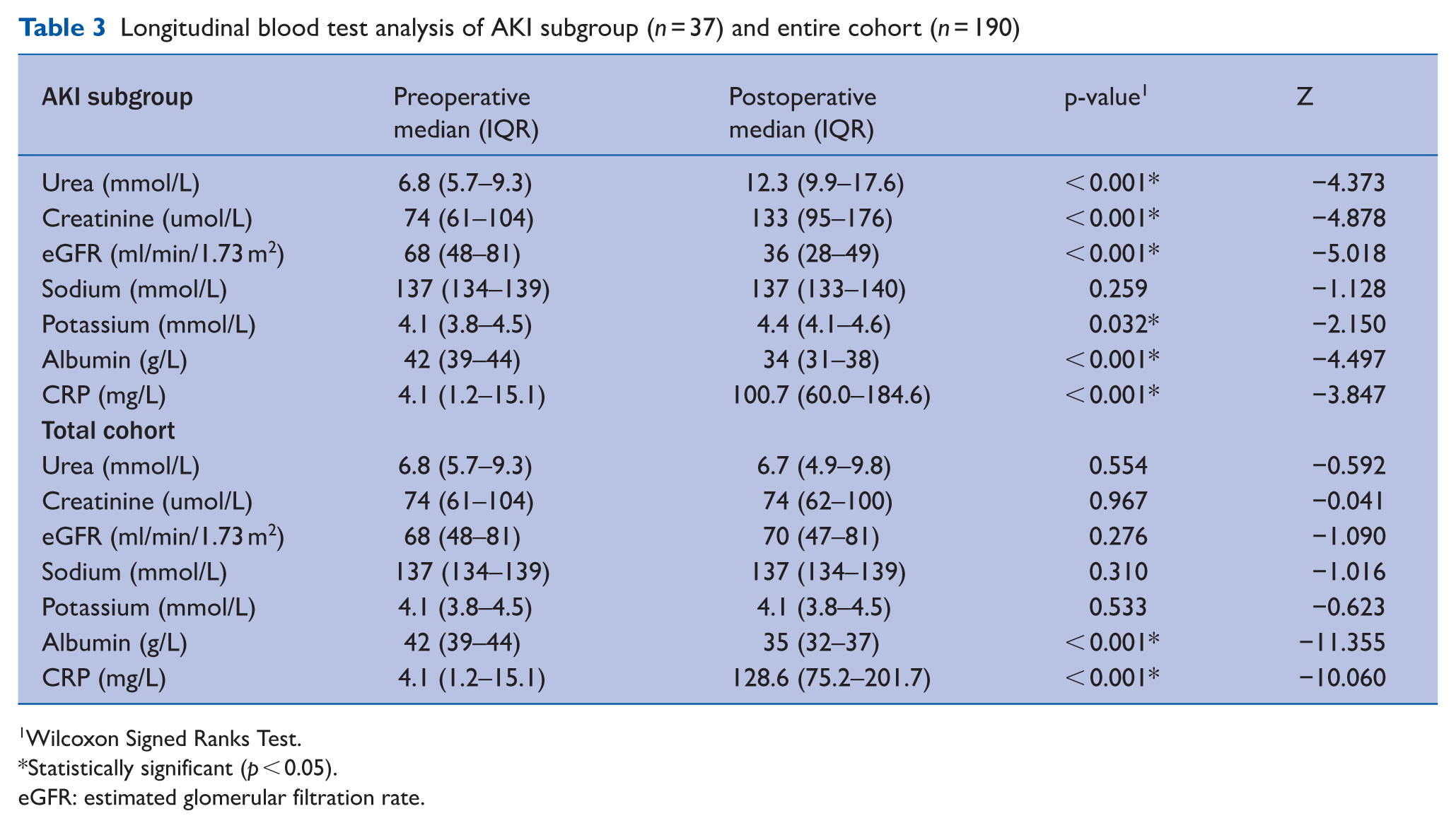
Wilcoxon Signed Ranks Test.
Statistically significant (p < 0.05).
eGFR: estimated glomerular filtration rate.
