Abstract
Objective
To evaluate the efficacy of intravenous (IV) amino acids in preventing acute kidney injury (AKI) in patients undergoing cardiac surgery.
Methods
PubMed, EMBASE, and Cochrane Library databases were systematically searched for all relevant randomised controlled trials (RCTs) published from inception to July 25, 2024. A random effects model was used to pool the risk ratios (RR) for dichotomous outcomes, and the combined data was visually represented using forest plots.
Results
3 studies involving 3646 patients were included in the meta-analysis. IV amino acids did not significantly lower the overall AKI incidence compared with control measures. However, they significantly reduced the risk of Stage 1 AKI and Stage 3 AKI. No differences were observed between intervention and control for Stage 2 AKI or need for kidney replacement therapy.
Conclusion
IV amino acids may offer protective benefits against severe AKI stages in cardiac surgery patients. Further studies are warranted to validate our findings.
Introduction
Kidney injury is the initial stage of a potentially catastrophic cascade that can result in kidney failure. 1 Therefore, it is considered an independent risk factor for mortality in the general population undergoing surgery. 2 Prompt identification of early signs of kidney dysfunction, together with preventive strategies and kidney protection, plays a critical role in patient outcome. 3 This could potentially reduce mortality, shorten hospital stays, and decrease healthcare costs.4,5
Acute kidney injury (AKI) is a relatively frequent complication of cardiac surgery, and is associated with increased morbidity and mortality. The incidence of kidney impairment following cardiovascular surgery varies substantially between studies. For example, one study found that 37% of patients undergoing coronary surgery, 49% undergoing valve surgery, and 55% undergoing aortic surgery experienced this condition. 6 The risk increases when cardiopulmonary bypass (CPB) is used. In some studies, the prevalence of AKI following cardiac surgery with CPB is as high as 35%, with approximately 1.5% of patients requiring dialysis. 7
The pathophysiology of AKI is complex and influenced by several factors. In the perioperative context, one of the most common causes is prolonged hypoperfusion (pre-kidney AKI), which is associated with septic shock, cardiogenic shock, hypovolemia, and haemorrhage. Furthermore, the use of perioperative nephrotoxic medications and contrast medium can further decrease kidney function. 8
Measures to decrease or mitigate the risk of cardiac surgery-associated acute kidney injury (CSA-AKI) can significantly reduce morbidity and healthcare costs. Various perioperative pharmacological prevention strategies have been proposed. These include treatment with corticosteroids, albumin, erythropoietin, statins, N-acetylcysteine, sodium bicarbonate, intraoperative/postoperative dopamine, anaesthetics (volatile or IV), mannitol, furosemide, or fenoldopam.9–11 Research has been conducted to assess whether amino acids can prevent AKI, but a conclusive answer is still unavailable. One study involving critically ill patients who received either IV amino acids or standard care, found no significant difference in the duration of kidney impairment. 12 However, another study in patients undergoing valve surgery and coronary artery bypass grafting (CABG), found that amino acid supplementation may help reduce the duration of AKI and improve estimated glomerular filtration rate (eGFR). 13
Considering the high prevalence and strong association between AKI and poor outcomes, developing strategies for kidney protection is essential, especially for high-risk groups such as patients undergoing cardiac surgery. Therefore, we conducted a systematic review and meta-analysis to assess the evidence regarding the potential of IV amino acids to protect the kidneys of patients undergoing cardiac surgery.
Methods
PubMed, Cochrane Library, and EMBASE were systematically searched for relevant studies published from inception to July 25, 2024. Key words/terms in both AND and OR combinations included: kidney; acute kidney injury; amino acids; infusion; intravenous; cardiac surgical procedures; thoracic surgery; cardiothoracic surgery. In addition, the reference lists of all included studies were reviewed for potential additional publications, and we performed forward citation searching via the Web of Science to obtain other relevant studies. The systematic review was conducted according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) regulations. 14 Ethical committee approval was not required and the review was registered with PROSPERO (CRD42024565982).
