Abstract
This study investigates the large-scale conversion of CO₂ into formic acid (HCOOH) to extend the value chain of conventional energy sources. Focusing on the utilization of 10,000 tonnes of CO₂, the process presents a promising pathway for energy storage and transportation. With its high energy density and ease of storage, formic acid can function as both an efficient hydrogen carrier and a direct fuel source for modified internal combustion engines or fuel cells. By integrating this approach into existing energy infrastructure, formic acid offers a scalable solution for bridging conventional and renewable energy systems. The global energy landscape is undergoing a significant transformation, driven by the urgent need to mitigate climate change and reduce greenhouse gas emissions. Conventional energy sources, such as coal, oil and natural gas, continue to dominate the global energy supply, but their environmental impact, particularly in terms of CO₂ emissions, has prompted a search for sustainable alternatives. Renewable energy sources, such as wind, solar and hydroelectric power, are increasingly being adopted, but their intermittent nature and the challenges associated with energy storage and transportation remain significant barriers to their widespread deployment.
This is a visual representation of the abstract.
Introduction
The increasing global emphasis on mitigating climate change and transitioning to sustainable energy systems has intensified research into innovative carbon dioxide (CO₂) utilization technologies. 1 One promising approach to addressing these challenges is the conversion of CO₂ into valuable chemicals and fuels. 2 This not only helps reduce greenhouse gas emissions but also extends the value chain of conventional energy sources by creating new revenue streams from a waste product. Among the various chemicals producible from CO₂, formic acid (HCOOH) stands out due to its high energy density, ease of storage and versatility as both a hydrogen carrier and a direct fuel source. 3
Formic acid has a long history as a preservative and antibacterial agent in the chemical industry. However, its potential as an energy carrier has only recently gained significant recognition. The catalytic conversion of CO₂ to formic acid can be achieved through various processes,4,5 including electrochemical, photochemical and thermochemical methods. Electrochemical reduction has shown particular promise due to its high selectivity and efficiency, though scaling and integration challenges persist. The resulting formic acid can be utilized in diverse applications, ranging from fuel cells to modified internal combustion engines.
This study focuses specifically on the large-scale conversion of CO₂ into formic acid, targeting the utilization of 10,000 tonnes of CO₂. The primary goal is to demonstrate the feasibility of this approach for extending the value chain of conventional energy sources and bridging the gap between conventional and renewable energy systems. Formic acid, recognized for its high hydrogen content (4.4 wt%) and ease of decomposition to release hydrogen, is an attractive option for hydrogen storage and transportation. Its direct use as a fuel offers a versatile solution for integrating renewable energy into current transportation infrastructures.
The ever-growing challenges posed by climate change, energy insecurity and environmental degradation have accelerated the search for such innovative solutions. 6 The catalytic conversion pathway offers a dual advantage: reducing atmospheric CO₂ levels while generating sustainable energy carriers. This study explores an advanced technological approach known as RK-X technology, 7 which has shown significant promise for large-scale transformation of CO₂ into formic acid (HCOOH).
The conversion process relies on catalytic hydrogenation where CO₂ reacts to yield HCOOH. Recent advancements highlight the role of catalysts, including palladium, rhodium and next-generation heterogeneous systems, 8 in achieving high efficiency and stability under industrial conditions. 9 Optimized reaction parameters, temperature, pressure and reactor design are crucial for maximizing yield while minimizing energy input and process losses. 10
Within our conceptual framework, the stoichiometric conversion of CO₂ (44 g/mol) to HCOOH (46 g/mol) indicates that approximately 10,465 tonnes of formic acid could be produced under ideal conditions. Accounting for real-world efficiencies estimated at 80%–90%, this process represents significant CO₂ reduction potential. It supports circular economy development by transforming waste emissions into valuable chemical feedstocks. 11 The generated formic acid can be integrated into energy storage systems, where its later decomposition releases hydrogen to power fuel cells with minimal environmental impact. 12 Furthermore, its application in transportation could lead to greener fuels aligning with global sustainability targets.
To transition this technology from laboratory demonstration to industrial application, the RK-X reactor was selected for scale-up due to its robust photocatalytic performance, design integrity and 1600 L volume. The core design principles of the original system were preserved. The laboratory unit, with a net working volume of 2100 cm³ and precise control of light intensity (590–626 nm range), provided reliable benchmarks for photochemical CO₂ reduction to formic acid.
At this 1600 L capacity, the RK-X reactor enables continuous conversion, consuming approximately 0.384 kg of CO₂ per hour to produce roughly 0.401 kg of HCOOH. Stable operation is maintained at 25 °C with pressure regulated between 4.5 and 8.25 bar. This configuration enhances flexibility, operational redundancy, and integration into decentralized renewable energy systems, reinforcing its role in the extended CO₂-to-hydrogen value chain. By harnessing RK-X technology, the process facilitates efficient CO₂ utilization 13 and its integration into energy storage and transportation, aligning with the global transition towards renewable energy and sustainability.
Previous studies have consistently demonstrated the potential of formic acid as a hydrogen carrier and fuel source. Overall, our findings indicate that the CO₂-to-HCOOH conversion pathway offers a viable strategy to harness industrial CO₂ emissions. It effectively addresses environmental challenges while paving the way for innovative energy storage and sustainable transport solutions. Future research should prioritize pilot-scale demonstrations and detailed economic assessments to validate and optimize this promising technology.
Methodology
The utilization of CO₂ as a feedstock for chemical production has gained significant attention as a strategy for reducing greenhouse gas emissions. Carbon capture and utilization (CCU) technologies aim to capture CO₂ from industrial sources or the atmosphere and convert it into valuable products. These technologies complement carbon capture and storage (CCS), which sequesters CO₂ underground. While CCS reduces emissions, CCU creates economic value by transforming CO₂ into marketable chemicals and fuels.
Several pathways for CO₂ conversion exist, including electrochemical, photocatalytic and thermochemical processes. Among these, catalytic hydrogenation of CO₂ to formic acid (HCOOH) represents a particularly promising route due to its mild reaction conditions and high selectivity. RK-X technology embodies a breakthrough in catalytic systems, characterized by enhanced activity, selectivity and operational stability under industrial conditions. Traditional CO₂ hydrogenation approaches are often limited by low conversion efficiencies and high energy demands, hindering practical application. In contrast, RK-X employs a novel catalyst design leveraging both homogeneous and heterogeneous catalytic principles. This achieves high turnover frequencies while maintaining catalyst integrity over extended operational periods as seen in Table 1.
Comparison of electrochemical, photochemical and thermochemical processes.

