Abstract
Objectives:
Acute kidney injury is common among the critically ill. However, the incidence, medication use, and outcomes of acute kidney injury have been variably described. We conducted a single-center, retrospective cohort study to examine the risk factors and correlates associated with acute kidney injury in critically ill adults with a particular focus on medication class usage.
Methods:
We reviewed the electronic medical records of all adult patients admitted to an intensive care unit between 1 February and 30 August 2020. Acute kidney injury was defined by the 2012 Kidney Disease: Improving Global Outcomes guidelines. Data included were demographics, comorbidities, symptoms, laboratory parameters, interventions, and outcomes. The primary outcome was acute kidney injury incidence. A Least Absolute Shrinkage and Selection Operator regression model was used to determine risk factors associated with acute kidney injury. Secondary outcomes including acute kidney injury recovery and intensive care unit mortality were analyzed using a Cox regression model.
Results:
Among 226 admitted patients, 108 (47.8%) experienced acute kidney injury. 37 (34.3%), 39 (36.1%), and 32 patients (29.6%) were classified as acute kidney injury stages I–III, respectively. Among the recovery and mortality cohorts, analgesics/sedatives, anti-infectives, and intravenous fluids were significant (
Conclusion:
Evaluation of vital signs, laboratory, and medication use data may be useful to determine acute kidney injury risk stratification. The influence of particular medication classes further impacts the risk of developing acute kidney injury, necessitating the importance of examining pharmacotherapeutic regimens for early recognition of renal impairment and prevention.
Introduction
Acute kidney injury (AKI) is a common diagnosis in the adult intensive care unit (ICU) due to substantial short- and long-term morbidity and mortality associated with its occurrence.1–4 AKI globally impacts more than 13 million patients, resulting in approximately 2 million deaths annually. 5 In the United States, it has been estimated that up to 10% of hospitalizations and up to 60% of patients admitted to an ICU experience AKI.1,6 AKI is an abrupt (within hours) decrease in kidney function defined as an increase in serum creatinine (SCr) or a decrease in urine output (UO) for a duration between 7 and 90 days after exposure to an AKI-causing event.7,8 The etiology is complex and routinely multifactorial. Known common risk factors associated with AKI include diabetes, hypertension, chronic kidney disease (CKD), cardiovascular, lung, and liver diseases, sepsis and surgical interventions, as well as exposure to nephrotoxic medications and diagnostic contrast agents.9–13 Despite the development of numerous prevention strategies, the most recent data suggest the incidence of AKI is increasing in the critically ill population. 6
Mortality rates in the ICU can be as high as 50% and incur upward of $24 billion upon the US health system annually.14,15 During the COVID-19 pandemic, studies have reported AKI incidence rates as high as 38.4 per 1000 patient-days and in-hospital mortality rates to be 18.9%, 46.2%, and 81.3% among those with AKI stages I–III, respectively.16,17 In another study of approximately 3100 critically ill COVID-19-infected patients, about 20% experienced AKI requiring renal replacement during their ICU admission. Among patients who developed AKI, 60% resulted in hospital-associated mortality. 18 However, existing evidence has not extensively explored medication use and their impact on AKI incidence, recovery, and outcomes among critically ill patients.
The Kidney Disease Improving Global Outcomes (KDIGO) 2012 workgroup classifies AKI into three stages based on

(a) Study population flowchart AKI, acute kidney injury (b) Kidney Disease Improving Global Outcomes (KDIGO) classification of AKI status.
This study aimed (1) to identify clinical characteristics, correlates, and outcomes of critically ill patients who experienced AKI and (2) to provide insight into the clinical characteristics regarding the incidence, severity, and influence of medication use on the injury. The primary outcome of AKI incidence was defined using the KDIGO classification method. Secondary outcomes analyzed were AKI recovery and ICU mortality.
Methods
Ethics approval
Ethical approval for this study was waived by Roger Williams Medical Center, IRB: 00000058 and The University of Rhode Island, IRB: 00000599 because of the retrospective, de-identified nature of the data. Informed consent was not sought for the present study because of the nature of the de-identified data.
Study design and setting
This retrospective, single-center cohort study was conducted using electronic health records (EHRs) data from a 220-bed acute care, community teaching hospital in Providence, Rhode Island, USA.