All references retrieved by our search were imported to Endnote v20 and duplicates were removed. Four authors [AH. TA, BK, BN] independently screened the titles and abstracts of all studies and selected relevant studies. Any discrepancy or disagreement was resolved by another author [AA]. The study design was limited to randomized controlled trials (RCTs) and the intervention was IV amino acids. Eligible procedures for cardiac surgery included: CABG; valve surgeries; thoracic or thoracoabdominal aortic replacement; aortic replacement procedures (e.g., ascending aorta replacement); congenital defect repairs (e.g., atrial septal defect closure); pulmonary valve replacement (PVR); arrhythmia-related procedures (e.g., atrial fibrillation ablation). Procedures unrelated to the primary cardiac structure (e.g., catheter-based interventions without surgical repair) were excluded.
Two authors [AH, AM] independently extracted data into a pre-established data collection form in Microsoft Excel. Any disagreement was settled by a third author [AA]. The following items, when available, were extracted: study characteristics (i.e., first author, publication date, study setting, study design and sample size); patient characteristics (i.e., age, sex, body mass index, serum creatinine, left ventricular ejection fraction (LVEF) and type of surgery); post-allocation characteristics (i.e., dose of amino acids, duration of infusion, duration of cardiopulmonary bypass [CBP] and cross-clamp time); outcomes (i.e., AKI, Kidney Disease: Improving Global Outcomes [KDIGO] stage of AKI and use of kidney replacement therapy). Primary outcome of the meta-analysis was number of patients with AKI, assessed as a binary outcome (yes/no) and using the KDIGO definition of AKI. KDIGO defines AKI as any of the following: an increase in serum creatinine by ≥0.3 mg/dl (≥26.5 µmol/l) within 48 h; an increase in serum creatinine to ≥1.5 times baseline within the previous 7 days; urine volume ≤0.5 ml/kg/h for 6 h. 15 The secondary outcomes were number of patients with KDIGO stage 1, 2 or 3 and use of kidney replacement therapy.
Statistical analysis
The meta-analysis was performed using Review Manager (RevMan) [Computer program] Version 5.4.1. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration 2020. A P value <0.05 was considered to indicate statistical significance.
The level of evidence quality of each study was estimated according to the guidelines of Grading of Recommendations, Assessment, Development, and Evaluation (GRADE). 16 The GRADEpro GDT software, a tool specifically designed for implementing GRADE criteria was used, which facilitates summarizing evidence in a transparent and standardized manner. The software allows for evaluating critical aspects of the evidence, including risk of bias, inconsistency, indirectness, imprecision, and publication bias. By applying these criteria, we derived the certainty ratings ranging from high to moderate based on the strength and limitations of the evidence.
The Cochrane risk-of-bias tool for randomized trials (ROB 2.0) was used to assess the quality of each included RCT. 17 ROB 2.0 evaluates the quality of the RCT across five domains (i.e., randomization process; deviations from intended interventions; missing outcome data; measurement of the outcome; selection of the reported result). Based on answers, risk of bias was categorized as high, low, or some concern. The risk of bias in each study was independently assessed by two authors [AMM, IAS]. Any disagreement was settled by a third author [AA].
The DerSimonian and Laird random effects model was used to pool the risk ratios (RR) for dichotomous outcomes, with 95% confidence intervals (CIs), and the combined data was visually represented using forest plots. Cochran's Q test and Higgins' I2 statistical test were used to assess the statistical heterogeneity of the pooled results. If I2 statistic ≥50% and P < 0.05, a random effects model was applied to the data. If no heterogeneity was observed, a fixed effect model was used. 18
Results
The literature search identified 6099 articles from which three articles13,19,20 ultimately met the eligibility criteria (Figure 1). The main features of the three studies are summarized in Table 1 and Table 2. In total, 3646 patients were involved. One study was carried out in Italy, Croatia, and Singapore, another in Japan, and a third in Australia. Ages across the three studies varied from 52 to 77 years, with median ages for the intervention group being 72, 66, and 71 years, respectively, and for the control group, 71, 67, and 72 years. Cardiac surgeries included: CABG; mitral valve repair; aortic valve repair; total aortic arch replacement (TAAR); thoracoabdominal aortic replacement (TAAAR). All three RCTs used KDIGO criteria for AKI assessment. Control groups received, Ringer’s solution, crystalloid infusion or standard care.

Flow diagram of included and excluded studies.