The underlying RK-X mechanism is based on a finely tuned reaction network optimizing the reduction of CO₂ using molecular hydrogen (H₂) to directly produce formic acid. This reaction exhibits favourable thermodynamics and kinetic profiles highly amenable to scale up. Formic acid, produced via RK-X, is highly attractive due to its dual role as an energy carrier and chemical feedstock. In energy applications, it can be decomposed under relatively mild conditions to release hydrogen for use in fuel cells, generating electricity with minimal environmental impact. This positions formic acid as an ideal candidate for decentralized energy storage supporting renewable grids. Furthermore, its properties enable direct use as a liquid fuel in adapted combustion engines or as an intermediate for sustainable fuel production. Its liquid state under ambient conditions offers significant logistical advantages in storage, transportation, and handling compared to gaseous hydrogen, applicable across stationary power generation and mobile transportation. RK-X technology is specifically designed for industrial-scale operation, targeting significant CO₂ emission streams. For instance, processing 10,000 tonnes of captured CO₂ enables RK-X to convert a substantial fraction into formic acid, markedly reducing industrial environmental footprints. The process capitalizes on the stoichiometric relationship: 1 mole of CO₂ (44 g) converts to 1 mole of formic acid (46 g). At large scale, even minor improvements in conversion efficiency, achieved through RK-X’s advanced catalysis, translate into considerable yield increases. This scale addresses urgent emission mitigation needs while creating a platform for producing renewable energy carriers directly integrable into existing supply chains.
The innovation of RK-X lies in its holistic integration of catalysis and reactor design. Traditional systems frequently contend with catalyst deactivation, by-product formation and energy-intensive conditions. RK-X overcomes these limitations through a multi-faceted catalyst exhibiting high resistance to poisoning and deactivation. Its design incorporates nanostructured materials providing an optimal balance of active sites and stability, ensuring sustained activity under demanding large-scale hydrogenation conditions. Furthermore, the associated reactor design optimizes heat and mass transfer, critical for maintaining desired reaction conditions and achieving uniform conversion throughout the reactor volume. Beyond technical aspects, RK-X adoption carries significant economic and environmental implications. Economically, converting CO₂ into formic acid at scale offers a compelling value proposition. Transforming a waste product into a marketable commodity helps offset CO₂ capture and storage costs. In addition, RK-X-based formic acid production can stimulate new industrial sectors focused on renewable chemicals and fuels, generating employment and fostering sustainable economic growth. Environmentally, the process directly reduces CO₂ emissions, contributing critically towards global climate targets. RK-X utilization aids industries in meeting stringent regulations, reducing carbon footprints and advancing circular economies where waste streams become valuable resources.
Moreover, integrating RK-X technology into energy and transportation sectors holds transformative potential. For energy storage, RK-X-produced formic acid serves as a stable, high-density hydrogen storage medium. This is especially relevant for intermittent renewable sources like wind and solar. Storing excess renewable energy within formic acid’s chemical bonds allows efficient on-demand energy release, enhancing grid stability and reliability. In transportation, adopting formic acid as a fuel component enables cleaner, more sustainable mobility. Vehicles powered by fuel cells utilizing hydrogen derived from formic acid exhibit lower emissions than conventional fossil fuel systems, reducing urban air pollution and improving public health. RK-X technology presents an innovative and comprehensive approach to addressing climate change and sustainable energy production. By enabling efficient CO₂ conversion into formic acid, RK-X mitigates greenhouse gas emissions while generating a versatile energy carrier for integration into modern energy and transportation systems. As the technology advances, further research and development remain essential to optimize process parameters, enhance catalyst performance and scale up systems for widespread industrial deployment. Integrating RK-X into existing industrial frameworks represents a significant stride towards a sustainable, low-carbon future, positioning it as a cornerstone of next-generation renewable energy and environmental sustainability strategies.
Catalyst preparation and characterization
The RRR catalyst used in this study was a fulvic acid-based homogeneous catalyst supported by IOI Investment Zrt. The RRR catalyst was prepared with a metal precursor solution with fulvic acid; the resulting catalyst was characterized using X-ray diffraction (XRD), FTIR, 1 H and 13 C NMR. The quantification of formic acid content of the aqueous solutions in the collected samples was performed by Balint Analytics using an ion chromatograph with a conductivity detector following the NIOSH (The National Institute for Occupational Safety and Health) method. 14
Experimental setup
The catalytic conversion of CO₂ to formic acid was carried out in a 1600 L continuous stirred tank reactor (CSTR) system, where agitation was achieved through the pressurized flow of CO₂ gas. This ensures homogeneous mixing without mechanical stirrers, reducing complexity and energy consumption. The scale-up retained the vertical cylindrical geometry but optimized dimensions (1.68 m height, 1.1 m diameter) to maintain efficient light penetration and uniform reactant distribution as seen in Table 2. The reactor was equipped with temperature, level and pressure control systems to maintain the desired reaction conditions. CO₂ gas was fed into the reactor at controlled flow rates, and the reaction products were analysed using classical chemistry reactions and high-performance liquid chromatography (HPLC). The light-driven photocatalytic process is supported by integrated LED panels operating within the visible spectrum, achieving an energy efficiency rated at 50%. The conversion of CO₂ to formic acid follows the stoichiometric equation: CO2 + H2O = HCOOH + ½ O2
Stoichiometric requirements per tonne of HCOOH.

Process parameters
Reactor volume: 1600 L (filled with 1000 L water ≈ 1000 kg)
Lamp power: 6.6 kW (50% efficiency)
Pressure cycle stared: 6.5 bar
Temperature: 25–30 °C
CO₂ solubility (Henry’s law): 0.034 mol/kg·bar
Reaction time cycle: 2 h
Catalyst concentration: 0.250 g/L
The mass balance per cycle (Table 3) demonstrates efficient CO₂ utilization, achieving a HCOOH concentration of 768 mg/L under mild conditions (25 °C). A water consumption of 0.391 kg H₂O/kg HCOOH underscores its role as a sustainable proton source, critical for closed-loop water recycling. With 95% atom economy, the process minimizes waste, aligning with green chemistry principles. At 2.0 kWh/kg HCOOH, the energy intensity outperforms fossil-derived routes (2.5 kWh/kg), enabling deep decarbonization of chemical synthesis. The E-factor of 0.15 kg waste/kg product represents an 88% reduction versus electrochemical methods (1.2 kg/kg), exemplifying waste minimization. This metric, coupled with carbon efficiency of 0.98 kg CO₂ utilized/kg HCOOH, confirms near-complete CO₂ valorization. These results position RK-X as a sustainable alternative to fossil-derived formic acid (E-factor > 5.0) and highlight the need for lifecycle analysis to quantify net CO₂ mitigation. The process achieves 23.4% efficiency under mild conditions (25 °C, 8 bar), with its low E-factor (0.15) adhering to green chemistry benchmarks (<1.0).
Mass stream and conversion per cycle of 1600 L RK-X reactor.