Table 4 displays patient demographics and hospital-related factors for those who did and did not develop AKI. Patients who developed AKI had a significantly higher body weight (p = 0.046) and a longer total hospital stay (p < 0.001) compared to those who did not. Time from admission to surgery did not significantly differ between patients who did and did not develop postoperative AKI (p = 0.477).
Patient factors contributing to development of postoperative AKI
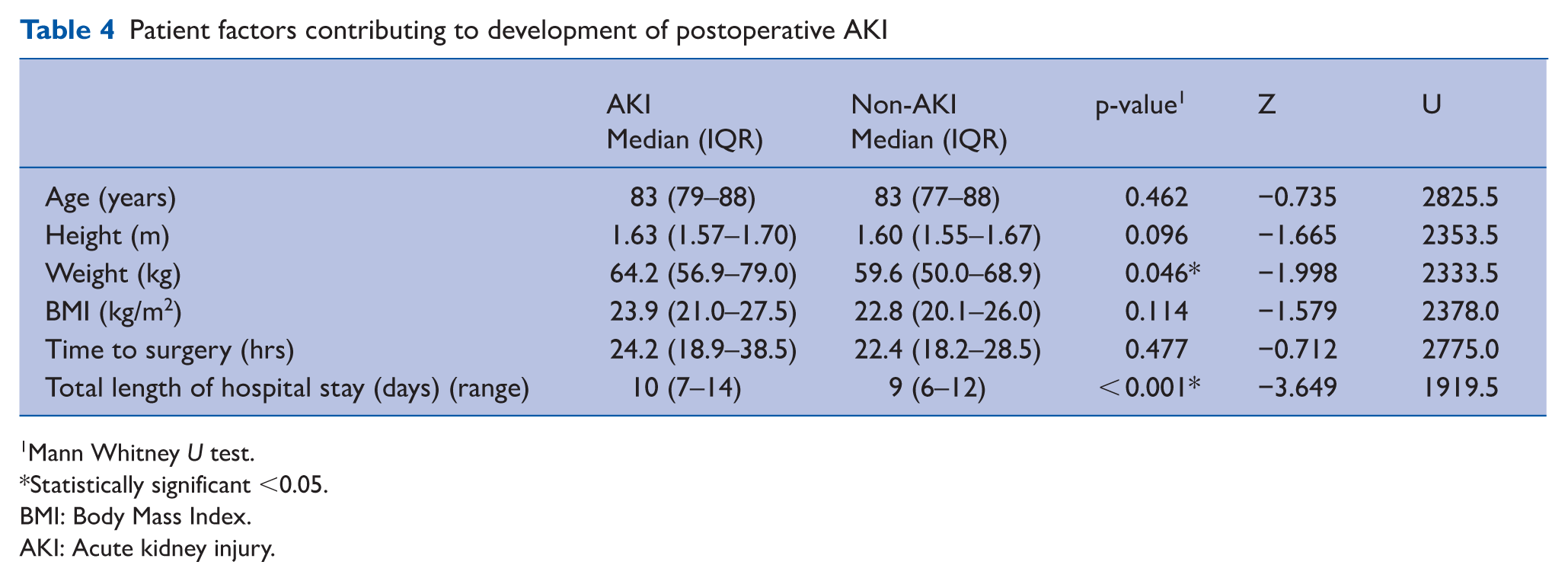
Mann Whitney U test.
Statistically significant <0.05.
BMI: Body Mass Index.
AKI: Acute kidney injury.
Table 5 presents the surgical and perioperative factors associated with the development of postoperative AKI. Admission route was significantly associated with postoperative AKI (p = 0.046), with 50% (4/8) of patients who sustained a hip fracture while already admitted as inpatients developing AKI. No significant associations were observed between ASA grade (p = 0.128), mode of anaesthesia (p = 0.764), or type of surgical procedure (p = 0.205) and the development of postoperative AKI.