Characteristics of the included studies.

Abbreviations: MC, multicentre; DB, double blind; RCT, Randomized Controlled Trial; SC, single centre; IV, intravenous; AKI, acute kidney injury; KDIGO, Kidney Disease: Improving Global Outcomes; eGFR, estimated glomerular filtration rate; ICU, intensive care unit; HDU, high dependency unit.
Clinical and demographic characteristics of the patients.
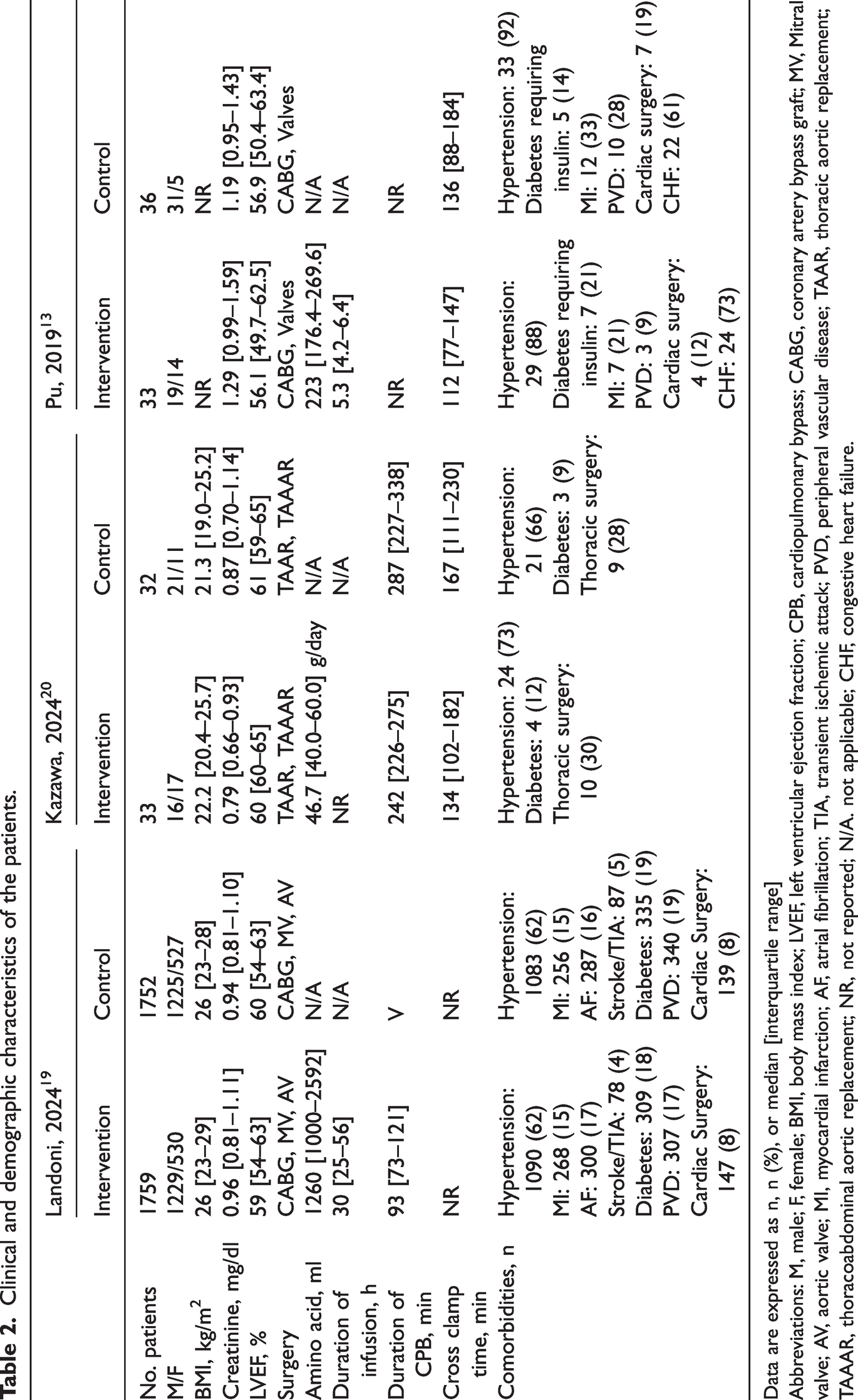
Data are expressed as n, n (%), or median [interquartile range]
Abbreviations: M, male; F, female; BMI, body mass index; LVEF, left ventricular ejection fraction; CPB, cardiopulmonary bypass; CABG, coronary artery bypass graft; MV, Mitral valve; AV, aortic valve; MI, myocardial infarction; AF, atrial fibrillation; TIA, transient ischemic attack; PVD, peripheral vascular disease; TAAR, thoracic aortic replacement; TAAAR, thoracoabdominal aortic replacement; NR, not reported; N/A. not applicable; CHF, congestive heart failure.
