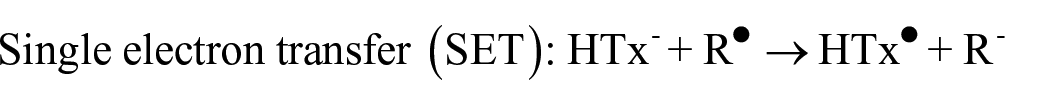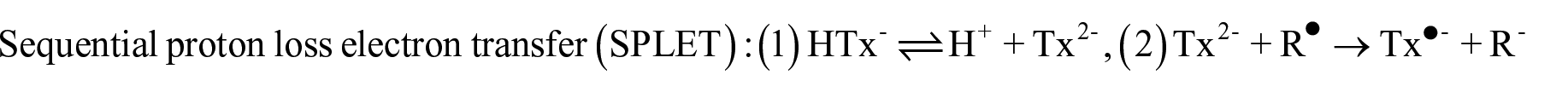Abstract
An adaptation of the DPPH and ABTS assays, commonly used in UV-Vis, to electrochemical techniques is presented as an analytical tool for determining antioxidant activity in non-aqueous media of a commercial Sacha Inchi oil of Amazonian origin. The results obtained through cyclic voltammetry and square wave voltammetry demonstrate that the proposed methodology allows for the determination of antioxidant capacity equivalent to Trolox (TEAC), using tetrafluoroborate of tetrabutylammonium and lithium perchlorate in isopropyl alcohol as supporting electrolytes. In addition, in the ABTS assay, the analysis time is reduced due to the in situ generation of the ABTS•+ radical through electrochemical oxidation. The results confirm that the commercial Amazonian Sacha Inchi oil exhibits antioxidant activity.

Introduction
Sacha Inchi (Plukenetia volubilis L.), a member of the Euphorbiaceae family, is also known as Inca peanut, mountain peanut, Inca nut, or Inca peanut. 1 It is native to the tropical rainforest of the Amazon region in South America, encompassing Colombia, Peru, and Brazil. 2 Due to its significant potential as an economic crop, Sacha Inchi is now cultivated in other parts of the world, such as Southeast Asia. 3 Sacha Inchi seeds are composed of lipids (35%–60%, ω-3, 6, and 9 fatty acids), proteins (25%–30%, cysteine, tyrosine, threonine, and tryptophan), and minerals (potassium, magnesium, calcium, iron, zinc, sodium, and copper). 4 The oil extracted from Sacha Inchi seeds is known to prevent cardiovascular diseases and help reduce cholesterol, triglycerides, and excess fat in the body.5 –7 In addition, Sacha Inchi holds significant economic value in the cosmetics and pharmaceutical industries and is witnessing growing demand for oil-rich food products, seeds, proteins, supplements, and beverages.8,9 This burgeoning interest can further drive the growth of the Sacha Inchi agro-industrial sector. In Colombia, the cultivation of Sacha Inchi aims to establish and consolidate value chains within a sustainable bioeconomy framework. This includes promoting biodiversity, strengthening socio-environmental and economic components, enhancing social fabric, and ensuring forest conservation.10 –12
A wide range of analytical tools are currently available for assessing antioxidant activity employing well-established techniques such as chromatography and spectroscopy, which remain highly versatile and accessible.13,14 These methods typically involve either the reduction in the concentration of a colored radical in solution or the formation of colored complexes. Among the most used analytical procedures are the DPPH (2,2-Diphenyl-1-picrylhydrazyl), the ORAC (Oxygen Radical Absorbance Capacity), the FCT (Ferric Thiocyanate), the FRAP (Ferric-Reducing Antioxidant Power), the CUPRAC (Cupric Ion-Reducing Antioxidant Capacity), and the ABTS ([2,2’-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt]) assays. In recent years, many of these assays have been adapted to electrochemical techniques due to their speed, simplicity, robustness, and the use of relatively inexpensive instrumentation and small sample volumes. Unlike optical methods, electrochemical signals are independent of electromagnetic radiation, unaffected by cell geometry or sample turbidity, and offer an extended dynamic range. Another key advantage of electrochemical approaches is the inherent electroactivity of most antioxidants, as electron transfer plays a central role in their reactions with free radicals. This enables rapid screening of antioxidant capacity across a wide range of organic bioactive compounds, even in complex or colored matrices.14 –17 These experiments are typically conducted in aqueous media, as many extracts are prepared in water. However, non-aqueous media are gaining interest because they allow direct analysis of a broader variety of samples with minimal pretreatment and reduced solvent use, an especially important benefit when working with oils. In this context, the present study describes the electrochemical adaptation of the ABTS and DPPH assays in non-aqueous media for the analysis of commercial Sacha Inchi oil produced in the Amazon region of Colombia.