Surgical factors contributing to development of postoperative AKI
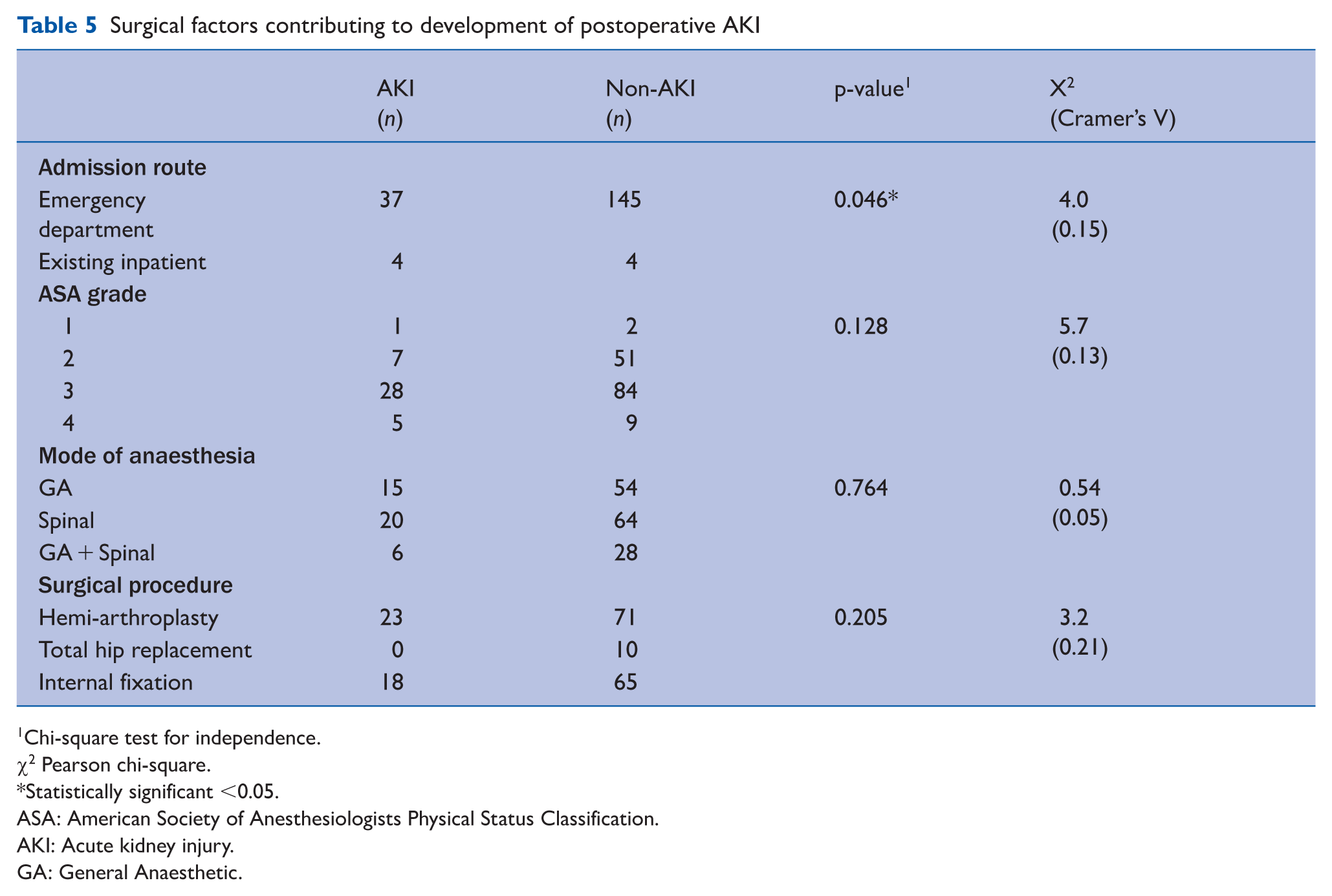
Chi-square test for independence.
χ2 Pearson chi-square.
Statistically significant <0.05.
ASA: American Society of Anesthesiologists Physical Status Classification.
AKI: Acute kidney injury.
GA: General Anaesthetic.