Participants
A total of 231 patients were considered for inclusion in the study. All adult patients admitted to the ICU between 1 February 2020, and 30 August 2020, were included if they met the following criteria: ⩾ 18 years of age and admission to the ICU > 24 h. Three patients were removed as they were considered long-term care patients and extreme outliers (> 4 standard deviations of ICU length of stay). Patients with a history of renal replacement therapy, kidney transplantation, or missing clinical information data upon enrollment were excluded (Figure 1(a)). Active COVID-19 illness was confirmed by a positive nucleic acid amplification test (NAAT) result.28,29 After exclusion, 226 patients were included in the final analysis.
Data sources and exposures
A range of factors were investigated to determine the risk of AKI including patient demographics, chronic conditions, vital signs, laboratory measures, and medication class usage. Demographic measures included were age, sex, BMI, ethnicity, and race. Vital signs data such as, systolic blood pressure (SBP), diastolic blood pressure (DBP), mean arterial pressure (MAP), heart rate (HR), respiratory rate (RR), and temperature measurements, were calculated by averaging for the first 2 days of admission. Laboratory values such as electrolytes, complete blood cell counts, arterial blood gasses, hepatic, and kidney function values were included. Severity of illness was assessed using the Acute Physiology and Chronic Health Evaluation (APACHE II) score based on the poorest variables recorded during the first 24 h of ICU admission. All severity of illness scoring tools (i.e. APACHE II and Glasgow Coma Scale (GCS)) were calculated using their respective formulas and reported as a numerical value.30,31 A complete list and description of variables are provided in Supplementary Table 1.
AKI definition and recovery
AKI status was determined by evaluating both
AKI recovery was defined using
Statistical analysis
Descriptive statistics are presented as percentages for categorical variables, and medians (interquartile ranges (IQRs)) for continuous variables. The independent
The Kaplan–Meier method was used to conduct survival modeling for AKI recovery and ICU mortality over time according to AKI staging. In the analysis, we observed violations of two statistical assumptions of the Cox proportional hazard model: (1) time-invariant hazards and (2) multiple competing outcomes (i.e. worsening condition, AKI recovery, and mortality). To address these statistical violations, we modeled the cause-specific hazards of recovery and mortality as a function of AKI as well as other demographic predictors. Schoenfeld residual analysis was utilized to determine time-varying variables. Cause-specific hazards (risk of event per time unit change) provided statistically valid measurements for competing outcomes (i.e. recovery and mortality). A competing risk is an event that either hinders the observed event of interest or modifies the chance of occurrence. In our analysis, we did not separate the COVID-19-infected cohort due to the small mortality sample size (n = 1).
33
To examine the survival functions of nonparametric data, stacked cumulative incidence functions were used in place of Kaplan–Meier curves to provide a comparison of cumulative probability of recovery and mortality over time according to different AKI staging cohorts.
34
The details of the improved models are located in the Supplementary File. The risk of AKI recovery and mortality was reported using a hazard rate (HR) and the risk of event per time unit change is presented as
Results
Clinical characteristics of AKI and non-AKI patients
AKI was identified in 47.8%, among them, 37 patients (34.3%) were classified as stage I AKI, 39 patients (36.1%), and 32 patients (29.6%) were classified as stages II and III, respectively. The mortality rate was 13.7%, and among those who died 51% had AKI. Thirty-three patients (30.6%) recovered from the AKI event. Baseline demographic, physiological status, and comorbidities were compared to those who developed AKI and those who did not. Patients who developed AKI had a significantly higher BMI 30.8 (24.1–36.5),
Clinical characteristics in non-AKI, AKI groups, and all patient cohorts.

AKI: acute kidney injury; BMI: body mass index; BUN: blood urea nitrogen; CO2: serum bicarbonate level; eGFR: estimated glomerular filtration rate; GCS: Glasgow Coma Scale; INR: international normalized ratio; K: serum potassium level; PO4: serum phosphate level; PT: prothrombin time; RR: respiratory rate; SBP: systolic blood pressure; SCr: serum creatinine; WBC: total white blood cell count. Data presented as n (%) or median [interquartile range].
Count (N), mortality (%), mean time on mechanical ventilation in hours (IQR) and ICU in hours (IQR) for AKI, COVID infected and AKI, and non-AKI patients.

AKI: acute kidney injury; COVID: coronavirus disease; ICU: intensive care unit; LOS: length of stay; TOMV: time on mechanical ventilation.