According to the Cochrane risk-of-bias tool for randomized trials (ROB 2.0) one study was judged to be at low risk of bias in all domains, 19 while the other two studies,13,20 showed some concerns of bias in one domain. However, all three RCTs showed a low risk of bias arising from the randomization process, a low risk of bias due to missing outcome data, and a low risk of bias in the measurement of the outcome. A graphical summary of the risk of bias assessment within each study as well as a combined graphical summary of risk of bias within each domain is shown in Figure 2. According to the GRADE assessment for level of evidence quality, there was moderate certainty for the majority of the outcomes, high certainty for stage 1 AKI, and low certainty for kidney replacement therapy.

Graphical summary of risk of bias within each domain according to Cochrane risk-of-bias tool for randomized trials (ROB 2.0). 17 (a) Summary Plot and (b) Traffic Light Plot.
In terms of the primary outcome, the incidence of AKI, the pooled outcome of the three studies showed that IV amino acids were comparable with control therapy (RR, 0.73; 95% CI, 0.52, 1.04; P = 0.08; I2 = 35%) (Figure 3).

Forest plot evaluating the effects of IV amino acids by comparison with control measures (i.e., Ringer’s solution, crystalloid infusion, standard care) on the incidence of acute kidney injury (AKI).
In terms of the secondary outcome, the number of patients with Stage 1 AKI, the pooled outcome of the three studies showed that IV amino acids were significantly superior to control therapy (RR, 0.87; 95% CI, 0.78, 0.97; P = 0.01; I2 = 0%) (Figure 4).
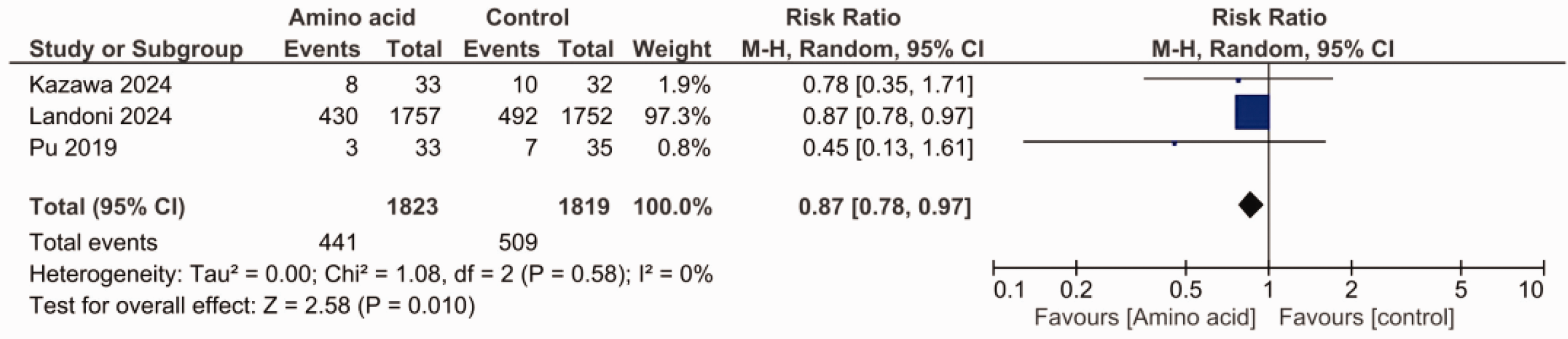
Forest plot evaluating the effects of IV amino acids by comparison with control measures (i.e., Ringer’s solution, crystalloid infusion, standard care) on the incidence of Stage 1 AKI.