Table 6 presents the association between AKI and mortality outcomes. Patients who developed AKI at any point during admission had significantly higher mortality rates compared to those who did not (p = 0.001). Similarly, the presence of postoperative AKI was also significantly associated with increased mortality (p = 0.002). Of the three patients managed nonoperatively, none developed AKI and two died. Among the four patients with preoperative AKI, three died (75% mortality), and one of these patients continued to have AKI postoperatively.
Effect of AKI on mortality
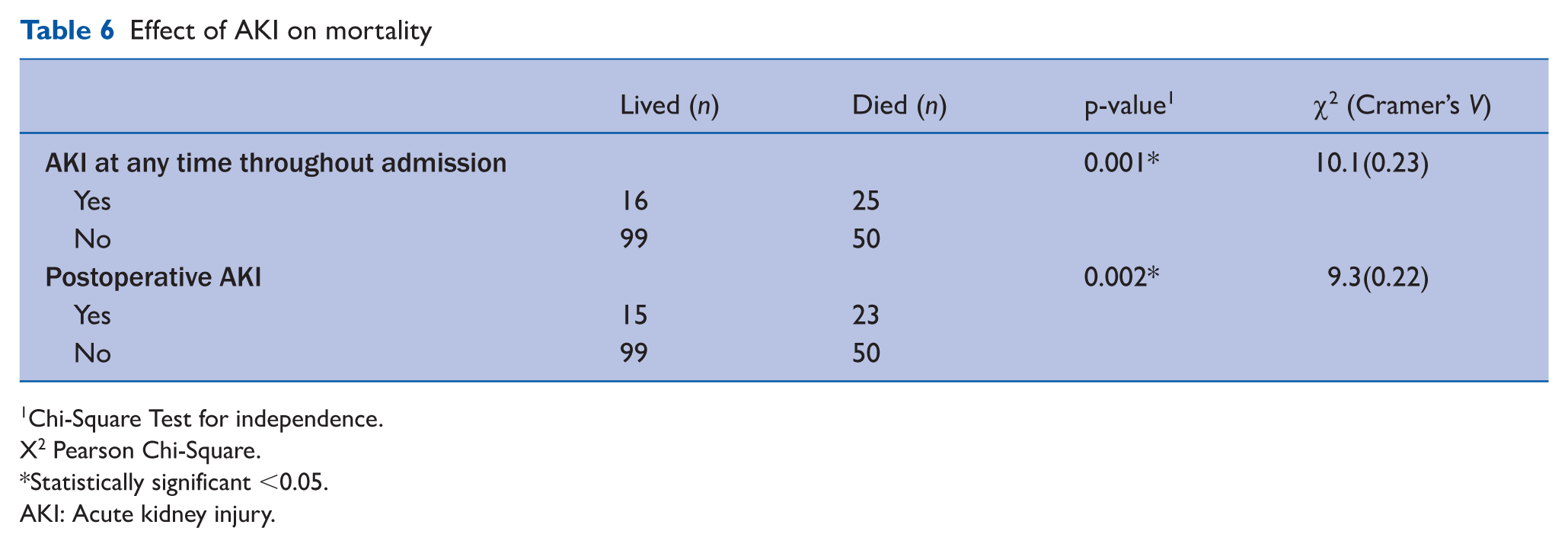
Chi-Square Test for independence.
X2 Pearson Chi-Square.
Statistically significant <0.05.
AKI: Acute kidney injury.
The overall 1-year postoperative mortality rate was 11.8% for the entire study cohort. After excluding patients with preoperative AKI (n = 4), nonoperative management (n = 3), and the one patient with both pre- and postoperative AKI (n = 1), the mortality rate among patients without AKI was 29.8% (48/161). In comparison, among those who developed postoperative AKI (n = 37), 22 died, yielding a mortality rate of 59.5%. These differences in mortality are illustrated in Figure 1. AKI was recognised and treated promptly in 89.7% of cases. Intravenous fluid administration was the most common intervention used to manage AKI.