The LASSO model identified 13 predictors associated with higher odds of experiencing AKI including BMI (OR: 1.01), hypo-osmolality and hyponatremia comorbidities (OR: 2.34), and COVID-19 infection (OR: 1.31) (Table 3).
Coefficients and OR of predictors of AKI classification, selected by LASSO regression.

AKI: acute kidney injury; BMI: body mass index; COVID: coronavirus disease; OR: odds ratio.
Medication class influence on AKI
In the AKI cohort, the use of anti-infective agents, vancomycin, and piperacillin/tazobactam were implicated in approximately double (23% and 27% versus 12% and 14%) the incidence of AKI when compared to the non-AKI cohort. In addition, we observed that the use of meropenem was frequently (15%) administered within the AKI cohort. Notably, use of albumin (27%), calcium gluconate (18%), fentanyl (18%), and midazolam (15%) were administered only in the AKI cohort (Supplementary Table 2). Interestingly, we found different medication classes were associated with a higher risk of AKI, including analgesics/sedatives (OR: 1.27), anti-infectives (OR: 1.71), diuretics (OR: 1.66), and gastrointestinal agents (OR: 1.70) (Table 3).
Significant differences between medication classes and the occurrence of AKI were found. Greater proportions of patients who were given anti-infectives, diuretics, endocrine, paralytic, pulmonary, and vasopressor agents had persistent AKI (Table 4). Among the recovery group, the most frequently used medication classes were analgesics/sedatives (93.5%), anti-infectives (90.3%), GI agents (90.3%), hematologic/anticoagulants (90.3%), intravenous fluids, electrolytes, and nutritional support agents (100%). Among the mortality group, the most frequently used medication classes were analgesics/sedatives (100%), anti-infectives (100%), endocrines (100%), intravenous fluids, electrolytes, nutritional support, and vasopressors agents (100%).
Medication classification association with AKI persistence, recovery, and mortality.

AKI: acute kidney injury.
The most frequently administered medications in the
AKI recovery and mortality modeling
AKI status over time, outcomes of recovery, and mortality were considered in the survival models. Figure 2 demonstrates the probability of survival by AKI staging. Survival was similar for all patients at any AKI stage and in the non-AKI cohort until 10 days. However, due to competing outcomes (i.e. patients may experience recovery, worsening of condition, or mortality), a single-outcome method application is not valid, yet it is shown for demonstration (Figure 2). Notably, the Kaplan–Meier curve and log-rank statistic of survival cannot capture significant differences in the survival model in part due to multiple outcomes and nonproportionality.
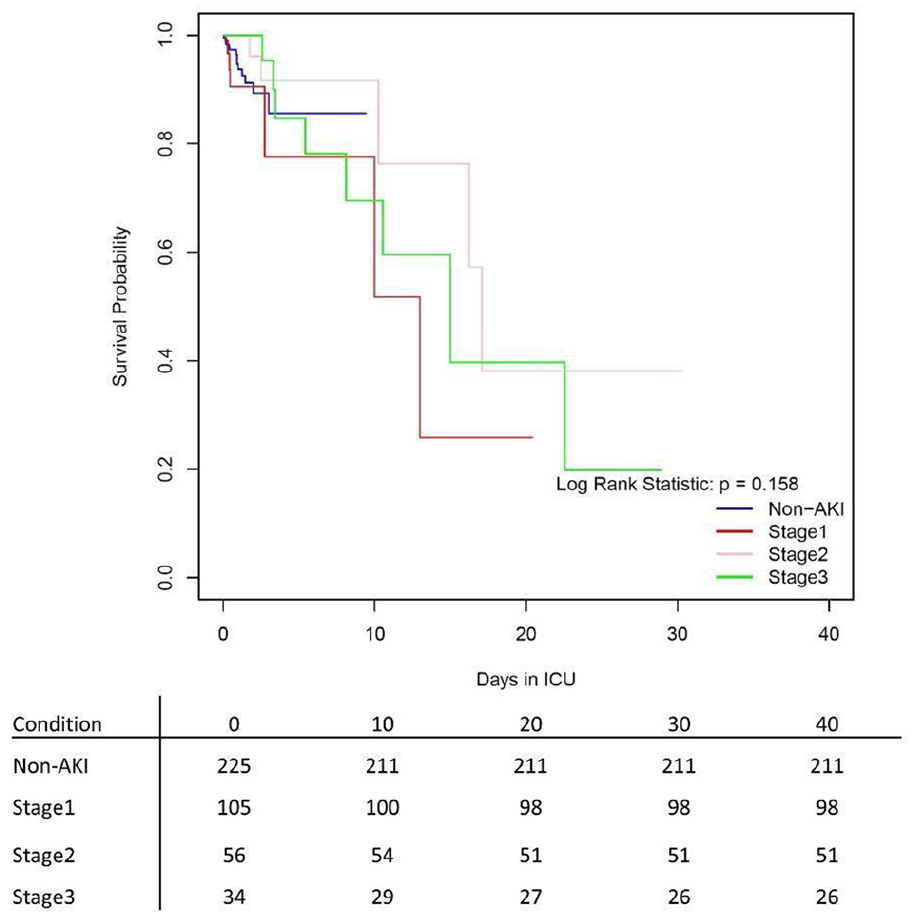
Kaplan–Meier survival curves for mortality based upon AKI status (non-AKI, stages I, II, and III).