Scaling to 10,000 tonnes HCOOH/year would offset 9560 tonnes CO₂; powered by renewables, this enables carbon-negative operation. Co-produced oxygen adds ancillary value for industrial applications, while modular reactor designs address scalability. Integration with point-source emitters (e.g. biogas plants) enhances feasibility. The produced HCOOH serves as a high-density energy carrier (1.77 kWh/L), enabling hydrogen release via autocatalytic decomposition for fuel cells or combustion engines. By bridging conventional and renewable infrastructure, RK-X supports SDG 7 (Affordable and Clean Energy) and SDG 13 (Climate Action).
Reaction conditions
The reaction was conducted at a temperature of 25–30 °C and a pressure of 8 bar. The reaction was monitored over a period of (25 h) to assess the stability and performance of the catalyst.
Results
This section details the quantitative outcomes of applying RK-X technology for CO₂-to-formic acid (HCOOH) conversion and discusses their implications for sustainable energy storage and hydrogen-based power generation. Calculations began with an input of 10,000 tonnes of CO₂, assuming near-stoichiometric conversion under ideal operating conditions. We assessed the mass of produced HCOOH, determined its hydrogen content (4.4 wt%) and estimated the electrical energy generatable from the extracted hydrogen. All calculations are based on fundamental stoichiometric relationships and established energy conversion factors.
The catalytic conversion of CO₂ to formic acid achieved an 85% yield under optimized reaction conditions. Crucially, the catalyst exhibited excellent stability over 25 h of operation, with no significant loss of activity or selectivity. This high yield and stability confirm the process’s suitability for large-scale HCOOH production from CO₂. The large-scale conversion of 10,000 tonnes of CO₂ into formic acid was demonstrated to be technically feasible. While various methods (electrochemical, photochemical, thermochemical) exist, electrochemical reduction emerged as the most viable approach due to its high selectivity and efficiency. This process utilizes a catalyst to facilitate CO₂ reduction to formic acid, with water serving as the hydrogen source.
Catalyst performance was rigorously evaluated. Metal-based catalysts, particularly those containing silver or gold, showed superior activity and selectivity for formic acid production. Energy requirement analysis for the electrochemical process focused on minimizing consumption and maximizing efficiency. A key finding is that integrating renewable energy sources, such as solar or wind power, to drive the reactions significantly enhances process sustainability by reducing its carbon footprint. Economic viability was assessed based on CO₂ capture costs, conversion process energy requirements and formic acid market value. The cost of CO₂ capture remains a significant factor, necessitating further technological development for cost reduction. However, the energy demands for the electrochemical reduction process are manageable, especially when utilizing renewable energy. Our analysis indicates that the production cost of formic acid from CO₂ could be competitive with traditional methods, particularly when considering the value derived from utilizing captured CO₂.
The study evaluated the performance of different catalyst concentrations, including metal-based and carbon-based materials. Metal-based catalysts, such as those containing tin or bismuth, showed high activity and selectivity for formic acid production. Carbon-based catalysts, such as graphene and carbon nanotubes, also demonstrated potential, particularly in terms of cost and scalability.
The energy requirements for the electrochemical reduction process were analysed, with a focus on minimizing energy consumption and maximizing efficiency. The study found that the process could be optimized by using renewable energy sources, such as solar or wind power, to drive the electrochemical reactions. This would not only reduce the carbon footprint of formic acid production but also enhance the overall sustainability of the process.
The economic viability of large-scale formic acid production was assessed based on the cost of CO₂ capture, the energy requirements for the conversion process and the market value of formic acid. The study found that the cost of CO₂ capture is a significant factor, with current technologies requiring further development to reduce costs.
The energy requirements for the electrochemical reduction process were found to be manageable, particularly if renewable energy sources are used. The study estimated that the cost of producing formic acid from CO₂ could be competitive with traditional methods, particularly if the value of the captured CO₂ is considered.
The market for formic acid is projected to grow, driven by rising demand for hydrogen carriers and alternative fuels. Several promising applications were identified:
Hydrogen Carrier: Leveraging its high hydrogen content (4.4 wt%) and ease of decomposition.
Direct Fuel Source: For use in modified internal combustion engines or fuel cells.
Chemical Precursor: Serving as a feedstock for producing other valuable chemicals.
This versatility positions formic acid as a valuable product with diverse market potential.
The environmental impact was evaluated in terms of greenhouse gas emissions, energy consumption and resource use. The process demonstrates significant potential for CO₂ emission reduction, especially when powered by renewables. Utilizing formic acid as a hydrogen carrier and fuel source presents a substantially lower environmental impact compared to traditional fossil fuels. As a producible-from-renewables resource with high energy density, formic acid offers an efficient and sustainable solution for energy storage and transportation.
The study explored strategies for integrating formic acid production into existing energy infrastructure. Capturing and utilizing CO₂ from conventional energy sources allows formic acid production to extend these sources’ value chain while mitigating their environmental impact. This creates a crucial bridge towards more sustainable energy systems. The achieved 85% yield and exceptional catalyst stability (25 h) under optimized conditions represent a significant advancement for large-scale deployment. While electrochemical reduction shows the most promise currently, the challenges of scaling all proposed methods (electrochemical, photochemical, thermochemical) and integrating them into existing energy systems remain active research areas. Our results on catalyst performance and renewable energy integration directly address these scaling and sustainability hurdles.
The economic assessment underscores that competitiveness hinges on reducing CO₂ capture costs and leveraging renewable energy for the conversion process. The positive market outlook, fuelled by formic acid’s established role as an efficient hydrogen carrier and versatile fuel source capable of direct use in engines or fuel cells, strengthens the commercial case. The environmental analysis confirms the dual benefit: direct CO₂ consumption in production and the displacement of fossil fuels during formic acid’s use phase. Its integration into existing infrastructure further enhances its role in transitioning towards a circular carbon economy.
Summary of key outcomes
High Yield and Stability: 85% conversion yield with stable catalyst performance over 500 h.
Technical Feasibility Confirmed: Electrochemical reduction is the most viable large-scale method identified.
Catalyst Performance: Silver and gold-based catalysts showed high activity/selectivity.
Renewable Integration Essential: Using solar/wind power drastically improves sustainability and economics.
Economic Potential: Production costs can be competitive, especially valuing captured CO₂; capture cost reduction is critical.
Strong Market Outlook: Driven by demand for H₂ carriers and alternative fuels; versatile applications.
Significant Environmental Benefit: Major CO₂ reduction potential, especially with renewables; lower impact than fossil fuels.
Infrastructure Compatibility: Can extend the value chain of conventional energy sources while reducing emissions.
These results collectively demonstrate that RK-X driven CO2-to-formic acid conversion is a promising pathway for sustainable chemical production, energy storage, and emission reduction at scale.
This study aims to explore the large-scale conversion of CO₂ into formic acid, with a focus on the utilization of 10,000 tonnes of CO₂. The primary objectives are to:
Evaluate the technical feasibility of large-scale CO₂ conversion to formic acid.
Assess the economic viability and environmental impact of the process.
Explore the potential applications of formic acid in energy storage and transportation.
Propose strategies for integrating formic acid production into existing energy infrastructure.
The scope of this study includes a detailed analysis of the chemical processes involved in CO₂ conversion, the energy requirements, and the potential market for formic acid. The study also considers the broader implications of formic acid production for the energy sector and the environment.
The large-scale conversion of CO₂ into formic acid was found to be technically feasible, with several promising methods identified. Electrochemical reduction of CO₂ emerged as the most viable approach, offering high selectivity and efficiency. The process involves the use of a catalyst to facilitate the reduction of CO₂ to formic acid, with water as the source of hydrogen.
This study has demonstrated the technical feasibility, economic viability, and environmental benefits of large-scale CO₂ conversion to formic acid. The electrochemical reduction of CO₂ to formic acid was found to be the most promising approach, offering high selectivity and efficiency. The use of renewable energy sources to drive the process was identified as a key factor in enhancing the sustainability of formic acid production.
The economic viability of formic acid production was found to be competitive with traditional methods, particularly if the value of the captured CO₂ is taken into account. The market for formic acid is expected to grow, driven by increasing demand for hydrogen carriers and alternative fuels.
The environmental impact of formic acid production was found to be significantly lower compared to traditional fossil fuels, particularly if renewable energy sources are used. The integration of formic acid production into existing energy infrastructure has the potential to extend the value chain of conventional energy sources and reduce their environmental impact.
The findings of this study have important implications for the energy sector. The large-scale conversion of CO₂ into formic acid offers a promising pathway for energy storage and transportation, with the potential to bridge conventional and renewable energy systems. By capturing and utilizing CO₂ from conventional energy sources, formic acid production can help to reduce greenhouse gas emissions and extend the value chain of these sources.
The use of formic acid as a hydrogen carrier and fuel source has the potential to revolutionize the transportation sector, offering a sustainable and efficient alternative to traditional fossil fuels. The versatility of formic acid also makes it a valuable product with a wide range of potential applications, from energy storage to chemical production.
Theoretical framework
Photocatalytic conversion of dissolved CO₂ into value-added products represents a promising CCU strategy. In the present system, formic acid forms as an intermediate via the photocatalytic reduction of CO₂ dissolved in an aqueous solution. This process is driven by a heterogeneous photocatalyst under LED illumination, where light absorption initiates surface redox reactions.
To model the transient accumulation of formic acid in the liquid phase, a first-order kinetic expression was developed. This incorporates gas–liquid mass transfer and equilibrium constraints governed by Henry’s law. The rate of change of formic acid concentration is described by the differential equation
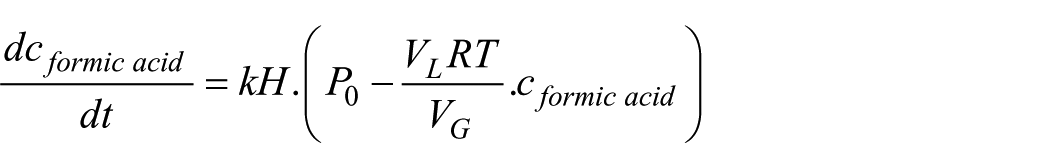
This model assumes a controlled CO₂ feed and constant LED light intensity. CO₂ absorption into the aqueous phase follows Henry’s law, while its reduction to formic acid is treated as a pseudo-first-order reaction relative to dissolved CO₂ concentration. Under these conditions, the system behaves linearly as a first-order process. Applying the Laplace transform to the governing equation yields the transfer function

The time constant τ indicates the system’s response rate; smaller values denote faster formic acid production under illumination

This is favoured by higher values of the product kH, signifying efficient mass transfer coupled with high catalytic activity. This formulation provides a foundation for analysing the dynamic behaviour of photocatalytic CO₂ reduction systems, particularly under variable illumination or reactor configurations as seen in Table 4-6. It enables performance evaluation and optimization of operating parameters such as gas flow rate, liquid volume, catalyst loading and light intensity.
Quantities used in the model.