Methodology
Reagents
Methanol (MeOH), ethanol (EtOH), 2-propanol (i-PrOH), tetrabutylammonium tetrafluoroborate (TBATBF4), lithium perchlorate (LiClO4), ABTS, DPPH, and 6-hydroxy-2,5,7,8-tetramethylchromane-2-carboxylic acid (Trolox) were all analytical grade. Fresh solutions were prepared for each analysis. Due to their limited solubility in organic solvents of lower polarity than methanol, acetone, or ethanol, ABTS and Trolox were not dissolved directly in i-PrOH. Instead, 2 mM stock solutions were prepared in ethanol, and the appropriate volume was added to achieve a final concentration of 0.2 mM in the sample. 18 In i-PrOH, the Sacha Inchi oil filtrate reached a concentration of 60 mg/mL. As supporting electrolytes, LiClO4 and TBATBF4 were selected based on their high solubility in organic media.19 –21 Supplementary Figure 1S shows CVs of 0.05 M TBATBF4 and 0.1 M LiClO4 electrolyte media in i-PrOH; the observed potential windows were −0.4 to 1.4 V and −0.3 to 1.4 V, respectively.
Instruments and equipment
Cyclic voltammetry (CV) and square wave voltammetry (SWV) measurements were performed on an Autolab PGSTAT302N potentiostat/galvanostat. A Teflon cell with a volume of 2.5 mL was used for electrochemical determination (Supplementary Figure 2S). A three-electrode system was used for CV and SWV. The working electrode was glassy carbon (GC), the auxiliary electrode was platinum wire, and the reference electrode was Ag|AgCl|LiCl (2 M). The GC electrode was polished on a microfiber cloth coated with an alumina suspension of particle size 0.3 µm and then washed in deionized water in an ultrasonic cleaner. All experiments were carried out at room temperature.
Electrochemical characterization
A stock solution of 2 mM DPPH in i-PrOH, 2 mM ABTS in EtOH, 20 mg/mL Sacha Inchi oil i-PrOH, and 2 mM Trolox in EtOH was prepared for electrochemical characterization. The solutions for the in-cell analysis were 0.2 mM DPPH + 100 mM LiClO4 and 0.2 mM ABTS + 50 mM TBATBF4 in i-PrOH.
Determination of antioxidant activity
For the DPPH assay, the solution consisted of 0.2 mM DPPH + 100 mM LiClO4 + 0–100 μM Trolox or 0–15 mg/mL Sacha Inchi oil. The solutions were prepared separately a few minutes prior to each measurement and stored in the dark for 30 min. The determination was conducted using SWV under the following conditions: 20 mV amplitude, 25 Hz frequency, 5 mV potential step, and an oxidation scan within a potential window of −0.1 to 0.7 V. For the ABTS assay, the solution was 0.2 mM ABTS + 50 mM TBATBF4 + 0–100 μM Trolox or 0–15 mg/mL Sacha Inchi oil. The solutions were prepared separately minutes before each measurement. The determination was performed by CV, under the following conditions: a potential stop of 1.0 V for 10 , and subsequent reduction sweep between 0.8 and 0.3 V at a scan rate of 100 mV/s.