Therefore, the use of a stacked cumulative incidence function serves as a nonparametric comparison of cause-specific hazard. These were modeled for each AKI condition and reveal major differences by AKI staging in both recovery and mortality. Regarding mortality outcomes, patients who experienced AKI stage III had the highest hazard and cumulative incidence (Figure 3(a)). The cumulative incidence function for mortality demonstrates that control patients have high mortality upon ICU admission (<3 days) and plateau in terms of hazard (Figure 3(a)). Most notably, the opposite is true among severe AKI cases.
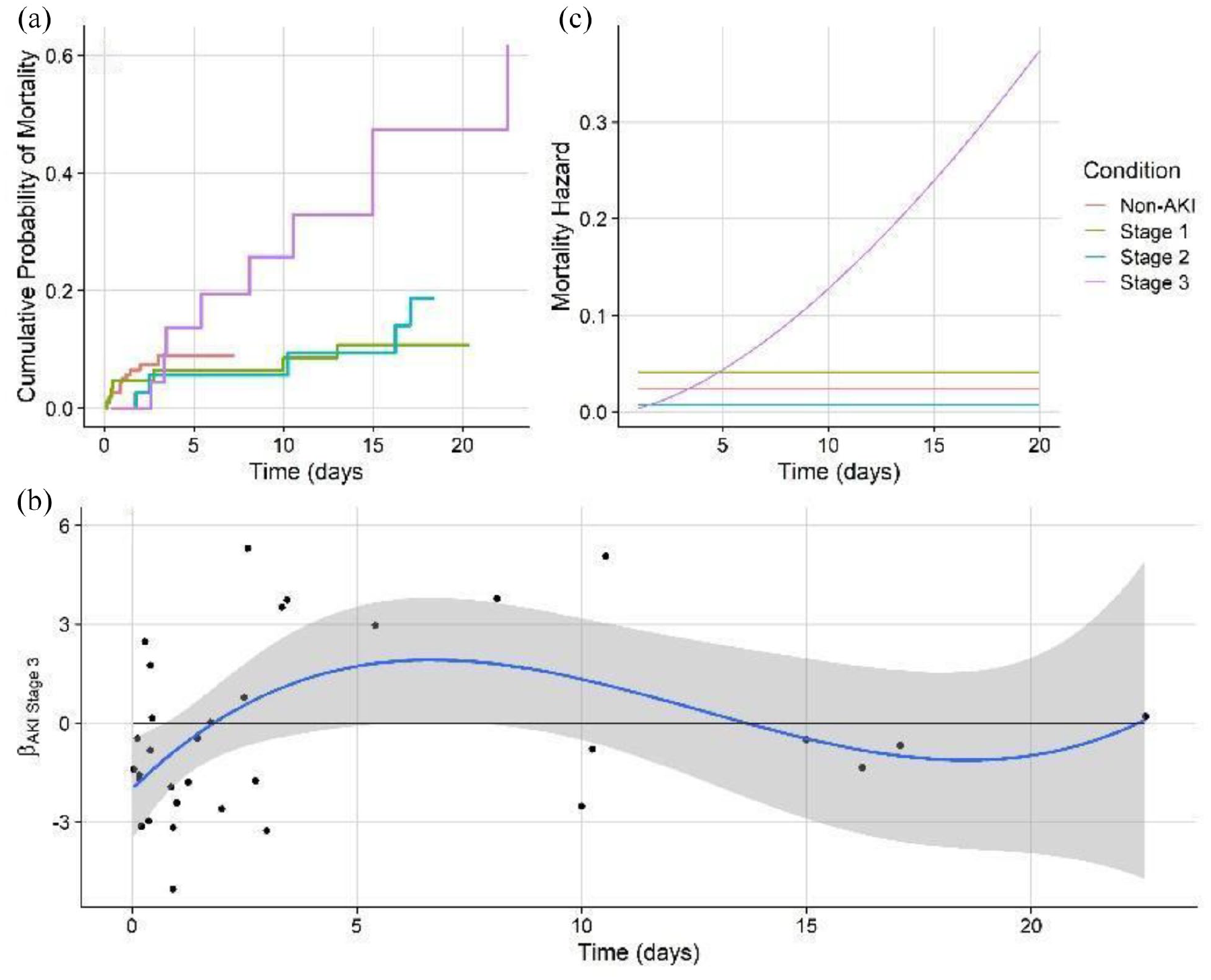
(a) Stacked cumulative incidence functions of AKI mortality for non-AKI, stage I, stage II, and stage III AKI cases. (b) Schoenfeld residuals (black points) for AKI stage III, fit over time, and fit with a third-order polynomial function for visualization (solid blue line: mean, gray shading: 95% CI). (c) Modeled cause-specific mortality hazards over time for all AKI conditions.
The Cox model was used to investigate predictors of cause-specific hazard for mortality. In the mortality model, examination of Schoenfeld residuals demonstrated significant time-varying effects and thus nonproportionality for AKI stage III patients (Figure 3(b)). Therefore, a log-linear time-varying effect for these variables was incorporated into the final model. Furthermore, mortality analysis using Cox regression repeatedly incorporates time-varying hazards for AKI stage III patients, thus allowing