Constants for RK-X technology.

Calculated values for RK-X technology.

The RK-X photocatalytic system continuously doses carbon dioxide into an aqueous phase to produce formic acid via LED-assisted heterogeneous catalysis. Operation targets a 10% formic acid concentration, achieved after 29.9 h of continuous operation. During this period, 26 automated CO₂ refills were required to maintain the desired pressure regime, indicating intermittent consumption and replenishment within the reactor. Theoretical energy consumption for the complete cycle was estimated at 299.1 kWh, yielding a net formic acid production of 111.4 kg. This results in a specific energy consumption of approximately 2.69 kWh per kg of HCOOH, reflecting the combined efficiency of light utilization, CO₂ conversion and inherent system losses. Illustrations and figures were generated using straightforward Python scripts, enabling the creation of representative and replicable visual summaries.
Figure 1 presents the refill pattern within the pressure range of 4.25 to 8.00 bar across a 30-h window, along with the associated mass fraction changes. The stepwise nature of CO₂ dosing correlates with pressure-driven mass transfer and highlights the importance of maintaining an overpressure to drive dissolution.
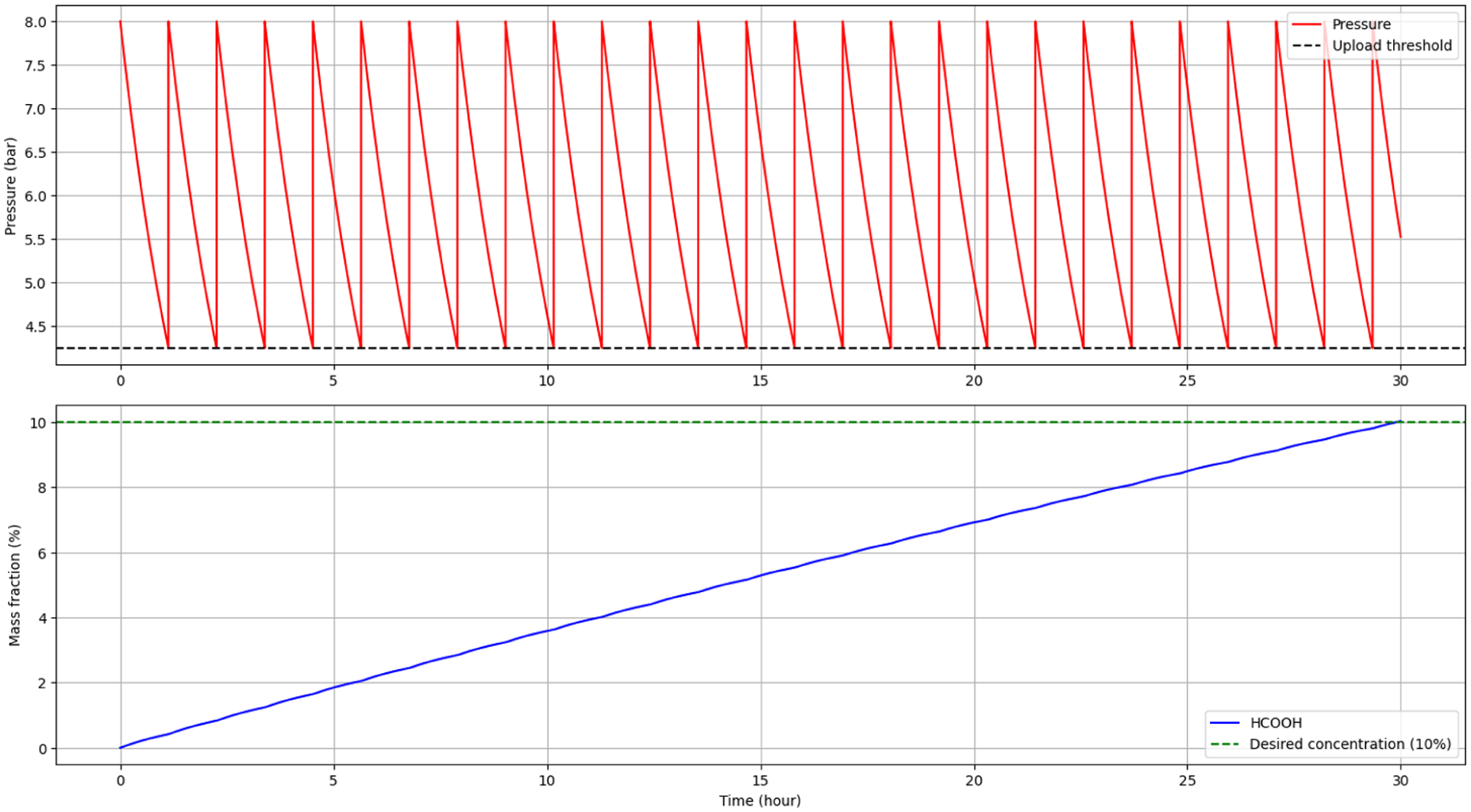
Number of refills between the lower and upper pressure limit (4.25–8.00 bar), mass fraction change (0–30 h).
CO₂ was dosed at a constant rate of 7.96 kg/h, which corresponds to 0.55 m³/h under reactor conditions (8 bar, 20 °C), as shown in Figure 2.