Results and discussion
Electrochemical characterization of ABTS
A solubility test for ABTS was conducted before its electrochemical characterization in i-PrOH, and it was observed that ABTS is soluble in i-PrOH up to a concentration of 0.2 mM. Due to its low solubility in i-PrOH, a 2-mM stock solution of ABTS was prepared in EtOH, the solvent commonly used for determining antioxidant capacity. 22 Figure 1(a) shows a CV of 0.2 mM ABTS + 0.05 M TBATBF4 in i-PrOH. Two anodic peaks were observed at potentials of 0.71 and 1.16 V, corresponding to the oxidation of ABTS ↔ ABTS•+ + e⁻ and ABTS•+ ↔ ABTS2+ + e⁻, respectively. An analysis of the effect of the scan rate revealed that at high scan rates (500 mV/s), two redox reduction processes were observed at 0.93 and 0.57 V. However, at a low scan rate (50 mV/s), the second redox reduction process, corresponding to ABTS2+ + e⁻ → ABTS•+, was not observed. Instead, the reduction peak current for the first redox process was approximately two times the oxidation peak current. This electrochemical behavior has been previously observed in aqueous solutions 23 and ionic liquids 24 corresponding to the co-proportioning reaction of ABTS + ABTS2+ ↔ 2ABTS•+. Figure 1(b) shows CVs carried out on the first redox couple. For an electrochemically reversible one-electron reaction, the peak potential separation (ΔEp) should be approximately 59 mV and independent of the scan rate. 25 However, the ΔEp value for the redox process of ABTS ↔ ABTS•+ + e⁻ is 110 mV and increases with the scan rate, indicating a quasi-reversible redox process. These results may suggest slow electron transfer kinetics or could be attributed to the uncompensated ohmic resistance of the electrolyte. 24 In addition, two straight lines were obtained when plotting the peak current against the square root of the scan rate, indicating a diffusion-controlled process (Figure 1(c)). SWV assessed the stability of the 0.2 mM ABTS solution in EtOH over 10 days following its preparation. Starting on the second day, a decrease in the peak oxidation current intensity at a potential of 0.65 V was observed, and this decrease continued progressively until the tenth day. Thus, the results indicate a low stability of the ABTS stock solution in EtOH.

0.2 mM ABTS + 0.05 M TBATBF in i-PrOH: (a) CV, (b) different scan rates, (c) current versus scan rate1/2, and (d) SWV.
Electrochemical characterization of DPPH
Electrochemical reactions in non-aqueous media require organic solvent and charge carriers. i-PrOH was the solvent of choice for electrochemical determinations from solubility tests. DPPH• was observed to dissolve up to 2 mM in i-PrOH without difficulty. Figure 2(a) shows the CV of DPPH• in 0.1 M LiClO4 at a sweep rate of 100 mV/s. Two quasi-reversible one-electron anodic redox processes at 0.36 V and 0.94 V are observed, corresponding to the oxidation of DPPH• ↔ DPPH + e⁻ and DPPH ↔ DPPH+ + e⁻, respectively. Other authors have observed similar electrochemical behavior.26,27 The first redox couple oxidation analysis at different scan rates (10–400 mV/s, Figure 2(b)) shows that the peak current changes linearly with the square root of the scan rate (Figure 2(c)), indicating a diffusion-controlled process. 28 Under these conditions, the peak current ratio (Ipa/Ipc) is close to unity, indicating that DPPH• is stable on the electrode surface under the experimental conditions. On the other contrary, the stability of the DPPH• radical solution over time is also a crucial parameter to evaluate. Figure 2(d) shows SWV under the same experimental conditions as the CV measured at different intervals (0–25 min). A decrease in the oxidation peak current of approximately 1.25% is observed 25 min after the solution is prepared. Some authors have reported that the DPPH• radical is unstable during storage and recommend using it within 24 h after preparation.29 –31

2 mM DPPH• + 0.1 M LiClO4 in i-PrOH: (a) CV, (b) different scan rates, (c) current versus scan rate1/2, and (d) SWV.