potentially nonproportional hazards with time (Figure 3(a)). Results revealed a significant increase in hazard HR: 4.72(95% CI: 1.0–22.33) for AKI stage III patients, which resulted in a significantly higher mortality hazard when compared to milder AKI stages and those without AKI (Table 5, Figure 3(c)). The mortality hazard was significant and proportional to GCS and BMI. Forward selection of the model revealed that COVID-19-infected patients with AKI stage I were significantly associated with mortality (HR: 16.77). Furthermore, we found medication classes such as analgesics/sedatives, intravenous fluids, electrolytes, nutritional, psychiatric, and vasopressors agents were significantly (
Cox regression model of the risk factors associated with ICU mortality.

BMI: body mass index; CI: confidence interval; GCS: Glasgow Coma Scale; HR: hazard ratio.
Coefficients, standard error (SE), and
An analogous model evaluating AKI recovery was performed incorporating the addition of dialysis treatment as a predictor, as well as time-varying effects for AKI stages I and II and COVID-19 infection, thus allowing potentially nonproportional hazards with time (Figure 4(a)). Examination of Schoenfeld residuals of the mortality model revealed significant time-varying effects and thus nonproportionality for COVID-19 and AKI stages I and III (Figure 4(b)). Results revealed a significant increase in HR: 9.14 (95% CI: 2.14–39.06) for AKI stage I, which resulted in a faster recovery hazard when compared to severe AKI patients (Table 4, Figure 4(c)).