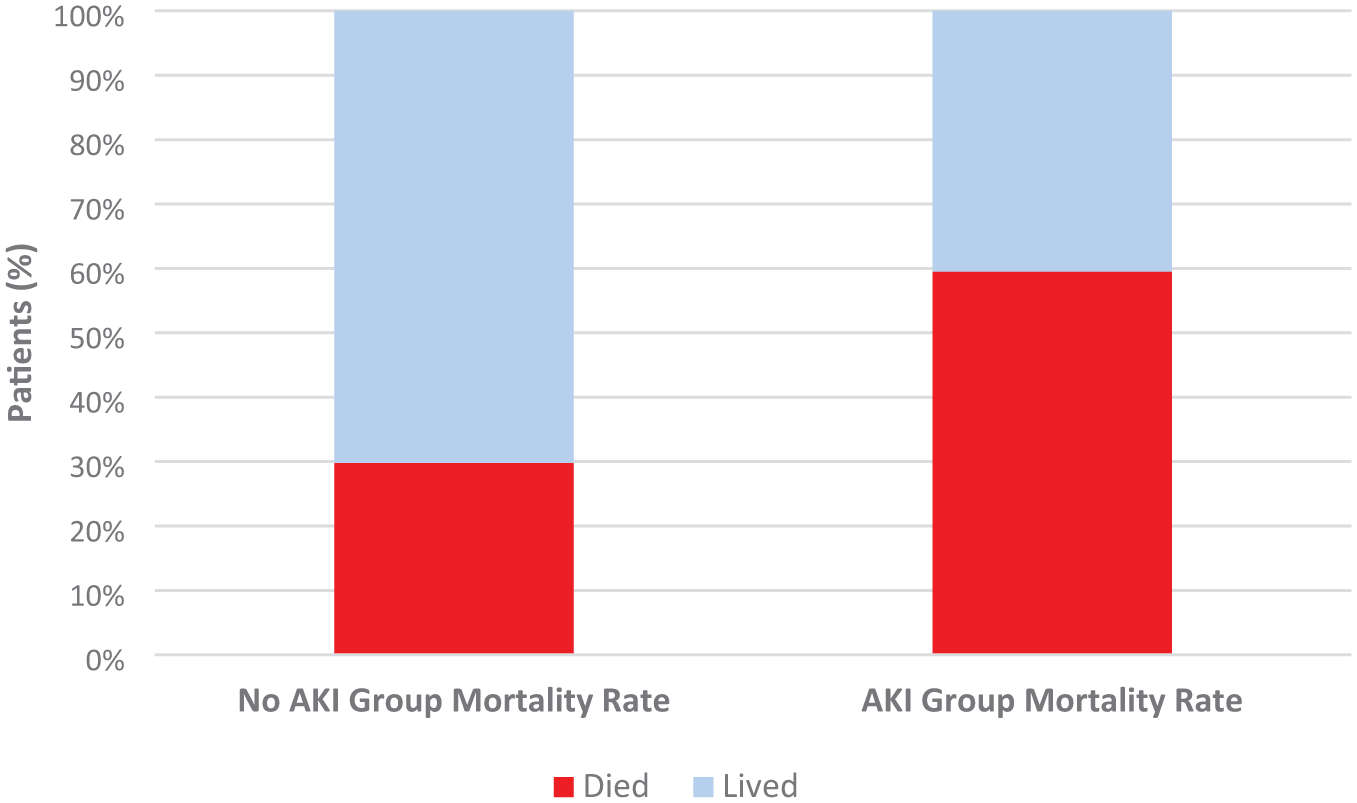
Baseline non-AKI hip fracture mortality rate vs AKI mortality rate
Discussion
The main findings of this study were that the overall AKI prevalence in patients with hip fractures in this cohort was 21.6%, with preoperative and postoperative rates of 2.1% and 19.8% respectively. Notably, higher body weight, prolonged length of hospital stay and sustaining a hip fracture while an existing inpatient on a medical ward (as opposed to a new patient emergency admission) were significant factors in the development of postoperative AKI. The 1 year mortality rate for the entire study cohort was 11.8% with a baseline mortality rate of 29.8% among non-AKI hip fracture patients, increasing to 59.5% in those with AKI.