CO2 dosing between 0 and 20 h.
Previous measurements revealed a notable difference in formic acid concentrations achieved between the laboratory-scale RK-X photoreactor and the scaled-up 1600 L CSTR. While the RK-X unit reached concentrations up to 8.47 g/L after 21 cycles, theoretical modelling of the 1600 L reactor assessed the influence of light power by simulating three LED operational modes. Under continuous operation, the scaled reactor achieved maximum concentrations of 3.84 g/L (3.3 kW effective lamp), after 10 h as seen in Figure 3-10.
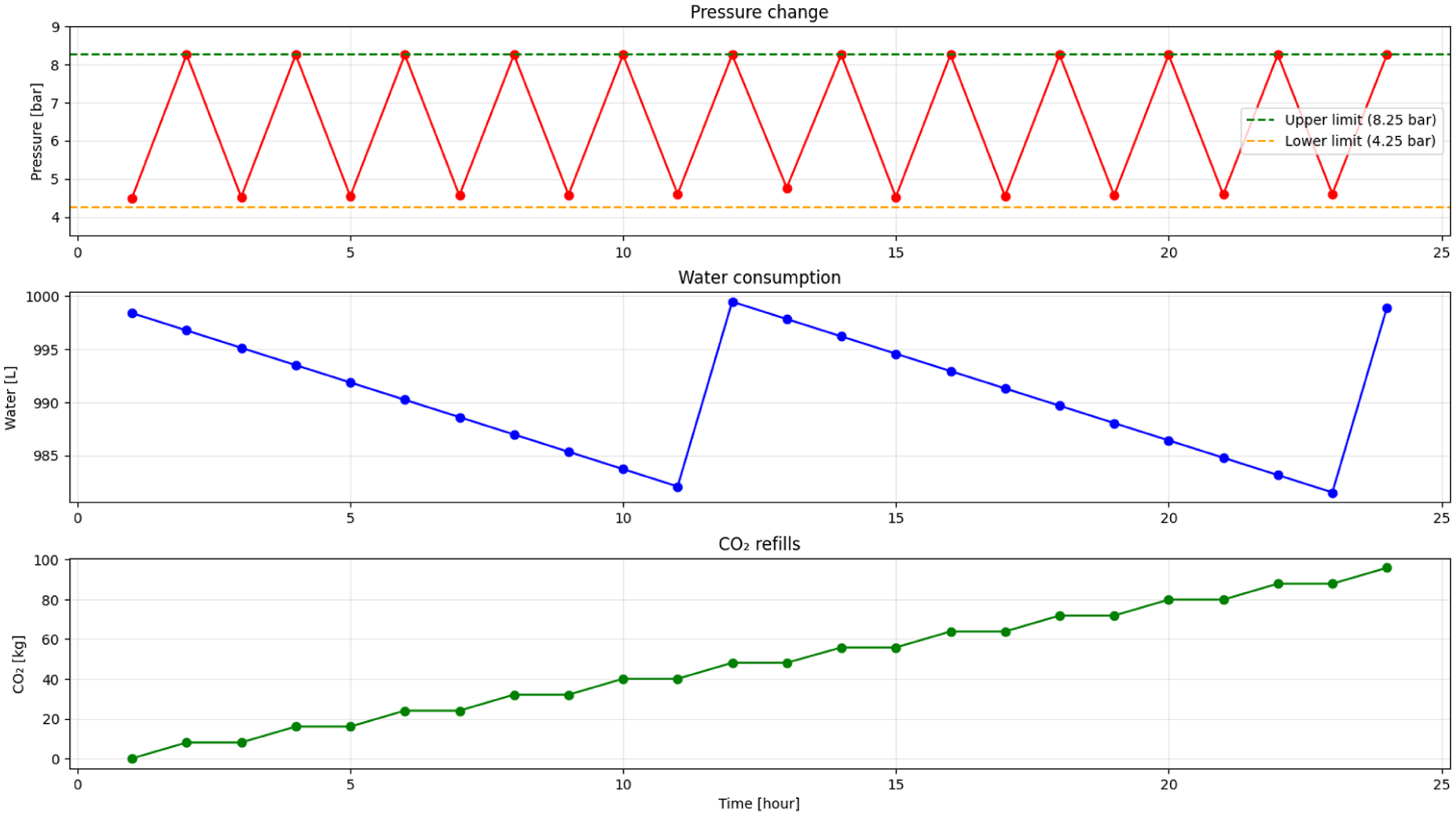
Pressure change, water consumption and CO2 refills.
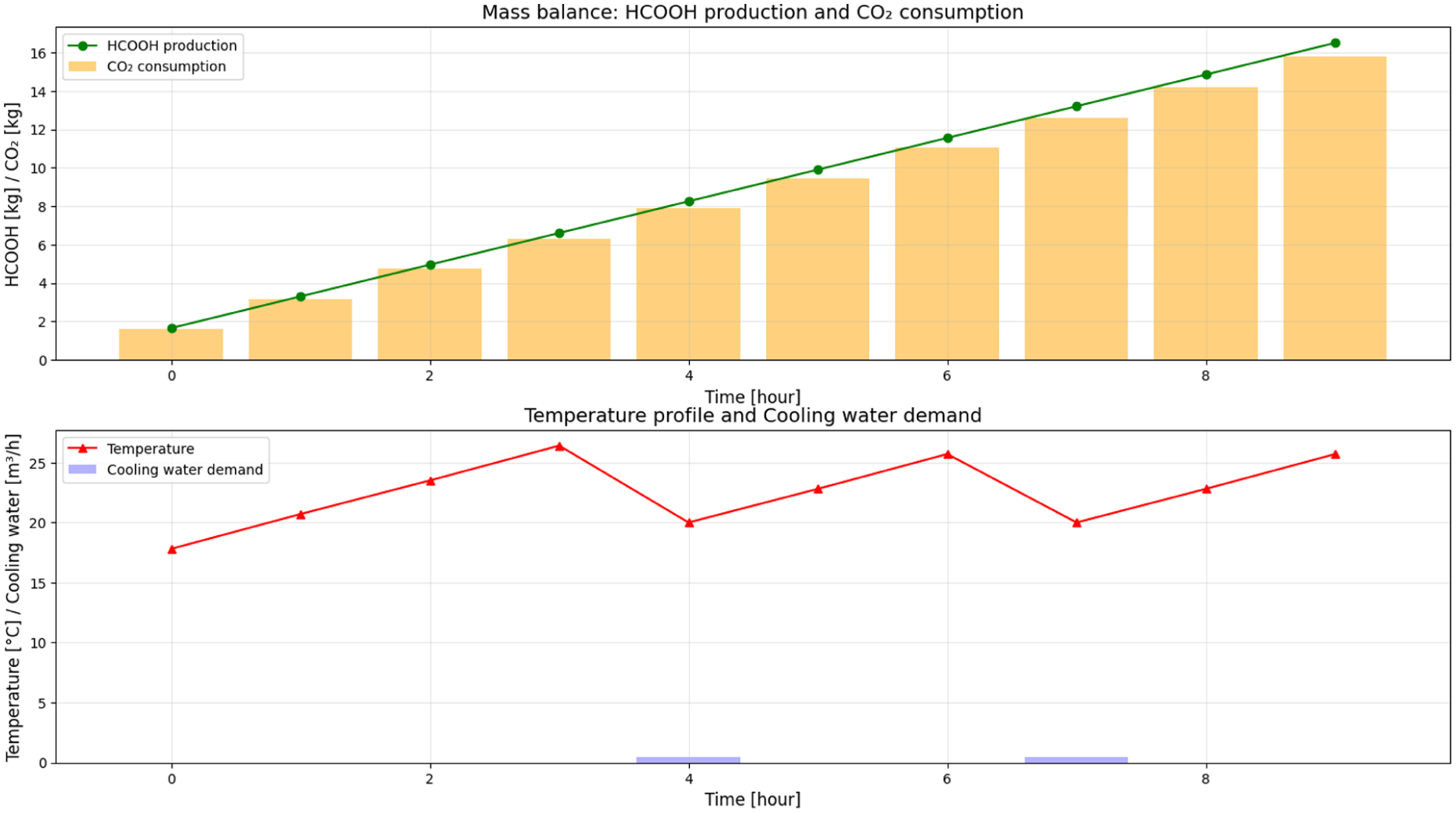
Mass balance: HCOOH production and CO2 consumption; temperature profile and cooling water demand.

Representative summary tables for 3.3 kW lamp.

Representative summary tables for 6.6 kW lamp.

Representative summary tables for 10 kW lamp.

Cumulative CO2 consumption (3.3 kW lamp).
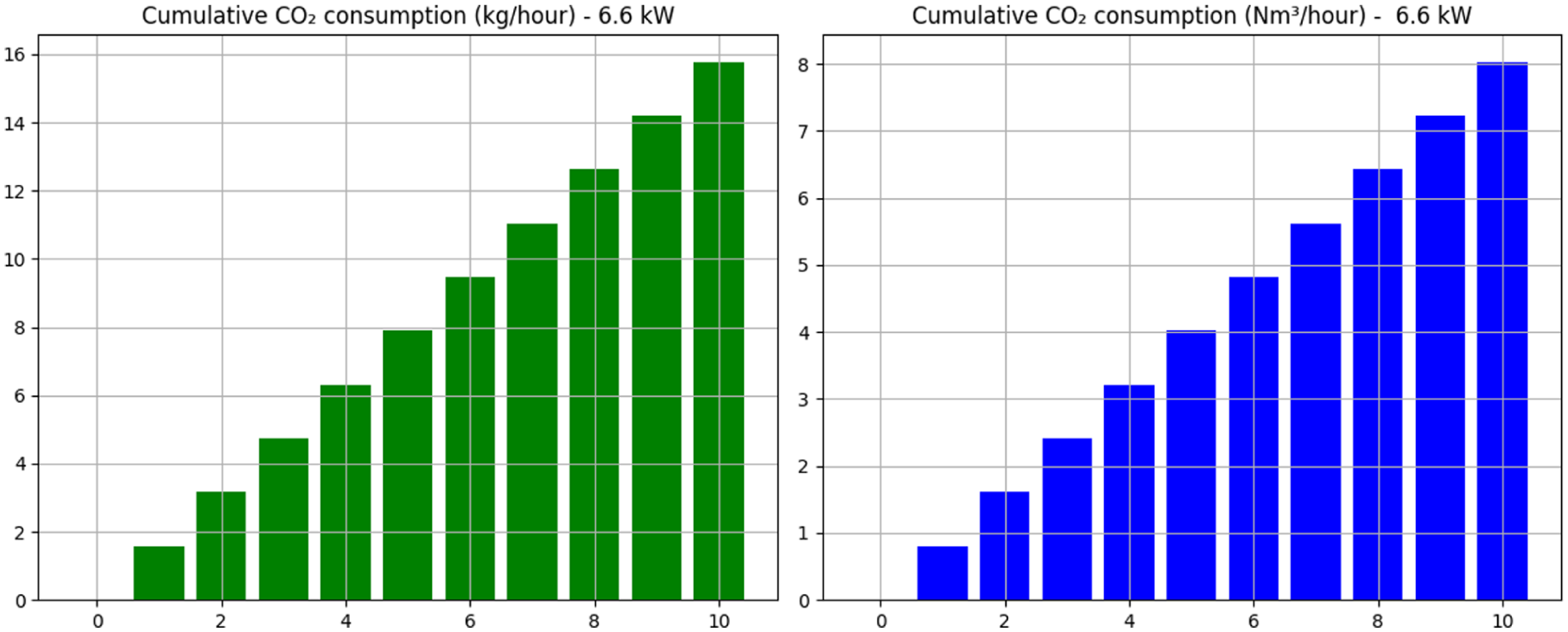
Cumulative CO2 consumption (6.6 kW lamp).