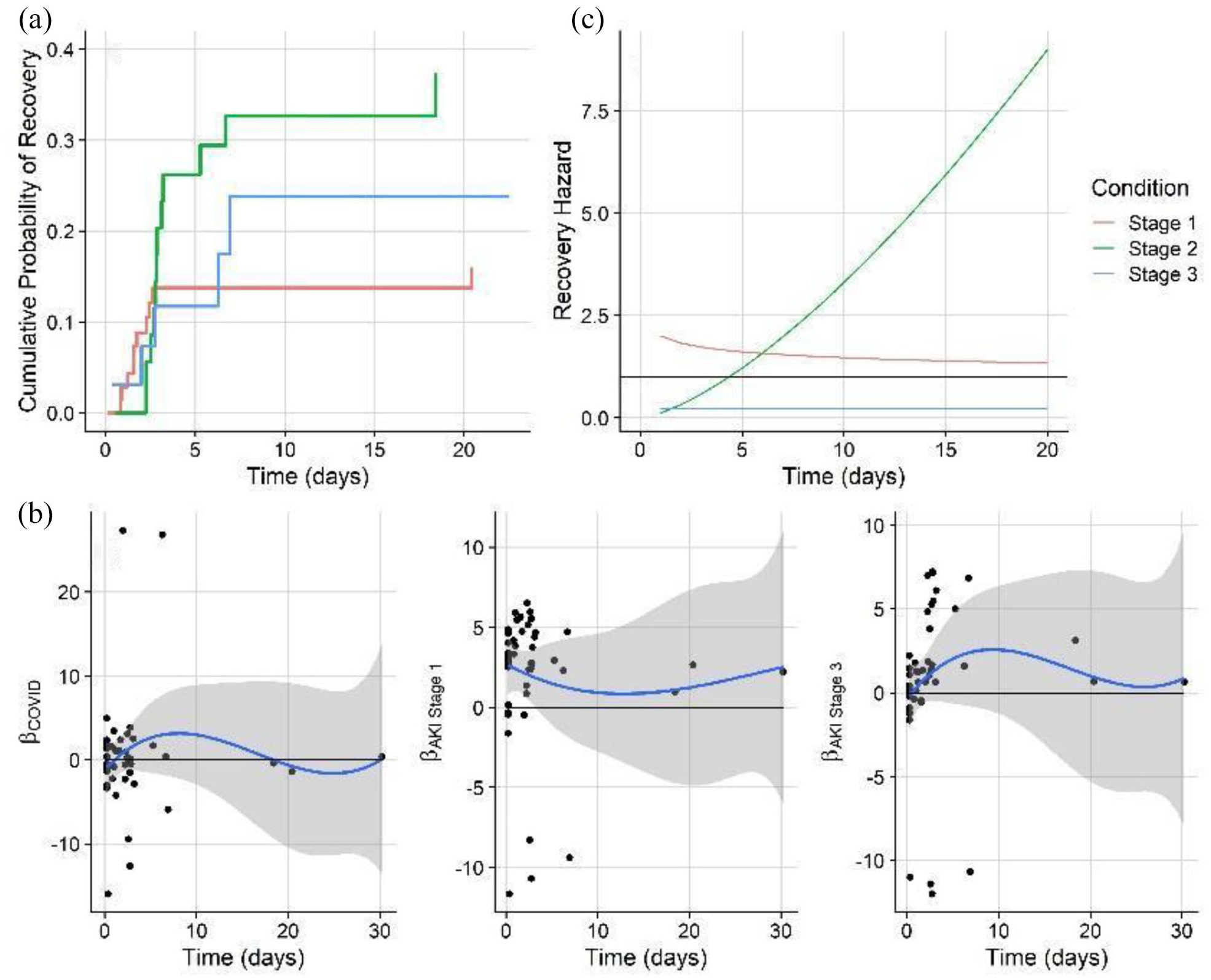
(a) Stacked cumulative incidence functions of AKI recovery for stage I, stage II, and stage III AKI cases. (b) Schoenfeld residuals (black points) for AKI Stage III, fit over time, and fit with a third-order polynomial function for visualization (solid line: mean, gray shading: 95% CI). (c) Modeled cause-specific AKI recovery hazards over time for all AKI conditions.
Forward model selection in predicting AKI recovery revealed that among all medication classes administered, diuretics and
Discussion
Summary of key findings
In the present study, we evaluated the incidence of AKI, correlates, recovery, and mortality. Of the 226 enrolled patients, 47.8% developed AKI based upon the KDIGO (combined)
Context in association with the published literature
We opted to combine both
To investigate the mortality hazard, we employed an improved method where we considered multiple competing outcomes in the Cox regressions model. We found that patients with AKI who were in stage III had a higher hazard of mortality (Table 5) when compared to milder AKI stages and patients without AKI. Considering multiple competing outcomes, survival analysis is appropriate for this nonproportional data. As previously, multiple outcomes are considered competitive hazards when using survival modeling and have the potential to mathematically alter the relationship between the hazard functions (instantaneous failure rate) and the survival function (defining probability of survival up to a given time).46,47 However, our refined models fit both the cause-specific hazards for mortality and recovery rather than survival directly. The cause-specific hazards were stated due to the 1:1 relationship between hazard and survival being lost in competitive hazard cases.33,47 Conversely, patients in stage I had higher rates of recovery compared to patients with severe AKI (Table 6).
Cox regression model of the risk factors associated with AKI recovery.