For the number of patients with Stage 2 AKI, the pooled outcome of the three studies showed that IV amino acids were comparable with control therapy (RR, 1.24; 95% CI, 0.60, 2.56; P = 0.57; I2 = 0%) (Figure 5).

Forest plot evaluating the effects of IV amino acids by comparison with control measures (i.e., Ringer’s solution, crystalloid infusion, standard care) on the incidence of Stage 2 AKI.
For the number of patients with Stage 3 AKI, the pooled outcome of the three studies showed that IV amino acids were significantly superior to control therapy (RR, 0.53; 95% CI, 0.34, 0.83; P = 0.005; I2 = 0%) (Figure 6).
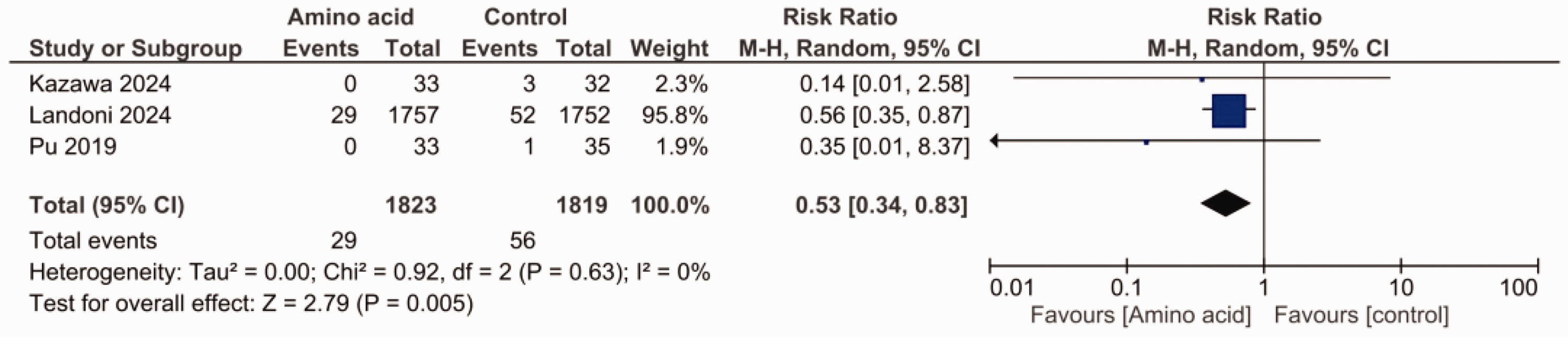
Forest plot evaluating the effects of IV amino acids by comparison with control measures (i.e., Ringer’s solution, crystalloid infusion, standard care) on the incidence of Stage 3 AKI.
In terms of the secondary outcome, the number of patients that used kidney replacement therapy, the pooled outcome of the three studies showed that IV amino acids were comparable with control therapy (RR, 0.86; 95% CI, 0.22, 3.37; P = 0.83; I2 = 48%) (Figure 7).

Forest plot evaluating the effects of IV amino acids by comparison with control measures (i.e., Ringer’s solution, crystalloid infusion, standard care) on the incidence of Kidney Replacement Therapy.
The analysis was repeated after excluding one study that was a significant contributor to the overall weight of the analysis. 19 Results of this sensitivity analysis showed that IV amino acids significantly reduced the risk of AKI (RR, 0.52; 95% CI, 0.30, 0.90; P = 0.02; I2 = 0%). However, no significant difference was observed between intervention and control for number of patients with Stage 1 AKI (RR, 0.67; 95% CI, 0.34, 1.31; P = 0.24; I2 = 0%), Stage 2 AKI (RR, 0.63; 95% CI, 0.08, 4.98; P = 0.66; I2 = 0%), Stage 3 AKI (RR, 0.21; 95% CI, 0.02, 1.83; P = 0.16, I2 = 0%), and use of kidney replacement therapy (RR, 0.89; 95% CI, 0.03, 25.97, P = 0.95, I2 = 71%).