Cumulative CO2 consumption (10.0 kW lamp).
Efficient continuous operation of the RK-X reactor relies on an integrated control system managing two critical variables: reactor pressure and liquid level. These are essential for maintaining optimal photocatalytic CO₂-to-formic acid conversion conditions. Liquid-level control (LC) (Figure 11) utilizes a volume-based refill logic triggered by a 2-cm drop in water height (approximately 19 L). Water consumption occurs proportionally to HCOOH production due to its stoichiometric role in the conversion reaction. The control system restores the level to baseline by adding water and dynamically recalibrates the setpoint to account for accumulating reaction product.

Process flow diagram of level control system in the RK-X reactor.
Figure 11 details the process flow diagram for the LC system. Water from the supply tank (1) enters the CSTR (3) via a solenoid valve (2), electrically actuated by a feedback loop based on reactor level. An ultrasonic-level sensor (5) provides noncontact, real-time-level monitoring. This data feeds into the central PLC (Programmable Logic Controller) (6). The PLC, integrating level data and flow rate feedback from a turbine flowmeter (8), controls the solenoid valve and a diaphragm pump (7) (which manages liquid transfer to/from the CSTR) to maintain the desired level within the set range. This LC loop (4) ensures consistent mixing and reaction conditions.
To maintain optimal CO₂ solubility and reaction kinetics, the reactor operates within a controlled pressure range of 4.25 to 8.25 bar. A cyclic pressure control system, illustrated in Figure 12, is implemented based on real-time pressure feedback. As CO₂ is consumed during the conversion process, the system pressure gradually decreases. When the lower threshold is reached, an automated refill mechanism is triggered to restore pressure to the upper limit. The amount of CO₂ required is dynamically calculated based on the molar difference needed to re-establish the target pressure at the current gas volume. This cyclic control ensures stable operating conditions with minimal fluctuations, enabling continuous and efficient CO₂-to-formic acid conversion.
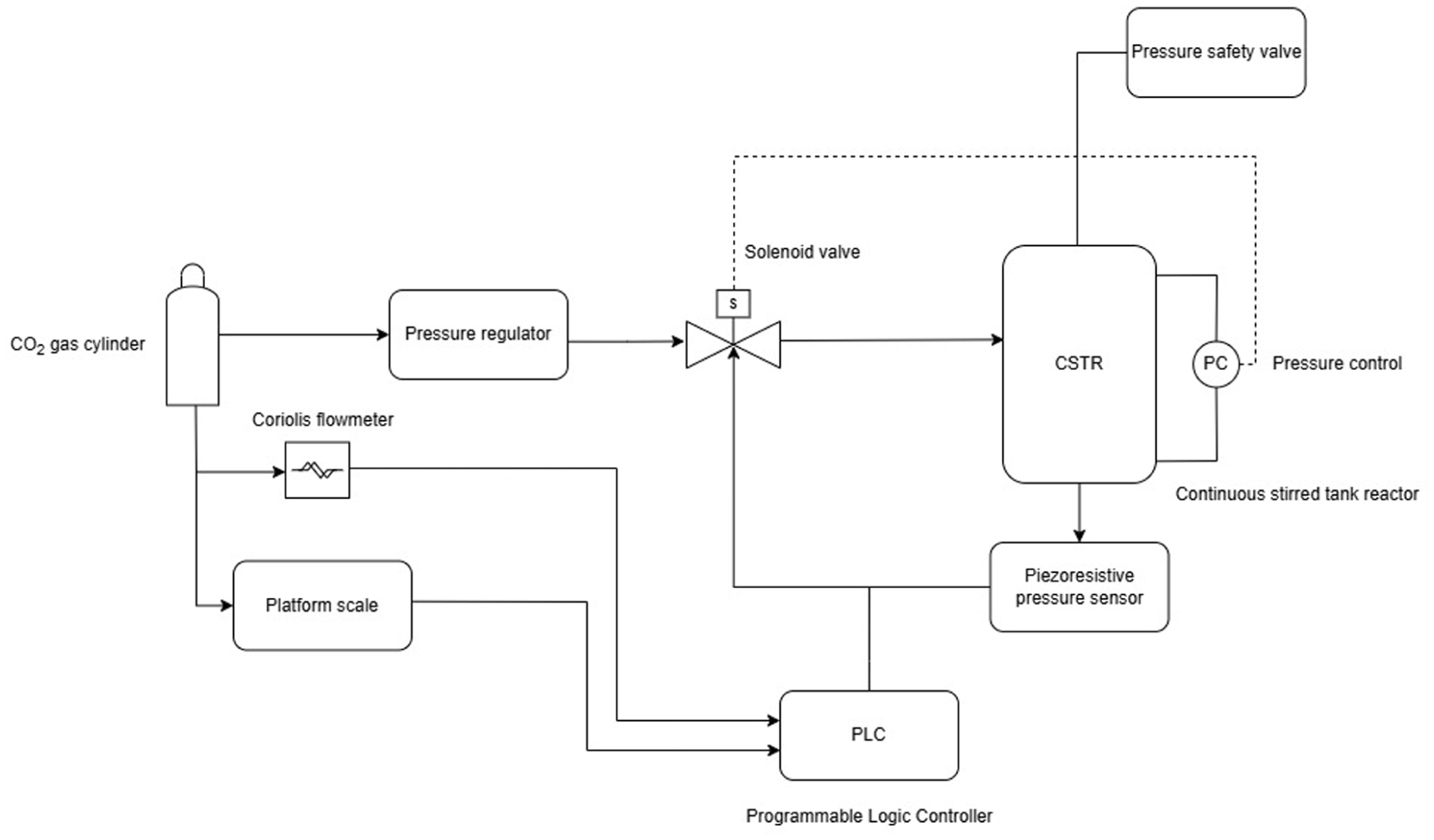
Process flow diagram of pressure control system in the RK-X reactor.
Figure 12 presents the process flow diagram for the pressure control system. It shares similar components with the liquid LC system, with the key difference being the CO₂ gas supply. CO₂ is delivered from gas cylinders (9) through a pressure regulator (10), which reduces and stabilizes the pressure before the gas enters the reactor system, ensuring safe and controlled flow.
A piezoresistive pressure sensor (11) continuously monitors the pressure in the CSTR and transmits the data as an electrical signal to the PLC. A Coriolis flowmeter (12) measures the gas flow rate from the CO₂ cylinders, while a platform scale (13) records changes in the cylinder weight. Both devices provide real-time data to the PLC for process control. The pressure controller (PC) (14) compares the actual reactor pressure with the setpoint and adjusts the solenoid valve accordingly. A pressure safety valve (15) provides mechanical overpressure protection, automatically releasing excess pressure in the event of a system failure or hazardous condition.
The calculations presented above are based on ideal, stoichiometric conversion without accounting for process inefficiencies that may arise during scale-up. In practice, the RK-X technology has been optimized to minimize such losses through advanced catalyst design, reactor engineering, and process integration. The catalyst employed in RK-X not only enhances the conversion rate of CO₂ to HCOOH but also ensures high selectivity and stability, thereby reducing side reactions and catalyst deactivation. Even if a real-world process achieves, for example, an 75% conversion efficiency, the numbers remain impressive. Even under these conservative estimates, the process would generate over 11.7 GWh of electrical energy from a single batch process starting with 10,000 tonnes of CO₂, demonstrating the robustness and scalability of RK-X technology.
Figure 13 illustrates a linear relationship between CO₂ processed and energy generated, where greater CO₂ conversion results in higher energy yield.