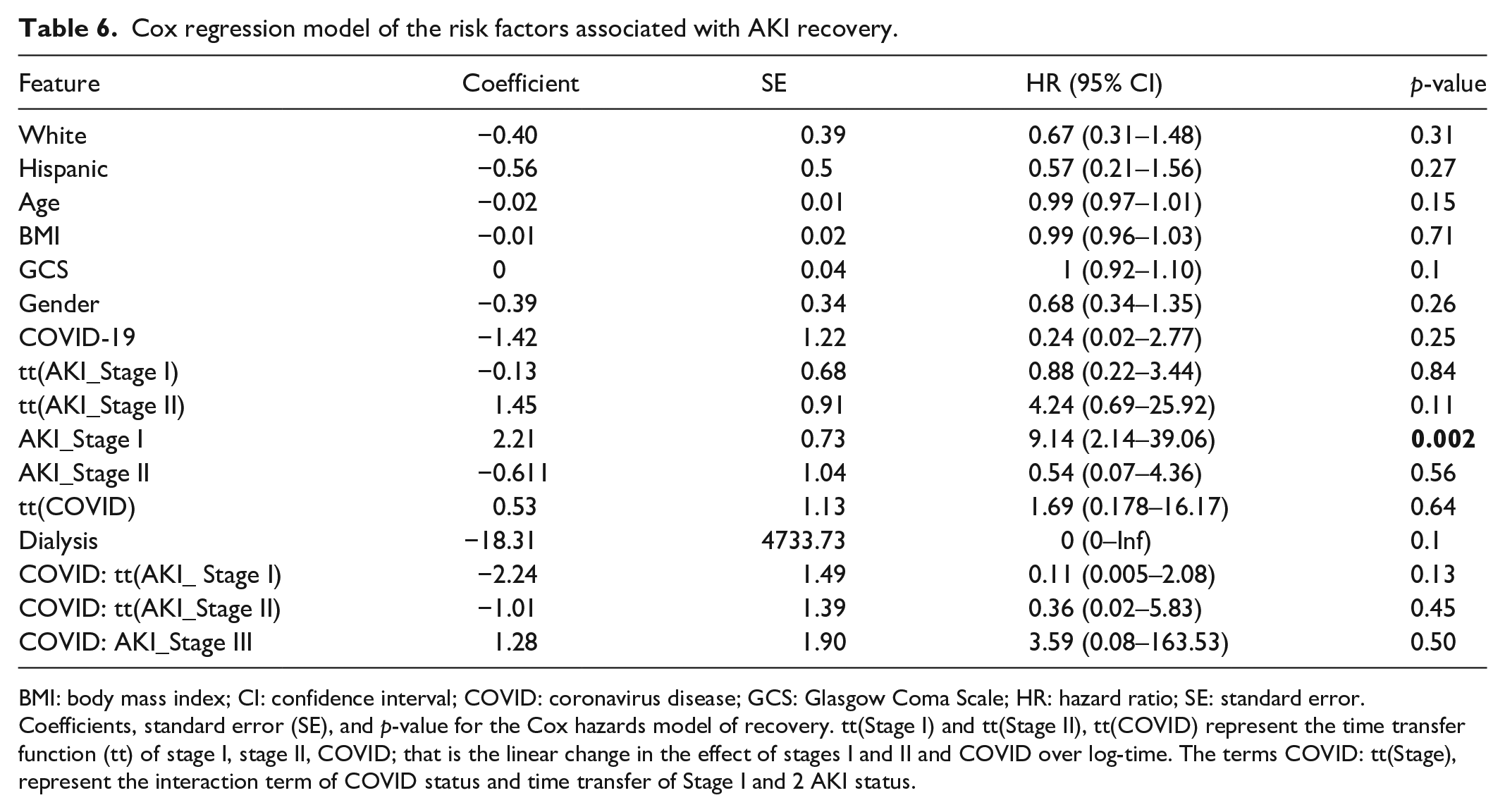
BMI: body mass index; CI: confidence interval; COVID: coronavirus disease; GCS: Glasgow Coma Scale; HR: hazard ratio; SE: standard error. Coefficients, standard error (SE), and
When reviewing the specific details of medication use among AKI patients, we found that higher mortality was exhibited among those administered vasopressors and “other” medications while lower mortality was seen for sedatives and psychiatric agents. However, diuretics and IV fluids were positively associated with recovery. Furthermore, consistent with published literature, patients receiving diuretics and gastrointestinal agents are associated with higher incidence of AKI.48–53 Of particular interest, the use of anti-infectives (i.e. vancomycin, piperacillin/tazobactam, and meropenem) as monotherapy or in combination increased the likelihood of experiencing AKI in the ICU.54,55 Finally, in our findings, albumin was notably used only within the AKI cohort. However, published studies have varied regarding the correlation of albumin use and increased incidence of AKI.56,57
The main strengths of the study include (1) granular details of the vitals (every 2 h), laboratory indices (every 6 h), and any prescribed medications, dosage, route of administration, route of interval, and duration during the ICU stay; (2) rigorous application of the KDIGO definition and classification in diagnosing AKI, (i.e. using both
Our study is not without limitations. First, the retrospective nature of the EHR chart review and the limited sample size expose the risk of missing data which may contribute to confounding bias. Second, the use of
Implications for clinicians
This innovative methodologic study confirms traditionally identified risk factors and investigates the influence of medications as they relate to AKI incidence, recovery, and outcomes. Clinical implications of our findings suggest the need for continued vigilant, proactive monitoring, and early recognition of AKI to minimize further insult and delayed renal recovery. Medication class use and their influence on AKI further warrants careful examination of all combined medications to cautiously select each agent weighing their risk versus benefit and their respective influence on AKI outcomes. Furthermore, this study will aid critical care pharmacy practitioners in their pursuit of improving safe medication use by not only considering the therapeutic class risk of kidney injury but importantly avoiding the combined use of particular agents in the complex comorbid health conditions to avoid further kidney injury. Finally, our research confirms the time-varying effect among different AKI stages relating to mortality and recovery using a novel methodological approach.
Conclusion