Discussion
Following cardiovascular surgery, AKI is a common complication that affects more than 40% of patients who have undergone CPB. 6 Our meta-analysis showed that IV amino acid infusions during cardiac surgery significantly lowered the risk of Stage 1 and Stage 3 AKI. However, the treatment had no significant impact on Stage 2 AKI and need for kidney replacement therapy. These findings suggest that IV amino acids provide targeted protection, particularly in the early and severe stages of AKI, and could be a valuable addition to strategies aimed at preventing severe kidney complications after cardiac surgery.
The main cause of AKI is kidney hypoperfusion. 21 Reduced cardiac output following surgery frequently makes AKI worse by decreasing kidney perfusion and raising afferent arteriolar resistance via vasopressor use. This can lead to oliguria and reduced glomerular filtration rate (GFR). The complex interaction of preoperative, intraoperative, and postoperative factors that impair kidney function contributes to the multifaceted nature of AKI. 22
Despite advances in our understanding and management of AKI, effective preventive measures remain limited. Established approaches concentrate on supportive measures, but specific and effective approaches are needed. 23 IV amino acid infusions are one such method that has attracted attention. Initial studies have suggested that these infusions may help preserve kidney function during critical illness 12 and after cardiac surgery. 13 Kidney blood flow and function may be improved by amino acid infusion through the activation of kidney functional reserves, 24 lowering of afferent arteriolar resistance, 25 and amplification of nitric oxide generation. 26 Studies in rats have demonstrated that amino acids infusion lowers afferent arteriolar resistance and nitric oxide has been demonstrated to mediate the hyperfiltration and vasodilation observed with amino acid infusion.25,26 While the precise mechanisms through which IV amino acids enhance kidney function remain unknown, it has been established in studies conducted in humans that the increase in GFR caused by amino acid infusion is accompanied by an increase in kidney plasma flow without a marked increase in filtration fraction (FF). 24 Therefore, it is believed the effect is due to afferent (pre-glomerular) arteriolar dilation. 24 The exact mechanisms causing this vasodilation are not fully understood but evidence indicates that neither the direct vasodilatory effects of amino acids on the blood vessels, nor their metabolism in the liver or the release of hormonal factors from the liver, are likely to be significant contributors. One suggested mechanism for this vasodilatory effect is that an increase in the filtered load of amino acids reduces distal tubular NaCl delivery by enhancing its reabsorption in the proximal tubules. This is detected by the macula densa which leads to inhibition of the tubule-glomerular feedback, thus causing vasodilatation of the afferent arterioles. 24
Pre-existing kidney dysfunction, such as chronic kidney disease (CKD), is recognized as a risk factor for AKI in patients undergoing cardiac surgery. 27 While IV amino acid infusion has shown potential for increasing kidney blood flow and GFR, this effect primarily benefits kidneys with sufficient residual nephron mass. Consequently, in the case of severe pre-existing structural nephropathy, where nephron mass is significantly reduced, amino acid infusion may not reduce the incidence of postoperative AKI. 28 Further research and large-scale studies are required to substantiate this observation and fully understand the role of amino acids in this patient population. However, in a study involving 474 critically ill patients in ICU who were randomized to either standard care or IV amino acid supplementation, in those with normal kidney function at baseline, amino acid supplementation significantly reduced 90-day mortality and improved eGFR. 29 In addition, long-term IV amino acid supplementation has been shown to improve kidney function, as evidenced by increased eGFR and urine output, although it does not appear to impact the duration of kidney dysfunction in critically ill patients. 12 Furthermore, animal studies indicate that high levels of aromatic amino acids can have a protective effect on CKD. 30 Given the established link between AKI and development of subsequent CKD, these findings highlight the potential of amino acid supplementation to reduce acute kidney injury and prevent long-term complications associated with CKD. 30
An important aspect of our meta-analysis is that all three RCTs used the KDIGO criteria to assess AKI. This consistency enhances the reliability and comparability of our meta-analysis, ensuring accurate conclusions about the impact of IV amino acids on kidney outcomes in cardiac surgery patients. Although not achieving statistical significance, the pooled analysis of three studies revealed an RR of 0.73 for AKI, showing a trend toward reduction of AKI risk with IV amino acids. Moreover, our findings are consistent with those of another meta-analysis that examined amino acid infusion for perioperative kidney protection, specifically focusing on AKI in a cardiac surgery subgroup. 31 The consistency of results across both meta-analyses reinforces the evidence suggesting a potential kidney-protective effect of amino acid infusion in cardiac surgery patients. However, since the results did not reach statistical significance, this conclusion cannot be made and further large scale RCTs are needed to make definitive conclusions. Following the exclusion of one study that was a significant contributor to the overall weight of the analysis, 19 our sensitivity analyses found that IV amino acids significantly reduced the risk of AKI. This result further underscores the need for additional large-scale, double-blind RCTs to determine the true effect of amino acids on AKI outcomes.
