Abstract
A novel method for the synthesis of isoflavones from coumarin analogues has been devised. This strategy involves the transformation of coumarins into halogenated hemiacetal ester analogues, followed by the installation of the B-ring of isoflavones via a classical Suzuki–Miyaura cross-coupling reaction. Subsequent simple hydrolysis affords the target isoflavones. The substrate scope with respect to both the A-ring and B-ring was evaluated, revealing that the method exhibits broad substrate tolerance. It was observed that the electronic properties of substituents on either ring A or ring B exerted no significant influence on the reaction efficiency, which can be rationalized by the push–pull electronic effect inherent to the hemiacetal ester intermediate. This approach enables the convergent synthesis of diverse isoflavone analogues and represents a valuable addition to the existing repertoire of synthetic methodologies for isoflavones.

Introduction
Isoflavonoids represent a significant class of natural products predominantly isolated from members of the Leguminosae family, with isoflavones constituting the largest subgroup.1,2 These compounds have garnered considerable attention due to their diverse pharmacological properties, including estrogenic activity,3,4 antioxidant potential,5,6 anticancer effects,7,8 and cardioprotective benefits.9,10 Owing to their broad spectrum of biological activities, isoflavonoids have remained a focal point of scientific investigation.2,11
From a medicinal chemistry perspective, the development of versatile synthetic methodologies for the efficient construction of isoflavone derivatives is of paramount importance. The design of a synthetic route of convergent cross-coupling of functionalized fragments would allow the rapid generation of isoflavone analogs. We recently developed a synthetic method for flavones based on such a strategy, which employed the Stork-Danheiser reaction applied to a coumarin system. 12 As an extension of this reaction paradigm, we wished to synthesize isoflavone analogs using a similar method. Interestingly, while the synthesis of isoflavones from 3-halochromone-based precursors has been extensively studied,13 –19 a coumarin-based approach remains unreported.
Results and discussion
We recently published our work on the synthesis of flavones based on the Stork-Danheiser reaction (Figure 1).
12
The method involved a strategy based on the 1,3-transposition of a coumarin adduct to forge the flavone scaffold. As an extension of this methodology, we anticipated that isoflavones

Our previous work 12 and current work.

The original route to the synthesis of isoflavone
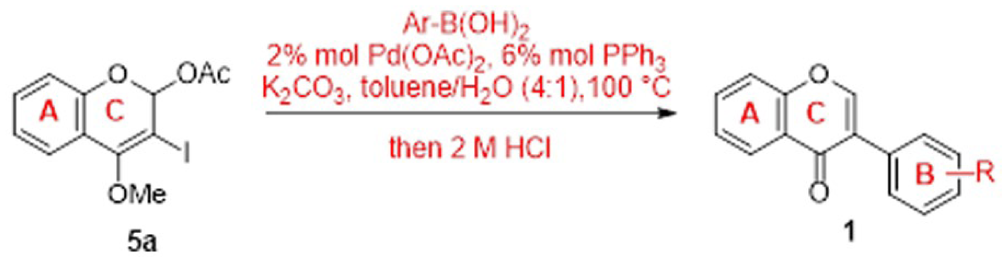
To validate this hypothesis, 4-hydroxycoumarin (where R =H) was employed as the starting material and successfully converted to the iodinated intermediate