In conclusion, we found that AKI incidence in the critical care unit remains extremely high and is associated with poorer outcomes. Commonly implicated AKI medication classes, diuretics, anti-infectives, and vasopressor agents were also strongly correlated to increasing the incidence of AKI. However, a direct cause and effect relationship between medication class and incidence or severity of AKI cannot be established. Furthermore, the influence of pharmacotherapy in the occurrence of AKI further supports the need for early identification or avoidance of potential nephrotoxic agents and combinations. Continued vigilant monitoring is warranted to prevent renal insufficiency, minimize progression, and reduce mortality risk.
Supplemental Material
sj-docx-1-smo-10.1177_20503121221099359 – Supplemental material for Survival and recovery modeling of acute kidney injury in critically ill adults
Supplemental material, sj-docx-1-smo-10.1177_20503121221099359 for Survival and recovery modeling of acute kidney injury in critically ill adults by Todd N Brothers, Jacob Strock, Traci J LeMasters, Jayne Pawasauskas, Ronald C Reed and Mohammad A Al-Mamun in SAGE Open Medicine
Footnotes
Acknowledgements
We would like to thank Dr Douglus Slain, PharmD, BCPS, FCCP, FASHP (University of West Virginia, School of Pharmacy) for his support in reviewing the manuscript. We would like to further thank all of our research interns who performed extensive manual data extraction: David Sabatino, Pharm D, James Farrell, Martina Boda, and Allie Lindo, Pharm D. Candidates, Class of 2022.
Author contributions
T.N.B. has full access to all the data in the study and takes responsibility for the integrity of the data. T.N.B., M.A.A., and J.S. contributed to study concept and design. T.N.B. and M.A.A. contributed to acquisition of data. T.N.B., J.S., M.A.A., and T.J.L. contributed to analysis and interpretation of data. T.N.B. and M.A.A. contributed to drafting of the manuscript. T.J.L., J.P., R.C.R., and J.S. contributed to critical revision of the manuscript for important intellectual content.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was waived by Roger Williams Medical Center, IRB: 00000058, and The University of Rhode Island, IRB: 00000599 because of the retrospective, de-identified nature of the data.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the North East Big Data Innovation Hub, National Science Foundation (NSF) (OAC-1916585). The funding organization did not contribute to the study design, collection, management, analysis, or interpretation of data; writing of the report or the decision to submit the report for publication.
Informed consent
Informed consent was not sought for the present study because of the nature of the de-identified data.
Poster presentation
This portion of the research was accepted for a poster presentation at the Society of Critical Care Medicine Annual Congress in February 2022.
Research data
The data supporting the findings from the study are available from the corresponding author on a reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