As demonstrated by a large multicentre study involving 3,500 patients undergoing cardiac surgery, Stage 1 AKI is a frequent complication following such procedures. 32 Any intervention that prevents Stage 1 AKI would benefit a significant number of patients undergoing cardiac surgery. Our meta-analysis showed a significant protective effect of IV amino acids in terms of Stage 1 AKI. However, a subsequent sensitivity analysis excluding one study, 19 found that IV amino acids no longer showed a beneficial effect indicating that this single study heavily influenced the overall effect of our analysis. Therefore, caution is required when interpreting our results, and emphasises the need for further well-powered studies to validate the protective effect of IV amino acids on Stage 1 AKI. With regard to Stage 2, AKI, we found that no difference between the IV amino acids and control measures. A subsequent sensitivity analysis confirmed these findings. For Stage 3 AKI, we found a significant protective effect of IV amino acids in patients undergoing cardiac surgery. Indeed, by comparison with the control group, patients receiving IV amino acids had a 47% lower risk of having Stage 3 AKI. However, a subsequent sensitivity analysis excluding one study 19 , found that IV amino acids no longer showed a beneficial effect suggesting that the inclusion that study influenced the significant effect on Stage 3 AKI.
With regard to kidney replacement therapy, our meta-analysis found no difference between IV amino acids and control measures. The moderate level of heterogeneity (i.e., I2 = 48%) suggested variability in study results, which may be due to differences in the criteria for initiating kidney replacement therapy or the severity of AKI. A previous study indicated that 1.4% of patients who underwent CABG required continuous kidney replacement therapy as a result of AKI. 33 In another study, kidney replacement therapy was required for three patients in the control group, and none in the IV amino acid group, suggesting the IV amino acids have a potential protective effect. 20 However, the low sample size and number of events in that study suggest it could be a random finding and warrants further investigation.
Our meta-analysis had several limitations. For example, it was based on an analysis of only three studies, which may limit the generalizability of the findings and reduce the statistical power to detect meaningful differences across the outcomes. In addition, most patients included in the meta-analysis were from one large RCT. 19 Consequently, this study made a large contribution to the overall results, which also limits the generalizability of our findings. Furthermore, we only included RCTs and excluded cohort studies. This decision may have excluded important patient data that could have offered additional context. Also, some of the individual studies had wide confidence intervals, which suggests that their effect size was uncertain. More research involving additional studies is required to validate our findings.
In conclusion, this systematic review and meta-analysis showed that IV amino acids may provide protective benefits against AKI Stages 1 and 3 in patients undergoing cardiac surgery. However, no significant difference between amino acids and control measures was shown for prevention of overall AKI, Stage 3 AKI or kidney replacement therapy. Although our meta-analysis was based on just three studies, its focus on including only RCTs ensured a high level of evidence, minimized bias, and offered a robust foundation for clinical guidelines.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251315919 - Supplemental material for Intravenous amino acids for kidney protection in patients undergoing cardiac surgery: A systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-pdf-1-imr-10.1177_03000605251315919 for Intravenous amino acids for kidney protection in patients undergoing cardiac surgery: A systematic review and meta-analysis of randomized controlled trials by Malik Waleed Zeb Khan, Aizaz Ali, Amna Hussain, Aban Masaud Mian, Abdul Moeez, Waqas Inayat, Bushra Noor, Izhar Ali Shah, Muhammad Osama, Bushra Khan, Muhammad Sajid, Touba Azeem, Mohammad Ebad Ur Rehman, Ashraf Ali and Ubaid Khan Afridi in Journal of International Medical Research
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