Determination of antioxidant activity
The antioxidant activity assay using ABTS is influenced by the method of generating the ABTS•+ radical cation. 32 Traditionally, ABTS•+ is produced via a chemical oxidation reaction of ABTS before UV-Vis spectrophotometric determination, typically using oxidizing agents such as K2S2O8 or MnO2 is. 33 This step is often the most time-consuming part of the assay. As an alternative, a simpler and faster approach involves the electrochemical in situ generation of ABTS•⁺ via a controlled potential step. As shown in Figure 1(c), the redox behavior of ABTS•⁺ is diffusion-controlled. Based on this, applying a potential of 1.0 V for a defined period generates a reproducible and approximately constant amount of the radical cation at the electrode surface. When the applied bias is stopped and a reverse potential is quickly applied, the resulting reduction current is proportional to the concentration of ABTS•⁺ remaining near the electrode surface. Under these conditions, that amount of ABTS•⁺ is influenced by both its diffusion into the bulk solution and its chemical reaction with antioxidant species present in the medium (e.g. Trolox or Sacha Inchi oil). Therefore, antioxidant activity can be evaluated by measuring the decrease in the cathodic peak current during the reduction scan after adding increasing concentrations of antioxidant compounds to the electrochemical cell (Figure 3(a) and (c)). One aspect to consider is the chemical reaction kinetics; for antioxidant species with slower reaction kinetics, the reaction may be incomplete due to the diffusion into the bulk solution and the rapid application of the reduction potential after generation of ABTS•+. However, for antioxidants with faster reaction kinetics, such as Trolox and tocopherol (present in Sacha Inchi oil), electrochemical determination offers significant advantages. 34 Figure 3(b) and (d) shows the linear relationship between the cathodic peak current (Icp), expressed as a percentage of inhibition (% inhibition ratio = [Icp ABTS•+ − Icp ABTS•++ sample/Icp ABTS•+]*100), and the concentration of Sacha Inchi oil and Trolox, respectively. It can be observed that the cathodic peak current decreases progressively as the antioxidant concentration in the medium increases, reflecting the dose-dependent scavenging activity of the antioxidants (Figure 3(a) and (c)).

CVs for 0.2 mM ABTS + 0.05 M TBATBF in i-PrOH: (a) 0–15 mg/mL Sacha Inchi oil and (c) 0–100 μM Trolox. (b) and (d) % inhibition ratio versus concentration of Sacha Inchi oil and Trolox, respectively.
On the contrary, the determination of antioxidant activity via the DPPH• method using SWV is an adaptation of the UV-Vis spectrophotometric technique, differing in how the remaining concentration of the DPPH• radical is measured after the chemical reaction. Under these conditions, the final concentration of DPPH• depends on the sample concentration, its intrinsic antioxidant activity, and the reaction time. 14 Figure 4(a) and (c) displays the SWV responses for 0.2 mM DPPH• with increasing additions of Sacha Inchi oil and Trolox, respectively. For both determinations, a decrease in the anodic peak current at 0.34 V, characteristic of the DPPH• radical, was observed. However, in the SWV of DPPH• + Sacha Inchi oil, a shift of the anodic peak potential to more positive values was noted. For instance, for 0.2 mM DPPH• + 5.0 mg/mL Sacha Inchi oil, the anodic peak potential was 0.41 V. This suggests an increase in solution resistance due to the rising concentration of the oil (Figure 4(a)). From the anodic peak current (Iap), the percentage of inhibition (% inhibition ratio = [Iap DPPH• − Iap DPPH •+ sample/Iap DPPH•]*100) was calculated relative to the concentration of Sacha Inchi oil and Trolox, shown in Figure 4(b) and (d), respectively.

SWV for 0.2 mM DPPH• + 0.1 M LiClO4 in i-PrOH: (a) 0–15 mg/mL Sacha Inchi oil and (c) 0–100 μM Trolox. (b) and (d) % inhibition ratio versus concentration of Sacha Inchi oil and Trolox, respectively.
Antioxidant activity results are commonly reported using the IC50 parameter, which indicates the sample concentration (antioxidant) required to reduce the initial signal (e.g. absorbance or peak current) of the ABTS•+ or DPPH• radical solution by 50%.35,36 This parameter is highly dependent on experimental conditions and is therefore unsuitable for comparisons between samples analyzed under different methodologies. On the other hand, the Trolox Equivalent Antioxidant Capacity (TEAC), defined as the ratio between the IC50 of Trolox and the IC50 of the sample of interest (TEAC = IC50 Trolox/IC50 sample), both determined under identical experimental conditions, is frequently used for comparative purposes.14,37,38 6-Hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox), an analog of vitamin C is the reference standard against which the antioxidant capacity of other compounds is compared and expressed. This is due to its well-established antioxidant activity, which has been extensively studied both theoretically and experimentally. 39 Trolox is known to act as a potent radical scavenger through at least four distinct reaction mechanisms, as follows:
Trolox can react with a wide variety of radical species. It has also been reported that, in non-aqueous media, HT and RAF are the most relevant pathways, a significant observation, as they are also the most kinetically favored34,40 and this is particularly relevant in the ABTS assay, as previously mentioned.