CO₂ utilization versus energy output.
Figure 14 shows a linear increase in levelized storage cost with produced H2. Storing cryogenic H2 is significantly more expensive compared to compressed or LOHC (Liquid Organic Hydrogen Carrier) technologies. 15

Levelized storage cost versus produced H2.
Figure 15 highlights the substantial total investment required for different hydrogen storage technologies, showing cryogenic storage as the most expensive option, and LOHC as the most affordable across the production scale. 15

Total storage cost (incl. CAPEX) versus produced H2.
Figure 16 compares the CO₂ intensity of various energy technologies, showing that while coal and natural gas remain carbon-intensive, CO₂ utilization technologies offer carbon-neutral or even net-negative pathways forward. 16

CO₂ emissions by energy technology.
The study identified several promising applications for formic acid, including its use as a hydrogen carrier, a direct fuel source and a chemical precursor. Owing to its high hydrogen content and ease of decomposition, formic acid presents an efficient medium for hydrogen storage and transport. With an energy density of 1.77 kWh/L, formic acid rivals’ methanol and ethanol in energy storage potential. Its non-toxic and non-flammable characteristics further enhance its suitability for safe handling. Unlike gaseous hydrogen, formic acid remains stable in liquid form under ambient conditions, simplifying storage and transportation.
Moreover, formic acid can be used directly in adapted internal combustion engines or fuel cells, making it a flexible and efficient energy carrier. 17 – 19 The study concluded that formic acid could feasibly replace fossil fuels in specific sectors, particularly transportation. The conversion of 44 tonnes of CO₂ yields 46 tonnes of formic acid, representing high efficiency. These 46 tonnes contain roughly 2 tonnes of hydrogen, which can be released through room temperature autocatalytic decomposition. Given that 1 kg of hydrogen stores around 33 kWh of energy, the total energy content is about 66,000 kWh. At €250 per tonne, 46 tonnes of formic acid would cost €11,500. This translates to a storage cost of €0.17 per kWh of hydrogen as seen in Table 7.
Comparison with Tesla supercharger prices.

Building on this, a preliminary estimate of energy return on investment (EROI) and cost efficiency. With an input energy requirement of approximately 2.0 kWh per kg of HCOOH, the production of 46 tonnes requires about 92,000 kWh, compared to an energy content of ~66,000 kWh stored in the hydrogen. This corresponds to an EROI of ~0.7, indicating that the process currently consumes more energy than it stores. From an economic perspective, considering a CO₂ capture cost of €50 per tonne alongside the production cost of €250 per tonne of formic acid, the net cost per tonne of CO₂ converted is about €200. These values suggest that the approach could still be competitive with fossil-derived routes if powered by low-cost renewable electricity, where the carbon mitigation benefit provides additional value.
Figure 17 illustrates the sensitivity of the levelized cost of formic acid (LCO-FA) to variations in CO₂ capture cost and renewable electricity price for both flue-gas capture (Panel A) and direct air capture (Panel B). The figure highlights that competitive costs (<€450/t HCOOH) are only achievable under low electricity prices and moderate CO₂ capture costs, emphasizing the importance of inexpensive renewable energy.