The present study found that 21.6% of hip fracture patients developed AKI, a rate consistent with the existing literature (Li et al 2021, Zhou et al 2021). A meta-analysis by Li et al (2021) reported a pooled postoperative AKI prevalence of 17%, with significant heterogeneity between studies (I² = 95%), while Zhou et al (2021) identified individual study rates ranging from 4.5% to 28.4%. Overall, the present study’s findings may reflect a more frail and comorbid population in the present study contributing to a higher AKI prevalence, with 66.3% of patients classified ASA grade 3 or above, and a mean age of 81.9 years (Porter et al 2017). These findings also demonstrate that the systemic physiological effects of surgical intervention within this frail and older cohort of patients, reflected by a significant postoperative rise in CRP (p < 0.001), may have been a major contributor to the development of AKI following hip fracture surgery. The 1-year mortality rate was 59.5% among patients who developed postoperative AKI, compared to 29.8% in those without, reinforcing AKI as a strong predictor of poor outcomes. This aligns with the findings of Rantalaiho et al (2019), who reported a 22.3% mortality rate increase in AKI patients. These results highlight the importance of early renal assessment on admission, enabling timely interventions such as fluid optimisation and avoidance of nephrotoxic medications, particularly in patients with established risk factors.
The present study found that patients who developed postoperative AKI had a significantly higher body weight (p = 0.046), but not a significantly higher BMI (23.9 in AKI vs 22.8 kg/m², p = 0.114). The relationship between BMI and AKI risk has been well documented in hip fracture cohorts (Pedersen et al 2017). Prior research often focuses on BMI categories, with obesity consistently linked to increased risk of AKI (Pedersen et al 2017). Although the difference in BMI in the present cohort was not statistically significant, even small shifts within weight categories may have clinical relevance. Increased weight is associated with heightened systemic inflammation, oxidative stress, and impaired fluid regulation, all of which may contribute to renal hypoperfusion and increased susceptibility to AKI (Shi et al 2020). In addition, perioperative fluid management may be more challenging in patients with higher body weight, further compounding this risk.
Length of stay was significantly longer in patients with postoperative AKI compared to those without (p < 0.001). This finding is consistent with previous studies reporting that extended hospitalisation increases the risk of AKI in surgical and older populations (Braüner Christensen et al 2020, Porter et al 2017). The nature of this relationship may be bidirectional. Prolonged admission may expose patients to hospital-acquired complications, nephrotoxic medications, and haemodynamic instability, all of which contribute to AKI. Conversely, the development of AKI itself often leads to delayed recovery, medical complications, and extended inpatient care. This creates a reinforcing cycle in which AKI prolongs hospital stay, and longer hospitalisation further increases the risk of AKI. Breaking this cycle is therefore crucial to improving outcomes. During the COVID-19 pandemic (overlapping with data collection period) NHS emergency funding prioritised accelerated discharge to community rehabilitation centres, nursing homes, or home care. This likely contributed to the shorter inpatient stays observed in the present cohort (Table 4) relative to typical pre-pandemic hip fracture admissions, which are often prolonged in frail, older patients. As a result, such strategies may have helped further reduce overall AKI prevalence in the cohort, highlighting the value of coordinated discharge planning in reducing AKI risk and improving outcomes in hip fracture patients.
To our knowledge, no previous study of AKI in hip fracture patients has examined the association between admission route and AKI risk. In the study cohort, sustaining a hip fracture following an inpatient fall was significantly associated with the development of postoperative AKI (p = 0.046). This novel finding may reflect greater baseline frailty of inpatients, along with the cumulative impact of nosocomial factors. These include prolonged immobility, increased exposure to nephrotoxic agents (e.g. certain intravenous antibiotics), and higher rates of hospital-acquired infections, all of which may collectively increase renal vulnerability. This finding identifies a high-risk subgroup that may benefit from targeted AKI prevention strategies. Future multicentre studies should aim to validate this association between AKI and admission route, and assess whether tailored interventions such as early nephrology input, fluid management protocols, and nephrotoxin avoidance can reduce AKI prevalence in this subgroup.