Table 1 presents the IC50 values for Trolox, Sacha Inchi oil, and their corresponding TEAC values. The antioxidant activities measured using the DPPH• and ABTS•+ radicals were 0.0076 and 0.0012, respectively. Comparative studies on the ABTS•+ and DPPH• assays have concluded that the observed differences arise from the complex reaction mechanisms associated with each radical, particularly in samples containing multiple antioxidant species.41,42 In such cases, specific reaction types, such as HAT or SET, may be favored.43,44 The antioxidant activity of Sacha Inchi oil has been primarily attributed to its high content of tocopherols, flavonoids, carotenoids, and phenolic compounds.45,46 These substances are especially effective in combating lipid oxidation due to their structural affinity with cell membranes and their ability to act in lipophilic media. 47
TEAC values determined using the DPPH• and ABTS•+ assays.

It is also well established that antioxidant activity strongly depends on the oil extraction process, particularly when seeds are exposed to high temperatures (>100 °C). Thermal treatment significantly reduces the phenol and tocopherol content, leading to a marked decrease in antioxidant activity.4,48,49
Conclusion
The electrochemical techniques employed to evaluate antioxidant activity through the adaptation of DPPH and ABTS free radical scavenging assays are straightforward and rapid. Moreover, they provide an analytical alternative to the classical spectrophotometric technique, where results are often compromised by the color and turbidity of complex natural samples. The electrochemical determination of antioxidant activity in non-aqueous media expands the applicability of the method to the analysis of oils without requiring additional pretreatments that could influence the outcomes. In this specific case, the findings support the potential of Amazonian Sacha Inchi oil as a natural product with significant antioxidant activity.
Supplemental Material
sj-docx-1-chl-10.1177_17475198251349265 – Supplemental material for Antioxidant activity of Sacha Inchi oil (Plukenetia volubilis): Electrochemical determination in a Non-Aqueous medium
Supplemental material, sj-docx-1-chl-10.1177_17475198251349265 for Antioxidant activity of Sacha Inchi oil (Plukenetia volubilis): Electrochemical determination in a Non-Aqueous medium by L Miguel Hernandez-Parra, Jhon I Maldonado-Rodriguez, Lisette D Ruiz-Bravo and Jaime F Martínez-Suárez in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank Sistema General de Regalías (SGR) and Universidad de la Amazonia for the support through the project “Fortalecimiento de vocaciones científicas en jóvenes mediante becas-pasantías en la región centro sur. Caquetá, Amazonas, Putumayo, Huila, Tolima” (BPIN: 2022000100076).
Ethical considerations
No experiments involving animals, human subjects, or any living organisms were conducted in this study. All experiments were performed using commercially available Sacha Inchi oil and chemical reagents.
Author contributions
Conceptualization: L. Miguel Hernandez-Parra and Jaime F. Martínez-Suárez; Methodology: L. Miguel Hernandez-Parra and Jaime F. Martínez-Suárez; Investigation: L. Miguel Hernandez-Parra; Formal analysis: L. Miguel Hernandez-Parra and Jaime F. Martínez-Suárez; Writing—original draft: L. Miguel Hernandez-Parra and Jaime F. Martínez-Suárez; Writing—review & editing: L. Miguel Hernandez-Parra, Jaime F. Martínez-Suárez, Jhon I. Maldonado-Rodriguez, and Lisette D. Ruiz-Bravo; and Supervision: Jaime F. Martínez-Suárez, Jhon I. Maldonado-Rodriguez, and Lisette D. Ruiz-Bravo.
Funding
This research was supported by Sistema General de Regalías (SGR) and Universidad de la Amazonia through the project “Fortalecimiento de vocaciones científicas en jóvenes mediante becas-pasantías en la región centro sur. Caquetá, Amazonas, Putumayo, Huila, Tolima” (BPIN: 2022000100076).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.