Synthesis of 3-iodo-4-methoxylcoumarin.
It was found, however, that the preparation of advanced intermediate
We later found that if the reduction step was implemented prior to the cross-coupling, the reaction profiles could be significantly improved. The details of our work are as follows. Accordingly, 3-iodo-4-methoxycoumarin
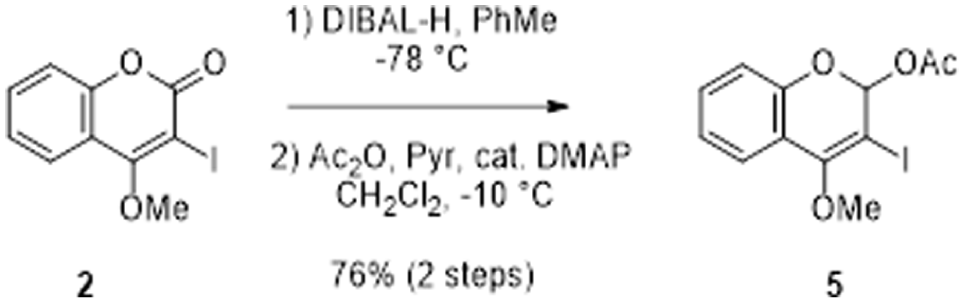
The preparation of the advanced intermediate
With 5 in hand, we advanced to the next step for the Suzuki-Miyaura cross-coupling reaction (Scheme 4).
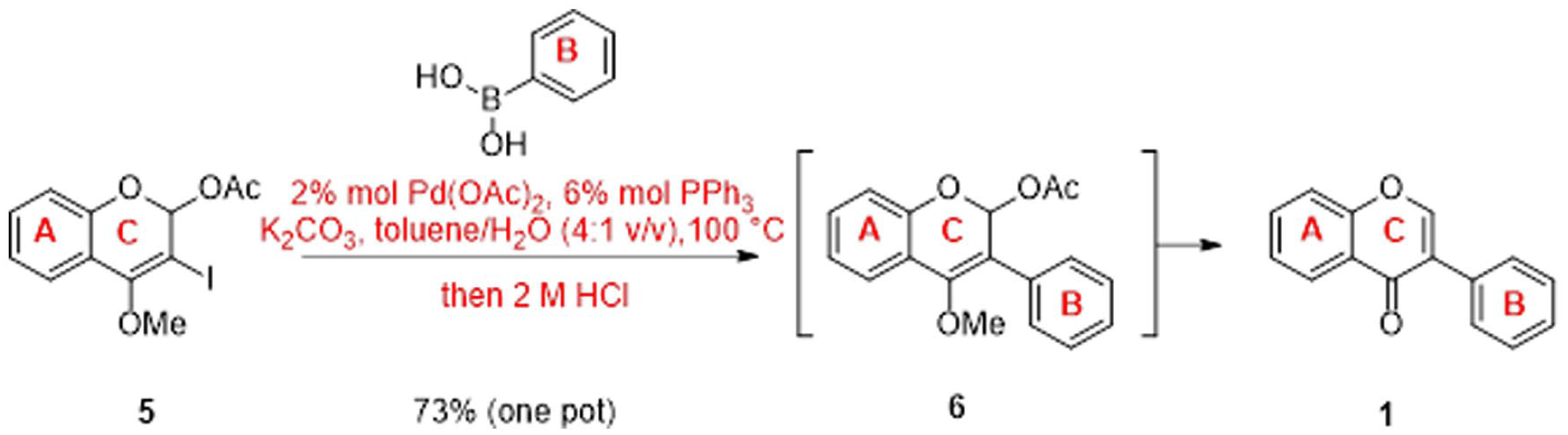
Revised method for the synthesis of isoflavone
Thus, coumarin derivative
The scope of variation at the B-ring.

After that, the survey of the scope of the A-ring was also conducted, and the results were tabulated in Table 2. It was found that the electronic nature of the A-rings does not significantly affect the outcome. The yields corresponding to the electron-donating substrates were 59%–64%; and those to the electron-withdrawing substrates 51%–76%.
The scope of variation at the A-ring.

It is interesting that the electronic nature of the substituents on either A-ring or B-ring substrates did not significantly affect the overall yields. This phenomenon was probably due to the excellent leaving ability of the acetate group at the 2-position with the assistance of the electron-donating methoxy group at the 4-position (Scheme 5). The push-and-pull mechanism operated so efficiently that the electronic nature of either the A-ring or the B-ring did not significantly impact the yields.