By contrast, several perioperative factors were not significantly associated with the development of postoperative AKI in the present cohort. These included ASA grade (p = 0.128), mode of anaesthesia (p = 0.764), and surgical procedure type (p = 0.205). In addition, there was no significant difference in time to surgery between AKI and non-AKI groups (median: 24.2 vs. 22.4 hours, p = 0.477). These findings contrast with existing literature. Pan et al (2023) reported ASA grade as a significant independent predictor of AKI, likely reflecting the impact of greater comorbidity burden. Conflicting evidence also exists around anaesthetic approach, where Chen et al (2023) found general anaesthesia to increase AKI risk, while Haddad et al (2023) identified spinal anaesthesia as a significant risk factor, suggesting that the relationship may be context- or population-dependent. Similarly, procedure type has been implicated, with Zhou et al (2021) reporting higher AKI incidence following arthroplasty procedures. In terms of timing, median time to surgery for both groups was within the national 36 hour target, which may explain the absence of a significant association (Aqil et al 2016). However, a previous study conducted by Berar Yanay et al (2021) found that delays beyond 48 hours are more strongly associated with increased AKI risk, suggesting that the benefits of timely surgery may be most apparent when recommended timing targets are exceeded. The lack of association in the present study may reflect consistent adherence to national perioperative care standards, which likely reduced variability in clinical management across patients and may have limited the ability to detect underlying associations.
The findings of the present study highlight the importance of early recognition and proactive management of AKI in patients presenting with hip fractures. Integrating key risk factors into standard hip fracture pathways may support earlier intervention and improved outcomes. Routine comparison of admission blood results with pre-admission baselines may help detect early renal decline. Close monitoring of biochemical trends and clinical signs, particularly in the postoperative period, is crucial in this frail population. Future research should further evaluate the role of nephrotoxic medications, nutritional status, and dynamic renal monitoring to guide more personalised and preventive care strategies. Many Trauma units use a generic hip fracture proforma during the clerking and hospital admission pathway. Including the parameters highlighted in the present study within hip fracture protocols may enhance AKI detection and prevention in this high-risk cohort.
A limitation of this study is the retrospective design, which meant urine output was not consistently recorded and AKI diagnosis depended solely on serum creatinine changes, potentially leading to both under- and overestimation of the true AKI incidence and severity. Another limitation concerns potential perioperative factors which were not captured in the cohort including intraoperative blood pressure variability, fluid balance and nephrotoxic drug exposure prior to surgery, which may collectively influence AKI risk. Power analysis was not performed as all eligible patients were included in the study. Data collection took place during the COVID-19 global pandemic, and while time to surgery remained within the national target, pandemic-related disruptions to staffing, preoperative screening and postoperative rehabilitation may have impacted AKI outcomes and limit generalisability to non-pandemic practice.
Key strengths of this study include its comprehensive AKI assessment using the KDIGO criteria, which ensures standardised case definitions and staging. An inclusive, consecutive cohort of every eligible hip-fracture admission over a full year further strengthens the study by minimising selection bias. Detailed longitudinal analysis of clinical variables and AKI prevalence allowed for the assessment of the impact of surgery on renal function over time. The study also included a comprehensive set of variables, encompassing demographic, biochemical, perioperative, anaesthetic and outcome measures, which supported multifactorial exploration of factors influencing AKI.
Conclusion
In conclusion, this study found that 21.6% of patients with hip fractures developed AKI, with the majority of cases occurring postoperatively. Higher body weight, longer hospital stays and inpatient status at the time of hip fracture were identified as key risk factors in the development of AKI. The presence of AKI was linked to increased 1-year mortality, rising from 29.8% in patients without AKI to 59.5% in those affected. These findings demonstrate that AKI is a common and serious complication in this population, closely associated with adverse outcomes. Early identification through recognition of key risk factors during the immediate postoperative recovery period may be critical in improving outcomes in this older and vulnerable population. Future prospective studies should aim to validate these risk factors and develop targeted interventions to reduce AKI incidence in patients with hip fractures.
Footnotes
Author contributions
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This was a retrospective observational cohort study which did not require IRB/ethics committee approval.
Informed consent
Written consent was obtained from all participants for their surgical procedure.