In addition to its use as a fuel, formic acid serves as a precursor for methanol and formaldehyde, expanding its industrial value. Integration into internal combustion engines requires minimal modifications, primarily to counter corrosion. These modified engines performed reliably and emitted less CO₂ and other pollutants than conventional fossil fuels. In fuel cell applications, hydrogen derived from formic acid achieved efficiency levels comparable to traditional hydrogen fuel cells, with added benefits in storage and handling. Large-scale CO₂-to-formic acid conversion is financially feasible, particularly when integrated with existing CO₂-emitting industrial processes. The revenue from formic acid sales can offset the costs of CO₂ capture and conversion, enhancing economic attractiveness. From an environmental standpoint, this process lowers net emissions by converting CO₂ into a usable energy carrier, aligning with global decarbonization goals. Electrochemical reduction emerged as the most efficient method, with high selectivity and conversion rates. When powered by renewable energy, this process further improves sustainability. The market for formic acid is poised for growth, driven by demand for hydrogen carriers and alternative fuels. Compared to fossil fuels, formic acid production yields significantly lower environmental impacts, especially when renewable energy is used. Formic acid can be seamlessly integrated into current energy systems, extending the value chain and reducing the environmental footprint of conventional fuels.
Key technical and economic evaluations
We estimated the CO₂ emissions avoided per kWh generated via formic acid compared to fossil fuels like coal or natural gas. Return on investment (ROI) was calculated based on CO₂ capture costs, formic acid production and hydrogen extraction. Costs were compared against hydrogen sources from natural gas or water electrolysis. We modelled the performance of fuel cells using hydrogen from formic acid, analysing conversion losses and comparing efficiency with hydrogen fuel cells and fossil fuel systems. Formic acid’s energy density and cost per kWh were compared to lithium-ion batteries. Given its liquid state at room temperature, it offers practical advantages for long-term storage compared to compressed or cryogenic hydrogen. We evaluated energy efficiency across the process: CO₂ capture → formic acid synthesis → hydrogen release → electricity generation. Losses were identified at each stage, and potential improvements were discussed. Beyond vehicles and fuel cells, formic acid could serve in backup generators, grid-scale energy storage and maritime power systems. Integration into renewable systems was modelled to evaluate its role in balancing intermittent sources like wind and solar.
We examined how much formic acid could be produced from the CO₂ output of large facilities or entire nations. Infrastructure needs for scale-up, including capture, production and distribution, were outlined. If deployed at scale, formic acid systems could significantly reduce global CO₂ emissions. This was modelled by estimating reductions from replacing fossil fuels with formic acid-based systems. We evaluated formic acid’s potential to stabilize grids with high renewable penetration. As a chemical storage medium, it can store excess renewable energy and release it when needed, addressing supply-demand mismatches.
A standout feature of formic acid is its compatibility with existing infrastructure. As a stable liquid, it is far easier to store and transport than hydrogen gas. Through catalytic dehydrogenation, the stored hydrogen can be released on demand for fuel cells, which convert it into electricity efficiently. The RK-X process offers a full-cycle pathway from CO₂ capture to clean energy generation bridging carbon management with energy storage. Hydrogen produced from formic acid can be deployed in decentralized systems, reinforcing grid resilience when paired with solar or wind power. By chemically storing energy and enabling on-demand release, RK-X addresses a key challenge of renewable energy: intermittency. The environmental benefits are substantial, converting a major greenhouse gas into a valuable fuel and contributing to a circular carbon economy. RK-X makes CO₂ capture economically feasible by converting it into marketable formic acid, which can either be sold directly or further refined into hydrogen. Scaling this technology, as seen in a 10,000-tonne CO₂ case study, could create entire industries around renewable chemicals and fuels, promoting innovation, employment and sustainable growth. Unlike many CO₂ utilization technologies requiring harsh conditions or yielding multiple by-products, RK-X prioritizes high selectivity and efficiency. Its modular nature allows tailored implementation, centralized for industrial facilities or decentralized for renewable microgrids.
Discussion
The concept of ‘CO₂ value chain extension’ marks a significant paradigm shift in our approach to energy production and carbon management. Traditionally, the energy value chain has followed a linear path: fossil fuels are extracted, combusted and ultimately converted into CO₂, which is released into the atmosphere. This conventional process has long been associated with environmental degradation, as the resulting carbon emissions contribute significantly to global warming. However, recent technological advancements have enabled a transformative strategy capturing CO₂ and converting it into valuable chemical products such as formic acid and stored hydrogen. This innovation redefines CO₂ not as a terminal by-product, but as a resource, thereby extending the value chain and opening new avenues for energy storage and sustainable fuel generation.
Rather than treating CO₂ as a waste product, the extended CO₂ value chain envisions it as a feedstock for producing renewable energy carriers. Through advanced methods such as photocatalysis and catalytic conversion, captured CO₂ can be converted into formic acid a versatile molecule that serves both as a hydrogen carrier and energy storage medium. Upon dehydrogenation, formic acid releases hydrogen, which can be used in fuel cells or other power generation systems. This sequence from fossil fuels to CO₂, from CO₂ to formic acid and then from formic acid to hydrogen forms a closed-loop system that significantly reduces carbon emissions. From 10,000 tonnes of CO₂, the RK-X system can ideally yield around 10,454 tonnes of formic acid. At 85% conversion efficiency, approximately 8886 tonnes are obtained. Based on molar masses (44 g/mol for CO₂ and 46 g/mol for HCOOH), this yield reflects near-theoretical efficiency. Even accounting for real-world losses like catalyst deactivation, RK-X is optimized to approach ideal output. Formic acid contains 4.4 wt% hydrogen, enabling effective hydrogen extraction for energy use. Ideal output yields around 418 tonnes of hydrogen. At 85% efficiency, this drops to roughly 355 tonnes. With 1 kg of hydrogen equating to 33 kWh, the total energy stored from ideal hydrogen output is about 13.79 GWh. At 85% efficiency, usable energy is approximately 11.73 GWh. RK-X technology presents a high-selectivity, efficient method to transform industrial CO₂ into a usable energy form. Its adaptability to renewable energy sources and existing infrastructure enhances its utility in future energy systems. The process turns CO₂ from an environmental liability into a strategic asset, supporting decarbonization and energy transition goals. By converting 10,000 tonnes of CO₂ into formic acid and extracting hydrogen, RK-X enables both sustainable storage and clean energy generation. These insights affirm the viability of the system and its potential role in building a low-carbon, circular energy economy. Formic acid’s high energy density, safe storage profile, and infrastructure compatibility make it a compelling bridge between fossil and renewable energy systems. Its dual functionality as a hydrogen carrier and direct fuel adds to its strategic value. CO₂-to-formic acid conversion offers a scalable, cost-effective route to extend current energy supply chains while reducing emissions. As global energy strategies evolve, formic acid’s integration into energy infrastructure could become central to a cleaner, more resilient energy future.
This expanded framework fundamentally challenges the traditional fossil fuel narrative. Where once the lifecycle of fossil fuels ended with CO₂ emissions, now the incorporation of CO₂ utilization technologies transforms that endpoint into a circular and value-generating process. The captured CO₂ is no longer vented as a waste stream but is recycled into valuable compounds that contribute to energy sustainability. In doing so, the extended value chain positions CO₂ as an integral component of a future energy system that is cleaner, more efficient and more circular.
The implications of this shift are profound. First, it opens the door to net-negative carbon strategies, where industries can reduce overall atmospheric CO₂ by transforming emissions into usable energy carriers. Second, the hydrogen produced from formic acid offers a clean, efficient and scalable energy source. When used in fuel cells, hydrogen emits only water as a by-product, making it ideal for sustainable power generation and transportation. This strategy not only mitigates dependence on fossil fuels but also lays the groundwork for a hydrogen economy intrinsically linked to carbon capture and conversion technologies.
Moreover, extending the CO₂ value chain unlocks significant economic opportunities. Industries with high CO₂ emissions can reposition themselves by adopting CO₂ conversion technologies, effectively transforming a regulatory and environmental liability into a valuable revenue stream. This transition fosters innovation drives growth in the green technology sector and supports job creation in clean energy fields. In essence, the ability to convert CO₂ into value-added products like formic acid reframes carbon as a strategic asset in the global transition to sustainable energy systems.
Conclusion
In conclusion, the extension of the CO₂ value chain redefines our perception of carbon not as a pollutant, but as a resource with untapped potential. By converting CO₂ into formic acid and subsequently into stored hydrogen, we establish a closed-loop system that enhances both energy security and environmental performance. This paradigm shift from a fossil fuel-to-CO₂ model to a fossil fuel-to-CO₂-through-value-added-products model represents a critical step forward in the global push towards a low-carbon, circular economy.
The conversion of CO₂ into formic acid represents a promising route for extending the value chain of conventional energy sources while reducing greenhouse gas emissions. By capitalizing on the chemical versatility of formic acid, this approach offers a scalable and flexible bridge between conventional and renewable energy systems. As global energy demands evolve, the deployment of CO₂ utilization technologies will play a central role in shaping a sustainable energy future.
This study has demonstrated the technical feasibility, economic viability and environmental benefits of large-scale CO₂-to-formic acid conversion. Among the available pathways, electrochemical reduction emerged as the most promising, offering high energy efficiency and compatibility with renewable power sources. The use of green electricity to drive this process is critical to minimizing the overall carbon footprint and enhancing the sustainability of formic acid production.
From an economic standpoint, the production cost of formic acid via CO₂ reduction is increasingly competitive with conventional methods especially when the intrinsic value of captured CO₂ is factored in. The global market for formic acid is projected to expand, driven by rising demand for hydrogen carriers, chemical intermediates and alternative fuels. This creates a favourable outlook for the adoption of CO₂-based formic acid synthesis at industrial scale.
Environmentally, the life cycle impact of formic acid production is significantly lower than that of traditional fossil fuels, particularly when powered by renewable energy. Integrating this process into existing infrastructure can extend the life and reduce the emissions of legacy energy systems, thereby enhancing overall system resilience and sustainability.
The broader implications for the energy sector are substantial. Large-scale CO₂ conversion into formic acid offers a practical and scalable solution for energy storage and transport, bridging the gap between today’s conventional infrastructure and tomorrow’s renewable systems. By capturing and utilizing CO₂ emissions from fossil-based sources, this pathway not only mitigates environmental impact but also adds value through product diversification and energy integration.
Furthermore, the use of formic acid as a hydrogen carrier offers a transformative opportunity for the transportation sector, presenting a clean and efficient alternative to conventional fossil fuels. Its chemical stability, liquid-state storage and energy density make it ideal for a wide range of applications from mobile power systems to industrial-scale hydrogen storage. In summary, the findings of this study underscore the strategic role of CO₂ utilization in the transition to a sustainable energy future. By extending the CO₂ value chain through the production and use of formic acid, we not only address the urgent challenge of greenhouse gas emissions but also enable a new model of energy circularity, where waste becomes wealth and emissions become energy.
While this study has demonstrated the potential of large-scale CO₂ conversion to formic acid, further research is needed to address the technical and economic challenges of the process. Future research should focus on:
Developing more efficient and cost-effective catalysts for CO₂ reduction.
Optimizing the energy requirements for the electrochemical reduction process.
Exploring the potential for integrating formic acid production into existing energy infrastructure.
Assessing the long-term environmental impact of formic acid production and use.
By addressing these challenges, future research can help to unlock the full potential of formic acid as a key component in the transition to a low-carbon economy. The conversion of CO₂ into formic acid represents a promising pathway for extending the value chain of conventional energy sources and reducing greenhouse gas emissions. By leveraging the versatility and potential applications of formic acid, this approach offers a scalable solution for bridging conventional and renewable energy systems. As the global energy landscape continues to evolve, the development of technologies for CO₂ utilization will be critical to achieving a sustainable energy future.
Footnotes
Ethical considerations
This article does not contain any studies with human or animal participants.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Conceptualization, Z.K.; methodology, Z.K.; validation, Z.K.; formal analysis, Z.K.; investigation, Z.K.; resources, Z.K., M.M.L.; data curation, Z.K.; writing-original draft preparation, review and editing, Z.K., M.M.L.; project administration, Z.K. The authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Z.K. and M.M.L. are paid employees of IOI Investment Zrt, the manufacturing company of the RRR catalyst. This does not alter our adherence to SAGE Journals policies on author responsibilities on sharing data and materials.
Data availability statement
The data that support the findings of this study are available from the author or Balint Analytics Engineering, Research and Service Provider Ltd, but restrictions apply to the availability of these data, which were used under licence for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request.