The push-and-pull mechanism of hydrolytic rearrangement.
Conclusions
In summary, we have developed a new method to synthesize isoflavones. A precursor derived from coumarin was cross-coupled with an aryl boronic acid. The resulting product was subsequently hydrolyzed under acidic conditions to forge the final isoflavones. The method allows the synthesis of isoflavones to be assembled in a convergent fashion. Due to the ready availability of commercial boronic acids, it has the advantage of quickly generating a wide array of isoflavones with various B-rings. We anticipate that it would find applications in areas such as medicinal chemistry research related to phytoestrogen.
Experimental section
General
Unless otherwise noted, all reactions were performed in oven-dried round-bottom flasks under argon atmosphere. All ambient temperatures are 25 °C unless otherwise stated. Et2O, THF, and toluene were freshly distilled with sodium/benzophenone ketyl under argon atmosphere. All other reagents were used from commercial sources without further purification, unless otherwise noted. The silica gel (100–200 meshes, Anhui LiangChen Silicon Material Co. Ltd., Huoshan, China) was used for column chromatography. Thin layer chromatography (TLC) was carried out on GF plates (0.25 mm layer thickness, Anhui LiangChen Silicon Material Co. Ltd., Huoshan, China). Visualization was accomplished with UV light (254 nm) and/or by phosphomolybdic acid (50 g/L) in ethanol following heating as developing agents. Yields reported were based on isolated and spectroscopically pure compounds.
1H and 13C NMR spectra were recorded on AVANCE III HD 500 MHz, AVANCE III HD 600 MHz spectrometers (Bruker, Germany) using chloroform-d (CDCl3) as the internal standard (1H NMR, CHCl3 at 7.26 ppm; 13C NMR, CDCl3 at 77.16 ppm) at ambient temperature. 1H NMR data are reported as follows: chemical shift, multiplicity (s = singlet, d = doublet, t = triplet, q = quartet, quint = quintet, sext = sextet, br = broad, m = multiplet), coupling constants (Hz), and integration. HRMS (ESI) was obtained on Thermo LTQ Orbitrap Discovery. IR spectroscopy was performed on a Perkin Elmer Spectrum Two™ spectrophotometer.
General procedure for preparation of isoflavone 1 from 5 (Scheme 6)
A 100-mL three-necked flask was charged with 3-iodo-4-methoxy-2H-chromen-2-yl acetate

Synthesis of isoflavones from
3-Phenyl-4H-chromen-4-one (1aa)

An orange-white solid, yield 73%, m.p. 126.3 °C –128.6 °C.
19
1
H NMR (500 MHz, Chloroform-d) δ 8.33 (dd, J = 8.0, 1.7 Hz, 1H), 8.03 (s, 1H), 7.69 (ddd, J = 8.5, 7.0, 1.7 Hz, 1H), 7.58 (d, J = 7.0 Hz, 2H), 7.49 (d, J = 8.4 Hz, 1H), 7.44 (q, J = 6.4, 5.9 Hz, 3H), 7.39 (t, J = 7.4 Hz, 1H). 13C NMR (126 MHz, CDCl3) δ 176.2, 156.2, 153.1, 133.6, 131.8, 129.0, 128.5, 128.2, 126.4, 125.4, 125.3, 124.6, 118.1. FT-IR(Film)
For more research data, please refer the supplemental material.
Supplemental Material
sj-docx-1-chl-10.1177_17475198261427611 – Supplemental material for Synthesis of isoflavones: A coumarin-based approach
Supplemental material, sj-docx-1-chl-10.1177_17475198261427611 for Synthesis of isoflavones: A coumarin-based approach by Yabin Liu, Liming Xiong, Ankun Zhou, Dongning Bao, Xiaoting Wang, Yonghui He, Xia Zhang and Jianfeng Li in Journal of Chemical Research
Footnotes
Ethical considerations
This study required no ethical approval as it involved neither human participants nor animal subjects.
Consent to participate
This study did not involve human or animal subjects, and no consent to participate was required
Consent for publication
All participants provided consent for the publication of their data in this journal.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was been supported by the following funds: Zhang Xia Expert Workstation of the Yunnan Province (grant number: 202305AF150038), the Open Research Fund of Yunnan Characteristic Plant Extraction Laboratory (grant number: YKKF2024008), the Central Guidance Fund for Local Science and Technology Development (grant number: 202407AB110021), and the Funding from Yunnan University of Chinese Medicine (grant number: BK-30270106806)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data are available in Supplementary file.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.